-
question_answer1)
A student poured an equal amount of water into \[4\] containers as shown below.

What result does this experiment show?
A)
Water has definite volume. done
clear
B)
Water has no definite shape. done
clear
C)
Water has definite mass. done
clear
D)
Water has no definite volume. done
clear
View Solution play_arrow
-
question_answer2)
Which of the following has a direct effect on the K.E. of particles?
A)
Pressure done
clear
B)
Potential energy done
clear
C)
Temperature done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer3)
Look at the Venn diagram given below.
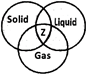
Which of the following can be placed in\['Z'\]?
A)
Water done
clear
B)
Dry ice done
clear
C)
Salt done
clear
D)
Jelly done
clear
View Solution play_arrow
-
question_answer4)
Based on the statements given below choose the correct answer.
| Some sugar can be added to a glassful of water without causing an overflow. |
| Liquids have spaces present between the molecules |
A)
\[P\] and \[Q\] are true and \[Q\] explains \[P\]. done
clear
B)
\[P\] and \[Q\] are true but \[Q\] does not explain\[P\] done
clear
C)
Only \[P\] is true. done
clear
D)
Only \[Q\] is true. done
clear
View Solution play_arrow
-
question_answer5)
What happens to a substance when the space between its particles is decreased?
A)
The volume of the substance increases. done
clear
B)
The volume of the substance decreases. done
clear
C)
The mass of the substance increases. done
clear
D)
The mass of the substance decreases. done
clear
View Solution play_arrow
-
question_answer6)
Match the entries in Column I with those in Column II.
| | Column-I | | Column-II |
| [a] | Evaporation | 1. | Liquid to gas at a fixed temperature |
| [b] | Boiling | 2. | Solid to gas |
| [c] | Sublimation | 3. | Gas to solid |
| [d] | Hoar frost | 4. | Liquid to gas at any temperature |
A)
a - 4, b - 1, c - 2, d - 3 done
clear
B)
a - 1, b - 2, c - 3, d - 4 done
clear
C)
a - 2, b - 3, c - 4, d - 1 done
clear
D)
a - 4, b - 1, c - 3, d - 2 done
clear
View Solution play_arrow
-
question_answer7)
Which of the following have a regular, repeated molecular pattern and large number of free surfaces in three dimensional space?
A)
Solids and liquids done
clear
B)
Liquids and gases done
clear
C)
Solids done
clear
D)
Gases done
clear
View Solution play_arrow
-
question_answer8)
Which of the following is used for determining the mass of a solid?
A)
Microscope done
clear
B)
Balance done
clear
C)
Thermometer done
clear
D)
Telescope done
clear
View Solution play_arrow
-
question_answer9)
Which element has neither a definite shape nor volume?
A)
Sodium done
clear
B)
Hydrogen done
clear
C)
Iodine done
clear
D)
Lithium done
clear
View Solution play_arrow
-
question_answer10)
Which of the following statements is NOT true?
A)
All forms of matter have volume. done
clear
B)
All forms of matter have colour. done
clear
C)
All forms of matter have mass. done
clear
D)
Matter can be living or non-living. done
clear
View Solution play_arrow
-
question_answer11)
Three containers \[X,\,\,Y\] and \[Z\] as shown below were filled with water and then left near an open window. The purpose was to find out whether the exposed surface area of the water affects the rate of evaporation of water.
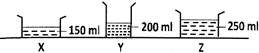
Why was the experiment NOT a fair one?
A)
The exposed surface areas were different. done
clear
B)
Different types of containers were used. done
clear
C)
The amount of water in the containers was different. done
clear
D)
The containers were not covered. done
clear
View Solution play_arrow
-
question_answer12)
What happens to the inter-particle attractive forces with an increase in the distance between the particles?
A)
They increase. done
clear
B)
They decrease. done
clear
C)
They increase moderately. done
clear
D)
They remain the same. done
clear
View Solution play_arrow
-
question_answer13)
Bromine has a melting point of \[-{{7.2}^{o}}C\] and a boiling point of\[{{59}^{o}}C\]. At what temperature will bromine have a definite volume but no definite shape?
A)
\[{{65}^{o}}C\] done
clear
B)
\[{{36}^{o}}C\] done
clear
C)
\[-{{26}^{o}}C\] done
clear
D)
\[98\,\,K\] done
clear
View Solution play_arrow
-
question_answer14)
What happens when a solid melts?
A)
Its molecules move farther apart. done
clear
B)
Its molecules move closer. done
clear
C)
The movement of its molecules decreases. done
clear
D)
The temperature decreases although heat is supplied. done
clear
View Solution play_arrow
-
question_answer15)
Calculate the temperature at which both the Celsius and Fahrenheit scales show the same reading?
A)
\[40\,\,K\] done
clear
B)
\[{{100}^{o}}F\] done
clear
C)
\[-{{40}^{o}}C\] done
clear
D)
\[-{{100}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer16)
Why does a gas from a small container when transferred to a larger container occupy all the space available?
A)
Gas has no definite mass. done
clear
B)
Gas has no definite shape. done
clear
C)
Gas has no definite volume. done
clear
D)
Gas has no definite density. done
clear
View Solution play_arrow
-
question_answer17)
Which of the following metals is NOT a solid at room temperature?
A)
Iron done
clear
B)
Aluminium done
clear
C)
Mercury done
clear
D)
Steel done
clear
View Solution play_arrow
-
question_answer18)
Why can't solids be compressed?
A)
The inter-particle attractive forces are very weak. done
clear
B)
The movement of the constituent particles are not restricted. done
clear
C)
The constituent particles are closely packed. done
clear
D)
None of the above. done
clear
View Solution play_arrow
-
question_answer19)
Find the rise in temperature of \[1\,\,kg\] of water if \[1000\,\,J\] of heat is supplied to it.
A)
\[{{\left( \frac{1000}{4186} \right)}^{o}}C\] done
clear
B)
\[{{\left( \frac{4186}{1000} \right)}^{o}}C\] done
clear
C)
\[{{(1000\times 4186)}^{o}}C\] done
clear
D)
\[{{(4186-1000)}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer20)
When the vapour pressure of a liquid is equal to the atmospheric pressure, what happens to the liquid?
A)
The liquid freezes. done
clear
B)
The liquid evaporates. done
clear
C)
The liquid boils. done
clear
D)
The liquid does not undergo any change. done
clear
View Solution play_arrow
-
question_answer21)
Which of the following water molecules \[W,\,\,X,\,\,Y\] or \[Z\] can easily escape to form water vapour?
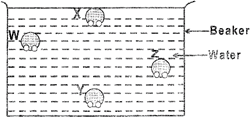
A)
\[W\] done
clear
B)
\[X\] done
clear
C)
\[Y\] done
clear
D)
\[Z\] done
clear
View Solution play_arrow
-
question_answer22)
Which of the following statements is TRUE of matter?
A)
The states of matter are inter-convertible. done
clear
B)
Force of attraction varies from one kind of matter to another done
clear
C)
The states of matter can be changed by changing the temperature or pressure, done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer23)
Which of the following will diffuse faster than the others?
A)
Water done
clear
B)
Petrol done
clear
C)
Perfume done
clear
D)
Milk done
clear
View Solution play_arrow
-
question_answer24)
Identify the normal temperature of the human body.
A)
\[{{40}^{o}}F\] done
clear
B)
\[{{40}^{o}}C\] done
clear
C)
\[{{37}^{o}}C\] done
clear
D)
\[{{37}^{o}}F\] done
clear
View Solution play_arrow
-
question_answer25)
What is the \[S.I.\] unit of temperature?
A)
Centigrade done
clear
B)
Fahrenheit done
clear
C)
Kelvin done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer26)
Which of the following processes involve absorption of energy?

A)
Only (i) and (ii) done
clear
B)
Only (ii) and (iii) done
clear
C)
Only (i) and (iii) done
clear
D)
(i), (ii) and (iii) done
clear
View Solution play_arrow
-
question_answer27)
When does the thermal energy of a body increase?
A)
When the K.E, of the body decreases. done
clear
B)
When the P.E. of the body increases done
clear
C)
When the mechanical energy of the body decreases done
clear
D)
When the K.E. of molecules in a body increases done
clear
View Solution play_arrow
-
question_answer28)
A student finds that as he blows air into a balloon, it becomes bigger. What inference can he make from the above?
A)
Air is a gas. done
clear
B)
Air has mass done
clear
C)
Air is matter done
clear
D)
Air occupies space done
clear
View Solution play_arrow
-
question_answer29)
What happens when ice is converted into water?
A)
Heat is absorbed done
clear
B)
Heat is released done
clear
C)
Temperature increases done
clear
D)
Temperature decreases done
clear
View Solution play_arrow
-
question_answer30)
Identify the freezing point of pure water,
A)
\[-{{4}^{o}}C\] done
clear
B)
\[{{100}^{o}}C\] done
clear
C)
\[{{10}^{o}}C\] done
clear
D)
\[{{0}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer31)
Under what conditions, can a gas be cooled and converted into liquid?
A)
At its critical temperature by decreasing pressure on it done
clear
B)
Above its critical temperature by increasing pressure on it done
clear
C)
Above its critical temperature by decreasing pressure on it done
clear
D)
Below its critical temperature by increasing pressure on it done
clear
View Solution play_arrow
-
question_answer32)
Which of the following substances can be compressed?
A)
Sponge done
clear
B)
Stone done
clear
C)
Magnet done
clear
D)
Diamond done
clear
View Solution play_arrow
-
question_answer33)
A class teacher demonstrates the effect of impurities on the boiling point of water using the arrangement of apparatus as shown below.
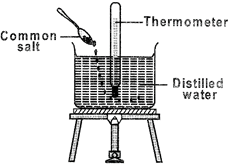
What could be the probable thermometer readings recorded during the experiment?
A)
\[{{97}^{o}}C\] done
clear
B)
\[{{100}^{o}}C\] done
clear
C)
\[{{102}^{o}}C\] done
clear
D)
\[{{99}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer34)
At what temperature should all the gases occupy zero volume?
A)
\[{{0}^{o}}C\] done
clear
B)
\[-{{273}^{o}}C\] done
clear
C)
\[{{273}^{o}}C\] done
clear
D)
\[{{100}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer35)
On which of the following factors/ does the molecular arrangement of a substance depend?
A)
Temperature and pressure done
clear
B)
Concentration and temperature done
clear
C)
Temperature, pressure and concentration done
clear
D)
Volume and pressure done
clear
View Solution play_arrow
-
question_answer36)
What does conversion of \[475\,\,K\] into celsius scale give?
A)
\[{{301.85}^{o}}C\] done
clear
B)
\[{{273}^{o}}C\] done
clear
C)
\[{{207}^{o}}C\] done
clear
D)
\[{{201.85}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer37)
Which of the following properties is different for solids, liquids and gases?
A)
Movement of molecules done
clear
B)
Particle size of the substance done
clear
C)
Mass of the substance done
clear
D)
Energy changes done
clear
View Solution play_arrow
-
question_answer38)
Non-reacting gases have the tendency to mix with each other. What is this phenomenon called?
A)
Evaporation done
clear
B)
Diffusion done
clear
C)
Effusion done
clear
D)
Explosion done
clear
View Solution play_arrow
-
question_answer39)
The figures given below show the state of molecules of different types of matter. Which figure shows that water is being heated?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer40)
What could be the diameters of the molecules of matter?
A)
\[{{10}^{-7}}m\] done
clear
B)
\[{{10}^{-11}}m\] done
clear
C)
\[{{10}^{-9}}m\] done
clear
D)
\[{{10}^{-15}}m\] done
clear
View Solution play_arrow

