question_answer 1)
A piece of chalk can be broken into small particles by hammering but a piece of iron cannot be broken into smaller particles by hammering. It illustrates that
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 2)
Which of the following represents the correct increasing order of the densities of given substances?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 3)
Direction: Study the given graph carefully and answer the following question.
What is the physical state of matter at points 1, 3 and 5 respectively?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 4)
Direction: Study the given graph carefully and answer the following question.
What is represented by straight lines QR and ST in the graph?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
Sneha set up the experiment for melting of ice in a beaker and noted down the temperature throughout the process. What do you think she observed when the ice was melting?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
Which of the following statements are correct?
I. Temperature changes during the change of a state. II. Dry ice gets converted directly into gaseous state under normal atmospheric conditions. III. Higher boiling point of liquid indicates weaker intermolecular forces. IV. Latent heat of vaporisation is generally higher than the latent heat of fusion for a substance.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 7)
Read the given statements and mark the correct option.
Statement 1: Solid \[\text{C}{{\text{O}}_{\text{2}}}\] changes its state when exposed to air.
Statement 2: \[\text{C}{{\text{O}}_{\text{2}}}\] undergoes sublimation.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
Match column I with column II and choose the correct option from the codes given below.
Column I Column II P. Hot tea poured in saucer gets cooled faster. (i) Evaporation increases with increase in temperature Q. We feel more cold after a hot water bath than a cold water bath. (ii) Evaporation increases with decrease in humidity. R. Water is cool in earthen pitcher during dry hot day. (iii) Evaporation increases with increase in wind speed. S. We feel comfortable under a moving fan in summer (iv) Evaporation increases with increase in surface area.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
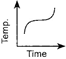
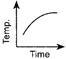
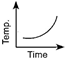
Which of the following curves would be obtained on heating solid naphthalene to a temperature which is above its melting point?
A)
B)
C)
D)
View Solution play_arrow
question_answer 10)
What is the correct increasing order of molecular motion (kinetic energy) of particles in the following?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
An organic compound ‘X’ has a melting point of - \[33{}^\circ C\] and a boiling point of \[66{}^\circ C.\]
The physical states of substance ‘X’ at temperature - \[100{}^\circ C,\] \[0{}^\circ C\] and \[100{}^\circ C\] are respectively
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
An experiment is performed as shown in the given figure.
The conclusion we can draw from the above experiment is that
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
Fill in the blanks P, Q, R and S left in the table with appropriate words.
Properties
Solid
Liquid
Gas
Inter particle spaces
Very less
Comparatively large
___P___
Intermolecular Forces
___Q___
Weak
Very weak
Compressibility
___R___
Very Small
High
Diffusion
Negligible
___S___
Very Fast
A)
B)
C)
D)
View Solution play_arrow
question_answer 14)
Ritwik was given four inflammable substances P, 0, R and S. He was asked to handover them to Ramesh who was the resident of a hot area (temperature, \[46{}^\circ C\]). Ritwik collected the data about the substances as shown in the table. Which substances Ritwik should not carry to the hot area where Ramesh lives?
Substance
Ignition Temperature
P
40°C
Q
38°C
R
52°C
S
59°C
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
Choose the correct statements about plasma state.
I. The state consists of super energetic and super excited particles. II. The particles of plasma state are in the form of ionised gases. III. The plasma is created on stars. IV. Colour of plasma glow depends on temperature of gas only.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
Observe the given figure carefully.
Which of the following statements is incorrect?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 17)
The given apparatus is used to study the diffusion of a number of gases at the same temperature and pressure.
Which of the following pairs of gases would diffuse into the vacuum at the same speed?
[Given: Atomic mass of H=1u, C=12u, N=14u, S=32u, O=16u]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
Diagrams W, X and Y show how the particles of a substance are packed at different temperatures.
The given graph shows the temperature changes which occur on warming the substance. In which region of the graph would all the particles be packed as in Y?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 19)
Observe the given diagram showing change in state carefully.
Which of the following is correct identification for processes P to U?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 20)
Match column I with column II and select the correct option from the codes given below.
Column I Column II (P) Liquid (i) Highly compressible (Q) Gas (ii) Definite volume (R) Plasma (iii) Super low density (S) Bose-Einstein condensate (iv) Super energetic
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
