-
question_answer1)
Which pair of substances given below are isotopes?
A)
\[_{6}^{12}C\]and\[_{6}^{14}C\] done
clear
B)
Carbon dioxide and carbon monoxide done
clear
C)
Diamond and graphite done
clear
D)
\[{{C}_{2}}{{H}_{4}}\]and\[{{C}_{3}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer2)
Which valence shell electron arrangement is that of a metallic element?
A)
\[2,\,\,1\] done
clear
B)
\[2,\,\,4\] done
clear
C)
\[2,\,\,5\] done
clear
D)
\[2,\,\,7\] done
clear
View Solution play_arrow
-
question_answer3)
An element \[X\] has two isotopes, which may be represented as \[^{238}X\] and\[^{235}X\]. How does \[^{238}X\] differ from\[^{235}X\]?
A)
\[^{238}X\]has \[3\] more protons and \[3\] more electrons than\[235\]. done
clear
B)
\[^{238}X\] has \[3\] more protons, but same number of electrons as\[235\]. done
clear
C)
\[^{238}X\] has \[3\] more neutrons, but the electrons or protons are same as\[235\]. done
clear
D)
\[^{238}X\] has \[3\] more protons, but same number of electrons as\[235\]. done
clear
View Solution play_arrow
-
question_answer4)
An atom of an element contains \[17\] protons, \[18\] neutrons and \[17\] electrons. Which of the following will be the atomic structure of another isotope of this element?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer5)
The diagram given below represents the atom of an element.
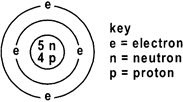
Which symbol gives the above information?
A)
\[_{4}^{9}Be\] done
clear
B)
\[_{4}^{9}F\] done
clear
C)
\[_{4}^{9}B\] done
clear
D)
\[_{4}^{9}O\] done
clear
View Solution play_arrow
-
question_answer6)
What does an atom and an ion of the isotope of sodium \[_{11}^{22}Na\] both contain?
A)
\[11\] electrons done
clear
B)
\[12\] neutrons done
clear
C)
\[23\] protons done
clear
D)
\[23\] neutrons done
clear
View Solution play_arrow
-
question_answer7)
What can be deduced from the symbol\[_{2}^{4}He\]?
A)
An atom of helium contains two electrons. done
clear
B)
An atom of helium has two protons and four neutrons in its nucleus. done
clear
C)
The proton (atomic) number of helium is four. done
clear
D)
Helium acts as a diatomic molecule. done
clear
View Solution play_arrow
-
question_answer8)
Which of the following ions has the same number of electrons as a krypton atom with atomic number\[36\]?
A)
Chlorine done
clear
B)
Sodium done
clear
C)
Rubidium done
clear
D)
Xenon done
clear
View Solution play_arrow
-
question_answer9)
In elements \[_{15}^{31}P\]and\[_{16}^{32}S\], which of the following are the same?
A)
Number of nucleons done
clear
B)
Number of electrons done
clear
C)
Number of neutrons done
clear
D)
Number of protons done
clear
View Solution play_arrow
-
question_answer10)
What is valency?
A)
The combining capacity of an element done
clear
B)
The atomicity of an element done
clear
C)
The oxidation number of an element done
clear
D)
All of the above done
clear
View Solution play_arrow
-
question_answer11)
An element \[X\] forms a un-positive ion with the electronic structure\[2,\,\,8,\,\,8\]. What is the atomic number of\[X\]?
A)
\[16\] done
clear
B)
\[17\] done
clear
C)
\[18\] done
clear
D)
\[19\] done
clear
View Solution play_arrow
-
question_answer12)
The letters \[X,\,\,Y\] and \[Z\] represent different atoms. \[_{19}^{40}X\,\,\,\,\,_{19}^{39}Y\,\,\,\,\,\,\,_{20}^{40}Z\] What can be deduced from the proton numbers and nucleon numbers of\[X,\,\,Y\] and\[Z\]?
A)
\[X\] and \[Y\] are the same elements. done
clear
B)
\[X\] and \[Z\] are the same elements. done
clear
C)
\[X\] has more protons than \[Y\]. done
clear
D)
\[Z\] has more neutrons than\[Y\]. done
clear
View Solution play_arrow
-
question_answer13)
What is the maximum number of electrons that can be accommodated in the 4th energy level?
A)
\[32\] done
clear
B)
\[18\] done
clear
C)
\[2\] done
clear
D)
\[8\] done
clear
View Solution play_arrow
-
question_answer14)
A neutral atom of an element has a nucleus with a nuclear charge \[13\] times and mass \[27\] times as that of hydrogen nucleus. What is the ratio of electrons to protons in its stable positively charged ion?
A)
\[27:13\] done
clear
B)
\[10:13\] done
clear
C)
\[20:24\] done
clear
D)
\[10:14\] done
clear
View Solution play_arrow
-
question_answer15)
What does Bohr's atomic theory explain?
A)
About quantum numbers done
clear
B)
About the shape of sub-levels done
clear
C)
About nucleus done
clear
D)
About stationary states done
clear
View Solution play_arrow
-
question_answer16)
Which of the following observations of \[\alpha -\]ray scattering experiment led to the discovery of a small positively charged nucleus in the centre?
A)
Most of the \[\alpha -\]particles passed straight. done
clear
B)
Most of the \[\alpha -\]particles rebounded after hitting the atoms. done
clear
C)
Only a few \[\alpha -\]particles deflected away from their path. done
clear
D)
Most of the \[\alpha -\]particles rebounded. done
clear
View Solution play_arrow
-
question_answer17)
Which of the following electronic configurations is incorrect7
A)
\[Be(4)=2,\,\,2\] done
clear
B)
\[O(8)=2,\,\,6\] done
clear
C)
\[S(16)=2,\,\,6,\,\,8\] done
clear
D)
\[P(15)=2,\,\,8,\,\,5\] done
clear
View Solution play_arrow
-
question_answer18)
An atom has mass number 23 and atomic number\[11\]. How many protons, electrons and neutrons are present in the atom?
A)
\[11,\,\,12,\,\,13\] done
clear
B)
\[11,\,\,11,\,\,12\] done
clear
C)
\[11,\,\,11,\,\,13\] done
clear
D)
\[11,\,\,14,\,\,15\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following electronic configurations represents a noble gas?
A)
\[2,\,\,8,\,\,2\] done
clear
B)
\[2,\,\,8,\,\,6\] done
clear
C)
\[2,\,\,8\] done
clear
D)
\[2,\,\,8,\,\,8,\,\,2\] done
clear
View Solution play_arrow
-
question_answer20)
An element has atomic number \[17\] and mass number\[37\]. There are \[17\] electrons outside the nucleus. Find the number of neutrons in it.
A)
\[20\] done
clear
B)
\[21\] done
clear
C)
\[17\] done
clear
D)
\[37\] done
clear
View Solution play_arrow
-
question_answer21)
What does the mass number of an atom represent?
A)
Only the number of protons done
clear
B)
The number of protons and neutrons done
clear
C)
The number of protons and electrons done
clear
D)
Only the number of neutrons done
clear
View Solution play_arrow
-
question_answer22)
The atom of an element has \[10\] electrons, \[10\] protons and \[12\] neutrons. Find the mass number of the element.
A)
\[10\] done
clear
B)
\[12\] done
clear
C)
\[22\] done
clear
D)
\[32\] done
clear
View Solution play_arrow
-
question_answer23)
\[_{17}^{35}Cl\]and \[_{17}^{37}Cl\] are examples of\['X'\]. What is\['X'\]?
A)
Isobars done
clear
B)
Isotopes done
clear
C)
Isotones done
clear
D)
Isomers done
clear
View Solution play_arrow
-
question_answer24)
Which of the following statements is false?
A)
Neutron has the highest mass among the fundamental particles. done
clear
B)
The mass of an electron is negligible. done
clear
C)
The value of e/m is the highest for a proton. done
clear
D)
Charge of a neutron is zero. done
clear
View Solution play_arrow
-
question_answer25)
Which of the following is true about the \[e/m\] value of positive particles?
A)
It is constant. done
clear
B)
It depends on the gas taken in the discharge tube. done
clear
C)
It depends on the nature of cathode and anode taken. done
clear
D)
It depends on the fluorescent material coated on the screen. done
clear
View Solution play_arrow
-
question_answer26)
Element \['X'\] contains \[9\] protons, \[10\] electrons and \[11\] neutrons. What is\['X'\]?
A)
A neutral atom done
clear
B)
An isotope done
clear
C)
A cation done
clear
D)
An anion done
clear
View Solution play_arrow
-
question_answer27)
Which of the following are present in an atom with atomic number \[82\] and mass number\[208\]?
A)
\[82\] protons and \[82\] electrons done
clear
B)
\[82\]protons and \[126\] neutrons done
clear
C)
\[82\]protons, \[82\] electrons and \[126\] neutrons done
clear
D)
\[82\]neutrons, \[126\] protons and \[126\] electrons done
clear
View Solution play_arrow
-
question_answer28)
Which of the following ions is the smallest in size?
A)
\[M{{g}^{2+}}\] done
clear
B)
\[N{{a}^{+}}\] done
clear
C)
\[{{O}^{2-}}\] done
clear
D)
\[{{F}^{-}}\] done
clear
View Solution play_arrow
-
question_answer29)
Which of the following gas when taken in the discharge tube produces maximum specific charge of positive rays?
A)
Oxygen done
clear
B)
Nitrogen done
clear
C)
Helium done
clear
D)
Hydrogen done
clear
View Solution play_arrow
-
question_answer30)
In an element, the number of protons, electrons and neutrons are \[1,\,\,0,\,\,1\] respectively. Identify the element.
A)
\[{{D}^{+}}\] done
clear
B)
\[{{H}^{+}}\] done
clear
C)
\[He\] done
clear
D)
\[L{{i}^{+}}\] done
clear
View Solution play_arrow
-
question_answer31)
What is the maximum number of electrons in the main shell of an atom?
A)
\[{{n}^{2}}\] done
clear
B)
\[2{{n}^{2}}\] done
clear
C)
\[2n\] done
clear
D)
\[4n+1\] done
clear
View Solution play_arrow
-
question_answer32)
How many protons, neutrons and electrons are contained in one atom of\[_{19}^{39}K\]?
A)
\[19\] protons \[+\,\,20\] neutrons \[+\,\,19\] electrons done
clear
B)
\[19\] protons \[+\,\,20\] neutrons \[+\,\,20\] electrons done
clear
C)
\[20\] protons \[+\,\,19\] neutrons \[+\,\,20\] electrons done
clear
D)
\[20\] protons \[+\,\,20\] neutrons \[+\,\,19\] electrons done
clear
View Solution play_arrow
-
question_answer33)
What do oxygen atom and oxide ion have in common?
A)
Same size done
clear
B)
Same number of electrons done
clear
C)
Same electronic configuration done
clear
D)
Same number of protons done
clear
View Solution play_arrow
-
question_answer34)
Identify the number of electrons present in the outermost shell of atoms of each of the following elements.

A)
(i) - 2, (ii) - 3, (iii) ? 4 done
clear
B)
(i) - 1, (ii) - 4, (iii) ? 6 done
clear
C)
(i) - 2, (ii) - 2, (iii) ? 6 done
clear
D)
(i) - 3, (ii) - 4, (iii) ? 5 done
clear
View Solution play_arrow
-
question_answer35)
The atomic numbers of three elements \[P,\,\,Q\] and \[R\] are \[5,\,\,9\] and \[13\] respectively. Which two elements show similar chemical properties?
A)
\[P\]and\[Q\] done
clear
B)
\[P\] and \[R\] done
clear
C)
\[Q\]and\[R\] done
clear
D)
\[P,\,\,Q\]and\[R\] done
clear
View Solution play_arrow
-
question_answer36)
Identify the element having two electrons in the K shell of its atom.
A)
Hydrogen done
clear
B)
\[H{{e}^{+}}\] done
clear
C)
Helium done
clear
D)
Deuterium done
clear
View Solution play_arrow
-
question_answer37)
The total number of electrons in a nitrogen atom and a chlorine atom are \[7\] and \[17\] respectively. Find the number of valence electrons in them.
A)
\[3,\,\,1\] done
clear
B)
\[5,\,\,8\] done
clear
C)
\[4,\,\,1\] done
clear
D)
\[5,\,\,7\] done
clear
View Solution play_arrow
-
question_answer38)
An element has an atomic number 20. How many electrons will be present in \[K,\,\,L,\,\,M\] and \[N\] shells of its atom?
A)
\[2,\,\,8,\,\,6,\,\,4\] done
clear
B)
\[2,\,\,8,\,\,7,\,\,3\] done
clear
C)
\[3,\,\,7,\,\,7,\,\,3\] done
clear
D)
\[2,\,\,8,\,\,8,\,\,2\] done
clear
View Solution play_arrow
-
question_answer39)
An atom and its ion differ in the number of
A)
electrons done
clear
B)
protons done
clear
C)
neutrons done
clear
D)
nucleons done
clear
View Solution play_arrow
-
question_answer40)
What is the ratio of the neutrons present in carbon and silicon atoms with respect to their atomic masses? (Atomic mass of carbon and silicon are \[12\] and \[28\] respectively)
A)
\[3:7\] done
clear
B)
\[7:3\] done
clear
C)
\[3:4\] done
clear
D)
\[6:28\] done
clear
View Solution play_arrow
-
question_answer41)
According to JJ. Thomson's atomic model, where are the negative charges of an atom embedded in?
A)
A lump of positive charge done
clear
B)
A lump of small atoms done
clear
C)
A lump of neutrons done
clear
D)
The nucleus done
clear
View Solution play_arrow
-
question_answer42)
Which of the following conclusions cannot be drawn on Rutherford's atomic model?
A)
The total mass of an atom is concentrated at the centre of the atom. done
clear
B)
Nucleus is located inside the atom containing positively charged particles. done
clear
C)
Most of the atom is empty in space. done
clear
D)
Electrons revolve around the nucleus in stationary, circular orbits. done
clear
View Solution play_arrow
-
question_answer43)
Which of the following is a pair of isobars?
A)
\[_{7}^{15}N,\,\,_{8}^{16}O\] done
clear
B)
\[_{6}^{13}C,\,\,_{7}^{14}N\] done
clear
C)
\[_{6}^{13}C,\,\,_{7}^{13}N\] done
clear
D)
\[_{6}^{13}C,\,\,_{8}^{15}O\] done
clear
View Solution play_arrow
-
question_answer44)
An isotope of helium is represented by the symbol\[_{2}^{3}He\]. How many protons, neutrons and electrons are there in an atom of the isotope?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer45)
How are the two atoms\[_{92}^{235}P\] and\[_{92}^{238}P\]similar?

A)
Only (i) done
clear
B)
Only (i) and (ii) done
clear
C)
Only (i) and (iii) done
clear
D)
(i), (ii) and (iii) done
clear
View Solution play_arrow
-
question_answer46)
Identify the least reactive element from the following.
A)
\[_{8}^{16}X\] done
clear
B)
\[_{10}^{20}X\] done
clear
C)
\[_{11}^{23}X\] done
clear
D)
\[_{9}^{19}X\] done
clear
View Solution play_arrow
-
question_answer47)
What is the ratio of the number of neutrons present in potassium and magnesium atoms with mass numbers \[39\] and \[24\] respectively?
A)
\[19:12\] done
clear
B)
\[5:3\] done
clear
C)
\[5:6\] done
clear
D)
\[4:3\] done
clear
View Solution play_arrow
-
question_answer48)
A beam of alpha-particles is fired at a piece of gold foil as shown below.
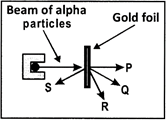
After striking the gold foil, in which direction do most of the alpha-particles travel?
A)
\[P\] done
clear
B)
\[Q\] done
clear
C)
\[R\] done
clear
D)
\[S\] done
clear
View Solution play_arrow
-
question_answer49)
Which of the following statements about the isotopes of an element is NOT correct?
A)
Isotopes have the same proton number. done
clear
B)
Isotopes have the same chemical properties. done
clear
C)
Isotopes have the same nucleon number. done
clear
D)
Atoms of the isotopes of the element have the same number of electrons. done
clear
View Solution play_arrow
-
question_answer50)
An element \[X\] combines with oxygen to form compounds \[P\] and\[Q\]. If the ratio of the valency of element \[X\] in \[P\] to element \[X\] in \[Q\] is \[3:5\] respectively, what could be the probable compounds \[P\] and\[Q\]?
A)
B)
C)
D)
View Solution play_arrow

