-
question_answer1)
Which of the following statements about \[{{H}_{3}}B{{O}_{3}}\] is not correct [CBSE PMT 1994]
A)
It is a strong tribasic acid done
clear
B)
It is prepared by acidifying an aqueous solution of borax done
clear
C)
It has a layer structure in which planar \[B{{O}_{3}}\] units are joined by hydrogen bonds done
clear
D)
It does not act as proton donor but acts as a Lewis acid by accepting hydroxyl ion done
clear
View Solution play_arrow
-
question_answer2)
The type of hybridisation of boron in diborane is [CPMT 1999]
A)
sp-hybridisation done
clear
B)
\[s{{p}^{2}}\text{-}\]hybridisation done
clear
C)
\[s{{p}^{3}}\text{-}\]hybridization done
clear
D)
\[s{{p}^{3}}{{d}^{2}}\text{-}\]hybridization done
clear
View Solution play_arrow
-
question_answer3)
In the reaction \[{{B}_{2}}{{O}_{3}}+C+C{{l}_{2}}\to A+CO.\] The A is [Pb. PMT 2000]
A)
\[BC{{l}_{3}}\] done
clear
B)
\[BC{{l}_{2}}\] done
clear
C)
\[{{B}_{2}}C{{l}_{2}}\] done
clear
D)
\[CC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer4)
The molecular formula of felspar is [MP PMT 2003]
A)
\[{{K}_{2}}O\,.\,A{{l}_{2}}{{O}_{3}}.\,6Si{{O}_{2}}\] done
clear
B)
\[{{K}_{2}}O\,.\,3A{{l}_{2}}{{O}_{3}}.\,6Si{{O}_{2}}\] done
clear
C)
\[N{{a}_{3}}Al{{F}_{6}}\] done
clear
D)
\[CaS{{O}_{4}}.\,2{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer5)
The most acidic of the following compounds is [Bihar CEE 1995]
A)
\[{{P}_{2}}{{O}_{3}}\] done
clear
B)
\[S{{b}_{2}}{{O}_{3}}\] done
clear
C)
\[{{B}_{2}}{{O}_{3}}\] done
clear
D)
\[A{{s}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer6)
Identify the statement that is not correct as far as structure of diborane is concerned [Pb. PMT 1998]
A)
There are two bridging hydrogen atoms in diborane done
clear
B)
Each boron atom forms four bonds in diborane done
clear
C)
The hydrogen atoms are not in the same plane in diborane done
clear
D)
All B - H bonds in diborane are similar done
clear
View Solution play_arrow
-
question_answer7)
Soft heavy metal melts at \[{{30}^{o}}C\] and is used in making heat sensitive thermometers the metal is [RPET 2000]
A)
Galium done
clear
B)
Sodium done
clear
C)
Potassium done
clear
D)
Caesium done
clear
View Solution play_arrow
-
question_answer8)
Which of the following is formed when aluminium oxide and carbon is strongly heated in dry chlorine gas [AFMC 2000]
A)
Aluminium chloride done
clear
B)
Hydrate aluminium chloride done
clear
C)
Anhydrous aluminium chloride done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer9)
Which metal burn in air at high temperature with the evolution of much heat [UPSEAT 1999, 2001]
A)
\[Cu\] done
clear
B)
\[Hg\] done
clear
C)
\[Pb\] done
clear
D)
\[Al\] done
clear
View Solution play_arrow
-
question_answer10)
Aluminium hydroxide is soluble in excess of sodium hydroxide forming the ion [AMU 2001]
A)
\[Be{{(OH)}_{2}}\] done
clear
B)
\[AlO_{2}^{-3}\] done
clear
C)
\[AlO_{2}^{-}\] done
clear
D)
\[AlO_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer11)
Boron form covalent compound due to [Pb. PMT 2000]
A)
Higher ionization energy done
clear
B)
Lower ionization energy done
clear
C)
Small size done
clear
D)
Both a and c done
clear
View Solution play_arrow
-
question_answer12)
In diborane, the two \[H-B-H\] angles are nearly [AIIMS 2005]
A)
\[60{}^\circ \,,\text{ }120{}^\circ \] done
clear
B)
\[95{}^\circ \,,\text{ }120{}^\circ \] done
clear
C)
\[95{}^\circ \,,\text{ }150{}^\circ \] done
clear
D)
\[120{}^\circ \,,\text{ }180{}^\circ \] done
clear
View Solution play_arrow
-
question_answer13)
Which of the following is a non-metal [MP PMT 1999]
A)
Gallium done
clear
B)
Indium done
clear
C)
Boron done
clear
D)
Aluminium done
clear
View Solution play_arrow
-
question_answer14)
Which of the following is most acidic [BHU 1998]
A)
\[N{{a}_{2}}O\] done
clear
B)
\[MgO\] done
clear
C)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
D)
\[CaO\] done
clear
View Solution play_arrow
-
question_answer15)
When orthoboric acid \[({{H}_{3}}B{{O}_{3}})\] is heated, the residue left is [Pb. PMT 2002]
A)
Metaboric acid done
clear
B)
Boron done
clear
C)
Boric anhydride done
clear
D)
Borax done
clear
View Solution play_arrow
-
question_answer16)
Which of the following form dimeric halides [Roorkee Qualifying 1998]
A)
\[Al\] done
clear
B)
\[Mg\] done
clear
C)
\[In\] done
clear
D)
\[Ga\] done
clear
View Solution play_arrow
-
question_answer17)
The liquid field metal expanding on solidification is [AIIMS 2004]
A)
Ga done
clear
B)
Al done
clear
C)
Zn done
clear
D)
Cu done
clear
View Solution play_arrow
-
question_answer18)
Aluminium chloride exists as dimer, \[A{{l}_{2}}C{{l}_{6}}\] in solid state as well as in solution of non-polar solvents such as benzene. When dissolved in water, it gives [AIEEE 2004]
A)
\[{{[Al{{(OH)}_{6}}]}^{3-}}+3HCl\] done
clear
B)
\[{{[Al{{({{H}_{2}}O)}_{6}}]}^{3+}}+3C{{l}^{-}}\] done
clear
C)
\[A{{l}^{3+}}+3C{{l}^{-}}\] done
clear
D)
\[A{{l}_{2}}{{O}_{3}}+6HCl\] done
clear
View Solution play_arrow
-
question_answer19)
The hardest substance amongst the following is [Kerala PMT 2004]
A)
\[B{{e}_{2}}C\] done
clear
B)
Graphite done
clear
C)
Titanium done
clear
D)
SiC done
clear
E)
\[{{B}_{4}}C\] done
clear
View Solution play_arrow
-
question_answer20)
Which of the following is known as inorganic benzene [Pb. CET 2001]
A)
Borazine done
clear
B)
Boron nitride done
clear
C)
p-dichlorobenzene done
clear
D)
Phosphonitrilic acid done
clear
View Solution play_arrow
-
question_answer21)
Which of the following is only acidic in nature [AIIMS 2004]
A)
\[Be{{(OH)}_{2}}\] done
clear
B)
\[Mg{{(OH)}_{2}}\] done
clear
C)
\[B{{(OH)}_{3}}\] done
clear
D)
\[Al{{(OH)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer22)
Moissan boron is [DCE 2003]
A)
Amorphous boron of ultra purity done
clear
B)
Crystalline boron of ultra purity done
clear
C)
Amorphous boron of low purity done
clear
D)
Crystalline boron of low purity done
clear
View Solution play_arrow
-
question_answer23)
Which of the following does not exist in free form [Kerala PMT 2004]
A)
\[B{{F}_{3}}\] done
clear
B)
\[BC{{l}_{3}}\] done
clear
C)
\[BB{{r}_{3}}\] done
clear
D)
\[B{{H}_{3}}\] done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer24)
Alumina is [DCE 2002]
A)
Acidic done
clear
B)
Basic done
clear
C)
Amphoteric done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer25)
The most abundant metal in the earth crust is [Pb. CET 2004]
A)
Al done
clear
B)
Ca done
clear
C)
Fe done
clear
D)
Na done
clear
View Solution play_arrow
-
question_answer26)
Crystalline metal can be transformed into metallic glass by [NCERT 1984]
A)
Alloying done
clear
B)
Pressing into thin plates done
clear
C)
Slow cooling of molten metal done
clear
D)
Very rapid cooling of a spray of the molten metal done
clear
View Solution play_arrow
-
question_answer27)
Which metal is protected by a layer of its own oxide [NCERT 1981; DPMT 1983; BHU 1998]
A)
\[Al\] done
clear
B)
\[Ag\] done
clear
C)
\[Au\] done
clear
D)
\[Fe\] done
clear
View Solution play_arrow
-
question_answer28)
Aluminium is a self-preserving metal, because
A)
It is not tarnished by air done
clear
B)
A thin film of basic carbonate on its surface done
clear
C)
A non-porous layer of oxide is formed on its surface done
clear
D)
It is not affected by salt water done
clear
View Solution play_arrow
-
question_answer29)
Anhydrous \[AlC{{l}_{3}}\] cannot be obtained from which of the following reactions [CPMT 1987]
A)
Heating \[AlC{{l}_{3}}.6{{H}_{2}}O\] done
clear
B)
By passing dry \[HCl\] over hot aluminium powder done
clear
C)
By passing dry \[C{{l}_{2}}\] over hot aluminium powder done
clear
D)
By passing dry \[C{{l}_{2}}\] over a hot mixture of alumina and coke done
clear
View Solution play_arrow
-
question_answer30)
An element A dissolves both in acid and alkali. It is an example of [NCERT 1972]
A)
Allotropic nature of A done
clear
B)
Dimorphic nature of A done
clear
C)
Amorphous nature of A done
clear
D)
Amphoteric nature of A done
clear
View Solution play_arrow
-
question_answer31)
Hydrogen gas will not reduce [IIT 1984]
A)
Heated cupric oxide done
clear
B)
Heated ferric oxide done
clear
C)
Heated stannic oxide done
clear
D)
Heated aluminium oxide done
clear
View Solution play_arrow
-
question_answer32)
Conc. \[HN{{O}_{3}}\]
A)
Reacts with aluminium vigrously done
clear
B)
Reacts with aluminium to form aluminium nitrate done
clear
C)
Does not react with aluminium done
clear
D)
Reacts with platinum done
clear
View Solution play_arrow
-
question_answer33)
Anhydrous \[AlC{{l}_{3}}\] is obtained from [BHU 1980; CPMT 1982]
A)
\[HCl\] and aluminium metal done
clear
B)
Aluminium and chlorine gas done
clear
C)
Hydrogen chloride gas and aluminium metal done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer34)
Which is true for an element \[R\] present in III group of the periodic table [EAMCET 1991]
A)
It is gas at room temperature done
clear
B)
It has oxidation state of \[+\,4\] done
clear
C)
It forms \[{{R}_{2}}{{O}_{3}}\] done
clear
D)
It forms \[R{{X}_{2}}\] done
clear
View Solution play_arrow
-
question_answer35)
When \[Al\] is added to \[KOH\] solution [NCERT 1974, 76; CPMT 1977]
A)
No action takes place done
clear
B)
Oxygen is evolved done
clear
C)
Water is produced done
clear
D)
Hydrogen is evolved done
clear
View Solution play_arrow
-
question_answer36)
Aluminium is more reactive than iron. But aluminium is less easily corroded than iron because [KCET 1993]
A)
Aluminium is a noble metal done
clear
B)
Oxygen forms a protective oxide layer done
clear
C)
Iron undergoes reaction easily with water done
clear
D)
Iron forms mono and divalent ions done
clear
View Solution play_arrow
-
question_answer37)
Aluminium vessels should not be washed with materials containing washing soda since [KCET 1993]
A)
Washing soda is expensive done
clear
B)
Washing soda is easily decomposed done
clear
C)
Washing soda reacts with aluminium to form soluble aluminate done
clear
D)
Washing soda reacts with aluminium to form insoluble aluminium oxide done
clear
View Solution play_arrow
-
question_answer38)
Which of the statements about anhydrous aluminium chloride is correct [IIT 1981]
A)
It exists as \[AlC{{l}_{3}}\] molecule done
clear
B)
It is not easily hydrolysed done
clear
C)
It sublimes at \[{{100}^{o}}C\] under vacuum done
clear
D)
It is a strong Lewis base done
clear
View Solution play_arrow
-
question_answer39)
Common alum is [DPMT 1982; CPMT 1978; AMU 1982, 83]
A)
\[{{K}_{2}}S{{O}_{4}}.A{{l}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\] done
clear
B)
\[{{K}_{2}}S{{O}_{4}}.C{{r}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\] done
clear
C)
\[{{K}_{2}}S{{O}_{4}}.F{{e}_{2}}{{(S{{O}_{4}})}_{3}}.24{{H}_{2}}O\] done
clear
D)
\[{{(N{{H}_{4}})}_{2}}S{{O}_{4}}.FeS{{O}_{4}}.6{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer40)
Which of the following is not true about potash alum [MNR 1993; UPSEAT 2002]
A)
Its empirical formula is \[KAl{{(S{{O}_{4}})}_{2}}.12{{H}_{2}}O\] done
clear
B)
Its aqueous solution is basic done
clear
C)
It is used in dyeing industries done
clear
D)
On heating it melts in its water of crystallization done
clear
View Solution play_arrow
-
question_answer41)
Which one of the following is correct statement
A)
The hydroxide of aluminium is more acidic than that of boron done
clear
B)
The hydroxide of boron is basic, while that of aluminium is amphoteric done
clear
C)
The hydroxide of boron is acidic, while that of aluminium is amphoteric done
clear
D)
The hydroxide of boron and aluminium are amphoteric done
clear
View Solution play_arrow
-
question_answer42)
\[AlC{{l}_{3}}\] is [AFMC 1995]
A)
Anhydrous and covalent done
clear
B)
Anhydrous and ionic done
clear
C)
Covalent and basic done
clear
D)
Coordinate and acidic done
clear
View Solution play_arrow
-
question_answer43)
Aluminium (III) chloride forms a dimer because [CBSE PMT 1995]
A)
Higher coordination number can be achieved by aluminium done
clear
B)
Aluminium has high ionization energy done
clear
C)
Aluminium belongs to III group done
clear
D)
It cannot form a trimer done
clear
View Solution play_arrow
-
question_answer44)
Aluminium has a great affinity for oxygen and its oxidation is an exothermic process. This fact is made use of in [MP PMT 1997]
A)
Preparing thin foils of aluminium done
clear
B)
Making utensils done
clear
C)
Preparing duralumin alloy done
clear
D)
Thermite welding done
clear
View Solution play_arrow
-
question_answer45)
Number of water molecules in Mohr's salt is [CPMT 1997; AIIMS 2001; JIPMER 2001]
A)
7 done
clear
B)
6 done
clear
C)
5 done
clear
D)
8 done
clear
View Solution play_arrow
-
question_answer46)
Which of the following is an amphoteric oxide [BHU 2001]
A)
\[MgO\] done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
C)
\[C{{l}_{2}}{{O}_{7}}\] done
clear
D)
\[T{{i}_{2}}{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer47)
Aluminium oxide is not reduced by chemical reactions since [KCET 2002]
A)
Aluminium oxide is reactive done
clear
B)
Reducing agents contaminate done
clear
C)
Aluminium oxide is highly stable done
clear
D)
The process pollutes the environment done
clear
View Solution play_arrow
-
question_answer48)
Aluminium is not used [DPMT 2002]
A)
In silvery paints done
clear
B)
For making utensils done
clear
C)
As a reducing agent done
clear
D)
As oxidizer in metallurgy done
clear
View Solution play_arrow
-
question_answer49)
In the thermite process the reducing agent is [Pb. PMT 2002]
A)
Al done
clear
B)
C done
clear
C)
Mg done
clear
D)
Na done
clear
View Solution play_arrow
-
question_answer50)
In Goldschmidt aluminothermic process, thermite contains [KCET 2003]
A)
3 parts of \[A{{l}_{2}}{{O}_{3}}\] and 4 parts of Al done
clear
B)
3 parts of \[F{{e}_{2}}{{O}_{3}}\]and 2 parts of Al done
clear
C)
3 parts of \[F{{e}_{2}}{{O}_{3}}\]and 1 part of Al done
clear
D)
1 part of \[F{{e}_{2}}{{O}_{3}}\] and 1 part of Al done
clear
View Solution play_arrow
-
question_answer51)
Bauxite containing impurities of iron oxide is purified by [CPMT 1987; AIIMS 1998]
A)
Hoop's process done
clear
B)
Serpeck's process done
clear
C)
Baeyer's process done
clear
D)
Electrolytic process done
clear
View Solution play_arrow
-
question_answer52)
In the purification of bauxite by Hall's process
A)
Bauxite ore is heated with \[NaOH\] solution at \[{{50}^{o}}C\] done
clear
B)
Bauxite ore is fused with \[N{{a}_{2}}C{{O}_{3}}\] done
clear
C)
Bauxite ore is fused with coke and heated at \[{{1800}^{o}}C\] in a current of nitrogen done
clear
D)
Bauxite ore is heated with \[NaHC{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer53)
Which one is used as a bye-product in Serpeck's process
A)
\[N{{H}_{3}}\] done
clear
B)
\[C{{O}_{2}}\] done
clear
C)
\[{{N}_{2}}\] done
clear
D)
\[P{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer54)
In the metallurgy of aluminium, cryolite is mixed in the molten state because it [Roorkee 1995]
A)
Increases the melting point of alumina done
clear
B)
Oxidises alumina done
clear
C)
Reduces alumina done
clear
D)
Decreases the melting point of alumina done
clear
View Solution play_arrow
-
question_answer55)
In the electrolytic extraction of aluminium, cryolite is used [NCERT 1981; CPMT 1989; RPMT 2000; MP PMT 2000, 02]
A)
To obtain more aluminium done
clear
B)
To decrease temperature to dissolve bauxite done
clear
C)
To protect the anode done
clear
D)
As reducing agent done
clear
View Solution play_arrow
-
question_answer56)
In the extraction of aluminium, bauxite is dissolved in cryolite because
A)
It acts as a solvent done
clear
B)
It reduces melting point of aluminium oxide done
clear
C)
It increases the resistance of aluminium oxide done
clear
D)
Bauxite becomes active done
clear
View Solution play_arrow
-
question_answer57)
In the extraction of aluminium the electrolyte is [CBSE PMT 1989; AIEEE 2002]
A)
Fused cryolite with felspar done
clear
B)
Fused cryolite with fluorspar done
clear
C)
Pure alumina in molten cryolite done
clear
D)
Pure alumina with bauxite and molten cryolite done
clear
View Solution play_arrow
-
question_answer58)
Aluminium is obtained by [KCET 1992; RPMT 2002]
A)
Reducing \[A{{l}_{2}}{{O}_{3}}\] with coke done
clear
B)
Electrolysing \[A{{l}_{2}}{{O}_{3}}\] dissolved in \[N{{a}_{3}}Al{{F}_{6}}\] done
clear
C)
Reducing \[A{{l}_{2}}{{O}_{3}}\] with chromium done
clear
D)
Heating alumina and cryolite done
clear
View Solution play_arrow
-
question_answer59)
In the electrolysis of alumina, cryolite is added to [IIT 1986; BHU 1987]
A)
Increase the melting point of alumina done
clear
B)
Increase the electrical conductivity done
clear
C)
Minimise the anodic effect done
clear
D)
Remove impurities from alumina done
clear
View Solution play_arrow
-
question_answer60)
The function of fluorspar in the electrolytic reduction of alumina dissolved in fused cryolite \[(N{{a}_{3}}Al{{F}_{6}})\] is [KCET 1993; IIT 1993]
A)
As a catalyst done
clear
B)
To lower the temperature of the melt and to make the fused mixture very conducting done
clear
C)
To decrease the rate of oxidation of carbon at the anode done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer61)
For purification of alumina, the modern processes most useful when (i) the impurity present is a lot of iron oxides and (ii) the impurity present is a lot of silica, are
A)
For (i) Hall's process; for (ii) Baeyer's process done
clear
B)
For (i) Hall's process; for (ii) Serpeck's process done
clear
C)
For (i) Serpeck's process; for (ii) Baeyer's process done
clear
D)
For (i) Baeyer's process; for (ii) Serpeck's process done
clear
View Solution play_arrow
-
question_answer62)
For the electrolytic production of aluminium, (i) the cathode and (ii) the anode are made of
A)
(i) Platinum and (ii) Iron done
clear
B)
(i) Copper and (ii) Iron done
clear
C)
(i) Copper and (ii) Carbon done
clear
D)
(i) Carbon and (ii) Carbon done
clear
View Solution play_arrow
-
question_answer63)
In the commercial electrochemical process for aluminium extraction, the electrolyte used is [IIT-JEE 1999]
A)
\[Al{{(OH)}_{3}}\] in \[NaOH\] solution done
clear
B)
An aqueous solution of \[A{{l}_{2}}{{(S{{O}_{4}})}_{3}}\] done
clear
C)
A molten mixture of \[A{{l}_{2}}{{O}_{3}}\] and \[N{{a}_{3}}Al{{F}_{6}}\] done
clear
D)
A molten mixture of \[AlO(OH)\] and \[Al{{(OH)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer64)
In electrolysis of aluminium oxide which of the following is added to accelerate the process [AFMC 1999; CPMT 1999]
A)
Silica done
clear
B)
Cryolite done
clear
C)
Nickel done
clear
D)
Silicate done
clear
View Solution play_arrow
-
question_answer65)
The purification of alumina is called [CPMT 1997; AFMC 1998; AIIMS 1999]
A)
Bosch process done
clear
B)
Caster process done
clear
C)
Baeyer's process done
clear
D)
Hoop's process done
clear
View Solution play_arrow
-
question_answer66)
Electrolytic reduction of alumina to aluminium by Hall-Heroult process is carried out in the presence of [IIT-JEE (Screening) 2000]
A)
\[NaCl\] done
clear
B)
Fluorite done
clear
C)
Cryolite which forms a melt with lower melting temperature done
clear
D)
Cryolite which forms a melt with higher melting temperature done
clear
View Solution play_arrow
-
question_answer67)
In the electrolytic method of obtaining aluminium from purified bauxite, cryolite is added to the charge in order to [KCET 2004]
A)
Minimize the heat loss due to radiation done
clear
B)
Protect aluminium produced from oxygen done
clear
C)
Dissolve bauxite and render it conductor of electricity done
clear
D)
Lower the melting point of bauxite done
clear
View Solution play_arrow
-
question_answer68)
Hoop's process is used for the purification of the metal [MP PET 1995; MP PMT 2001]
A)
\[Al\] done
clear
B)
\[Zn\] done
clear
C)
\[Ag\] done
clear
D)
\[Cu\] done
clear
View Solution play_arrow
-
question_answer69)
Purification of aluminium done by electrolytic refining is known as [CPMT 1989; CBSE PMT 1999; RPET 2003; BCECE 2005]
A)
Serpeck's process done
clear
B)
Hall's process done
clear
C)
Baeyer's process done
clear
D)
Hoop's process done
clear
View Solution play_arrow
-
question_answer70)
In the Hoope's process for refining of aluminium, the fused materials form three different layers and they remain separated during electrolysis also. This is because [MP PET 1996]
A)
The upper layer is kept attracted by the cathode and the lower layer is kept attracted by the anode done
clear
B)
There is special arrangement in the cell to keep the layers separate done
clear
C)
The 3 layers have different densities done
clear
D)
The 3 layers are maintained at different temperatures done
clear
View Solution play_arrow
-
question_answer71)
During metallurgy of aluminium bauxite is dissolved in cryolite because
A)
Bauxite is non-electrolyte done
clear
B)
Cryolite is a flux done
clear
C)
Cryolite acts as an electrolyte done
clear
D)
All are correct done
clear
View Solution play_arrow
-
question_answer72)
For the electrolytic refining of aluminium, the three fused layers consist of
A)
B)
C)
D)
View Solution play_arrow
-
question_answer73)
Heating an aqueous solution of aluminium chloride to dryness will give [AIEEE 2005]
A)
\[AlC{{l}_{3}}\] done
clear
B)
\[A{{l}_{2}}C{{l}_{6}}\] done
clear
C)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
D)
\[Al(OH)C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer74)
The structure of diborane \[({{B}_{2}}{{H}_{6}})\] contains [AIEEE 2005]
A)
Four 2c-2e bonds and two 3c-2e bonds done
clear
B)
Two 2c-2e bonds and four 3c-2e bonds done
clear
C)
Two 2c-2e bonds and two 3c-3e bonds done
clear
D)
Four 2c-2e bonds and four 3c-2e bonds done
clear
View Solution play_arrow
-
question_answer75)
Which of the following is the electron deficient molecule [CBSE PMT 2005]
A)
\[{{B}_{2}}{{H}_{6}}\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[P{{H}_{3}}\] done
clear
D)
\[Si{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer76)
In Hall's process, the main reagent is mixed with [AFMC 2005]
A)
NaF done
clear
B)
\[N{{a}_{3}}Al{{F}_{6}}\] done
clear
C)
\[Al{{F}_{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer77)
Acedic strength of Boron trihalide are in order of [Kerala CET 2005]
A)
\[B{{F}_{3}}<BC{{l}_{3}}<BB{{r}_{3}}<B{{I}_{3}}\] done
clear
B)
\[B{{I}_{3}}<BB{{r}_{3}}<BC{{l}_{3}}<B{{F}_{3}}\] done
clear
C)
\[BB{{r}_{3}}<BC{{l}_{3}}<B{{F}_{3}}<B{{I}_{3}}\] done
clear
D)
\[B{{F}_{3}}<B{{I}_{3}}<BC{{l}_{3}}<BB{{r}_{3}}\] done
clear
View Solution play_arrow
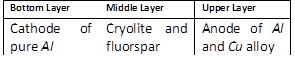 done
clear
done
clear
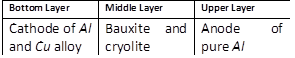 done
clear
done
clear
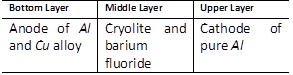 done
clear
done
clear
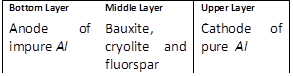 done
clear
done
clear
