-
question_answer1)
The velocity constant of a reaction at 280 K is \[1.6\times {{10}^{-4}}\]. The velocity constant of the same reaction at 300 K is
A)
\[1.6\times {{10}^{-4}}\] done
clear
B)
\[0.8\times {{10}^{-4}}\] done
clear
C)
\[6.4\times {{10}^{-4}}\] done
clear
D)
\[3.2\times {{10}^{-4}}\] done
clear
View Solution play_arrow
-
question_answer2)
| Decomposition of A follows first order kinetics by the following equation. |
| \[4A(g)\xrightarrow{{}}B(g)+2C(g)\] |
| If initially, total pressure was 800 mm of Hg and after 10 minutes it is found to be 650 mm of Hg. What is half-life of A? (Assume only A is present initially). |
A)
10 mins done
clear
B)
5 mins done
clear
C)
7.5 mins done
clear
D)
15 mins done
clear
View Solution play_arrow
-
question_answer3)
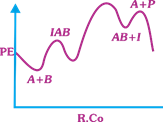
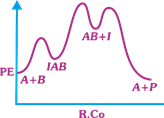
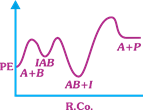
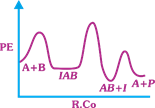
| The following mechanism has been proposed for the exothermic catalyzed complex reaction |
| \[A+BIAB\xrightarrow[{}]{{{k}_{1}}}AB+I\xrightarrow[{}]{{{k}_{2}}}Q+A\] |
| If \[{{k}_{1}}\] is much smaller than \[{{k}_{2}},\] the most suitable qualitative plot of Potential Energy (PE) versus reaction coordinate (R.Co) for the above reaction. |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer4)
| For the reaction \[A+2B\to \] products(started with concentrations taken in stoichiometric proportion), the experimentally determined rate law is \[-\frac{d[B]}{dt}=k\sqrt{[A]}\sqrt{[B]}\] |
| The half time of the reaction would be |
A)
\[\frac{\sqrt{2}\times 0.693}{k}\] done
clear
B)
\[\frac{0.693}{1/k}\] done
clear
C)
\[\frac{0.693}{\sqrt{2}k}\] done
clear
D)
not defined done
clear
View Solution play_arrow
-
question_answer5)
| Two consecutive irreversible first order reaction can be represented by \[A\xrightarrow[{}]{{{k}_{1}}}B\xrightarrow[{}]{{{k}_{2}}}C\]. |
| The rate equation for A is integrated to obtain |
| \[{{[A]}_{t}}={{[A]}_{0}}{{e}^{-{{k}_{1}}t}}\] and \[{{[B]}_{t}}=\frac{{{k}_{1}}[{{A}_{0}}]}{{{k}_{2}}-{{k}_{1}}}[{{e}^{-{{k}_{1}}t}}-{{e}^{-{{k}_{2}}t}}]\]. |
| At what time will B be present in the greatest concentration? |
A)
\[{{t}_{\max }}=\frac{1}{{{k}_{1}}+{{k}_{2}}}\ln \frac{{{k}_{2}}}{{{k}_{1}}}\] done
clear
B)
\[{{t}_{\max }}=\frac{1}{{{k}_{1}}+{{k}_{2}}}\ln \frac{{{k}_{2}}}{{{k}_{1}}}\] done
clear
C)
\[{{t}_{\max }}=\frac{1}{{{k}_{2}}+{{k}_{1}}}\ln \frac{{{k}_{1}}}{{{k}_{2}}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer6)
| Figure shows a graph in \[{{\log }_{10}}\,k\,vs\,\frac{1}{T}\] where, k is rate constant and T is temperature. The straight line BC has slope, \[\tan \,\theta \,=-\frac{1}{2.303}\] and an intercept of 5 on y-axis. Thus, \[{{E}_{a}},\] the energy of activation, is |
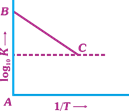 |
A)
4.606 cal done
clear
B)
\[\frac{0.2}{2.303}cal\] done
clear
C)
2 cal done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer7)
| In a gaseous phase reaction, |
| \[{{A}_{2}}(g)\,\xrightarrow{\,}\,B(g)\,+\frac{1}{2}\,(g),\] |
| the increase in pressure from 100 mm to 120 mm is noticed in 5 min. The rate of disappearance of \[{{A}_{2}}\] in mm \[{{\min }^{-1}}\] is |
A)
4 done
clear
B)
8 done
clear
C)
16 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer8)
| A reactant [a] forms two products |
| \[A\xrightarrow{{{k}_{1}}}B\] Activation energy \[{{E}_{{{a}_{1}}}}\] |
| \[A\xrightarrow{{{k}_{2}}}C\] Activation energy \[E{{\,}_{{{a}_{2}}}}\] |
| If \[{{E}_{{{a}_{2}}}}=2{{E}_{{{a}_{1}}}},\] then \[{{k}_{1}}\] and \[{{k}_{2}}\] will be related as |
A)
\[{{k}_{2}}={{k}_{1}}{{e}^{-{{E}_{{{a}_{1}}}}/RT}}\] done
clear
B)
\[{{k}_{2}}={{k}_{1}}{{e}^{-{{E}_{{{a}_{2}}}}/RT}}\] done
clear
C)
\[{{k}_{1}}={{k}_{2}}{{e}^{-{{E}_{{{a}_{1}}}}/RT}}\] done
clear
D)
\[{{k}_{1}}=2{{k}_{2}}{{e}^{{{E}_{{{a}_{2}}}}/RT}}\] done
clear
View Solution play_arrow
-
question_answer9)
Two substances A and B are present such that\[[{{A}_{0}}]=4[{{B}_{0}}]\] and half-life of A is 5 minute and that of B is 15 min. If they start decaying at the same time following 1st order kinetics, how much time later will the concentration of both of them would be same?
A)
15 min done
clear
B)
10 min done
clear
C)
5 min done
clear
D)
12 min done
clear
View Solution play_arrow
-
question_answer10)
| In the reversible reaction, |
| \[2N{{O}_{2}}{{N}_{2}}{{O}_{4}}\] |
| the rate of disappearance of \[N{{O}_{2}}\] is equal to |
A)
\[\frac{2{{k}_{1}}}{{{k}_{2}}}\,{{[N{{O}_{2}}]}^{2}}\] done
clear
B)
\[2{{k}_{1}}\,{{[N{{O}_{2}}]}^{2}}-2{{k}_{2}}\,[{{N}_{2}}{{O}_{4}}]\] done
clear
C)
\[2{{k}_{1}}{{[N{{O}_{2}}]}^{2}}-{{k}_{2}}[{{N}_{2}}{{O}_{4}}]\] done
clear
D)
\[(2{{k}_{1}}-{{k}_{2}})\,[N{{O}_{2}}]\] done
clear
View Solution play_arrow
-
question_answer11)
For the\[2A(g)\xrightarrow{\,}\,3B(g),\,{{t}_{1/2}}=12\,\min \]. Initial pressure exerted by A is 640 mm of Hg. The pressure of the reaction mixture after the time period of 36 min will be
A)
560 mm Hg done
clear
B)
680 mm Hg done
clear
C)
920 mm Hg done
clear
D)
600 mm Hg done
clear
View Solution play_arrow
-
question_answer12)
| A decomposes as |
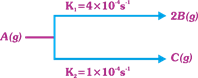 |
| The rate of disappearance of B, taking 4M concentration of A is equal to |
A)
\[1.6\times {{10}^{-3}}M{{s}^{-1}}\] done
clear
B)
\[6.4\times {{10}^{-3}}M{{s}^{-1}}\] done
clear
C)
\[3.2\times {{10}^{-3}}M{{s}^{-1}}\] done
clear
D)
insufficient to calculate done
clear
View Solution play_arrow
-
question_answer13)
| \[(aq)\xrightarrow{{}}B(aq)+C(aq)\] is a first order reaction. |
| Time | t | \[\infty \] | | Moles of reagent | \[{{x}_{1}}\] | \[{{x}_{2}}\] | |
| Reaction progress is measured with the help of titration of reagent P, if all A, B and C reacted with reagent have n factors \[\left[ \text{n factor}:n=\frac{mol.wt.}{eq.wt.} \right]\] in the ratio 1 : 2 : 3 with the reagent. The k in terms of t, \[{{x}_{1}}\] and \[{{x}_{2}}\] is |
A)
\[k=\frac{1}{t}\ln \left( \frac{{{x}_{2}}}{{{x}_{2}}-{{x}_{1}}} \right)\] done
clear
B)
\[k=\frac{1}{t}\ln \left( \frac{2{{x}_{2}}}{{{x}_{2}}-{{x}_{1}}} \right)\] done
clear
C)
\[k=\frac{1}{t}\ln \left( \frac{4{{x}_{2}}}{5({{x}_{2}}-{{x}_{1}})} \right)\] done
clear
D)
\[k=\frac{1}{t}\ln \left( \frac{8{{x}_{2}}}{{{x}_{2}}-{{x}_{1}}} \right)\] done
clear
View Solution play_arrow
-
question_answer14)
The reaction cis\[-X\text{trans}-X\] is first order in both directions. At \[\text{25}{}^\circ \text{C,}\] the equilibrium constant is 0.10 and the rate constant \[{{k}_{f}}=3\times {{10}^{-4}}{{s}^{-1}}\]. In an experiment starting with the pure cis-form, how long would it take for half of the equilibrium amount of the trans-isomer to be formed?
A)
150 s done
clear
B)
200 s done
clear
C)
240 s done
clear
D)
210 s done
clear
View Solution play_arrow
-
question_answer15)
| \[A\to B;\,\,\,\,\,\,\,\,\,\,\,\,\,{{k}_{A}}={{10}^{15}}{{e}^{-2000/T}}\] |
| \[C\to D;\,\,\,\,\,\,\,\,\,\,\,\,\,{{k}_{C}}={{10}^{14}}{{e}^{-1000/T}}\] |
| Temperature T kelvin at which \[(\,{{k}_{A}}={{k}_{C}})\] is: |
A)
1000 K done
clear
B)
2000 K done
clear
C)
(2000/2.303) K done
clear
D)
(1000/2.303) K done
clear
View Solution play_arrow
-
question_answer16)
The acid catalyzed hydrolysis of an organic, compound 'A' at \[30{}^\circ C\] has a time for half change of 100 minute when carried out in a buffer solution at \[(pH=5)\] and 10 minute when carried out at \[(pH=4)\]. Both times of half change are independent of the initial concentration of A. If rate constant K is defined by \[\frac{-d[A]}{dt}=K{{[A]}^{a}}{{[{{H}^{+}}]}^{b}},\] the values of a and b respectively are
A)
1, 1 done
clear
B)
1, 2 done
clear
C)
0, 1 done
clear
D)
1, 0 done
clear
View Solution play_arrow
-
question_answer17)
Which of the following statements regarding the molecularity of a reaction are correct?
A)
It is the number of molecules of the reactants taking part in a single step chemical reaction done
clear
B)
It is calculated from reaction mechanism done
clear
C)
It depends on the rate determining step in the reaction done
clear
D)
It always whole number. done
clear
View Solution play_arrow
-
question_answer18)
A reaction that is of the first order with respect to reactant A has a rate constant \[6{{\min }^{-1}}\]. If we start with [A]\[0.5\,mol\,{{l}^{-1}},\] when would [A] reach the value \[0.05\,mol\,{{l}^{-1}}\]
A)
0.384 min done
clear
B)
0.15 min done
clear
C)
3 min done
clear
D)
3.84 min done
clear
View Solution play_arrow
-
question_answer19)
For the reaction \[X+Y\to Z,\] following kinetic data was obtained :
| Initial [X]M | Initial [Y]M | Initial rate M \[\mathbf{mi}{{\mathbf{n}}^{\mathbf{-1}}}\] |
| 0.4 | 2.0 | \[6.2\times {{10}^{-3}}\] |
| 0.8 | 2.0 | \[2.48\times {{10}^{-2}}\] |
| 0.4 | 4.0 | \[1.24\times {{10}^{-2}}\] |
| 0.5 | 1.5 | ? |
Calculate initial rate in the last experiment
A)
\[\text{4}.\text{36}\times \text{l}{{0}^{-\text{3}}}\] done
clear
B)
\[\text{5}.\text{81}\times \text{l}{{0}^{-\text{3}}}\] done
clear
C)
\[\text{7}.\text{27}\times \text{l}{{0}^{-\text{3}}}\] done
clear
D)
\[\text{9}.\text{69}\times \text{l}{{0}^{-\text{3}}}\] done
clear
View Solution play_arrow
-
question_answer20)
\[6{{I}^{-}}(aq)+Br{{O}_{3}}^{-}(aq)+6{{H}^{+}}(aq)\xrightarrow{{}}3{{I}_{2}}(aq)+\] \[B{{r}^{-}}(aq)+3{{H}_{2}}O(\ell )\] these data were obtained when this reaction was studies.
| \[[{{I}^{-}}],M\] |
\[[Br{{O}_{3}}^{-}],M\] |
\[[{{H}^{+}}],M\] |
Reaction rate \[(mol\,{{L}^{-1}}{{s}^{-1}})\] |
| 0.0010 |
0.0020 |
0.010 |
\[8.0\times {{10}^{-5}}\] |
| 0.0020 |
0.0020 |
0.010 |
\[1.6\times {{10}^{-4}}\] |
| 0.0020 |
0.0040 |
0.010 |
\[1.6\times {{10}^{-4}}\] |
| 0.0010 |
0.0040 |
0.020 |
\[1.6\times {{10}^{-4}}\] |
What is the units of the rate constant for this reaction?
A)
\[{{s}^{-1}}\] done
clear
B)
\[mol\,\,{{L}^{-1}}{{s}^{-1}}\] done
clear
C)
\[L\,\,mo{{l}^{-1}}{{s}^{-1}}\] done
clear
D)
\[{{L}^{2}}\,\,mo{{l}^{-2}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer21)
For a first-order homogeneous gaseous reaction \[A\to 2B+C,\] if the total pressure after time t was \[{{P}_{t}}\] and after long time \[(t\to \infty )\] was \[{{P}_{\infty }}\] then k in terms of \[{{P}_{t}},\,{{P}_{\infty }}\] and t is:
A)
\[k=\frac{2.303}{t}\log \left( \frac{{{P}_{\infty }}}{{{P}_{\infty }}-{{P}_{t}}} \right)\] done
clear
B)
\[k=\frac{2.303}{t}\log \left( \frac{2{{P}_{\infty }}}{{{P}_{\infty }}-{{P}_{t}}} \right)\] done
clear
C)
\[k=\frac{2.303}{t}\log \left( \frac{2{{P}_{\infty }}}{3\left( {{P}_{\infty }}-{{P}_{t}} \right)} \right)\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer22)
The main function of a catalyst in speeding up a reaction is
A)
To increase the rate of the forward reaction done
clear
B)
To change the reaction path so as to decrease the energy of activation for the reaction done
clear
C)
To reduce the temperature at which the reaction can occur done
clear
D)
To increase the energy of the molecules of the reactants done
clear
View Solution play_arrow
-
question_answer23)
In a certain reaction, 10% of the reactant decomposes in 1 h, 20% in 2 h, 30% in 3 h, and so on. The dimensions of the rate constant is:
A)
\[{{h}^{-1}}\] done
clear
B)
\[mol/\left( L\,s \right)\] done
clear
C)
L / (mol s) done
clear
D)
mol/s done
clear
View Solution play_arrow
-
question_answer24)
| A hypothetical reaction: |
| \[{{A}_{2}}+{{B}_{2}}\xrightarrow{{}}2AB\] follows mechanism as given below: |
| \[{{A}_{2}}A+A\]............ (fast) |
| (\[{{k}_{c}}\] is equilibrium constant) |
| \[{{A}_{2}}+{{B}_{2}}\xrightarrow{{{k}_{1}}}AB+B\] ..............(slow) |
| (\[{{k}_{1}}\] rate constant) |
| \[A+BAB\] .................. (fast) |
| (\[{{k}_{2}},{{k}_{3}}\] are rate constant) |
| Give the rate law. |
A)
\[r={{k}_{1}}\sqrt{{{k}_{c}}}\,{{[{{A}_{2}}]}^{1/2}}[{{B}_{2}}]\] done
clear
B)
\[r=\frac{{{k}_{1}}}{{{k}_{c}}}\,{{[{{A}_{2}}]}^{1/2}}[{{B}_{2}}]\] done
clear
C)
\[r=\sqrt{{{k}_{1}}{{k}_{c}}}\,\,{{[{{A}_{2}}]}^{1/2}}[{{B}_{2}}]\] done
clear
D)
\[r=\frac{{{k}_{1}}}{\sqrt{{{k}_{c}}}}\,{{[{{A}_{2}}]}^{1/2}}[{{B}_{2}}]\] done
clear
View Solution play_arrow
-
question_answer25)
In presence of catalyst, the activation energy is lowered by 3 kcal at \[27{}^\circ C\]. Hence, the rate of reaction will increase by
A)
32 times done
clear
B)
243 times done
clear
C)
2 times done
clear
D)
148 times done
clear
View Solution play_arrow
-
question_answer26)
| A gaseous compound A reacts by three independent first-order processes (as shown in the figure) with rate constants \[2\times {{10}^{-3}},\,3\times {{10}^{-3}}\] and \[1.93\times {{10}^{-3}}{{s}^{-1}}\] products B, C and D respectively. If initially pure A was taken in a closed container with P = 8 atm, then the partial pressure of B (in atm) after 100 s from start of experiment will be: |
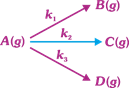 |
A)
0.288 done
clear
B)
0.577 done
clear
C)
1.154 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer27)
In a first-order reaction, the reacting substance has half-life period of 10 min. What fraction of the substance will be left after an hour the reaction has occurred?
A)
1/6 of initial concentration done
clear
B)
1/64 of initial concentration done
clear
C)
1/12 of initial concentration done
clear
D)
1/32 of initial concentration done
clear
View Solution play_arrow
-
question_answer28)
| A graph of volume of hydrogen released vs time for the reaction between zinc and dil. HCl is given in figure. On the basis of this mark the correct option. |
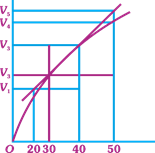 |
A)
Average rate upto 40s is \[\frac{{{V}_{3}}-{{V}_{2}}}{40}\] done
clear
B)
Average rate upto 40 seconds is \[\frac{{{V}_{3}}-{{V}_{2}}}{40-30}\] done
clear
C)
Average rate upto 40 seconds is \[\frac{{{V}_{3}}}{40}\] done
clear
D)
Average rate upto 40 seconds is \[\frac{{{V}_{3}}-{{V}_{1}}}{40-20}\] done
clear
View Solution play_arrow
-
question_answer29)
If a first-order reaction is completed to the extent of 60% and 20% in time intervals, \[{{t}_{1}}\] and \[{{t}_{2}},\] what is the ratio \[{{t}_{1}}:{{t}_{2}}\]
A)
6.32 done
clear
B)
5.58 done
clear
C)
4.11 done
clear
D)
8.33 done
clear
View Solution play_arrow
-
question_answer30)
In any unimolecular reaction
A)
only one reacting species is involved in the rate determining step done
clear
B)
the order and the molecularity of slowest step are equal to one done
clear
C)
the molecularity of the reaction is one and order is zero done
clear
D)
both molecularity and order of the reaction are one done
clear
View Solution play_arrow
-
question_answer31)
| Consider the following case of completing 1st order reactions. |
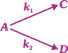 |
| After the start of the reaction at t = 0 with only A, the [C] is equal to the [D] at all times. The time in which all three concentrations will be equal is given by - |
A)
\[t=\frac{1}{2{{k}_{1}}}\ell n3\] done
clear
B)
\[t=\frac{1}{2{{k}_{2}}}\ell n\,3\] done
clear
C)
\[t=\frac{1}{3{{k}_{1}}}\ell n2\] done
clear
D)
\[t=\frac{1}{3{{k}_{1}}}\ell n2\] done
clear
View Solution play_arrow
-
question_answer32)
The forward rate constant for the reversible gaseous reaction \[{{C}_{2}}{{H}_{6}}2C{{H}_{3}}\] is\[3.14\times {{10}^{+2}}{{s}^{-1}}\] at 200 K. What is the rate constant for the backward reaction at this temperature, if \[{{10}^{-5}}\] moles of \[C{{H}_{3}}\] and 100 mol of \[{{C}_{2}}{{H}_{6}}\] are present in 10 L vessel at equilibrium?
A)
\[1.57\times {{10}^{14}}Lmo{{l}^{-1}}{{s}^{-1}}\] done
clear
B)
\[3.14\times {{10}^{15}}Lmo{{l}^{-1}}{{s}^{-1}}\] done
clear
C)
\[1.57\times {{10}^{7}}Lmo{{l}^{-1}}{{s}^{-1}}\] done
clear
D)
\[3.14\times {{10}^{10}}Lmo{{l}^{-1}}{{s}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer33)
In a reaction involving one single reactant, the fraction of the reactant consumed may be defined as \[f=\left[ 1-\left( C/{{C}_{0}} \right) \right],\] where \[{{C}_{0}}\] and C are the concentrations of the reactant at the start and after time t. For a first-order reaction:
A)
\[\frac{df}{dt}=k\left( 1-f \right)\] done
clear
B)
\[-\frac{df}{dt}=kf\] done
clear
C)
\[-\frac{df}{dt}=k\left( 1-f \right)\] done
clear
D)
\[\frac{df}{dt}=kf\] done
clear
View Solution play_arrow
-
question_answer34)
The inversion of cane sugar proceeds with half-life of 500 min at pH = 5 for any concentration of sugar. However, if pH = 6, the half-life changes to 50 min. The rate law expression for sugar inversion can be written as:
A)
\[r=K{{\left[ sugar \right]}^{2}}{{\left[ {{H}^{+}} \right]}^{0}}\] done
clear
B)
\[r=K{{\left[ sugar \right]}^{1}}{{\left[ {{H}^{+}} \right]}^{0}}\] done
clear
C)
\[r=K{{\left[ sugar \right]}^{1}}{{\left[ {{H}^{+}} \right]}^{1}}\] done
clear
D)
\[r=K{{\left[ sugar \right]}^{0}}{{\left[ {{H}^{+}} \right]}^{0}}\] done
clear
View Solution play_arrow
-
question_answer35)
| Given is the information: |
| Reaction 1 : \[k=A{{e}^{-{{E}_{1}}/RT}}\] |
| Reaction 2 : \[k'=A{{e}^{-{{E}_{2}}/RT}}\] |
| If \[{{E}_{1}}=2{{E}_{2}},\] then the ratio \[\ell n\left[ \frac{{{k}_{T}}+10k}{{{k}_{T}}} \right]\] for the reaction will be |
A)
Equal to that of reaction 2 done
clear
B)
greater than that of reaction 2 done
clear
C)
lesser than that of reaction 2 done
clear
D)
may be greater or lesser than reaction 2 depending upon the temperature T. done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
