-
question_answer1)
Arrange the following compounds in order of increasing dipole moment:
| (I) Toluene |
| (II) m-dichlorobenzene |
| (III) o-dichlorobenzene |
| (IV) p-dichlorobenzene |
A)
\[I<IV<II<III\] done
clear
B)
\[IV<I<II<III\] done
clear
C)
\[IV<I<III<II\] done
clear
D)
\[IV<II<I<III\] done
clear
View Solution play_arrow
-
question_answer2)
Pure chloroform is prepared by
A)
distilling chloral hydrate with aqueous sodium hydroxide. done
clear
B)
heating ethanol with bleaching powder. done
clear
C)
heating acetone with bleaching powder. done
clear
D)
reducing carbon tetrachloride. done
clear
View Solution play_arrow
-
question_answer3)
The reaction conditions leading to the best yields of \[{{C}_{2}}{{H}_{5}}Cl\] are:
A)
\[{{C}_{2}}{{H}_{6}}(excess)+C{{l}_{2}}\xrightarrow{UV\,light}\] done
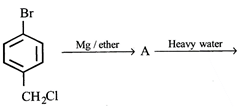
clear
B)
\[{{C}_{2}}{{H}_{6}}+C{{l}_{2}}\xrightarrow[room\text{ }temperature]{dark}\] done
clear
C)
\[{{C}_{2}}{{H}_{6}}+C{{l}_{2}}(excess)\xrightarrow{UV\,light}\] done
clear
D)
\[{{C}_{2}}{{H}_{6}}+C{{l}_{2}}\xrightarrow{UV\,light}\] done
clear
View Solution play_arrow
-
question_answer4)
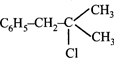
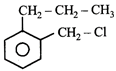
A compound A with molecular formula \[{{C}_{10}}{{H}_{13}}Cl\] gives a white precipitate on adding silver nitrate solution. A on reacting with alcoholic KOH gives compound B as the main product. B on ozonolysis gives C and D. C gives Cannizaro reaction but not aldol condensation. D gives aldol condensation but not Cannizaro reaction. A is:
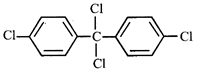
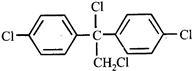
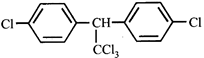
A)
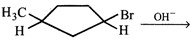
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-Cl\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-C{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\] done
clear
C)
D)
View Solution play_arrow
-
question_answer5)
The Wurtz-Fittig reaction involves condensation of
A)
two molecules of aryl halides. done
clear
B)
one molecule of each of aryl-halide and alkyl-halide. done
clear
C)
one molecule of each of aryl-halide and phenol. done
clear
D)
two molecules of aralkyl-halides. done
clear
View Solution play_arrow
-
question_answer6)
For the compounds \[C{{H}_{3}}Cl,C{{H}_{3}}Br,C{{H}_{3}}I\] and \[C{{H}_{3}}F,\] the correct order of increasing C-X bond length is:
A)
\[C{{H}_{3}}F<C{{H}_{3}}Cl<C{{H}_{3}}Br<C{{H}_{3}}I\] done
clear
B)
\[C{{H}_{3}}F<C{{H}_{3}}Br<C{{H}_{3}}Cl<C{{H}_{3}}I\] done
clear
C)
\[C{{H}_{3}}F<C{{H}_{3}}I<C{{H}_{3}}Br<C{{H}_{3}}Cl\] done
clear
D)
\[C{{H}_{3}}Cl<C{{H}_{3}}Br<C{{H}_{3}}F<C{{H}_{3}}I\] done
clear
View Solution play_arrow
-
question_answer7)
The order of reactivity of the given haloalkanes towards nucleophile is:
A)
\[RI>RBr>RCl\] done
clear
B)
\[RCl>RBr>RI\] done
clear
C)
\[RBr>RCl>RI\] done
clear
D)
\[RBr>RI>RCl\] done
clear
View Solution play_arrow
-
question_answer8)
\[AgN{{O}_{3}}\] does not give precipitate with chloroform because:
A)
\[CHC{{l}_{3}}\] does not ionise in water. done
clear
B)
\[CHC{{l}_{3}}\] is insoluble in water. done
clear
C)
\[AgN{{O}_{3}}\] is insoluble in \[CHC{{l}_{3}}\]. done
clear
D)
\[CHC{{l}_{3}}\] is an organic compound. done
clear
View Solution play_arrow
-
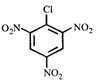
question_answer9)
A)
\[I>II>III>IV\] done
clear
B)
\[I>II>IV>III\] done
clear
C)
\[IV>I>II>III\] done
clear
D)
\[IV>III>II>I\] done
clear
View Solution play_arrow
-
question_answer10)
\[{{C}_{2}}{{H}_{5}}Br\xrightarrow{AgCN}X\xrightarrow[Zn-Hg/HCl]{Reduction}Y,Here\] Y is
A)
Ethyl methyl amine done
clear
B)
n-propylamine done
clear
C)
Isopropylamine done
clear
D)
Ethylamine done
clear
View Solution play_arrow
-
question_answer11)
The chief reaction product of reaction between n-butane and bromine at 573K is:
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Br\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,HBr\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}\underset{C{{H}_{2}}Br}{\mathop{\underset{|}{\mathop{C}}\,}}\,HBr\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,Br\] done
clear
View Solution play_arrow
-
question_answer12)
Aryl fluoride may be prepared from arene diazonium chloride using:
A)
\[HB{{F}_{4}}/\Delta \] done
clear
B)
\[HB{{F}_{4}}/NaN{{O}_{2}},Cu,\Delta \] done
clear
C)
\[CuF/HF\] done
clear
D)
\[Cu/HF\] done
clear
View Solution play_arrow
-
question_answer13)
When \[C{{H}_{3}}C{{H}_{2}}CHC{{l}_{2}}\] is treated with \[NaN{{H}_{2}}\] product formed is
A)
\[C{{H}_{3}}-CH=C{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-C\equiv CH\] done
clear
C)
D)
View Solution play_arrow
-
question_answer14)
\[C{{H}_{3}}Br+N{{u}^{-}}\xrightarrow{{}}C{{H}_{3}}-Nu+B{{r}^{-}}\] The decreasing order of the rate of the above reaction with nucleophiles \[\left( N{{u}^{-}} \right)\] A to D is \[[N{{u}^{-}}=(A)Ph{{O}^{-}},(B)Ac{{O}^{-}},(C)H{{O}^{-}},(D)C{{H}_{3}}{{O}^{-}}]\]
A)
\[A>B>C>D\] done
clear
B)
\[B>D>C>A\] done
clear
C)
\[~D>C>A>B\] done
clear
D)
\[D>C>B>A\] done
clear
View Solution play_arrow
-
question_answer15)
In a nucleophilic substitution reaction: \[R-Br+C{{l}^{-}}\xrightarrow{DMF}R-Cl+Br{{~}^{-}},\] Which one of the following undergoes complete inversion of configuration?
A)
\[{{C}_{6}}{{H}_{5}}CH{{C}_{6}}{{H}_{5}}Br\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Br\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHC{{H}_{3}}Br\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}CC{{H}_{3}}{{C}_{6}}{{H}_{5}}Br\] done
clear
View Solution play_arrow
-
question_answer16)
Which of the following statements is wrong?
A)
Ethyl chloride on reduction with Zn-Cu couple and alcohol gives ethane. done
clear
B)
The reaction of methyl magnesium bromide with acetone gives butanol-2. done
clear
C)
Alkyl halides follow the following reactivity sequence on reaction with alkenes. \[R-I>R-Br>R-Cl>R-I\] done
clear
D)
\[{{C}_{2}}{{H}_{4}}C{{l}_{2}}\] may exist in two isomeric forms. done
clear
View Solution play_arrow
-
question_answer17)
Rate of \[{{S}_{N}}2\] will be negligible in:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer18)
A major component of Borsch reagent is obtained by reacting hydrazine hydrate with which of the following?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer19)
Chlorobenzne reacts with trichloro acetaldehyde in the presence of \[{{H}_{2}}S{{O}_{4}}\]
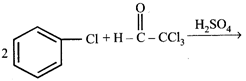
The major product formed is:
A)
B)
C)
D)
View Solution play_arrow
-
question_answer20)
The total number of acyclic isomers including the stereoisomers with the molecular formula \[{{C}_{4}}{{H}_{7}}Cl\]
A)
11 done
clear
B)
12 done
clear
C)
9 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer21)
The major product formed when 1,1,1 -trichloro- propane is treated with aqueous potassium hydroxide is:
A)
Propyne done
clear
B)
1-Propanol done
clear
C)
2-Propanol done
clear
D)
Propionic acid done
clear
View Solution play_arrow
-
question_answer22)
Which one of the following reagents is not suitable for the elimination reaction?
A)
NaI/acetone done
clear
B)
NaOEt/EtOH done
clear
C)
\[NaOH/{{H}_{2}}O\] done
clear
D)
\[NaOH/{{H}_{2}}O-EtOH\] done
clear
View Solution play_arrow
-
question_answer23)
Identify Z in the following serie's \[{{C}_{2}}{{H}_{5}}\xrightarrow{Alc.KOH}X\xrightarrow{B{{r}_{2}}}Y\xrightarrow{KCN}Z\]
A)
\[C{{H}_{3}}C{{H}_{2}}CN\] done
clear
B)
\[NCC{{H}_{2}}-C{{H}_{2}}CN\] done
clear
C)
\[BrC{{H}_{2}}-C{{H}_{2}}CN\] done
clear
D)
\[BrCH=CHCN\] done
clear
View Solution play_arrow
-
question_answer24)
Among the following, the molecule with the lowest dipole moment is
A)
\[CHC{{l}_{3}}\] done
clear
B)
\[C{{H}_{3}}Cl\] done
clear
C)
\[C{{H}_{2}}C{{l}_{2}}\] done
clear
D)
\[CC{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer25)
Which chloride is least reactive with the hydrolysis point of view?
A)
\[C{{H}_{3}}Cl\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\] done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{3}}CCl\] done
clear
D)
\[C{{H}_{2}}=CH-Cl\] done
clear
View Solution play_arrow
-
question_answer26)
The reaction is described as

A)
\[{{S}_{E}}2\] done
clear
B)
\[{{S}_{N}}1\] done
clear
C)
\[{{S}_{N}}2\] done
clear
D)
\[{{S}_{N}}0\] done
clear
View Solution play_arrow
-
question_answer27)
Replacement of \[Cl\] of chlorobenzene to give phenol requires drastic conditions but chlorine of 2,4-dinitrochlorobenzene is readily replaced. This is because
A)
\[N{{O}_{2}}\] makes the ring electron rich at ortho and para positions. done
clear
B)
\[N{{O}_{2}}\] withdraws \[{{e}^{-}}\] from meta-position. done
clear
C)
\[N{{O}_{2}}\] donates \[{{e}^{-}}\] at meta-position. done
clear
D)
\[N{{O}_{2}}\] withdraws \[{{e}^{-}}\] from ortho/para-positions. done
clear
View Solution play_arrow
-
question_answer28)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\] obtained by chlorination of n-butane, will be
A)
meso-form done
clear
B)
racemic mixture done
clear
C)
d-form done
clear
D)
l-form done
clear
View Solution play_arrow
-
question_answer29)
Which compound in each of the following pairs is most reactive to the conditions indicated?
A)
A and C done
clear
B)
B and C done
clear
C)
A and D done
clear
D)
B and D done
clear
View Solution play_arrow
-
question_answer30)
2-Bromopentane is heated with potassium ethoxide in ethanol. The major product obtained is
A)
2-Ethoxypentane done
clear
B)
Pentene-1 done
clear
C)
cis-Pentene-2 done
clear
D)
trans-Pentene-2 done
clear
View Solution play_arrow
-
question_answer31)
In the following reaction, compound [B] is
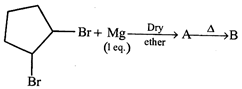
A)
B)
C)
D)
View Solution play_arrow
-
question_answer32)
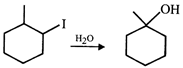
Which of the following is not expected to be intermediate of the following reaction?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer33)
Among the given halides, which one will give same product in both \[{{S}_{N}}1\] and \[{{S}_{N}}2\] reactions?
| (I) \[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-\underset{Br}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\] |
(II) 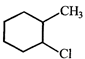 (III) (III)  |
| (IV)\[C{{H}_{3}}-\underset{Et}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-Br\] |
A)
(III) only done
clear
B)
(I) and (II) done
clear
C)
(III) and (IV) done
clear
D)
(I), (III) and (IV) done
clear
View Solution play_arrow
-
question_answer34)
\[C{{H}_{3}}C{{H}_{2}}Cl\xrightarrow{NaCN}X\xrightarrow{Ni/{{H}_{2}}}\]\[Y\xrightarrow[anhydride]{Acetic}Z\] Z in the above reaction sequence is
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}NHCOC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}N{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CONHC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CONHCOC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer35)
Which of the following is most reactive towards \[{{S}_{N}}2\] reaction?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer36)
Identify correct reactivity order for \[{{S}_{N}}1\] reaction
A)
i>ii>iii done
clear
B)
ii>iii>i done
clear
C)
i>iii>ii done
clear
D)
iii>ii>i done
clear
View Solution play_arrow
-
question_answer37)
Which compound undergoes nucleophilic substitution with \[NaCN\] at the fastest rate?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer38)
Identify correct reactivity order for \[{{S}_{N}}1\] reaction
A)
i>ii>iii done
clear
B)
ii>iii>i done
clear
C)
i>iii>ii done
clear
D)
ii>i>iii done
clear
View Solution play_arrow
-
question_answer39)
Which one of the sequences below is the best synthesis of(E)-3-hexene?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer40)
Which of the following reacts at the fast rate with \[C{{H}_{3}}OK\] in \[C{{H}_{3}}OH\] ?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer41)
In the following sequence of reactions \[\underset{A}{\mathop{C{{H}_{3}}C{{H}_{2}}I}}\,\xrightarrow[ether]{Mg}B\xrightarrow{HCHO}C\xrightarrow{{{H}_{2}}O}D\] the compound D is
A)
propanal done
clear
B)
butanal done
clear
C)
n-butyl alcohol done
clear
D)
n-propyl alcohol done
clear
View Solution play_arrow
-
question_answer42)
The correct product obtained in the reaction
A)
B)
C)
D)
View Solution play_arrow
-
question_answer43)
2-phenyl-2-hexanol can be prepared by Grignard synthesis. The pair of compounds giving the desired product is
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer44)
In the following reaction sequence: \[\underset{({{C}_{3}}\overset{{}}{\mathop{{{H}_{6}}}}\,C{{l}_{2}})}{\mathop{I}}\,\xrightarrow{KOH(aq)}II\xrightarrow[(ii){{H}_{2}}O/{{H}^{+}}]{(i)C{{H}_{3}}MgBr}III\] \[\xrightarrow{Anhy.ZnC{{l}_{2}}+Conc.HCl}given\,turbidity\,immediately\] The compound I is:
A)
\[\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\] done
clear
B)
\[\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,{{H}_{2}}-\underset{{}}{\mathop{\underset{{}}{\mathop{C}}\,}}\,{{H}_{2}}-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,{{H}_{3}}\] done
clear
C)
\[Cl-\underset{Cl}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-\underset{Cl}{\mathop{\overset{Cl}{\mathop{\underset{|}{\mathop{\overset{|}{\mathop{C}}\,}}\,}}\,}}\,-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer45)
Compound that on hydrogenation produces optically inactive compound is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer46)
Which of the following order is not correct?
A)
\[MeBr>M{{e}_{2}}CHBr>\] \[M{{e}_{3}}CBr>E{{t}_{3}}CBr\left( {{S}_{N}}2 \right)\] done
clear
B)
\[PhC{{H}_{2}}Br>Ph\,CHBrMe>PhCBrM{{e}_{2}}>\]\[PhCBrMePh\left( {{S}_{N}}1 \right)\] done
clear
C)
\[MeI>MeBr>MeCl>MeF({{S}_{N}}2)\] done
clear
D)
All are correct done
clear
View Solution play_arrow
-
question_answer47)
The major product of the following reaction is: \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}-\underset{Br}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-C{{H}_{2}}-C{{H}_{3}}\xrightarrow[{{C}_{2}}{{H}_{5}}OH]{{{C}_{2}}{{H}_{5}}ONa}\]
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}-\underset{O{{C}_{2}}{{H}_{5}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-C{{H}_{2}}-C{{H}_{3}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}CH=\underset{C{{H}_{3}}}{\overset{{}}{\mathop{\underset{|}{\overset{{}}{\mathop{C}}}\,}}}\,-C{{H}_{2}}-C{{H}_{3}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,=CHC{{H}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}-\underset{C{{H}_{2}}C{{H}_{3}}}{\overset{{}}{\mathop{\underset{|}{\overset{{}}{\mathop{C}}}\,}}}\,=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer48)
The compound most reactive towards \[{{S}_{N}}1\] reaction
A)
\[M{{e}_{3}}COC{{H}_{2}}Cl\] done
clear
B)
\[MeOC{{H}_{2}}Cl\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}C{{H}_{2}}Cl\] done
clear
D)
View Solution play_arrow
-
question_answer49)
Which chloro derivative of benzene among the following would undergo hydrolysis most readily with an aqueous sodium hydroxide to furnish the corresponding hydroxy derivative?
A)
B)
C)
D)
\[{{C}_{6}}{{H}_{5}}Cl\] done
clear
View Solution play_arrow
-
question_answer50)
In the following sequence of reactions, which is incorrect \[C{{H}_{2}}=CH-CH=C{{H}_{2}}\xrightarrow[80{}^\circ C]{HBr}\text{ }A\xrightarrow{HBr}B\] \[\xrightarrow{Mg}C\]
A)
A is \[C{{H}_{3}}-CH=CH-C{{H}_{2}}Br\] done
clear
B)
B is \[C{{H}_{3}}CHBr-C{{H}_{2}}-C{{H}_{2}}-Br\] done
clear
C)
C is cyclopropyi methane done
clear
D)
B on \[{{S}_{N}}2\] with aqueous \[NaOH\] gives \[C{{H}_{3}}CHOH.C{{H}_{2}}C{{H}_{2}}Br\] and not \[C{{H}_{3}}CHBr.C{{H}_{2}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer51)
The final product obtained in the reaction
A)
B)
C)
D)
View Solution play_arrow
-
question_answer52)
The following reaction proceeds through the intermediate formation of \[RCOOAg+B{{r}_{2}}\xrightarrow{{}}RBr+C{{O}_{2}}+AgBr\]
A)
\[RCO{{O}^{\bullet }}\] done
clear
B)
\[{{R}^{\bullet }}\] done
clear
C)
\[B{{r}^{\bullet }}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer53)
The compound \[{{C}_{7}}{{H}_{8}}\xrightarrow{3C{{l}_{2}}/\Delta }A\xrightarrow{B{{r}_{2}}/Fe}B\xrightarrow{Zn/HCl}C\]. The compound C is
A)
o-Bromotoluene done
clear
B)
m-Bromotoluene done
clear
C)
p-Bromotoluene done
clear
D)
3-Bromo-2,4,6-trichlorotoluene done
clear
View Solution play_arrow
-
question_answer54)
The product of 1-bromo-3-chloro cyclobutane with Na in presence ofdioxane
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer55)
Which of the following has the weakest carbon - chlorine bond?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer56)
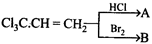
Which of the following is correct?
A)
A on reaction with aq. KOH gives \[HOC{{H}_{2}}C{{H}_{2}}COOK\] done
clear
B)
B can be resolved into d- and l-forms done
clear
C)
Both [a] and [b] done
clear
D)
Neither [a] nor [b] done
clear
View Solution play_arrow
-
question_answer57)
Although hexafluoroethane \[({{C}_{2}}{{F}_{6}},b.p.-79{}^\circ C)\] and ethane \[({{C}_{2}}{{H}_{6}},b.p.-89{}^\circ C)\] differ very much in their molecular weights, their boiling points differ only by\[10{}^\circ C\]. This is due to
A)
lowpolarizability of F done
clear
B)
nearly similar size of F and H done
clear
C)
both [a] and [b] done
clear
D)
Neither of the two done
clear
View Solution play_arrow
-
question_answer58)
Which of the following is a key intermediate in the reaction shown below?
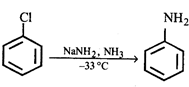
A)
B)
C)
D)
View Solution play_arrow
-
question_answer59)
Which of the following structure is more stable?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer60)
Which of the following is (are) true concerning the intermediate in the addition-elimination mechanism of the following reaction?
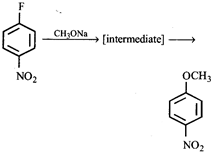
A = The intermediate is aromatic, B = The intermediate is a resonance stabilised anion, C = Electron withdrawing groups on the benzene ring stabilises the intermediate
A)
only A done
clear
B)
only B done
clear
C)
A and C done
clear
D)
B and C done
clear
View Solution play_arrow
-
question_answer61)
Identify Z in \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\xrightarrow{aq.\,NaOH}X\] \[\xrightarrow{A{{l}_{2}}{{O}_{3}}}Y\xrightarrow{C{{l}_{2}}/{{H}_{2}}O}Z\]
A)
Mixture of \[C{{H}_{3}}CHClC{{H}_{2}}Cl\] and \[C{{H}_{3}}CHOHC{{H}_{2}}Cl\] done
clear
B)
\[C{{H}_{3}}CHOHC{{H}_{2}}Cl\] done
clear
C)
\[C{{H}_{3}}CHClC{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}CHClC{{H}_{2}}Cl\] done
clear
View Solution play_arrow
-
question_answer62)
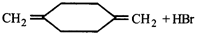
\[\xrightarrow{peroxide}A,\] then A is-
A)
B)
C)
D)
View Solution play_arrow
-
question_answer63)
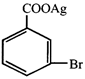
Silver benzoate reacts with bromine to form
A)
B)
C)
D)
\[{{C}_{6}}{{H}_{5}}Br\] done
clear
View Solution play_arrow
-
question_answer64)
\[PhCOCHB{{r}_{2}}\xrightarrow{O{{H}^{-}}}A\xrightarrow{O{{H}^{-}}}B\xrightarrow{{{H}^{+}}}C\] The compound C is -
A)
\[PhCH\left( OH \right)CHO\] done
clear
B)
\[PhCH\left( OH \right)COOH\] done
clear
C)
\[PhCO\underset{H}{\mathop{\underset{|}{\mathop{C}}\,}}\,B{{r}_{2}}\] done
clear
D)
\[Ph-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,-C{{H}_{2}}-OH\] done
clear
View Solution play_arrow
-
question_answer65)
A)
B)
C)
Both [a] and [b] done
clear
D)
No reaction done
clear
View Solution play_arrow
-
question_answer66)
X in the following reaction is -
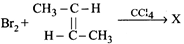
A)
(+) 2, 3-Dibromobutane done
clear
B)
(-) 2, 3-Dibromobutane done
clear
C)
(\[\pm \])- 2, 3-Dibromobutane done
clear
D)
meso-2, 3-Dibromobutane done
clear
View Solution play_arrow
-
question_answer67)
The missing reagents \[{{R}_{1}}\] and \[{{R}_{2}}\] in the following series of reactions are \[C{{H}_{3}}C{{H}_{2}}Br\xrightarrow{{{R}_{1}}}[\,\,\,\,]\xrightarrow{{{R}_{2}}}C{{H}_{3}}\overset{-}{\mathop{C}}\,H{{P}^{+}}P{{h}_{3}}\]
A)
PhLi and \[P{{h}_{3}}P\] respectively done
clear
B)
\[P{{h}_{3}}P\] and PhLi respectively done
clear
C)
\[P{{h}_{3}}P\] and \[{{C}_{2}}{{H}_{5}}ONa\] respectively done
clear
D)
Either [b] or [c] done
clear
View Solution play_arrow
-
question_answer68)
The reaction of toluene with chlorine in presence of ferric chloride gives predominantly:
A)
benzoyl chloride done
clear
B)
m-chlorotoluene done
clear
C)
benzyl chloride done
clear
D)
o- and p-chlorotoluene done
clear
View Solution play_arrow
-
question_answer69)
Arrange the following in order of decreasing rate of solvolysis with aqueous ethanol (fastest \[\to \] slowest)
A)
2>1>3 done
clear
B)
1>2>3 done
clear
C)
2>3>1 done
clear
D)
1>3>2 done
clear
View Solution play_arrow
-
question_answer70)
Which of the following alkyl halide undergo rearrangement in \[{{S}_{N}}1\] reaction?
A)
B)
C)
D)
All of these done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
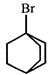 done
clear
done
clear
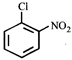 done
clear
done
clear
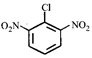 done
clear
done
clear
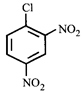 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
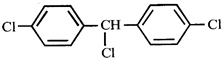 done
clear
done
clear
![]()

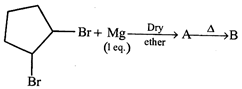
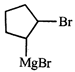 done
clear
done
clear
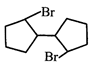 done
clear
done
clear
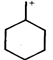 done
clear
done
clear
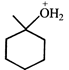 done
clear
done
clear
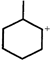 done
clear
done
clear
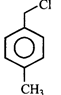 done
clear
done
clear
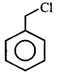 done
clear
done
clear
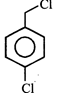 done
clear
done
clear
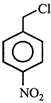 done
clear
done
clear
![]()
![]()
![]()
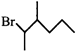 done
clear
done
clear
![]()
![]()
![]()
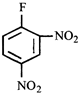 done
clear
done
clear
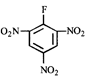 done
clear
done
clear
![]()
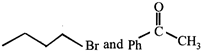 done
clear
done
clear
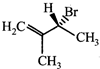 done
clear
done
clear
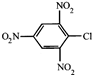 done
clear
done
clear

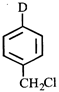 done
clear
done
clear
 done
clear
done
clear
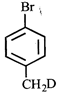 done
clear
done
clear
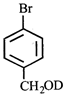 done
clear
done
clear
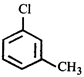 done
clear
done
clear
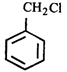 done
clear
done
clear
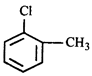 done
clear
done
clear
![]() Which of the following is correct?
Which of the following is correct? 
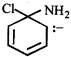 done
clear
done
clear
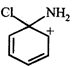 done
clear
done
clear
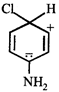 done
clear
done
clear
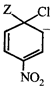 done
clear
done
clear
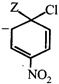 done
clear
done
clear
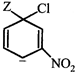 done
clear
done
clear
![]() \[\xrightarrow{peroxide}A,\] then A is-
\[\xrightarrow{peroxide}A,\] then A is- 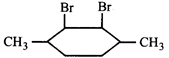 done
clear
done
clear
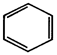 done
clear
done
clear
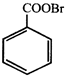 done
clear
done
clear
 done
clear
done
clear
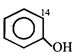 done
clear
done
clear
![]()
 done
clear
done
clear
 done
clear
done
clear
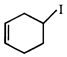 done
clear
done
clear
