-
question_answer1)
Which has lowest and highest first ionization enthalpy in 3d series?
A)
Cu and Cr done
clear
B)
Cu and Se done
clear
C)
Cu and Zn done
clear
D)
Sc and Zn done
clear
View Solution play_arrow
-
question_answer2)
For the four successive transition elements (Cr, Mn, Fe and Co), the stability of+2 oxidation state will be there in which of the following order?
A)
Fe > Mn > Co > Cr done
clear
B)
Cr > Mn > Co > Fe done
clear
C)
Mn > Cr > Fe > Co done
clear
D)
Co > Mn > Fe > Cr done
clear
View Solution play_arrow
-
question_answer3)
Which of the following is correct about basic strength of hydroxide?
A)
\[Sc{{(OH)}_{3}}>Y{{(OH)}_{3}}>La{{(OH)}_{3}}\] done
clear
B)
\[La{{(OH)}_{3}}>Y{{(OH)}_{3}}>Sc{{(OH)}_{3}}\] done
clear
C)
\[La{{(OH)}_{3}}>Y{{(OH)}_{3}}>Sc{{(OH)}_{3}}\] done
clear
D)
\[Y{{(OH)}_{3}}>La{{(OH)}_{3}}>Sc{{(OH)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer4)
AgCl and NaCl are colourless NaBr and Nal are also colourless but AgBr and AgI are coloured. This is due to
A)
\[A{{g}^{+}}\] has half-filled d-orbital done
clear
B)
\[A{{g}^{+}}\]polarises \[B{{r}^{-}}\] and \[{{I}^{-}}\] done
clear
C)
\[A{{g}^{+}}\]depolarises \[B{{r}^{-}}\] and \[{{I}^{-}}\] done
clear
D)
None is correct done
clear
View Solution play_arrow
-
question_answer5)
The basic character of the transition metal monoxides follows the order (Atomic number, Ti = 22, V = 23, Cr = 24, Fe = 26)
A)
TiO > VO > CrO > FeO done
clear
B)
VO > CrO > TiO > FeO done
clear
C)
TiO > FeO > VO > CrO done
clear
D)
CrO > VO > FeO > TiO done
clear
View Solution play_arrow
-
question_answer6)
The radii (metallic) of Fe, Co and Ni are nearly same. This is due to
A)
lanthanide contraction done
clear
B)
the fact that successive addition of d-electrons screen the outer electrons (4s) from the inward pull of the nucleus done
clear
C)
increase in radii due to increase in 'n' is compensated by decrease in radii due to increase in effective nuclear charge (Z) done
clear
D)
atomic radii do not remain constant but decrease in a normal gradation done
clear
View Solution play_arrow
-
question_answer7)
The melting point of Cu, Ag and Au follow the order
A)
Cu>Ag>Au done
clear
B)
Au>Ag>Cu done
clear
C)
Cu>Au>Ag done
clear
D)
Ag>Au>Cu done
clear
View Solution play_arrow
-
question_answer8)
Compound that is both paramagnetic and coloured is
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
B)
\[{{(N{{H}_{4}})}_{2}}[TiC{{l}_{6}}]\] done
clear
C)
\[VOS{{O}_{4}}\] done
clear
D)
\[{{K}_{3}}[Cu{{(CN)}_{4}}]\] done
clear
View Solution play_arrow
-
question_answer9)
\[MnO_{4}^{-}\] is of intense pink colour, though Mn is in (+7) oxidation state. It is due to
A)
oxygen gives colour to it done
clear
B)
charge transfer when Mn gives its electron to oxygen done
clear
C)
charge transfer when oxygen gives its electron to mb making it Mn(+VI) hence coloured done
clear
D)
none is correct done
clear
View Solution play_arrow
-
question_answer10)
\[C{{r}_{2}}Cr{{O}_{7}}^{2-}\xrightarrow{pH=X}Cr{{O}_{4}}^{2-}\xrightarrow{pH=Y}C{{r}_{2}}O_{4}^{2-}\] pH Value of X and Fare
A)
4 and 5 done
clear
B)
4 and 8 done
clear
C)
8 and 4 done
clear
D)
8 and 9 done
clear
View Solution play_arrow
-
question_answer11)
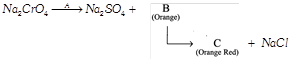
A, B and C respectively are:
A)
\[{{K}_{2}}S{{O}_{4}}\],\[{{K}_{2}}Cr{{O}_{4}}\]and \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
B)
\[{{K}_{2}}S{{O}_{4}}\],\[N{{a}_{2}}Cr{{O}_{4}}\] and \[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\],\[NaCr{{O}_{2}}\] and \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
D)
\[{{H}_{2}}S{{O}_{4}}\],\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\] and \[{{K}_{2}}C{{r}_{2}}{{0}_{7}}\] done
clear
View Solution play_arrow
-
question_answer12)
Pyrolusite in \[Mn{{O}_{2}}\] is used to prepare\[KMn{{O}_{4}}\]. Steps are \[Mn{{O}_{2}}\xrightarrow{I}Mn{{O}_{4}}^{2-}\xrightarrow{II}Mn{{O}_{4}}^{-}\] Here, I and II are
A)
fused with KOH/air, electrolytic oxidation done
clear
B)
fuse with cone. HN03/air, electrolytic oxidation done
clear
C)
fused with KOH/air, electrolytic reduction done
clear
D)
All are correct done
clear
View Solution play_arrow
-
question_answer13)
Identify P and Q products in given reaction \[KI+Mn{{O}_{4}}^{-}\xrightarrow{{{H}^{-}}}P+M{{n}^{2+}}\] \[KI+Mn{{O}_{4}}^{-}\xrightarrow{O{{H}^{-}}}I{{O}_{3}}^{-}+Q\]
A)
\[I{{O}_{3}}^{-}\],\[Mn{{O}_{2}}\]respectively done
clear
B)
\[{{I}_{2}}\],\[M{{n}^{2+}}\]respectively done
clear
C)
\[I{{O}_{3}}^{-},M{{n}^{2+}}\]respectively done
clear
D)
\[{{I}_{2}}\],\[Mn{{O}_{2}}\]respectively done
clear
View Solution play_arrow
-
question_answer14)
\[KMn{{O}_{4}}\] acts as an oxidizing agent in acidic medium. The number of moles of \[KMn{{O}_{4}}\] needed to react with one mole of sulphide ions in acidic solution is
A)
2/5 done
clear
B)
3/5 done
clear
C)
1/5 done
clear
D)
4/5 done
clear
View Solution play_arrow
-
question_answer15)
\[FeC{{r}_{2}}{{O}_{4}}\] (chromite) is converted to Cr by following steps: Chromite \[\xrightarrow{I}N{{a}_{2}}Cr{{O}_{4}}\]\[\xrightarrow{II}C{{r}_{2}}{{O}_{3}}\xrightarrow{III}Cr,\,\] I, II and III are
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
Across the lanthanide series, the basicity of the lanthanoid hydroxides
A)
increases done
clear
B)
decreases done
clear
C)
first increases and then decreases done
clear
D)
does not change done
clear
View Solution play_arrow
-
question_answer17)
The main reason for larger number of oxidation state exhibited by the actinides than that corresponding lanthanides, is
A)
lesser energy difference between 5\[f\]and 6d orbitals than between 4\[f\]and 5d-orbitals done
clear
B)
larger atomic size of actinides than the lanthanides done
clear
C)
more energy difference between 5\[f\]and 6d orbitals than between 4\[f\]and 5d-orbitals done
clear
D)
greater reactive nature of the actinides than the lanthanides. done
clear
View Solution play_arrow
-
question_answer18)
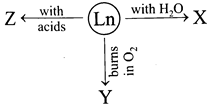
The X, Y and Z respectively are
A)
\[L{{n}_{2}}{{O}_{3}},{{H}_{2}},Ln{{(OH)}_{3}}+{{H}_{2}}\] done
clear
B)
\[Ln{{(OH)}_{3}}+{{H}_{2}},L{{n}_{2}}{{O}_{3}},{{H}_{2}}\] done
clear
C)
\[{{H}_{2}},L{{n}_{2}}{{O}_{3}},Ln{{(OH)}_{3}}+{{H}_{2}}\] done
clear
D)
\[Ln{{(OH)}_{3}}+{{H}_{2}},LN{{O}_{3}},{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer19)
A red solid is insoluble in water. However it becomes soluble if some KI is added to water. Heating the red solid in a test tube results in liberation of some violet colored fumes and droplets of metal appear on the cooler part of the test tube. The red solid is
A)
\[HgO\] done
clear
B)
\[P{{b}_{3}}{{O}_{4}}\] done
clear
C)
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
D)
\[Hg{{I}_{2}}\] done
clear
View Solution play_arrow
-
question_answer20)
Excess of KI reacts with \[CuS{{O}_{4}}\] solution and then \[N{{a}_{2}}{{S}_{2}}{{O}_{3}}\] solution is added to it. Which of the statements is incorred for this reaction?
A)
Evolved \[{{I}_{2}}\] is reduced done
clear
B)
\[C{{u}_{2}}{{I}_{2}}\] is formed done
clear
C)
\[N{{a}_{2}}{{S}_{2}}{{O}_{3}}\]is oxidized done
clear
D)
\[Cu{{I}_{2}}\]is formed done
clear
View Solution play_arrow
-
question_answer21)
Out of the following, how many of these have magnetic moment value \[2\sqrt{12}\] \[M{{n}^{2+}},M{{n}^{1+}},{{\operatorname{Re}}^{2+}},{{\operatorname{Re}}^{1+}},T{{e}^{1+}},Cr,W\]
View Solution play_arrow
-
question_answer22)
Out of following how many oxides are amphoteric in nature. \[MnO,M{{n}_{2}}O,Mn{{O}_{2}},Mn{{O}_{4}},M{{n}_{2}}{{O}_{7}},M{{n}_{3}}{{O}_{4}}\]
View Solution play_arrow
-
question_answer23)
Following steps are involved in manufacturing of potassium dichromate:
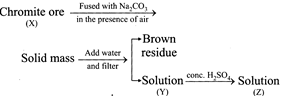
What is difference in the O.N. of Cr between X and Z?
View Solution play_arrow
-
question_answer24)
\[KMn{{O}_{4}}\] oxidises thiosulphate to \[S{{O}_{4}}^{2-}\] as per reaction \[xMn{{O}_{4}}^{-}+3{{S}_{2}}{{O}_{3}}^{2-}+{{H}_{2}}O\to xMn{{O}_{2}}+6S{{O}_{4}}^{2-}+2O{{H}^{-}}\]Find the value of x in above reaction.
View Solution play_arrow
-
question_answer25)
Number of good reducing agents among \[C{{e}^{4+}},E{{u}^{2+}},S{{m}^{2+}},Y{{b}^{2+}},T{{b}^{3+}},L{{a}^{3+}},C{{e}^{3+}},are\]
View Solution play_arrow
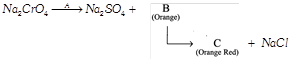 A, B and C respectively are:
A, B and C respectively are:  The X, Y and Z respectively are
The X, Y and Z respectively are 