| Observe the given figure carefully. |
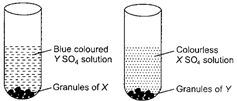
|
| X and Y respectively are |
A) Zn and Ag
B) Au and Cu
C) Fe and Cu
D) Zn and Cu
Correct Answer: D
Solution :
Zinc is more reactive than copper so, Zn displaces Cu from its blue, \[\text{CuS}{{\text{O}}_{\text{4}}}\] solution and forms colourless, \[\text{ZnS}{{\text{O}}_{\text{4}}}\] solution. \[\underset{(X)}{\mathop{\text{Zn}}}\,+\underset{(Blue)}{\mathop{CuS{{O}_{4}}}}\,\to \underset{(Colourless)}{\mathop{ZnS{{O}_{4}}}}\,+\underset{(Y)}{\mathop{Cu}}\,\]You need to login to perform this action.
You will be redirected in
3 sec
