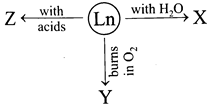
 The X, Y and Z respectively are
The X, Y and Z respectively are
A) \[L{{n}_{2}}{{O}_{3}},{{H}_{2}},Ln{{(OH)}_{3}}+{{H}_{2}}\]
B) \[Ln{{(OH)}_{3}}+{{H}_{2}},L{{n}_{2}}{{O}_{3}},{{H}_{2}}\]
C) \[{{H}_{2}},L{{n}_{2}}{{O}_{3}},Ln{{(OH)}_{3}}+{{H}_{2}}\]
D) \[Ln{{(OH)}_{3}}+{{H}_{2}},LN{{O}_{3}},{{H}_{2}}\]
Correct Answer: B
Solution :
[b] Lanthanoids react with dilute acids to liberate \[{{H}_{2}}\]gas and they form oxides and hydroxides of the type \[{{M}_{2}}{{O}_{3}}\] and \[M{{(OH)}_{3}}+{{H}_{2}}\] respectively.You need to login to perform this action.
You will be redirected in
3 sec
