-
question_answer1)
Which of the following amine will not react with nitrous acid to give nitrogen [NCERT 1984]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}\] done
clear
C)
\[\underset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,}{\mathop{C{{H}_{3}}-CH-N{{H}_{2}}}}\,}}\,\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}N\] done
clear
View Solution play_arrow
-
question_answer2)
Which of the following compound is expected to be most basic [NCERT 1982]
A)
Aniline done
clear
B)
Methylamine done
clear
C)
Hydroxylamine done
clear
D)
Ethylamine done
clear
View Solution play_arrow
-
question_answer3)
Which of the following compounds is an amino acid [Manipal MEE 1995]
A)
\[C{{H}_{3}}-C{{H}_{2}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-O-N{{H}_{4}}\] done
clear
B)
\[C{{H}_{3}}-\underset{N{{H}_{2}}\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,\,}{\mathop{CH-}}\,}}\,\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-OH\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-N{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}-\underset{N{{H}_{2}}\,\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,}{\mathop{CH-}}\,}}\,\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-Cl\] done
clear
View Solution play_arrow
-
question_answer4)
Nitro group in nitrobenzene is a [MNR 1986]
A)
Ortho director done
clear
B)
Meta director done
clear
C)
Para director done
clear
D)
Ortho and para director done
clear
View Solution play_arrow
-
question_answer5)
The alkyl cyanides are
A)
Acidic done
clear
B)
Basic done
clear
C)
Neutral done
clear
D)
Amphoteric done
clear
View Solution play_arrow
-
question_answer6)
The alkyl cyanides when hydrolysed to the corresponding acid, the gas evolved is
A)
\[{{N}_{2}}\] done
clear
B)
\[{{O}_{2}}\] done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
\[C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer7)
Aniline when treated with \[HN{{O}_{2}}\] and HCl at \[{{0}^{o}}C\] gives [CPMT 1982, 89; RPMT 2000]
A)
Phenol done
clear
B)
Nitrobenzene done
clear
C)
A diazo compound done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer8)
Nitrosobenzene can be isolated from nitrobenzene under [DPMT 1982]
A)
Metal and acid done
clear
B)
Zn dust and \[N{{H}_{4}}Cl\] done
clear
C)
Alkaline sodium arsenite done
clear
D)
Cannot be isolated done
clear
View Solution play_arrow
-
question_answer9)
Alkyl cyanides when react with Grignard reagent, the product on hydrolysis found, is [MP PMT 1980]
A)
Aldehyde done
clear
B)
Ketone done
clear
C)
Alcohol done
clear
D)
Acid done
clear
View Solution play_arrow
-
question_answer10)
The product formed when benzene is nitrated by fuming nitric acid is [MP PMT 1979]
A)
m-dinitrobenzene done
clear
B)
Nitrobenzene done
clear
C)
sym-trinitrobenzene done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer11)
Ethyl amine undergoes oxidation in the presence of \[KMn{{O}_{4}}\] to form [CPMT 1985]
A)
An acid done
clear
B)
An alcohol done
clear
C)
An aldehyde done
clear
D)
A nitrogen oxide done
clear
View Solution play_arrow
-
question_answer12)
Which of the following amines would undergo diazotisation
A)
Primary aliphatic amines done
clear
B)
Primary aromatic amines done
clear
C)
Both A and B done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer13)
Reaction of primary amines with aldehyde yields [NCERT 1984; Manipal MEE 1995]
A)
Amides done
clear
B)
Aldimines done
clear
C)
Nitriles done
clear
D)
Nitro compounds done
clear
View Solution play_arrow
-
question_answer14)
When acetamide is treated with \[HN{{O}_{2}},\] the gas is evolved [CPMT 1993]
A)
\[{{H}_{2}}\] done
clear
B)
\[{{O}_{2}}\] done
clear
C)
\[{{N}_{2}}\] done
clear
D)
\[C{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer15)
Nitrobenzene on nitration gives [NCERT 1978; CPMT 1989]
A)
o-dinitrobenzene done
clear
B)
p-dinitrobenzene done
clear
C)
m-dinitrobenzene done
clear
D)
o- and p-nitrobenzene done
clear
View Solution play_arrow
-
question_answer16)
Reduction of alkyl nitrites yields
A)
Alcohol done
clear
B)
Base done
clear
C)
Amine done
clear
D)
Acid done
clear
View Solution play_arrow
-
question_answer17)
When primary amines are treated with HCl, the product obtained is
A)
An alcohol done
clear
B)
A cyanide done
clear
C)
An amide done
clear
D)
Ammonium salt done
clear
View Solution play_arrow
-
question_answer18)
Which one is weakest base [BHU 1982; RPMT 2000]
A)
Ammonia done
clear
B)
Methylamine done
clear
C)
Dimethylamine done
clear
D)
Trimethylamine done
clear
View Solution play_arrow
-
question_answer19)
Chloroform when treated with aniline and alcoholic KOH gives [CPMT 1986; EAMCET 1992; MP PMT 1997; Pb. PMT 1999]
A)
Phenyl cyanide done
clear
B)
Phenyl isocyanide done
clear
C)
Chlorobenzene done
clear
D)
Phenol done
clear
View Solution play_arrow
-
question_answer20)
Which of following do not react with \[HN{{O}_{2}}\]
A)
Primary nitroalkanes done
clear
B)
Secondary nitroalkanes done
clear
C)
Tertiary nitroalkanes done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer21)
Primary amines can be distinguished from secondary and tertiary amines by reacting with [CPMT 1983]
A)
Chloroform and alcoholic KOH done
clear
B)
Methyl iodide done
clear
C)
Chloroform alone done
clear
D)
Zinc dust done
clear
View Solution play_arrow
-
question_answer22)
Which of following is not an usual method for preparation of primary amine
A)
Hofmann's method done
clear
B)
Curtius reaction done
clear
C)
Schmidt reaction done
clear
D)
Friedel-Craft's reaction done
clear
View Solution play_arrow
-
question_answer23)
A solution of methyl amine
A)
Turns blue litmus red done
clear
B)
Turns red litmus blue done
clear
C)
Does not affect red or blue litmus done
clear
D)
Bleaches litmus done
clear
View Solution play_arrow
-
question_answer24)
Mark the correct statement [CPMT 1974; DPMT 1983; MP PMT 1994]
A)
Methyl amine is slightly acidic done
clear
B)
Methyl amine is less basic than \[N{{H}_{3}}\] done
clear
C)
Methyl amine is stronger base than \[N{{H}_{3}}\] done
clear
D)
Methyl amine forms salts with alkalies done
clear
View Solution play_arrow
-
question_answer25)
The product of mustard oil reaction is
A)
Alkyl isothiocyanate done
clear
B)
Dithio carbonamide done
clear
C)
Dithio ethylacetate done
clear
D)
Thioether done
clear
View Solution play_arrow
-
question_answer26)
Which of the following is azo- group
A)
\[-N=\] done
clear
B)
\[-N=N-\] done
clear
C)
\[-NH-\] done
clear
D)
\[-CO-NH-\] done
clear
View Solution play_arrow
-
question_answer27)
'Oil of mirbane' is
A)
Aniline done
clear
B)
Nitrobenzene done
clear
C)
p-nitroaniline done
clear
D)
p-aminoazobenzene done
clear
View Solution play_arrow
-
question_answer28)
The maximum number of \[-N{{O}_{2}}\] groups that can be introduced by nitration in benzene is usually
A)
4 done
clear
B)
2 done
clear
C)
3 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer29)
Nitrobenzene at room temperature is
A)
Gas done
clear
B)
Liquid done
clear
C)
Solid done
clear
D)
Solution done
clear
View Solution play_arrow
-
question_answer30)
In the explosive amatol, TNT is mixed with [CPMT 1988]
A)
Ammonium citrate done
clear
B)
Ammonium nitrate done
clear
C)
Ammonium oxalate done
clear
D)
Ammonium sulphate done
clear
View Solution play_arrow
-
question_answer31)
By reduction of nitrosobenzene which of the following is not obtained
A)
B)
C)
D)
View Solution play_arrow
-
question_answer32)
By the presence of a halogen atom in the ring, basic properties of aniline is
A)
Increased done
clear
B)
Decreased done
clear
C)
Unchanged done
clear
D)
Doubled done
clear
View Solution play_arrow
-
question_answer33)
In the mustard oil reaction, an amine is treated with
A)
\[Na\,/\,{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[Sn\,/\,HCl\] done
clear
C)
\[C{{S}_{2}}\] done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}/{{H}_{2}}S{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer34)
Primary nitro compounds when react with \[HN{{O}_{2}}\] forms crystalline solids which on treatment with NaOH gives
A)
Red solution done
clear
B)
Blue solution done
clear
C)
White precipitate done
clear
D)
Yellow coloration done
clear
View Solution play_arrow
-
question_answer35)
Secondary nitro compounds when react with \[HN{{O}_{2}}\] forms crystalline solids which one on treatment with NaOH gives
A)
Red solution done
clear
B)
Blue solution done
clear
C)
White precipitate done
clear
D)
Yellow colouration done
clear
View Solution play_arrow
-
question_answer36)
Which of the following possess powerful mustard smell (and are called mustard oils)
A)
Alkyl isocyanates done
clear
B)
Alkyl cyanates done
clear
C)
Alkyl isothiocyanates done
clear
D)
Alkyl thiocyanates done
clear
View Solution play_arrow
-
question_answer37)
On heating acetamide in presence of \[{{P}_{2}}{{O}_{5}},\] which of the following is formed [MP PMT 1992; MP PET 1994; Kurukshetra CEE 1998]
A)
Ammonium acetate done
clear
B)
Acetonitrile done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
Methylamines done
clear
View Solution play_arrow
-
question_answer38)
When chloroform reacts with ethyl amine in presence of alcoholic KOH, the compound formed is [CPMT 1983; MP PMT 1993; CBSE PMT 1997; BHU 1999; AIEEE 2002]
A)
Ethyl cyanide done
clear
B)
Ethyl isocyanide done
clear
C)
Formic acid done
clear
D)
An amide done
clear
View Solution play_arrow
-
question_answer39)
When methyl cyanide is hydrolysed in presence of alkali, the product is [MP PMT 1993; BCECE 2005]
A)
Acetamide done
clear
B)
Methane done
clear
C)
\[C{{O}_{2}}+{{H}_{2}}O\] done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer40)
Hofmann's hypobromite reaction affords a method of [MP PMT 1993]
A)
Preparing a tertiary amine done
clear
B)
Preparing a mixture of amines done
clear
C)
Stepping down a series done
clear
D)
Stepping up a series done
clear
View Solution play_arrow
-
question_answer41)
The compound which on reaction with aqueous nitrous acid on \[HN{{O}_{2}}\] at low temperature produces an oily nitrosoamine is [IIT 1981; CPMT 1989; MP PET/PMT 1998; Kurukshetra CEE 1998; MP PMT 2001]
A)
Diethylamine done
clear
B)
Ethylamine done
clear
C)
Aniline done
clear
D)
Methylamine done
clear
View Solution play_arrow
-
question_answer42)
Identify the product Z in the series \[C{{H}_{3}}CN\xrightarrow{Na+{{C}_{2}}{{H}_{5}}OH}X\xrightarrow{HN{{O}_{2}}}Y\underset{{{H}_{2}}S{{O}_{4}}}{\mathop{\xrightarrow{{{K}_{2}}C{{r}_{2}}{{O}_{7}}}}}\,Z\] [AIIMS 1983; JIPMER 2001]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}NHOH\] done
clear
View Solution play_arrow
-
question_answer43)
The end product of the reactions is \[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\xrightarrow{HN{{O}_{2}}}A\xrightarrow{PC{{l}_{5}}}B\xrightarrow{H.N{{H}_{2}}}C\] [CPMT 1988, 89, 93; DCE 1999; JIPMER 2000]
A)
Ethyl cyanide done
clear
B)
Ethyl amine done
clear
C)
Methyl amine done
clear
D)
Acetamide done
clear
View Solution play_arrow
-
question_answer44)
Primary and secondary amines are distinguished by [AMU 1988; MP PMT 1996]
A)
\[B{{r}_{2}}/KOH\] done
clear
B)
\[HCl{{O}_{4}}\] done
clear
C)
\[HN{{O}_{2}}\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer45)
Which one of the following will give a primary amine on hydrolysis [BHU 1982]
A)
Nitroparaffin done
clear
B)
Alkyl cyanide done
clear
C)
Oxime done
clear
D)
Alkyl isocyanide done
clear
View Solution play_arrow
-
question_answer46)
Methyl amine reacts with \[HN{{O}_{2}}\] giving [RPMT 1997]
A)
\[C{{H}_{3}}O-N=O\] done
clear
B)
\[C{{H}_{3}}-O-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}OH\] done
clear
D)
A and B both done
clear
View Solution play_arrow
-
question_answer47)
Nitrobenzene on reduction by zinc and \[N{{H}_{4}}Cl\] gives [CPMT 1989, 94; BHU 1996; Pb. PMT 1999]
A)
Aniline done
clear
B)
Nitrosobenzene done
clear
C)
Hydrazobenzene done
clear
D)
Phenylhydroxyl amine done
clear
View Solution play_arrow
-
question_answer48)
The decreasing order of the basic character of the three amines and ammonia is [MP PET/PMT 1988; KCET 1990]
A)
\[N{{H}_{3}}>C{{H}_{3}}N{{H}_{2}}>{{C}_{2}}{{H}_{5}}N{{H}_{2}}>{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}>C{{H}_{3}}N{{H}_{2}}>N{{H}_{3}}>{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}>{{C}_{2}}{{H}_{5}}N{{H}_{2}}>C{{H}_{3}}N{{H}_{2}}>N{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}N{{H}_{2}}>{{C}_{2}}{{H}_{5}}N{{H}_{2}}>{{C}_{6}}{{H}_{5}}N{{H}_{2}}>N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer49)
Correct order of increasing basicity is [CBSE PMT 1992]
A)
\[N{{H}_{3}}<{{C}_{6}}{{H}_{5}}N{{H}_{2}}<{{({{C}_{2}}{{H}_{5}})}_{2}}NH<{{C}_{2}}{{H}_{5}}N{{H}_{2}}<{{({{C}_{2}}{{H}_{5}})}_{3}}N\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}<N{{H}_{3}}<{{({{C}_{2}}{{H}_{5}})}_{3}}N<{{({{C}_{2}}{{H}_{5}})}_{2}}NH<{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}<N{{H}_{3}}<{{C}_{2}}{{H}_{5}}N{{H}_{2}}<{{({{C}_{2}}{{H}_{5}})}_{3}}N<{{({{C}_{2}}{{H}_{5}})}_{2}}NH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}<{{({{C}_{2}}{{H}_{5}})}_{3}}N<N{{H}_{3}}<{{C}_{2}}{{H}_{5}}N{{H}_{2}}<{{({{C}_{2}}{{H}_{5}})}_{2}}NH\] done
clear
View Solution play_arrow
-
question_answer50)
Among the following compounds nitrobenzene, benzene, aniline and phenol, the strongest basic behaviour in acid medium is exhibited by [KCET 1993]
A)
Phenol done
clear
B)
Aniline done
clear
C)
Nitrobenzene done
clear
D)
Benzene done
clear
View Solution play_arrow
-
question_answer51)
Aniline on treatment with excess of bromine water gives [AFMC 1990; MP PMT 1991; RPMT 1997]
A)
Aniline bromide done
clear
B)
o-bromoaniline done
clear
C)
p-bromoaniline done
clear
D)
2, 4, 6-tribromoaniline done
clear
View Solution play_arrow
-
question_answer52)
Unpleasant smelling carbylamines are formed by heating alkali and chloroform with [KCET 1987, 2000, 01]
A)
Any amine done
clear
B)
Any aliphatic amine done
clear
C)
Any aromatic amine done
clear
D)
Any primary amine done
clear
View Solution play_arrow
-
question_answer53)
When an organic compound was treated with sodium nitrite and hydrochloric acid in the ice cold, nitrogen gas was evolved copiously. The compound is [KCET 1986]
A)
A nitro compound done
clear
B)
A primary amine done
clear
C)
An aliphatic primary amine done
clear
D)
An aromatic primary amine done
clear
View Solution play_arrow
-
question_answer54)
Aniline reacts with alkyl halide to give [KCET 1984]
A)
Amino compound done
clear
B)
Tertiary compound done
clear
C)
Quaternary ammonium compound done
clear
D)
Azomethane done
clear
View Solution play_arrow
-
question_answer55)
Aniline on treatment with conc. \[HN{{O}_{3}}+\] conc. \[{{H}_{2}}S{{O}_{4}}\] mixture yields [AIIMS 1992]
A)
o- and p-nitroanilines done
clear
B)
m-nitroanilines done
clear
C)
A black tarry matter done
clear
D)
No reaction done
clear
View Solution play_arrow
-
question_answer56)
Which statement is not correct [MP PMT 1995]
A)
Amines form hydrogen bond done
clear
B)
Ethyl amine has higher boiling point than propane done
clear
C)
Methyl amine is more basic than ammonia done
clear
D)
Dimethyl amine is less basic than methyl amine done
clear
View Solution play_arrow
-
question_answer57)
Which of the following is not used as an explosive [MP PET 1996]
A)
Trinitrotoluene done
clear
B)
Trinitrobenzene done
clear
C)
Picric acid done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer58)
Primary amines react with nitrous acid to yield
A)
Insoluble nitrite salts done
clear
B)
Yellow oily layer done
clear
C)
Nitrogen gas done
clear
D)
Azo dye done
clear
View Solution play_arrow
-
question_answer59)
Which of the following has the smell of bitter almonds
A)
Nitromethane done
clear
B)
Nitroethane done
clear
C)
Nitrobenzene done
clear
D)
Aniline done
clear
View Solution play_arrow
-
question_answer60)
The reaction of \[HN{{O}_{2}}\] with 'A' gives quaternary ammonium salt. A is [MP PMT 1997]
A)
Methyl amine done
clear
B)
Dimethyl amine done
clear
C)
Trimethyl amine done
clear
D)
Aniline done
clear
View Solution play_arrow
-
question_answer61)
Reaction of nitrous acid with aliphatic primary amine in the cold gives [MP PET/PMT 1998; CBSE PMT 1994]
A)
A diazonium salt done
clear
B)
An alcohol done
clear
C)
A nitrite done
clear
D)
A dye done
clear
View Solution play_arrow
-
question_answer62)
In presence of acid, hydrolysis of methyl cyanide gives [MP PET/PMT 1998]
A)
Acetic acid done
clear
B)
Methylamine done
clear
C)
Methyl alcohol done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer63)
The amine which does not react with acetyl chloride is or Which of the following cannot be acetylated [MP PET 1999; MP PMT 1999]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer64)
The fusion of sodium with amine gives mainly [MP PMT 1999; CPMT 2002]
A)
NaCN done
clear
B)
\[Na{{N}_{3}}\] done
clear
C)
NaSCN done
clear
D)
\[NaN{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer65)
Which of the following is most basic [MP PMT 1999]
A)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer66)
In reaction \[C{{H}_{3}}CN+2H\underset{\text{Ether}}{\mathop{\xrightarrow{HCl}}}\,X\xrightarrow{\text{Boiling }{{H}_{2}}O}Y;\] the term Y is [CBSE PMT 1999]
A)
Acetone done
clear
B)
Ethylamine done
clear
C)
Acetaldehyde done
clear
D)
Dimethylamine done
clear
View Solution play_arrow
-
question_answer67)
The following compound can be classified as N-N dimethyl propanamine, N-methyl aniline and aniline [Bihar MEE 1996]
A)
Primary, secondary, tertiary done
clear
B)
Primary, tertiary, secondary done
clear
C)
Secondary, tertiary, primary done
clear
D)
Tertiary, primary, secondary done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer68)
Which of the following compounds does not react with \[NaN{{O}_{2}}\] and HCl [KCET 1996]
A)
\[{{C}_{6}}{{H}_{5}}OH\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}CN{{O}_{2}}\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}CHN{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer69)
In the reduction of nitrobenzene, which of the following is the intermediate [CPMT 1999]
A)
\[{{C}_{6}}{{H}_{5}}N=O\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}NH-NH-{{C}_{6}}{{H}_{5}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}-N=N-{{C}_{6}}{{H}_{5}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N=\overset{O}{\mathop{\overset{\uparrow }{\mathop{N}}\,}}\,-{{C}_{6}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer70)
Aniline when treated with conc. \[HN{{O}_{3}}\] gives [KCET 1996]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer71)
Which one of the following is not a base [EAMCET 1997]
A)
\[{{N}_{2}}{{H}_{4}}\] done
clear
B)
\[N{{H}_{2}}OH\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N\] done
clear
D)
\[H{{N}_{3}}\] done
clear
View Solution play_arrow
-
question_answer72)
p-Nitrobromobenzene can be converted to p-nitroaniline by using \[NaN{{H}_{2}}\]. The reaction proceeds through the intermediate named [Orissa JEE 2005]
A)
Carbocation done
clear
B)
Carbanion done
clear
C)
Benzyne done
clear
D)
Dianion done
clear
View Solution play_arrow
-
question_answer73)
If methyl is alkyl group, then which order of basicity is correct [RPMT 1997]
A)
\[{{R}_{2}}NH>RN{{H}_{2}}>{{R}_{3}}N>N{{H}_{3}}\] done
clear
B)
\[{{R}_{2}}NH>{{R}_{3}}N>RN{{H}_{2}}>N{{H}_{3}}\] done
clear
C)
\[RN{{H}_{2}}>N{{H}_{3}}>{{R}_{2}}NH>{{R}_{3}}N\] done
clear
D)
\[N{{H}_{3}}>RN{{H}_{2}}>{{R}_{2}}NH>{{R}_{3}}N\] done
clear
View Solution play_arrow
-
question_answer74)
Which of the following has the minimum heat of dissociation [Roorkee Qualifying 1998]
A)
\[{{(C{{H}_{3}})}_{3}}N\to B{{F}_{3}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{3}}N\to B(C{{H}_{3}}){{F}_{2}}\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N\to B{{(C{{H}_{3}})}_{2}}F\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}N\to B{{(C{{H}_{3}})}_{3}}\] done
clear
View Solution play_arrow
-
question_answer75)
The major product (70% to 80%) of the reaction between m-dinitrobenzene with \[N{{H}_{4}}HS\] is [AIIMS 1997]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer76)
Which one is less alkaline [CPMT 1997]
A)
B)
C)
D)
All of these done
clear
View Solution play_arrow
-
question_answer77)
In the diazotisation of aniline with sodium nitrite and hydrochloric acid, an excess of hydrochloric acid is used primarily to [Pb. PMT 1998]
A)
Suppress the concentration of free aniline available for coupling done
clear
B)
Suppress hydrolysis of phenol done
clear
C)
Insure a stoichiometric amount of nitrous acid done
clear
D)
Neutralize the base liberated done
clear
View Solution play_arrow
-
question_answer78)
A primary amine can be converted to an alcohol by the action of [CET Pune 1998]
A)
Alkali done
clear
B)
Nitrous acid done
clear
C)
Reducing agent done
clear
D)
Oxidizing agent done
clear
View Solution play_arrow
-
question_answer79)
Arrange the following in increasing order of basicity \[C{{H}_{3}}N{{H}_{2}},\,\,{{(C{{H}_{3}})}_{2}}NH,\,{{C}_{6}}{{H}_{5}}N{{H}_{2}},\,{{(C{{H}_{3}})}_{3}}N\] [AFMC 1997]
A)
\[{{(C{{H}_{3}})}_{3}}N<{{(C{{H}_{3}})}_{2}}NH<C{{H}_{3}}N{{H}_{2}}<{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{3}}N>{{(C{{H}_{3}})}_{2}}NH>C{{H}_{3}}N{{H}_{2}}>{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}<{{(C{{H}_{3}})}_{3}}N<C{{H}_{3}}N{{H}_{2}}<{{(C{{H}_{3}})}_{2}}NH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}>{{(C{{H}_{3}})}_{3}}N>C{{H}_{3}}N{{H}_{2}}>{{(C{{H}_{3}})}_{2}}NH\] done
clear
View Solution play_arrow
-
question_answer80)
In the reaction \[C{{H}_{3}}CN+C{{H}_{3}}MgI\to A\xrightarrow{{{H}_{2}}O/{{H}^{+}}}B\] The compound B is [KCET 1999]
A)
Acetic acid done
clear
B)
Acetone done
clear
C)
Acetaldehyde done
clear
D)
Ethyl alcohol done
clear
View Solution play_arrow
-
question_answer81)
\[C{{H}_{3}}CN\] is known as acetonitrile because [AMU 1999]
A)
It contains an aceto group done
clear
B)
On hydrolysis it gives acetic acid done
clear
C)
Both A and B done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer82)
What is formed, when nitrobenzene is reduced using zinc and alkali [BHU 2000; AIIMS 2000; CBSE PMT 2000; MH CET 2003]
A)
Phenol done
clear
B)
Aniline done
clear
C)
Nitrosobenzene done
clear
D)
Hydrazobenzene done
clear
View Solution play_arrow
-
question_answer83)
\[RCOCl+2M{{e}_{2}}NH\to A+M{{e}_{2}}\overset{+\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{N{{H}_{2}}C{{l}^{-}}}}\,\]Here A is [RPET 2000]
A)
B)
\[RCON{{H}_{2}}\] done
clear
C)
\[RCONHMe\] done
clear
D)
\[{{(RCO)}_{2}}NH\] done
clear
View Solution play_arrow
-
question_answer84)
Decreasing order of basicity is [RPET 2000] (1) \[C{{H}_{3}}CON{{H}_{2}}\] (2) \[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] (3) Ph?\[C{{H}_{2}}CON{{H}_{2}}\]
A)
1 > 2 > 3 done
clear
B)
2 > 1 > 3 done
clear
C)
3 > 2 > 1 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer85)
Among the following, the strongest base is [UPSEAT 2000; IIT-JEE (Screening) 2000]
A)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[p-N{{O}_{2}}{{C}_{6}}{{H}_{4}}N{{H}_{2}}\] done
clear
C)
\[m-N{{O}_{2}}-{{C}_{6}}{{H}_{4}}N{{H}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer86)
Aniline and methyl amine can be differentiated by [DPMT 2000]
A)
Reaction with chloroform and aqueous solution of KOH done
clear
B)
Diazotisation followed by coupling with phenol done
clear
C)
Reaction with \[HN{{O}_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer87)
The amine which can react with \[{{C}_{6}}{{H}_{5}}-S{{O}_{2}}-Cl\] to form a product insoluble in alkali shall be [AMU 2000]
A)
Primary amine done
clear
B)
Secondary amine done
clear
C)
Tertiary amine done
clear
D)
Both primary and secondary amines done
clear
View Solution play_arrow
-
question_answer88)
A mixture of benzene and aniline can be separated by [KCET (Engg.) 2001]
A)
Hot water done
clear
B)
dil. HCl done
clear
C)
dil. NaOH done
clear
D)
Alcohol done
clear
View Solution play_arrow
-
question_answer89)
Nitrobenzene on further excessive nitration gives [AFMC 2001]
A)
Trinitrobenzene done
clear
B)
m-dinitrobenzene done
clear
C)
p-dinitrobenzene done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer90)
The compound A with following sequence of reaction gave benzoic acid \[A\xrightarrow{NaN{{O}_{2}}/HCl}B\xrightarrow{KCN}C\xrightarrow{{{H}_{3}}{{O}^{+}}}\] benzoic acid. The compound A is [AMU 2001]
A)
Nitrobenzene done
clear
B)
Aniline done
clear
C)
Benzaldehyde done
clear
D)
Amides done
clear
View Solution play_arrow
-
question_answer91)
Which of the following chemicals are used to manufacture methyl isocyanate that caused ?Bhopal Tragedy? (i) Methylamine (ii) Phosgene (iii) Phosphine (iv) Dimethylamine [AIIMS 2005]
A)
(i) and (iii) done
clear
B)
(iii) and (iv) done
clear
C)
(i) and (ii) done
clear
D)
(ii) and (iv) done
clear
View Solution play_arrow
-
question_answer92)
An isocyanide on hydrolysis gives [AMU 2001]
A)
An amide done
clear
B)
A carboxylic acid and ammonia done
clear
C)
A N-substituted amide done
clear
D)
A 1o-amine and formic acid done
clear
View Solution play_arrow
-
question_answer93)
Methyl isocyanide on hydrolysis gives [UPSEAT 2001]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
HCOOH done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
Both A and B done
clear
View Solution play_arrow
-
question_answer94)
Pure aniline is a [UPSEAT 2001]
A)
Colourless solid done
clear
B)
Brown coloured solid done
clear
C)
Colourless liquid done
clear
D)
Brown coloured liquid done
clear
View Solution play_arrow
-
question_answer95)
Reduction of methyl isocyanide gives [RPMT 2002]
A)
Ethylamine done
clear
B)
Methylamine done
clear
C)
Dimethylamine done
clear
D)
Trimethylamine done
clear
View Solution play_arrow
-
question_answer96)
Reaction of aniline with benzaldehyde is [RPMT 2002]
A)
Polymerisation done
clear
B)
Condensation done
clear
C)
Addition done
clear
D)
Substitution done
clear
View Solution play_arrow
-
question_answer97)
In the reaction \[{{C}_{6}}{{H}_{5}}CHO+{{C}_{6}}{{H}_{5}}N{{H}_{2}}\to \]\[{{C}_{6}}{{H}_{5}}N=HC{{C}_{6}}{{H}_{5}}+{{H}_{2}}O,\] the compound \[{{C}_{6}}{{H}_{5}}N=CH{{C}_{6}}{{H}_{5}}\] is known as [RPMT 2000; AIIMS 2002; AMU 2001]
A)
Aldol done
clear
B)
Schiff?s reagent done
clear
C)
Schiff?s base done
clear
D)
Benedict reagent done
clear
View Solution play_arrow
-
question_answer98)
The unshared pair of electrons on a cyanide ion can acts as [Kerala (Med.) 2002]
A)
Isocyanide centre done
clear
B)
Amido centre done
clear
C)
Cationic centre done
clear
D)
Nucleophilic centre done
clear
View Solution play_arrow
-
question_answer99)
Electrophilic substitution of

with bromine gives [Kerala (Med.) 2002]
A)
1, 4, 6-tribromo aniline done
clear
B)
2, 4, 6-tribromo aniline done
clear
C)
4-bromo aniline done
clear
D)
3-bromo aniline done
clear
View Solution play_arrow
-
question_answer100)
Mustard gas is obtained by [MP PET 2002]
A)
The action of dilute acids on mustard seeds done
clear
B)
Treating ethylene with mustard oil done
clear
C)
Treating sulphur chloride with ethylene done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer101)
Which of the following is capable of forming a zwitter ion [JIPMER 2002]
A)
\[{{C}_{6}}{{H}_{5}}-OH\] done
clear
B)
\[{{C}_{6}}{{H}_{4}}{{(N{{H}_{2}})}_{2}}\] done
clear
C)
\[\underset{C{{H}_{2}}OH\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{C{{H}_{2}}OH}}\,}}\,\] done
clear
D)
\[{{H}_{2}}N-C{{H}_{2}}-COOH\] done
clear
View Solution play_arrow
-
question_answer102)
Which one of the following reducing agents is likely to be the most effective in bringing about the following change \[R-\overset{O\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{\overset{||\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{C\,N{{H}_{2}}\to RC{{H}_{2}}N{{H}_{2}}}}\,}}\,\] [AMU 2002]
A)
\[{{H}_{2}}-Ni\] done
clear
B)
\[NaB{{H}_{4}}\] done
clear
C)
\[LiAl{{H}_{4}}\] done
clear
D)
Na-alcohol done
clear
View Solution play_arrow
-
question_answer103)
During acetylation of amines what is replaced by acetyl groups [UPSEAT 2002]
A)
Hydrogen atom attached to nitrogen atom done
clear
B)
One or more hydrogen atoms attached to carbon atom done
clear
C)
One or more hydrogen atoms attached to nitrogen atom done
clear
D)
Hydrogen atoms attached to either carbon atom or nitrogen atom done
clear
View Solution play_arrow
-
question_answer104)
Hydrolysis of acetonitrile in acidic medium produces [CPMT 2003; RPMT 2003]
A)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}NC\] done
clear
D)
\[C{{H}_{3}}COOC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer105)
Which has a pyramidal structure [UPSEAT 2003]
A)
Trimethylamine done
clear
B)
Methanol done
clear
C)
Acetylene done
clear
D)
Water done
clear
View Solution play_arrow
-
question_answer106)
Ethyl amine on acetylation gives [BHU 2002; BVP 2003]
A)
N-ethyl acetamide done
clear
B)
Acetamide done
clear
C)
Methyl acetamide done
clear
D)
None done
clear
View Solution play_arrow
-
question_answer107)
The refluxing of \[{{(C{{H}_{3}})}_{2}}NCOC{{H}_{3}}\]with acid gives [BHU 2002; BVP 2003]
A)
\[{{(C{{H}_{3}})}_{2}}NH+C{{H}_{3}}COOH\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NCOOH+C{{H}_{4}}\] done
clear
C)
\[2C{{H}_{3}}OH+C{{H}_{3}}CON{{H}_{2}}\] done
clear
D)
\[2C{{H}_{3}}N{{H}_{2}}+C{{H}_{3}}COOH\] done
clear
View Solution play_arrow
-
question_answer108)
p-chloro aniline and anilinium hydrogen chloride can be distinguished by [UPSEAT 2003]
A)
Sandmaeyer reaction done
clear
B)
Carbyl amine reaction done
clear
C)
Hinsberg?s reaction done
clear
D)
\[AgN{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer109)
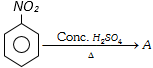
Product ?A? in above reaction is [RPMT 2003]
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer110)
Product obtained by electrolytic reduction of nitrobenzene in presence of \[{{H}_{2}}S{{O}_{4}}\] is [RPMT 2003]
A)
o-amino phenol done
clear
B)
m-amino phenol done
clear
C)
p-amino phenol done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer111)
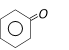
\[+N{{H}_{2}}OH\to A\underset{{{H}_{3}}{{O}^{+}}}{\mathop{\xrightarrow{PPA}}}\,B\]. The product ?B? is [RPMT 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer112)
Identify the product Z in the following reaction \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow{{{(AC)}_{2}}O}\]\[X\xrightarrow{B{{r}_{2}}/CC{{l}_{4}}}Y\xrightarrow{HOH}Z\] [Kerala (Med.) 2003]
A)
p-Bromoaniline done
clear
B)
p -Bromoacetophenone done
clear
C)
o-Bromoacetophenone done
clear
D)
o-Bromoacetonilide done
clear
View Solution play_arrow
-
question_answer113)
Benzaldehyde condenses with N, N-dimethylaniline in presence of anhydrous \[ZnC{{l}_{2}}\] to give [Kerala (Med.) 2003]
A)
Michler?s ketone done
clear
B)
Azo dye done
clear
C)
Malachite green done
clear
D)
Buffer yellow done
clear
View Solution play_arrow
-
question_answer114)
The correct order of reactivity towards the electrophilic substitution of the compounds aniline (I) benzene (II) and nitrobenzene (III) is [CBSE PMT 2003]
A)
I > II > III done
clear
B)
III > II > I done
clear
C)
II > III > I done
clear
D)
I < II > III done
clear
View Solution play_arrow
-
question_answer115)
The final product C, obtained in this reaction
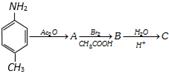
Would be [CBSE PMT 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer116)
The correct order of increasing basic nature for the bases \[N{{H}_{3}},\,C{{H}_{3}}N{{H}_{2}}\] and \[{{(C{{H}_{3}})}_{2}}NH\] is [AIEEE 2003]
A)
\[C{{H}_{3}}N{{H}_{2}}<N{{H}_{3}}<{{(C{{H}_{3}})}_{2}}NH\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH<N{{H}_{3}}<C{{H}_{3}}N{{H}_{2}}\] done
clear
C)
\[N{{H}_{3}}<C{{H}_{3}}N{{H}_{2}}<{{(C{{H}_{3}})}_{2}}NH\] done
clear
D)
\[C{{H}_{3}}N{{H}_{2}}<{{(C{{H}_{3}})}_{2}}NH<N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer117)
Nitrobenzene gives N-phenylhydroxylamine by [AIIMS 2003]
A)
\[Sn/HCl\] done
clear
B)
\[{{H}_{2}}/Pd-C\] done
clear
C)
\[Zn/NaOH\] done
clear
D)
\[Zn/N{{H}_{4}}Cl\] done
clear
View Solution play_arrow
-
question_answer118)
Among the following the weakest base is [AIIMS 2003]
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\] done
clear
C)
\[{{O}_{2}}N\,C{{H}_{2}}N{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}NH\,CHO\] done
clear
View Solution play_arrow
-
question_answer119)
The correct order of basicity of amines in water is : [Pb. CET 2003]
A)
\[{{(C{{H}_{3}})}_{2}}NH>{{(C{{H}_{3}})}_{3}}N>C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}N{{H}_{2}}>{{(C{{H}_{3}})}_{2}}NH>{{(C{{H}_{3}})}_{3}}N\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N>{{(C{{H}_{3}})}_{2}}NH>C{{H}_{3}}N{{H}_{2}}\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}N>C{{H}_{3}}N{{H}_{2}}>{{(C{{H}_{3}})}_{2}}NH\] done
clear
View Solution play_arrow
-
question_answer120)
Complete the following reaction : [MHCET 2004] \[R\,\,N{{H}_{2}}+{{H}_{2}}S{{O}_{4}}\to \]
A)
\[{{[R\,\,N{{H}_{3}}]}^{+}}HSO_{4}^{-}\] done
clear
B)
\[[R\,\,N{{H}_{3}}]_{2}^{+}SO_{4}^{2-}\] done
clear
C)
\[R\,\,N{{H}_{2}}.{{H}_{2}}S{{O}_{4}}\] done
clear
D)
No reaction done
clear
View Solution play_arrow
-
question_answer121)
Which of the following compound reacts with chloroform and a base to form phenyl isocyanide? [MHCET 2003]
A)
Phenol done
clear
B)
Aniline done
clear
C)
Benzene done
clear
D)
Nitro benzene done
clear
View Solution play_arrow
-
question_answer122)
Which one doesn't liberate \[N{{H}_{3}}\] when undergoes hydrolysis [Orissa JEE 2005]
A)
Acetanilide done
clear
B)
Acetonitrile done
clear
C)
Acetamide done
clear
D)
Phenyl isocyanide done
clear
View Solution play_arrow
-
question_answer123)
A nitrogen containing organic compound gave an oily liquid on heating with bromine and potassium hydroxide solution. On shaking the product with acetic anhydride, an antipyretic drug was obtained. The reactions indicate that the starting compound is [KCET 2004]
A)
Aniline done
clear
B)
Benzamide done
clear
C)
Acetamide done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer124)
Benzamide on reaction with \[POC{{l}_{3}}\] gives [IIT-JEE 2004]
A)
Aniline done
clear
B)
Chlorobenzene done
clear
C)
Benzyl amine done
clear
D)
Benzonitrile done
clear
View Solution play_arrow
-
question_answer125)
Among the following which one does not act as an intermediate in Hofmann rearrangement [AIIMS 2005]
A)
\[RNCO\] done
clear
B)
\[RCO\overset{\,.\ .}{\mathop{N}}\,\] done
clear
C)
\[RCO\overset{\,.\ .}{\mathop{N}}\,HBr\] done
clear
D)
\[RNC\] done
clear
View Solution play_arrow
-
question_answer126)
Aniline reacts with which of these to form Schiff base [AFMC 2004]
A)
Acetic acid done
clear
B)
Benzaldehyde done
clear
C)
Acetone done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer127)
Which of the following does not reduce Tollen?s reagent [Kerala PMT 2004]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}NHOH\] done
clear
C)
\[HCOOH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}\] done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer128)
Which one of the following compound is most basic? [UPSEAT 2004]
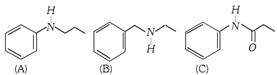
A)
A done
clear
B)
B done
clear
C)
C done
clear
D)
All are equally basic done
clear
View Solution play_arrow
-
question_answer129)
Which one of the following methods is neither meant for the synthesis nor for separation of amines [AIEEE 2005]
A)
Hinsberg method done
clear
B)
Hofmann method done
clear
C)
Wurtz reaction done
clear
D)
Curtius reaction done
clear
View Solution play_arrow
-
question_answer130)
Aniline in a set of reactions yielded a product D.
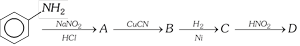
The structure of product D would be [CBSE PMT 2005]
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}NH\,C{{H}_{2}}C{{H}_{3}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}NHOH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer131)
Electrolytic reduction of nitrobenzene in weakly acidic medium gives [CBSE PMT 2005]
A)
Aniline done
clear
B)
Nitrosobenzene done
clear
C)
N-Phenylhydroxylamine done
clear
D)
p-Hydroxylaniline done
clear
View Solution play_arrow
-
question_answer132)
Among the following compounds \[{{C}_{3}}{{H}_{7}}N{{H}_{2}},\] \[N{{H}_{3}},C{{H}_{3}}N{{H}_{2}},\]\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] and \[{{C}_{6}}{{H}_{5}}N{{H}_{2}},\] the least basic compound is
A)
\[{{C}_{3}}{{H}_{7}}N{{H}_{2}}\] done
clear
B)
\[N{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
E)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer133)
The reduction of which of the following compound would yield secondary amine? [DCE 2004]
A)
Alkyl nitrite done
clear
B)
Carbylamine done
clear
C)
Primary amine done
clear
D)
Secondary nitro compound done
clear
View Solution play_arrow
-
question_answer134)
Azo dye is prepared by the coupling of phenol and : [Pb. CET 2000]
A)
Diazonium chloride done
clear
B)
o-nitro aniline done
clear
C)
Benzoic acid done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
-
question_answer135)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow{NaN{{O}_{2}}HCl}X\xrightarrow{C{{u}_{2}}{{(CN)}_{2}}}Y\xrightarrow{{{H}_{2}}O/{{H}^{+}}}Z\] Z is identified as : [Pb. PMT 2004]
A)
\[{{C}_{6}}{{H}_{5}}-NH-C{{H}_{3}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}-COOH\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-N{{H}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-COOH\] done
clear
View Solution play_arrow
-
question_answer136)
When acetamide reacts with \[B{{r}_{2}}\] and caustic soda, then we get : [CPMT 2004]
A)
Acetic acid done
clear
B)
Bromoacetic acid done
clear
C)
Methyl amine done
clear
D)
Ethyl amine done
clear
View Solution play_arrow
-
question_answer137)
In the reaction \[C{{H}_{3}}CN+2H\underset{\text{Ether}}{\mathop{\xrightarrow{HCl}}}\,X\underset{{{H}_{2}}O}{\mathop{\xrightarrow{\text{Boiling}}}}\,Y;\] the term Y is: [BHU 2004]
A)
Acetone done
clear
B)
Ethyl amine done
clear
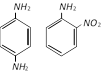
C)
Acetaldehyde done
clear
D)
Dimethyl amine done
clear
View Solution play_arrow
-
question_answer138)
Reaction of cyclohexanone with dimethylamine in the presence of catalytic amount of an acid forms a compounds if water during the reaction is continuously removed. The compound formed is generally known as [AIEEE 2005]
A)
A Schiff?s base done
clear
B)
An enamine done
clear
C)
An imine done
clear
D)
An amine done
clear
View Solution play_arrow
-
question_answer139)
\[R-NH-COH\underset{\text{pyridine}}{\mathop{\xrightarrow{POC{{l}_{3}}}}}\,\]product In the given reaction what will be the product [BHU 2005]
A)
R ? N = C = O done
clear
B)
\[R-\overset{+}{\mathop{N}}\,\equiv {{C}^{-}}\] done
clear
C)
\[R-C\equiv N\] done
clear
D)
None of these. done
clear
View Solution play_arrow
-
question_answer140)
Which of the following is secondary pollulant. [BHU 2005]
A)
CO2 done
clear
B)
N2O done
clear
C)
PAN done
clear
D)
SO2 done
clear
View Solution play_arrow
-
question_answer141)
Nitration of aniline also gives m-nitro aniline, in strong acidic medium because [Kerala CET 2005]
A)
In electrophilic substitution reaction amino group is meta directive done
clear
B)
Inspite of substituents nitro group always goes to m- position done
clear
C)
In strong acidic medium, nitration of aniline is a nucleophic substitution reaction done
clear
D)
In strong acidic medium aniline present as anilinium ion done
clear
E)
Strong acid, gives nitrate anion, which attacks at m-position done
clear
View Solution play_arrow
-
question_answer142)
Identify the product in following order 3,4,5-Tribromoaniline\[\underset{\text{(ii) }{{H}_{3}}P{{O}_{2}}}{\mathop{\xrightarrow{\text{(i) diazotization}}}}\,\,?\] [Kerala CET 2005]
A)
3, 4,5 ?Tribromobenzene done
clear
B)
1, 2, 3 ? Tribromobenzene done
clear
C)
2, 4, 6 ? Tribromobenzene done
clear
D)
3, 4, 5 ? Tribromo nitro benzene done
clear
E)
3, 4, 5 ? Tribromo phenol done
clear
View Solution play_arrow
-
question_answer143)
The correct order of basicity in amines (i) \[{{C}_{4}}{{H}_{5}}N{{H}_{2}}\] (ii) \[C{{H}_{3}}N{{H}_{2}}\] (iii) \[{{(C{{H}_{3}})}_{2}}NH\] (iv) \[{{(C{{H}_{3}})}_{3}}N\] [Kerala CET 2005]
A)
(i) < (iv) < (ii) < (iii) done
clear
B)
(iv) < (iii) < (ii) < (i) done
clear
C)
(i) < (ii) < (iii) < (iv) done
clear
D)
(ii) < (iii) < (iv) < (i) done
clear
E)
(iv) < (iii) < (ii) < (i) done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
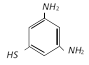 done
clear
done
clear
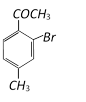
![]() with bromine gives [Kerala (Med.) 2002]
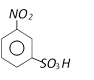
with bromine gives [Kerala (Med.) 2002] 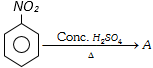 Product ?A? in above reaction is [RPMT 2003]
Product ?A? in above reaction is [RPMT 2003] 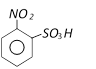 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
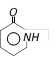 done
clear
done
clear
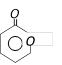 done
clear
done
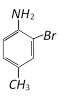
clear
 Would be [CBSE PMT 2003]
Would be [CBSE PMT 2003] 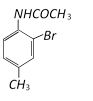 done
clear
done
clear
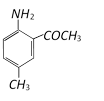 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

