-
question_answer1)
Mark the correct statement in a reversible reaction [CPMT 1974; EAMCET 1978, 79; MP PMT 1993]
A)
The catalyst catalyses the forward reaction done
clear
B)
The catalyst catalyses the backward reaction done
clear
C)
The catalyst influences the direct and the reverse reaction to the same extent done
clear
D)
The catalyst increases the rate of forward reaction and decreases the rate of backward reaction done
clear
View Solution play_arrow
-
question_answer2)
A catalyst is used [CPMT 1989]
A)
Only for increasing the velocity of the reaction done
clear
B)
For altering the velocity of the reaction done
clear
C)
Only for decreasing the velocity of the reaction done
clear
D)
All A, B and C are correct done
clear
View Solution play_arrow
-
question_answer3)
A catalyst is a substance which [NCERT 1981; CPMT 1996]
A)
Alters the equilibrium in a reaction done
clear
B)
Is always in the same phase as the reactants done
clear
C)
Participates in the reaction and provides easier pathway for the same done
clear
D)
Does not participate in the reaction but speeds it up done
clear
View Solution play_arrow
-
question_answer4)
In Haber's process for the manufacture of ammonia [AMU 1984; CPMT 1974, 90]
A)
Finely divided iron is used as catalyst done
clear
B)
Finely divided molybdenum is used as catalyst done
clear
C)
Finely divided nickel is used as catalyst done
clear
D)
No catalyst is necessary done
clear
View Solution play_arrow
-
question_answer5)
When \[KCl{{O}_{3}}\] is heated, it decomposes into \[KCl+{{O}_{2}}\]. If some \[Mn{{O}_{2}}\] is added, the reaction goes much faster because [CPMT 1971,76,80,94]
A)
\[Mn{{O}_{2}}\] decomposes to give \[{{O}_{2}}\] done
clear
B)
\[Mn{{O}_{2}}\] provides heat by reacting done
clear
C)
Better contact is provided by \[Mn{{O}_{2}}\] done
clear
D)
\[Mn{{O}_{2}}\] acts as a catalyst done
clear
View Solution play_arrow
-
question_answer6)
In the reaction \[2S{{O}_{2}}+{{O}_{2}}\underset{A{{s}_{2}}{{O}_{3}}}{\mathop{\xrightarrow{Pt}}}\,2S{{O}_{3}}\], \[A{{s}_{2}}{{O}_{3}}\] acts as a [MP PET 1995]
A)
Autocatalyst done
clear
B)
Poison done
clear
C)
Promotor done
clear
D)
Positive catalyst done
clear
View Solution play_arrow
-
question_answer7)
Reactions in Zeolite catalysts depend on [BHU 2000]
A)
Pores done
clear
B)
Apertures done
clear
C)
Size of cavities done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer8)
What is the role of a catalyst in a catalysed reaction [MP PMT 1996; Pb. PMT 2000;UPSEAT 2001,02]
A)
Lowers the activation energy done
clear
B)
Increases the activation energy done
clear
C)
Affects the free energy change done
clear
D)
Affects the enthalpy change done
clear
View Solution play_arrow
-
question_answer9)
The catalyst used in the lead chamber process of sulphuric acid manufacture is [CPMT 1977]
A)
Platinum done
clear
B)
Oxide of nitrogen done
clear
C)
Nickel done
clear
D)
Vanadium compounds done
clear
View Solution play_arrow
-
question_answer10)
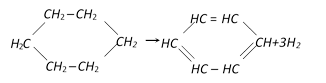
In the following reaction the catalyst used is [AMU (Engg.) 1999]
A)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
B)
\[C{{r}_{2}}{{O}_{3}}\] done
clear
C)
\[C{{r}_{2}}{{O}_{3}}\] and \[A{{l}_{2}}{{O}_{3}}\] done
clear
D)
Zn dust done
clear
View Solution play_arrow
-
question_answer11)
Enzymes with two sites are called [AIIMS 2002]
A)
Apoenzyme done
clear
B)
Holoenzyme done
clear
C)
Allosteric enzyme done
clear
D)
Conjugate enzyme done
clear
View Solution play_arrow
-
question_answer12)
Which of the following types of metals make the most efficient catalysts [DPMT 1985]
A)
Alkali metals done
clear
B)
Transition metals done
clear
C)
Alkaline?earth metals done
clear
D)
Radioactive metals done
clear
View Solution play_arrow
-
question_answer13)
An example of autocatalytic reaction is [NCERT 1983]
A)
The decomposition of nitroglycerine done
clear
B)
Thermal decomposition of \[KCl{{O}_{3}}\] and \[Mn{{O}_{2}}\] mixture done
clear
C)
Break down of \[_{6}{{C}^{14}}\] done
clear
D)
Hydrogenation of vegetable oil using nickel catalysts done
clear
View Solution play_arrow
-
question_answer14)
In the case of auto catalysis [KCET (Med.) 2002]
A)
Solvent catalyses done
clear
B)
Product catalyses done
clear
C)
Reactant catalyses done
clear
D)
Heat produced in the reaction catalyses done
clear
View Solution play_arrow
-
question_answer15)
In a reversible reaction, a catalyst will affect the rate of [KCET (Med.) 2002]
A)
Forward reaction done
clear
B)
Reverse reaction done
clear
C)
Forward and reverse reaction done
clear
D)
Neither A nor B done
clear
View Solution play_arrow
-
question_answer16)
The role of a catalyst in a reversible reaction is to [KCET (Med.) 2001]
A)
Increase the rate of forward reaction done
clear
B)
Decrease the rate of backward reaction done
clear
C)
Alter the equilibrium constant of the reaction done
clear
D)
Allow the equilibrium to be achieved quickly done
clear
View Solution play_arrow
-
question_answer17)
The catalyst used in the contact process for manufacturing of sulphuric acid is [MP PMT 1987]
A)
Copper done
clear
B)
Iron/aluminum oxide done
clear
C)
Vanadium pent oxide done
clear
D)
Platinized asbestos done
clear
View Solution play_arrow
-
question_answer18)
For the functioning of enzymes which of the following statements is not correct [MP PMT 2000]
A)
An optimum temperature is needed done
clear
B)
An optimum pH is needed done
clear
C)
They are substrate specific done
clear
D)
They always increase activation energy done
clear
View Solution play_arrow
-
question_answer19)
When a catalyst is added to a system the [JIPMER 2000]
A)
Value of equilibrium constant is decreased done
clear
B)
The rate of forward reaction is increased and that of backward reaction is decreased done
clear
C)
Equilibrium concentrations are unchanged done
clear
D)
Equilibrium concentrations are increased done
clear
View Solution play_arrow
-
question_answer20)
A catalyst can affect reversible reaction by [CPMT 2002]
A)
Changing equilibrium done
clear
B)
Slowing forward reaction done
clear
C)
Attaining equilibrium in both direction done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer21)
\[\underset{\text{Sucrose}}{\mathop{{{C}_{12}}{{H}_{22}}{{O}_{11}}}}\,+{{H}_{2}}O\xrightarrow{dil.\,{{H}_{2}}S{{O}_{4}}}\underset{\text{Fructose}}{\mathop{{{C}_{6}}{{H}_{12}}{{O}_{6\,}}(aq)}}\,+\underset{\text{Glucose}}{\mathop{{{C}_{6}}{{H}_{12}}{{O}_{6}}(aq)}}\,\] In this reaction, dilute \[{{H}_{2}}S{{O}_{4}}\] is called [AFMC 1997]
A)
Homogeneous catalysis done
clear
B)
Homogeneous catalyst done
clear
C)
Heterogeneous catalysis done
clear
D)
Heterogeneous catalyst done
clear
View Solution play_arrow
-
question_answer22)
Which one of the following statement is wrong in case of enzyme catalysis [MP PMT 1985, 2001]
A)
Enzymes work best at an optimum temperature done
clear
B)
Enzymes work at an optimum \[pH\] done
clear
C)
Enzymes are highly specific for substances done
clear
D)
An enzyme raises activation energy done
clear
View Solution play_arrow
-
question_answer23)
Which of the following catalyses the conversion of glucose into ethanol [CPMT 1983, 84; CBSE PMT 1989; KCET 1993]
A)
Zymase done
clear
B)
Invertase done
clear
C)
Maltase done
clear
D)
Diastase done
clear
View Solution play_arrow
-
question_answer24)
Which of the following is used as a catalyst in the manufacture of toluene from benzene with \[C{{H}_{3}}Cl\] [CPMT 1985]
A)
\[Ni\] done
clear
B)
Anhydrous \[AlC{{l}_{3}}\] done
clear
C)
\[Pd\] done
clear
D)
\[Pt\] done
clear
View Solution play_arrow
-
question_answer25)
Hydrolysis of ethyl acetate is catalysed by aqueous [MP PMT 2002]
A)
\[N{{a}_{2}}S{{O}_{4}}\] done
clear
B)
\[{{K}_{2}}S{{O}_{4}}\] done
clear
C)
\[{{H}_{2}}S{{O}_{4}}\] done
clear
D)
\[BaS{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer26)
Which of the following statements about a catalyst is true [AIIMS 1996]
A)
It lowers the energy of activation done
clear
B)
The catalyst altered during the reaction is regenerated done
clear
C)
It does not alter the equilibrium done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer27)
Which of the following statements is true for a catalyst
A)
It increases the energy of the reactants done
clear
B)
It decreases the energy of the products done
clear
C)
It decreases the energy of the reactants done
clear
D)
It does not change the enthalpy of the reactants done
clear
View Solution play_arrow
-
question_answer28)
Which is not a characteristic of a catalyst [AFMC 1992]
A)
It changes the equilibrium constant done
clear
B)
It alters the reaction path done
clear
C)
It increases the rate of reaction done
clear
D)
It increases the average K.E. of the molecules done
clear
View Solution play_arrow
-
question_answer29)
Which one of the following statements is correct in reversible reaction. A catalyst [MP PET 1994; EAMCET 1987]
A)
Increases the rate of forward reaction done
clear
B)
Decreases the rate of forward reaction done
clear
C)
Increases the rate of backward and forward reactions done
clear
D)
Alters the equilibrium constant of the reaction done
clear
View Solution play_arrow
-
question_answer30)
A catalyst [MNR 1987; UPSEAT 2002]
A)
Increases the free energy change in the reaction done
clear
B)
Decreases the free energy change in the reaction done
clear
C)
Does not increase or decrease the free energy change in the reaction done
clear
D)
Can either increase or decrease the free energy change depending on what catalyst we use done
clear
View Solution play_arrow
-
question_answer31)
Which one of the following changes when catalyst is used in a reaction
A)
Heat of reaction done
clear
B)
Product of reaction done
clear
C)
Equilibrium constant done
clear
D)
Activation energy done
clear
View Solution play_arrow
-
question_answer32)
In the reversible reaction a catalyst is the substance which [CBSE PMT 1992]
A)
Increases the rate of the forward reaction done
clear
B)
Decreases the value of enthalpy change in the reaction done
clear
C)
Reduces the time required for reaching the equilibrium state in the reaction done
clear
D)
Decreases the rate of the reverse reaction done
clear
View Solution play_arrow
-
question_answer33)
In the titration between oxalic acid and acidified potassium permanganate, the manganous salt formed catalyses the reaction. The manganous salt is [KCET 1992]
A)
A promoter done
clear
B)
A positive catalyst done
clear
C)
An autocatalyst done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer34)
Which one of the following statements is incorrect in the case of heterogeneous catalysis [CPMT 1990]
A)
The catalyst lowers the energy of activation done
clear
B)
The catalyst actually forms a compound with the reactant done
clear
C)
The surface of the catalyst plays a very important role done
clear
D)
There is no change in the energy of activation done
clear
View Solution play_arrow
-
question_answer35)
Regarding criteria of catalysis which one of the following statements is not true [CPMT 1990]
A)
The catalyst is unchanged chemically at the end of the reaction done
clear
B)
A small quantity of catalyst is often sufficient to bring about a considerable amount of reaction done
clear
C)
In a reversible reaction the catalyst alters the equilibrium position done
clear
D)
The catalyst accelerates the reaction done
clear
View Solution play_arrow
-
question_answer36)
Which of the following reaction is catalysed by enzyme maltase [MP PMT 2003]
A)
Starch ® maltose done
clear
B)
Maltose ® glucose done
clear
C)
Lactose ® maltose done
clear
D)
Maltose ® glucose + fructose done
clear
View Solution play_arrow
-
question_answer37)
The efficiency of an enzyme in catalysing a reaction is due to its capacity [NCERT 1982]
A)
To form a strong enzyme-substrate complex done
clear
B)
To decrease the bond energies of substrate molecule done
clear
C)
To change the shape of the substrate molecule done
clear
D)
To lower the activation energy of the reaction done
clear
View Solution play_arrow
-
question_answer38)
A catalyst in a chemical reaction [BHU 1998]
A)
Does not initiate a reaction done
clear
B)
Increases the activation energy of the reaction done
clear
C)
Changes the equilibrium constant of a reaction done
clear
D)
Does not change the rate of the reaction done
clear
View Solution play_arrow
-
question_answer39)
Platinized asbestos is used as a catalyst in the manufacture of \[{{H}_{2}}S{{O}_{4}}\]. It is an example of [CPMT 1975]
A)
Heterogeneous catalyst done
clear
B)
Autocatalyst done
clear
C)
Homogenous catalyst done
clear
D)
Induced catalyst done
clear
View Solution play_arrow
-
question_answer40)
Catalyst used in hydrogenation of oils is [CPMT 1975; MNR 1986; DPMT 1982, 85;BHU 1973, 87; EAMCET 1987; AFMC 1993; CET Pune 1998]
A)
\[Pt\] done
clear
B)
\[Mo\] done
clear
C)
\[Fe\] done
clear
D)
\[Ni\] done
clear
View Solution play_arrow
-
question_answer41)
Addition of catalyst in a system [MP PMT 1992]
A)
Increases equilibrium concentrations done
clear
B)
No effect on equilibrium concentrations done
clear
C)
Decreases equilibrium concentrations done
clear
D)
Increases rate of forward reaction and decreases rate of backward reaction done
clear
View Solution play_arrow
-
question_answer42)
In which of the following processes, platinum is used as a catalyst [NCERT 1978, 79]
A)
Oxidation of ammonia to form nitric acid done
clear
B)
Hardening of oils done
clear
C)
Production of synthetic rubber done
clear
D)
Synthesis of methanol done
clear
View Solution play_arrow
-
question_answer43)
Enzymes are [CPMT 1974, 81]
A)
Micro-organisms done
clear
B)
Proteins done
clear
C)
Inorganic compounds done
clear
D)
Moulds done
clear
View Solution play_arrow
-
question_answer44)
Protons accelerate the hydrolysis of esters. This is an example of [MP PMT 1987]
A)
A heterogeneous catalysis done
clear
B)
An acid-base catalysis done
clear
C)
A promoter done
clear
D)
A negative catalyst done
clear
View Solution play_arrow
-
question_answer45)
Which of the following processes does not involve a catalyst [KCET 1991; AIIMS 1996]
A)
Haber's process done
clear
B)
Thermite process done
clear
C)
Ostwald process done
clear
D)
Contact process done
clear
View Solution play_arrow
-
question_answer46)
Which of the statement is wrong among the following [AFMC 1993]
A)
Haber's process of \[N{{H}_{3}}\] requires iron as catalyst done
clear
B)
Friedel?Craft's reaction uses anhydrous \[AlC{{l}_{3}}\] done
clear
C)
Hydrogenation of oils uses iron as catalyst done
clear
D)
Oxidation of \[S{{O}_{2}}\] to \[S{{O}_{3}}\] requires \[{{V}_{2}}{{O}_{5}}\] done
clear
View Solution play_arrow
-
question_answer47)
A catalyst is a substance which
A)
Increases the rate of a reaction done
clear
B)
Increases the amount of the products formed in a reaction done
clear
C)
Decreases the temperature required for the reaction done
clear
D)
Alters the speed of the reaction remaining unchanged chemically at the end of the reaction done
clear
View Solution play_arrow
-
question_answer48)
In the Ostwald's process for the manufacture of \[HN{{O}_{3}}\], the catalyst used is [AMU 1982, 83; MP PET 1999]
A)
\[Mo\] done
clear
B)
\[Fe\] done
clear
C)
\[Ni\] done
clear
D)
\[Pt\] done
clear
View Solution play_arrow
-
question_answer49)
A biological catalyst is essentially [NCERT 1978; AFMC 1998]
A)
An amino acid done
clear
B)
A carbohydrate done
clear
C)
The nitrogen molecule done
clear
D)
An enzyme done
clear
View Solution play_arrow
-
question_answer50)
A catalyst added to a reaction mixture
A)
Increases the equilibrium constant done
clear
B)
Decreases the equilibrium constant done
clear
C)
Does not change the equilibrium constant done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer51)
The components of Zigler Natta catalyst, used in the polymerisation of propylene, are [MP PMT 2003]
A)
\[TiC{{l}_{3}}+Al{{({{C}_{2}}{{H}_{5}})}_{3}}\] done
clear
B)
\[TiC{{l}_{4}}+Al{{({{C}_{2}}{{H}_{5}})}_{3}}\] done
clear
C)
\[Ti{{({{C}_{2}}{{H}_{5}})}_{3}}+AlC{{l}_{3}}\] done
clear
D)
\[Ti{{({{C}_{2}}{{H}_{5}})}_{4}}+AlC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer52)
Which of the following statements regarding catalyst is not true [CPMT 1983, 84; MNR 1993; KCET 1999]
A)
A catalyst remains unchanged in composition and quantity at the end of the reaction done
clear
B)
A catalyst can initiate a reaction done
clear
C)
A catalyst does not alter the equilibrium in a reversible reaction done
clear
D)
Catalyst are sometimes very specific in respect of reaction done
clear
View Solution play_arrow
-
question_answer53)
The enzyme ptylin used for the digestion of food is present in [CPMT 1981]
A)
Saliva done
clear
B)
Blood done
clear
C)
Intestine done
clear
D)
Adrenal glands done
clear
View Solution play_arrow
-
question_answer54)
Amongst the following chemical reactions, the one representing homogeneous catalysis is [MP PMT 1999]
A)
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\xrightarrow{Fe}2N{{H}_{3}}(g)\] done
clear
B)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\xrightarrow{2NO}2S{{O}_{3}}(g)+2NO(g)\] done
clear
C)
\[CO(g)+3{{H}_{2}}(g)\xrightarrow{Ni}C{{H}_{4}}(g)+{{H}_{2}}O\] done
clear
D)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\xrightarrow{{{V}_{2}}{{O}_{5}}}2S{{O}_{3}}(g)\] done
clear
View Solution play_arrow
-
question_answer55)
Platinised asbestos helps in the formation of \[S{{O}_{3}}\] form \[S{{O}_{2}}\] and \[{{O}_{2}}\]. But, if even a small amount of \[A{{s}_{2}}{{O}_{3}}\] is present the platinised asbestos does not help in the formation of \[S{{O}_{3}}\]. \[A{{s}_{2}}{{O}_{3}}\] acts here as [MP PMT 1997]
A)
A positive catalyst done
clear
B)
A negative catalyst done
clear
C)
An auto catalyst done
clear
D)
A poison done
clear
View Solution play_arrow
-
question_answer56)
Which of the following statements is wrong
A)
Catalysts can aid a rapid reaching of the equilibrium position, but do not change the position of the equilibrium done
clear
B)
Homogeneous catalysis generally involves an equilibrium reaction between at least one of the reactants and the catalyst done
clear
C)
Heterogeneous catalysis involves chemisorption on the surface of the catalyst done
clear
D)
Positive catalysts raise the energy of activation of the reaction they catalyse done
clear
View Solution play_arrow
-
question_answer57)
Which one is false in the following statement [MP PET 1997]
A)
A catalyst is specific in its action done
clear
B)
A very small amount of the catalyst alters the rate of a reaction done
clear
C)
The number of free valencies on the surface of the catalyst increases on subdivision done
clear
D)
\[Ni\] is used as catalyst in the manufacture of ammonia done
clear
View Solution play_arrow
-
question_answer58)
In the redox reaction \[2MnO_{4}^{-}+5{{C}_{2}}O_{4}^{2-}+16{{H}^{+}}\]⇌\[2M{{n}^{2+}}+10C{{O}_{2}}+8{{H}_{2}}O\] The ion acting as autocatalyst is [MP PMT 1986,94]
A)
\[MnO_{4}^{-}\] done
clear
B)
\[{{C}_{2}}O_{4}^{2-}\] done
clear
C)
\[{{H}^{+}}\] done
clear
D)
\[M{{n}^{2+}}\] done
clear
View Solution play_arrow
-
question_answer59)
In a homogeneous catalysis
A)
The catalyst and the reactants should be gases done
clear
B)
The catalyst and the reactants should form a single phase done
clear
C)
Catalyst and the reactants are all solids done
clear
D)
The catalyst and the reactions are all liquids done
clear
View Solution play_arrow
-
question_answer60)
Which of the following statements is incorrect [CPMT 1985]
A)
Enzymes are in colloidal state done
clear
B)
Enzymes are catalysts done
clear
C)
Enzymes can catalyse any reaction done
clear
D)
Urease is an enzyme done
clear
View Solution play_arrow
-
question_answer61)
Enzymes are [BHU 1982]
A)
Substances made by chemists to activate washing powder done
clear
B)
Very active vegetable catalysts done
clear
C)
Catalysts found in organism done
clear
D)
Synthetic catalysts done
clear
View Solution play_arrow
-
question_answer62)
Catalyst used in the oxidation of \[S{{O}_{2}}\to S{{O}_{3}}\] [AIIMS 1996]
A)
Nickel done
clear
B)
\[ZnO.C{{r}_{2}}{{O}_{3}}\] done
clear
C)
\[{{V}_{2}}{{O}_{5}}\] done
clear
D)
Iron done
clear
View Solution play_arrow
-
question_answer63)
Which requires catalyst [AFMC 1987; MP PET 1999]
A)
\[S+{{O}_{2}}\to S{{O}_{2}}\] done
clear
B)
\[2S{{O}_{2}}+{{O}_{2}}\to 2S{{O}_{3}}\] done
clear
C)
\[C+{{O}_{2}}\to C{{O}_{2}}\] done
clear
D)
All done
clear
View Solution play_arrow
-
question_answer64)
The process which is catalysed by one of the products is called [MP PET 1999; AIIMS 2000; J & K 2005]
A)
Acid-base catalysis done
clear
B)
Autocatalysis done
clear
C)
Negative catalysis done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer65)
Adam's catalyst is [Pb.CET 2001]
A)
Platinum done
clear
B)
Iron done
clear
C)
Molybdenum done
clear
D)
Nickel done
clear
View Solution play_arrow
-
question_answer66)
A catalyst remains unchanged at the end of the reaction regarding [MP PET 1995]
A)
Mass done
clear
B)
Physical state done
clear
C)
Physical state and chemical composition done
clear
D)
Mass and chemical composition done
clear
View Solution play_arrow
-
question_answer67)
Wilhem Ostwald redefined the action of [Kerala (Med.) 2002]
A)
Anamers done
clear
B)
Isomers done
clear
C)
Catalyst done
clear
D)
Geometry of monomers done
clear
View Solution play_arrow
-
question_answer68)
In a reversible reaction, a catalyst used
A)
Increases the speed of the forward reaction done
clear
B)
Decreases the speed of the backward reaction done
clear
C)
Does not alter the final state of equilibrium done
clear
D)
Increases the amount of the products formed done
clear
View Solution play_arrow
-
question_answer69)
Enzyme activity is maximum at [KCET 1989]
A)
300 K done
clear
B)
310 K done
clear
C)
320 K done
clear
D)
330 K done
clear
View Solution play_arrow
-
question_answer70)
A catalyst is used to [Pb.CET 2000]
A)
Increase the product done
clear
B)
Increase or decrease the rate of reaction done
clear
C)
Increase or decrease the products done
clear
D)
Decrease the products done
clear
View Solution play_arrow
-
question_answer71)
The transition metal used as a catalyst is [Pb. PMT 2004]
A)
Nickel done
clear
B)
Platinum done
clear
C)
Cobalt done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer72)
Which of the following is true about catalyst [Pb.CET 2000]
A)
It initiates reaction done
clear
B)
It changes equilibrium point done
clear
C)
It increase average kinetic energy done
clear
D)
It accelerates the rate of reaction done
clear
View Solution play_arrow
-
question_answer73)
Which of the following types of metals form the most efficient catalysts [KCET 2005]
A)
Alkali metals done
clear
B)
Alkaline earth metals done
clear
C)
Transition metals done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer74)
Formation of ammonia from \[{{H}_{2}}\] and \[{{N}_{2}}\] by Haber's process using \[Fe\] is an example of [J & K 2005]
A)
Heterogeneous catalysis done
clear
B)
Homogeneous catalysis done
clear
C)
Enzyme catalysis done
clear
D)
Non-catalytic process done
clear
View Solution play_arrow
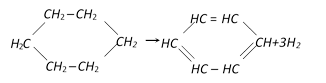 In the following reaction the catalyst used is [AMU (Engg.) 1999]
In the following reaction the catalyst used is [AMU (Engg.) 1999] 