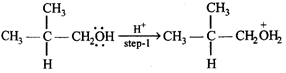
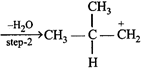
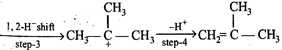 The slowest and fastest steps in the above reaction are
The slowest and fastest steps in the above reaction are
A) step 1 is slowest, while 3 is fastest.
B) step 2 is slowest, while 3 is fastest.
C) step 2 is slowest, while 4 is fastest.
D) all steps proceed at equal rate.
Correct Answer: C
Solution :
[c] Step 2 involves the formation of carbonium ion by the loss of weakly basic \[{{H}_{2}}O\] molecule. It is slowest step. Step 4 involves the conversion of an unstable (or intermediate) into a quite stable product, hence it is fastest step.You need to login to perform this action.
You will be redirected in
3 sec
