Answer:
(a)
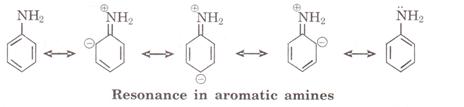
Aromatic amines are less basic than aliphatic amines because in aromatic amines
lone pair of nitrogen is involved in conjugation due to resonance. Thus the
availability of lone pair of donation decreases and acts as weak
base. ![]()
 (b)
The amine group strongly deactivates the benzene ring towards electrophilic
substitution. Hence it does not undergo Fridel-Crafts
alkylation. 1
(b)
The amine group strongly deactivates the benzene ring towards electrophilic
substitution. Hence it does not undergo Fridel-Crafts
alkylation. 1
You need to login to perform this action.
You will be redirected in
3 sec
