A) Compound (66) is prepared by the reaction of chlorobenzene with \[(C{{l}_{2}}+FeC{{l}_{3}})\] (excess) in dark.
B) Compound (666) is prepared by the reaction of benzene with \[C{{l}_{2}}\](excess) in light.
C) Carbolic acid is prepared by the reaction of benzene with aq. \[NaOH,\]at \[623\text{ }K,\] \[300\text{ }atm\]pressure followed by hydrolysis.
D) Formic acid is obtained as one of the product when phenyl carbyl amine is hydrolysed.
Correct Answer: C
Solution :
[c] Statement (3) is INCORRECT CORRECT statement: carbolic acid (phenol) is obtained by the mentioned reaction in (3) with chlorobenzene not benzene. (1)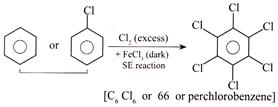 (2)
(2) 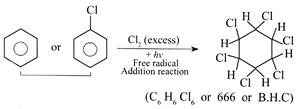 (3)
(3) 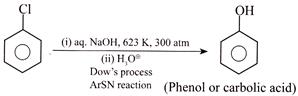 (4)
(4) 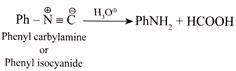 First \[{{E}^{\oplus }}\] adds to the C-atom and then \[N{{u}^{\bigcirc -}}\] adds to the same C-atom
First \[{{E}^{\oplus }}\] adds to the C-atom and then \[N{{u}^{\bigcirc -}}\] adds to the same C-atom
You need to login to perform this action.
You will be redirected in
3 sec
