 Select the INCORRECT statement.
Select the INCORRECT statement.
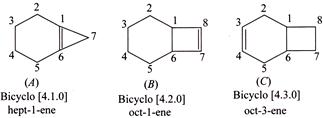
A) The rate of catalytic hydrogenation is faster in than in and
B) The rate of catalytic hydrogenation is faster in than in .
C) has a cyclopropene ring whose strain on going to less strained cyclopropane ring is reduced on hydrogenation. Similarly, cyclobutene ring in on hydrogenation is reduced to less-strained cyclobutane ring. But cyclopropene ring is more strained than cyclobutene ring, so faster hydrogenation is in than in .
D) There is a little change in the ring strain when the cyclohexene ring is reduced.
Correct Answer: A
Solution :
[a] CORRECT statement (I): Faster in[a] than [b] and [c]:You need to login to perform this action.
You will be redirected in
3 sec
