 The compound (A) , (B) and (C) are
The compound (A) , (B) and (C) are
A) A - \[Mn{{O}_{2}}\] B - \[{{K}_{2}}Mn{{O}_{4}}\] C - \[KMn{{O}_{4}}\]
B) A - \[M{{n}_{2}}{{O}_{3}}\] B - \[Mn{{O}_{2}}\] C - \[{{K}_{2}}Mn{{O}_{4}}\]
C) A - \[{{K}_{2}}Mn{{O}_{4}}\] B - \[KMn{{O}_{4}}\] C - \[M{{n}_{2}}{{O}_{3}}\]
D) A - \[KMn{{O}_{4}}\] B - \[M{{n}_{2}}{{O}_{3}}\] C - \[Mn{{O}_{2}}\]
Correct Answer: A
Solution :
[a] The reaction is a preparation of \[KMn{{O}_{4}}\] from the pyrolusite ore, \[Mn{{O}_{2}}\] (blackish brown) (i) \[2Mn{{O}_{2}}+4KOH+{{O}_{2}}/\] or oxidizing agent \[KN{{O}_{3}}\] [a]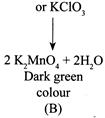 (ii) \[{{K}_{2}}Mn{{O}_{4}}\] disproportionates in a neutral or acidic solution to give \[KMn{{O}_{4}}\]
(ii) \[{{K}_{2}}Mn{{O}_{4}}\] disproportionates in a neutral or acidic solution to give \[KMn{{O}_{4}}\] 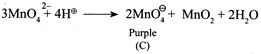
You need to login to perform this action.
You will be redirected in
3 sec
