A) 5
B) 6
C) 10
D) infinite
Correct Answer: B
Solution :
The wavelengths present in emission spectra are shown in the figure below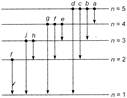 \[(\Delta \phi ={{\phi }_{2}}-{{\phi }_{1}})\] one atom\[T=\frac{2\pi }{\omega }\] one atom Remaining all by individual atoms.
\[(\Delta \phi ={{\phi }_{2}}-{{\phi }_{1}})\] one atom\[T=\frac{2\pi }{\omega }\] one atom Remaining all by individual atoms.
You need to login to perform this action.
You will be redirected in
3 sec
