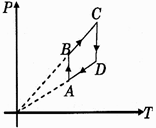
| Pressure versus temperature graph of an ideal gas is as shown in figure corresponding density \[(\rho )\] versus volume (V) graph will be - |
 |
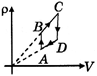
A)
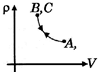
B)
C)
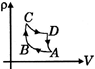
D)
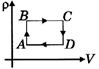
Correct Answer: B
Solution :
AD and BC is constant volume process so P v/s V graph is a point as V cannot change in the graph.You need to login to perform this action.
You will be redirected in
3 sec
