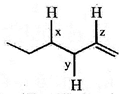
 Arrange the (C - H) bonds x, y and z in decreasing order of their bond dissociation energies in homolysis.
Arrange the (C - H) bonds x, y and z in decreasing order of their bond dissociation energies in homolysis.
A) \[y>x>z\]
B) \[z>x>y\]
C) \[z>y>x\]
D) \[y>z>x\]
Correct Answer: B
Solution :
 |
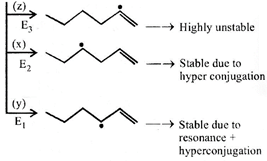 |
| So bond dissociation enegy order is |
| \[{{\operatorname{E}}_{1}}<{{E}_{2}}<{{E}_{3}}i.e,\left( z \right)>\left( x \right)>\left( y \right)\] |
You need to login to perform this action.
You will be redirected in
3 sec
