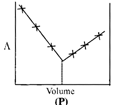
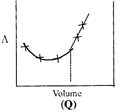
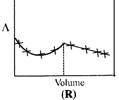
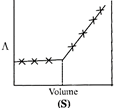
| \[AgNO{{~}_{3}}\](aq.) was added to an aqueous KCl solution gradually and the conductivity of the solution was measured. The plot of conductance \[\left( \wedge \right)\]versus the volume of \[AgNO{{~}_{3}}\] is | |
 |  |
 |  |
A) (P)
B) (Q)
C) (R)
D) (S)
Correct Answer: D
Solution :
[d] \[AgN{{O}_{3}}(aq)+KCl(aq)\xrightarrow{{}}AgCl(s)\,+KN{{O}_{3}}(aq)\] Conductivity or the solution is almost compensated due to formation of \[KN{{O}_{3}}(aq)\]however, after at end point, conductivity increases more rapidly due to addition of excess \[AgN{{O}_{3}}\] solution.You need to login to perform this action.
You will be redirected in
3 sec
