A) \[PO_{4}^{3\,-}\]
B) \[NH_{4}^{+}\]
C) \[SCl_{4}^{{}}\]
D) \[SO_{4}^{2\,-}\]
Correct Answer: C
Solution :
[c] \[SC{{l}_{4}}\] \[SC{{l}_{4}}\] is tetrahedral. All other given molecules/ ions are tetrahedral except \[SC{{l}_{4}}\] viz. trigonal bipyramid with one equatorial position occupied by a lone pair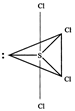
You need to login to perform this action.
You will be redirected in
3 sec
