The Van der Waal equation for n moles of a real gas is\[\left( p+\frac{a}{{{V}^{2}}} \right)(V-b)=nRT\]. Where P is the pressure, V is the volume. T is the absolute temperature. R is the molar gas constant and a and b are Van der Waal constant. The dimensions of a are the same as those of
A)
PV
done
clear
B)
\[P{{V}^{2}}\]
done
clear
C)
\[{{P}^{2}}V\]
done
clear
D)
\[\frac{P}{V}\]
done
clear
View Answer play_arrow
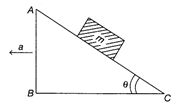
A particle of mass m is resting on a wedge of angle \[\theta \] as shown in the figure. The wedge is given an acceleration a. What is the value of a so that the mass m just falls freely?
A)
g
done
clear
B)
\[g\,\cot \,\theta \]
done
clear
C)
\[g\,\sin \theta \]
done
clear
D)
\[g\,\tan \,\theta \]
done
clear
View Answer play_arrow
Rain is falling vertically downwards with a velocity of 3 km/h. A man walks in the rain with a velocity of 4 km/h. The raindrops will fall on the man with a velocity of:
A)
1 km/h
done
clear
B)
3 km/h
done
clear
C)
4 km/h
done
clear
D)
\[5\,km/h\]
done
clear
View Answer play_arrow
A projectile is given an initial velocity of \[\hat{i}+2\hat{j}.\] The Cartesian equation of its path is: (\[g=10\,m/{{s}^{2}}\])
A)
\[~y=2x-5{{x}^{2}}\]
done
clear
B)
\[~y=x-5{{x}^{2}}\]
done
clear
C)
\[~4y=2x-5{{x}^{2}}\]
done
clear
D)
\[~y=2x-25{{x}^{2}}\]
done
clear
View Answer play_arrow
The potential energy of a certain spring when stretched through a distance S is 10 joule. The amount of work (in joule) that must be done on this spring to stretch it through an additional distance S will be
A)
30
done
clear
B)
40
done
clear
C)
10
done
clear
D)
20
done
clear
View Answer play_arrow
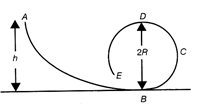
A frictionless track ABCDE ends in a circular loop of radius R. A body slides down the track from point A, which is at a height \[h=5\,cm.\]Maximum value of R for the body to successfully complete the loop is
A)
5 cm
done
clear
B)
\[\frac{15}{4}\,cm\]
done
clear
C)
\[\frac{10}{3}\,cm\]
done
clear
D)
\[2\,cm\]
done
clear
View Answer play_arrow
A body of mass 4 kg moving with velocity 12 m/s collides with another body of mass 6 kg at rest. If the two bodies stick together after collision, then the loss of kinetic energy of the system is
A)
0
done
clear
B)
288 J
done
clear
C)
172.8 J
done
clear
D)
144 J
done
clear
View Answer play_arrow
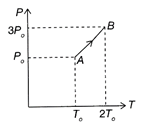
Pressure versus temperature graph of an ideal gas is as shown in the figure. Density of the gas at point A is \[{{\rho }_{0}}.\]Density at point B will be:
A)
\[\frac{3}{4{{\rho }_{0}}}\]
done
clear
B)
\[\frac{3}{2{{\rho }_{0}}}\]
done
clear
C)
\[\frac{4}{3{{\rho }_{0}}}\]
done
clear
D)
\[2{{\rho }_{0}}\]
done
clear
View Answer play_arrow
Equal volume of monoatomic and diatomic gases at the same temperature are given equal quantities of heat. Then:
A)
the temperature of diatomic gas will be more.
done
clear
B)
the temperature of monoatomic gas will be more.
done
clear
C)
the temperature of both will be zero.
done
clear
D)
nothing can be said.
done
clear
View Answer play_arrow
70 calories of heat are required to raise the temperature of 2 moles of an ideal gas at constant pressure from \[30{}^\circ C\] to \[35{}^\circ C\]. The amount of heat required (in calories) to raise the temperature of the same gas through the same range (\[30{}^\circ C\] to \[35{}^\circ C\]) at constant volume is
A)
30
done
clear
B)
50
done
clear
C)
70
done
clear
D)
90
done
clear
View Answer play_arrow
A spherical black body with a radius of 12 cm radiates 450 W power of 500 K. If the radius were half and temperature doubled, the power radiated in watt would be
A)
225
done
clear
B)
450
done
clear
C)
900
done
clear
D)
1800
done
clear
View Answer play_arrow
There are two hollow concentric spheres with radii \[{{R}_{1}}\]and R for the inner and outer spheres respectively. The inner sphere is given a charge \[{{Q}_{1}}\]and the outer a charge\[{{Q}_{2}}.\]Then the potential at the surface of the inner sphere is
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\left( \frac{{{Q}_{1}}}{{{R}_{1}}}+\frac{{{Q}_{2}}}{{{R}_{2}}} \right)\]
done
clear
B)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\left( \frac{{{Q}_{1}}}{{{R}_{1}}}-\frac{{{Q}_{2}}}{{{R}_{2}}} \right)\]
done
clear
C)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\left( \frac{{{Q}_{1}}}{{{R}_{1}}} \right)\]
done
clear
D)
0
done
clear
View Answer play_arrow
The electric field in a region is given by E\[E=\frac{3}{5}{{E}_{0}}i+\frac{4}{5}{{E}_{0}}j\]with\[{{E}_{0}}=2.0\times {{10}^{3}}N/C.\] The flux of this field through a rectangular surface area of \[0.2\,{{m}^{2}}\]parallel to the Y-Z plane is
A)
\[60\,N{{m}^{2}}/C\]
done
clear
B)
\[120\,N{{m}^{2}}/C\]
done
clear
C)
\[180\,N{{m}^{2}}/C\]
done
clear
D)
\[240\,N{{m}^{2}}/C\]
done
clear
View Answer play_arrow
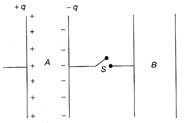
Consider the situation shown in the figure. The capacitor A has a charge q on it whereas B is uncharged, The charge appearing on the capacitor B a long time after the switch is closed is
A)
0
done
clear
B)
\[\frac{q}{2}\]
done
clear
C)
q
done
clear
D)
2q
done
clear
View Answer play_arrow
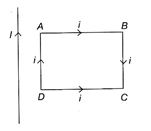
A rectangular loop carrying a current I is situated near a long straight wire such that the wire is parallel to one of the sides of the loop and is in the plane of the loop. If a steady current / is established in the wire as shown in the figure, the loop will:
A)
rotate about an axis parallel to the wire.
done
clear
B)
move away from the wire.
done
clear
C)
move towards the wire.
done
clear
D)
remain stationary.
done
clear
View Answer play_arrow
An electron of mass m is accelerated through a potential difference of V and then it enters a magnetic field of induction B normal to the lines. Then, the radius of the circular path is:
A)
\[\sqrt{(2\,eV/m)}\]
done
clear
B)
\[\sqrt{(2Vm/e{{B}^{2}})}\]
done
clear
C)
\[\sqrt{(2Vm/eB)}\]
done
clear
D)
\[\sqrt{(2Vm/{{e}^{2}}B)}\]
done
clear
View Answer play_arrow
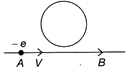
An electron moves along the line AB, which lies in the same plane as that of a circular loop of conducting wire as shown in the figure. What will be the direction of the current induced if any in the loop?
A)
No current will be induced.
done
clear
B)
The current will be clockwise.
done
clear
C)
The current will be anticlockwise.
done
clear
D)
The current will change direction as the electron passes by.
done
clear
View Answer play_arrow
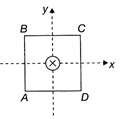
A square coil ABCD is lying in the \[x-y\]plane with its centre at origin. A long straight passing through origin carries a current \[i=2t\]It in negative z-direction. The induced current in the coil
A)
clockwise
done
clear
B)
anticlockwise
done
clear
C)
alternating
done
clear
D)
0
done
clear
View Answer play_arrow
A spherical drop of water has radius 1 mm. If surface tension of water is \[70\times {{10}^{-3}}N/m\]then the difference of pressures between the inside and outside of the spherical drop is:
A)
\[~35N/{{m}^{-2}}\]
done
clear
B)
\[~70\text{ }N/{{m}^{2}}\]
done
clear
C)
\[~140\text{ }N/{{m}^{2}}\]
done
clear
D)
0
done
clear
View Answer play_arrow
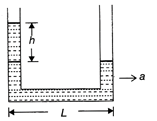
When at rest, a liquid stands at the same level in the tubes shown in the figure. But as indicated a height difference h occurs when the system is given an acceleration a towards the right. Here h is equal to:
A)
\[\frac{aL}{2g}\]
done
clear
B)
\[\frac{gL}{2a}\]
done
clear
C)
\[\frac{gL}{a}\]
done
clear
D)
\[\frac{aL}{g}\]
done
clear
View Answer play_arrow
In a metre bridge with standard resistance of\[5\Omega \]. In the left gap, the ratio of balancing lengths of metre bridge wire is 2:3. The unknown resistance is:
A)
\[10\Omega \]
done
clear
B)
\[15\Omega \]
done
clear
C)
\[10\Omega \]
done
clear
D)
\[3.3\Omega \]
done
clear
E)
\[7.5\Omega \]
done
clear
View Answer play_arrow
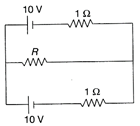
Maximum power developed across variable resistance R in the circuit shown in the figure is
A)
50 watt
done
clear
B)
75 watt
done
clear
C)
25 watt
done
clear
D)
100 watt
done
clear
View Answer play_arrow
A man measures time period of a pendulum (T) in stationary lift. If the lift moves upwards with acceleration g/4, then the new time period will be:
A)
\[\frac{2T}{\sqrt{5}}\]
done
clear
B)
\[\left( \frac{\sqrt{5T}}{2} \right)\]
done
clear
C)
\[\frac{\sqrt{5}}{(2T)}\]
done
clear
D)
\[\frac{2}{\sqrt{5T}}\]
done
clear
View Answer play_arrow
A luminous object is placed at a distance of 30 cm from a convex lens of focal length 20 cm. At what distance from the lens, on the other side, must a convex mirror of radius of curvature 10 cm be placed, in order to have an upright image of the object coincident with it?
A)
12 cm
done
clear
B)
30 cm
done
clear
C)
50 cm
done
clear
D)
60 cm
done
clear
View Answer play_arrow
A concave lens forms the image of an object such that the distance between the object and the image is 10 cm and the magnification produced is 1/4. The focal length of the lens is:
A)
8.6 cm
done
clear
B)
6.2 cm
done
clear
C)
10 cm
done
clear
D)
4.4 cm
done
clear
View Answer play_arrow
If two waves of same frequency and same amplitude respectively, on superposition, produce a resultant disturbance of the same amplitude, the waves differ in phase by:
A)
\[\pi \]
done
clear
B)
\[\frac{2\pi }{3}\]
done
clear
C)
\[\frac{2\pi }{3}\]
done
clear
D)
\[3\pi \]
done
clear
View Answer play_arrow
The light waves from two independent monochromatic light sources are given by: \[{{y}_{1}}=2\,\sin \omega t\]and \[{{y}_{2}}=3\,\cos \omega t.\]Then the correct statement is:
A)
Both the waves are coherent
done
clear
B)
Both the waves are incoherent
done
clear
C)
Both the waves have different time periods
done
clear
D)
None of the above
done
clear
View Answer play_arrow
When a beam of accelerated electrons hits a target a continuous X-ray spectrum is emitted from the target. Which one of the following wavelength is absent in the X-ray spectrum if the X-ray tube is operating at 40,000 volt?
A)
\[1.5\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[~0.5\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[0.25\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1.0\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
A freshly prepared radioactive source of half life 2 hr emits radiation of intensity, which is 64 times the permissible safe level. The minimum time after which it would be possible to work safely with this source is
A)
6 hr
done
clear
B)
12 hr
done
clear
C)
42 hr
done
clear
D)
128 hr
done
clear
View Answer play_arrow
Given that a photon of light of wavelength 10,000 angstrom has an energy equal to 1.23 eV. When light of wavelength 5000 angstrom and intensity\[{{I}_{0}}\] falls on a photoelectric cell, the saturation current is\[0.40\times {{10}^{-6}}\]ampere and the stopping potential is volt; then the work function is:
A)
0.43 eV
done
clear
B)
1.10 eV
done
clear
C)
1.36 eV
done
clear
D)
2.47 eV
done
clear
View Answer play_arrow
The de Broglie wavelength of an electron moving with a velocity \[1.5\times {{10}^{8}}m\,{{s}^{-1}}\]is equal to that of a photon. The ratio of the kinetic energy of the electron to that of the photon is
A)
2
done
clear
B)
4
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\frac{1}{4}\]
done
clear
View Answer play_arrow
The angular momentum of an electron in an orbit is quantized because it is a necessary condition for the compatibility with
A)
the wave nature of electron.
done
clear
B)
particle nature of electron.
done
clear
C)
Palli's exclusion behavior
done
clear
D)
None of these
done
clear
View Answer play_arrow
An LCR circuit contains resistance of 100 ohm and a supply of 200 volt at 300 radian angular frequency. If only capacitance is taken out from the circuit and the rest of the circuit is joined, current lags behind the voltage by \[60{}^\circ \]. If on the other hand, only inductor is taken out the current leads by \[60{}^\circ \] with the applied voltage. The current flowing in the circuit is:
A)
1 amp
done
clear
B)
1.5 amp
done
clear
C)
2 amp
done
clear
D)
2.5 amp
done
clear
View Answer play_arrow
Zener diode functions in:
A)
forward biased condition
done
clear
B)
reverse biased condition
done
clear
C)
both forward and reverse biased conditions
done
clear
D)
none of the above
done
clear
View Answer play_arrow
An artificial satellite is moving in a circular orbit around the earth with a speed equal to the escape velocity from the earth of radius R. What is the height of the satellite above the surface of the earth?
A)
\[\frac{R}{2}\]
done
clear
B)
R
done
clear
C)
3R
done
clear
D)
6R
done
clear
View Answer play_arrow
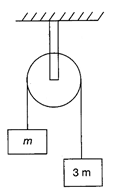
If the system is released, then the acceleration of the centre of mass of the system (as shown in the figure) is:
A)
\[\frac{g}{4}\]
done
clear
B)
\[\frac{g}{2}\]
done
clear
C)
\[g\]
done
clear
D)
\[2g\]
done
clear
View Answer play_arrow
The upper half of an inclined plane with inclination\[\phi \] is perfectly smooth, while the lower half is rough. A body starting from rest at the top will again come to rest at the bottom if coefficient of friction for the lower half is given by
A)
\[2\sin \phi \]
done
clear
B)
\[2\cos \phi \]
done
clear
C)
\[2\tan \phi \]
done
clear
D)
\[\tan \phi \]
done
clear
View Answer play_arrow
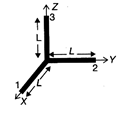
Three thin rods each of length L and mass M are placed along\[X,Y,\]and Z-axes in such a way that one end of each of the rods is at the origin. The moment of inertia of this system about Z-axis is:
A)
\[~2\text{ }M{{L}^{2}}/3\]
done
clear
B)
\[~4\text{ }M{{L}^{2}}/3\]
done
clear
C)
\[~5\text{ }M{{L}^{2}}/3\]
done
clear
D)
\[~M{{L}^{2}}/3\]
done
clear
View Answer play_arrow
A thin circular ring of mass M and radius R is rotating about its axis with a constant angular velocity\[\omega \]. Two objects each of mass m, are attached gently to the opposite ends of a diameter of the ring. The ring now rotates with an angular velocity
A)
\[\frac{\omega M}{M+m}\]
done
clear
B)
\[\frac{\omega (M-2m)}{M+2m}\]
done
clear
C)
\[\frac{\omega M}{M+2m}\]
done
clear
D)
\[\frac{\omega (M+2m)}{M}\]
done
clear
View Answer play_arrow
The bob of a simple pendulum is hanging vertically down from a fixed identical bob by means of a string of length\[l.\] If both bobs are charged with a charge q each, time period of the pendulum is: (ignore the radii of the bobs)
A)
\[2\pi \sqrt{\frac{\ell }{g+\sqrt{\frac{{{q}^{2}}}{{{\ell }^{2}}m}}}}\]
done
clear
B)
\[2\pi \sqrt{\frac{\ell }{g-\sqrt{\frac{{{q}^{2}}}{{{\ell }^{2}}m}}}}\]
done
clear
C)
\[2\pi \sqrt{\frac{\ell }{g}}\]
done
clear
D)
\[2\pi \sqrt{\frac{\ell }{g-\left( \frac{{{q}^{2}}}{\ell } \right)}}\]
done
clear
View Answer play_arrow
A string is wrapped several times round a solid cylinder and then the end of the string is held stationary while the cylinder is released from rest. The acceleration of the cylinder and tension in the string will be
A)
\[\frac{2g}{3}\]and\[\frac{mg}{3}\]
done
clear
B)
\[g\]and \[\frac{mg}{2}\]
done
clear
C)
\[\frac{g}{3}\]and \[\frac{mg}{2}\]
done
clear
D)
\[\frac{g}{2}\]and\[\frac{mg}{3}\]
done
clear
View Answer play_arrow
A uniform rod AB of mass m and length la is falling freely without rotation under gravity with AB horizontal. Suddenly the end A is fixed when the speed of the rod is v. The angular speed with which the rod begins to rotate is
A)
\[\frac{v}{2a}\]
done
clear
B)
\[\frac{4v}{3a}\]
done
clear
C)
\[\frac{v}{3a}\]
done
clear
D)
\[\frac{3v}{4a}\]
done
clear
View Answer play_arrow
A ray of light is incident on the surface of a glass plate of refractive index \[\sqrt{3}\]at the polarizing angle. The angle of refraction is
A)
\[60{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[50{}^\circ \]
done
clear
View Answer play_arrow
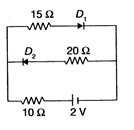
The current through \[10\Omega \] resistor in the circuit given below is (the diodes are ideal)
A)
50 mA
done
clear
B)
20 mA
done
clear
C)
40 mA
done
clear
D)
80 mA
done
clear
View Answer play_arrow
Hydrogen atoms are excited from ground state to the principal quantum number 4. Then the number of spectral lines observed will be
A)
3
done
clear
B)
6
done
clear
C)
5
done
clear
D)
2
done
clear
View Answer play_arrow
The value of Kp for the reaction, \[2{{H}_{2}}S(g)\leftrightarrow 2{{H}_{2}}\]\[(g)+{{S}_{2}}(g)\]is \[1.2\times {{10}^{-2}}\]at \[1065{{\,}^{o}}C.\]The value for Kc is
A)
\[<1.2\times {{10}^{-2}}\]
done
clear
B)
\[>1.2\times {{10}^{-2}}\]
done
clear
C)
\[1.2\times {{10}^{-2}}\]
done
clear
D)
\[0.12\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
1 mol of\[C{{H}_{3}}COOH\]and 1 mol of \[{{C}_{2}}{{H}_{5}}OH\]reacts to produce \[\frac{2}{3}\]mol of \[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}.\]The equilibrium constant is
A)
2
done
clear
B)
+ 2
done
clear
C)
- 4
done
clear
D)
+ 4
done
clear
View Answer play_arrow
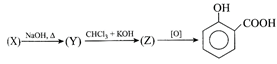
Identify (Z) in the following reaction
A)
Benzoic acid
done
clear
B)
Carbolic acid
done
clear
C)
Phenol
done
clear
D)
Salicylaldehyde
done
clear
View Answer play_arrow
The normality of solution obtained by mixing 10 ml of \[\frac{N}{5}HCl\] and 30 ml of \[\frac{N}{10}HCl\] is
A)
\[\frac{N}{5}\]
done
clear
B)
\[\frac{N}{8}\]
done
clear
C)
\[\frac{N}{15}\]
done
clear
D)
\[\frac{N}{7.5}\]
done
clear
View Answer play_arrow
The Beilstein test for organic compounds is used to detect
A)
Nitrogen
done
clear
B)
Sulphur
done
clear
C)
Carbon
done
clear
D)
Halogens
done
clear
View Answer play_arrow
Internal energy does not include
A)
Vibrational energy
done
clear
B)
Energy due to gravitational pull distillation
done
clear
C)
Rotational energy
done
clear
D)
Vibrational energy
done
clear
View Answer play_arrow
Which group of periodic table contains no metal
A)
VIIA
done
clear
B)
IIIA
done
clear
C)
VIII
done
clear
D)
IA
done
clear
View Answer play_arrow
Transition elements form coloured ions due to
A)
Fully filled d-orbitals
done
clear
B)
Availability of s-electrons
done
clear
C)
d-d transition
done
clear
D)
Smaller atomic radii
done
clear
View Answer play_arrow
Turpentine oil can be purified by
A)
Vacuum distillation
done
clear
B)
Fractional distillation
done
clear
C)
Steam distillation
done
clear
D)
Sublimation
done
clear
View Answer play_arrow
The chief ore of Hg is
A)
Cinnabar
done
clear
B)
Galena
done
clear
C)
Pyrolusite
done
clear
D)
Barunite
done
clear
View Answer play_arrow
Oxidation number of Os in\[\text{Os}{{\text{O}}_{\text{4}}}\] is
A)
+4
done
clear
B)
+8
done
clear
C)
+10
done
clear
D)
+18
done
clear
View Answer play_arrow
Hydrogen has high ionization energy than alkali metals because it has
A)
Small size
done
clear
B)
Large size
done
clear
C)
Ionic bond
done
clear
D)
Covalent bond
done
clear
View Answer play_arrow
If \[\text{e=1}\text{.60206}\times \text{1}{{\text{0}}^{-19}}C,\]\[e/m=1.75875\]\[\times {{10}^{11}}C\]\[k{{g}^{-1}}\] then the mass of electron is
A)
\[19.091\times {{10}^{-31}}kg\]
done
clear
B)
\[11.5830\times {{10}^{-31}}kg\]
done
clear
C)
\[6.9091\times {{10}^{-31}}kg\]
done
clear
D)
\[9.1091\times {{10}^{-31}}kg\]
done
clear
View Answer play_arrow
The transition element which shows the highest oxidation state is
A)
Manganese
done
clear
B)
Iron
done
clear
C)
Chromium
done
clear
D)
Vanadium
done
clear
View Answer play_arrow
The pH of a solution having the\[{{\text{H}}^{\text{+}}}\]ion concentration of \[1\times {{10}^{-4}}g\]ions/litre is
A)
5
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
Benzaldehyde can be prepared by the hydrolysis of
A)
Benzal chloride
done
clear
B)
Benzotrichloride
done
clear
C)
Benzonitrile
done
clear
D)
Benzyl chloride
done
clear
View Answer play_arrow
\[\text{Phenol}\xrightarrow{NaOH}(X)\xrightarrow{C{{O}_{2}}}(Y)\xrightarrow{{{H}^{+}}}(Z).\] (Z) is identified as
A)
Benzaldehyde
done
clear
B)
Benzoic acid
done
clear
C)
Salicylic acid
done
clear
D)
Sodium benzoate
done
clear
View Answer play_arrow
Alkynes usually show which type of reaction?
A)
Addition
done
clear
B)
Replacement
done
clear
C)
Substitution
done
clear
D)
Elimination
done
clear
View Answer play_arrow
The general molecular formula for disaccharide is
A)
\[{{C}_{10}}{{H}_{20}}{{O}_{10}}\]
done
clear
B)
\[{{C}_{12}}{{H}_{22}}{{O}_{10}}\]
done
clear
C)
\[{{C}_{12}}{{H}_{20}}{{O}_{10}}\]
done
clear
D)
\[{{C}_{12}}{{H}_{22}}{{O}_{11}}\]
done
clear
View Answer play_arrow
A gas expands isothermally against a constant external pressure of 1 atm from a volume of \[\text{10 d}{{\text{m}}^{\text{3}}}\]to a volume of \[\text{20 d}{{\text{m}}^{\text{3}}}\text{.}\] It absorbs 300 J of thermal energy from its surroundings. The \[\Delta U\]is
A)
-312 J
done
clear
B)
-213 J
done
clear
C)
-321 J
done
clear
D)
-123 J
done
clear
View Answer play_arrow
Chemical [A] is used for softening of water to remove temporary hardness. [A] reacts with sodium carbonates to produce caustic soda. When\[C{{O}_{2}}\]is bubble through [A] it turns cloudy. Chemically [A] is
A)
\[CaC{{O}_{3}}\]
done
clear
B)
\[Ca{{(HC{{O}_{3}})}_{2}}\]
done
clear
C)
\[Ca{{(OH)}_{2}}\]
done
clear
D)
\[CaO\]
done
clear
View Answer play_arrow
Bell metal is an alloy of
A)
Cu and Pb
done
clear
B)
Sn and Cu
done
clear
C)
Sn and Pb
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[CuS{{O}_{4}}\]and KCN reacts to produce
A)
\[K[Cu{{(CN)}_{6}}]\]
done
clear
B)
\[CuCN\]
done
clear
C)
\[CuC{{N}_{2}}\]
done
clear
D)
\[{{K}_{3}}[Cu{{(CN)}_{4}}]\]
done
clear
View Answer play_arrow
The empirical formula of a compound is \[C{{H}_{2}}O.\]Its molecular weight is 180. The molecular formula of that compound is
A)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
B)
\[{{C}_{6}}H{{O}_{4}}\]
done
clear
C)
\[{{C}_{3}}{{H}_{6}}{{O}_{3}}\]
done
clear
D)
\[{{C}_{5}}{{H}_{10}}{{O}_{5}}\]
done
clear
View Answer play_arrow
Nitrolium is
A)
\[Ca{{(CN)}_{2}}\]
done
clear
B)
\[CaC{{N}_{2}}\]
done
clear
C)
\[Ca{{(N{{O}_{3}})}_{2}}\]
done
clear
D)
\[CaC{{N}_{2}}+C\]
done
clear
View Answer play_arrow
\[{{\,}_{12}}{{A}^{27}}+{{\,}_{2}}H{{e}^{4}}\to {{\,}_{14}}S{{i}^{30}}+{{\,}_{1}}{{H}^{1}}+Q\]Mass of\[{{\,}_{13}}A{{l}^{27}}=26.9815\]amu and mass of \[{{\,}_{14}}S{{i}^{30}}=29.9738.\]The Q is equal to
A)
2.329 MeV
done
clear
B)
3.298 MeV
done
clear
C)
12.98 MeV
done
clear
D)
5.478 MeV
done
clear
View Answer play_arrow
\[\text{A}{{\text{l}}_{\text{2}}}{{\text{O}}_{\text{3}}}\]on heating with carbon in an atmosphere of\[{{\text{N}}_{\text{2}}}\]at high temperature produces
A)
\[\text{Al+C}{{\text{O}}_{2}}+NO\]
done
clear
B)
\[Al+C{{O}_{2}}\]
done
clear
C)
\[AlN+CO\]
done
clear
D)
\[A{{l}_{4}}{{C}_{3}}+C{{O}_{2}}\]
done
clear
View Answer play_arrow
The product obtained by treating: \[C{{H}_{3}}-CH=C{{H}_{2}}+HBr\to \]
A)
\[C{{H}_{3}}-CH=C{{H}_{2}}Br\]
done
clear
B)
\[C{{H}_{2}}BrC{{H}_{2}}=C{{H}_{2}}\]
done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}Br\]
done
clear
D)
\[C{{H}_{3}}-\underset{Br}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-C{{H}_{3}}\]
done
clear
View Answer play_arrow
The transport of matter in the absence of bulk flow is known as
A)
Transfusion
done
clear
B)
Diffusion
done
clear
C)
Rotation
done
clear
D)
Translation
done
clear
View Answer play_arrow
Purple of Cassius is a colloidal solution of
A)
Lead
done
clear
B)
Mercury
done
clear
C)
Silver
done
clear
D)
Gold
done
clear
View Answer play_arrow
Which of the following is not the characteristic of interhalogen compounds?
A)
More reactive than halogens
done
clear
B)
They are covalent
done
clear
C)
Quite unstable but not explosive
done
clear
D)
Have low boiling point and highly volatile
done
clear
View Answer play_arrow
The process of decomposition of organic compound by the application of heat is
A)
Evaporation
done
clear
B)
Sublimation
done
clear
C)
Pyrolysis
done
clear
D)
Condensation
done
clear
View Answer play_arrow
Gun metal is
A)
\[Cu+Sn+Zn\]
done
clear
B)
\[Zn+Sn\]
done
clear
C)
\[Cu+Zn\]
done
clear
D)
\[Cu+Sn\]
done
clear
View Answer play_arrow
The monomer of Teflon is
A)
Trifluoroethene
done
clear
B)
Tetrafluoroethene
done
clear
C)
Monofluoroethene
done
clear
D)
Difluoroethene
done
clear
View Answer play_arrow
The correct decreasing order of basic strength is
A)
\[P{{H}_{3}}>As{{H}_{3}}>Sb{{H}_{3}}>N{{H}_{3}}\]
done
clear
B)
\[~N{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}>Sb{{H}_{3}}\]
done
clear
C)
\[As{{H}_{3}}>Sb{{H}_{3}}\text{ }>P{{H}_{3}}>N{{H}_{3}}\]
done
clear
D)
\[Sb{{H}_{3}}>As{{H}_{3}}>P{{H}_{3}}>N{{H}_{3}}\]
done
clear
View Answer play_arrow
Iodide of Millon's base is
A)
\[{{K}_{2}}Hg{{I}_{4}}\]
done
clear
B)
\[HI{{O}_{3}}\]
done
clear
C)
\[Hg(N{{H}_{2}})I\]
done
clear
D)
\[N{{H}_{2}}HgO.HgI\]
done
clear
View Answer play_arrow
Sodium on heating with moist air produces
A)
\[NaO\]
done
clear
B)
\[N{{a}_{2}}O\]
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[NaOH\]
done
clear
View Answer play_arrow
The product obtained by treating benzene with chlorine in presence of ultraviolet light
A)
\[{{C}_{6}}C{{l}_{6}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
C)
\[CC{{l}_{4}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{6}}C{{l}_{6}}\]
done
clear
View Answer play_arrow
Gammexane is
A)
BHC
done
clear
B)
HCB
done
clear
C)
Chloral
done
clear
D)
DDT
done
clear
View Answer play_arrow
Which of the following produces ketone on treatment with Grignard reagent?
A)
Acetic acid
done
clear
B)
Methyl alcohol
done
clear
C)
Methyl cyanide
done
clear
D)
Acetaldehyde
done
clear
View Answer play_arrow
Vinegar is represented by
A)
HCOOH
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}COOH\]
done
clear
D)
\[~C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
Which shows electrical conductance?
A)
Graphite
done
clear
B)
Diamond
done
clear
C)
Potassium
done
clear
D)
Sodium
done
clear
View Answer play_arrow
Which of the following cannot displace hydrogen from its compound?
A)
Hg
done
clear
B)
Pb
done
clear
C)
Al
done
clear
D)
Fe
done
clear
View Answer play_arrow
The natural gas mainly contains
A)
Propane
done
clear
B)
Pentane
done
clear
C)
Butane
done
clear
D)
Methane
done
clear
View Answer play_arrow
Which compound can be sulphonated easily?
A)
Benzene
done
clear
B)
Nitrobenzene
done
clear
C)
Chlorobenzene
done
clear
D)
Toluene
done
clear
View Answer play_arrow
The energy of electron in the first energy level is \[-21.79\times {{10}^{-12}}\,\text{erg}\] per atom. The energy of electron in second energy level is
A)
\[-15.47\times {{10}^{-12}}\,\text{erg}\,\text{ato}{{\text{m}}^{-1}}\]
done
clear
B)
\[-0.00547\times {{10}^{-12}}\,\text{erg}\,\text{ato}{{\text{m}}^{-1}}\]
done
clear
C)
\[-4.557\times {{10}^{-12}}\,\text{erg}\,\text{ato}{{\text{m}}^{-1}}\]
done
clear
D)
\[-5.447\times {{10}^{-12}}\text{erg}\,\text{ato}{{\text{m}}^{-1}}\]
done
clear
View Answer play_arrow
Oxidation is
A)
Loss of electrons
done
clear
B)
Gain of electrons
done
clear
C)
Loss of neutrons
done
clear
D)
Decrease in positive valency
done
clear
View Answer play_arrow
When two halogen atoms are attached to same carbon atom then it is
A)
\[\alpha -\omega -\] halide
done
clear
B)
\[\alpha -\beta -\] halide
done
clear
C)
Gem dihalide
done
clear
D)
Vicinal dihalide
done
clear
View Answer play_arrow
Flux is used to remove
A)
Acidic impurities
done
clear
B)
Basic impurities
done
clear
C)
Both acidic and basic impurities
done
clear
D)
All types of impurities
done
clear
View Answer play_arrow
Geometrical isomerism is possible in case of
A)
Propene
done
clear
B)
2-butene
done
clear
C)
Tartaric acid
done
clear
D)
1-butene
done
clear
View Answer play_arrow
In five kingdom system, the main basis of classification is
A)
Structure of nucleus
done
clear
B)
Mode of nutrition
done
clear
C)
Structure of cell wall
done
clear
D)
Asexual reproduction
done
clear
View Answer play_arrow
The main difference in Gram-positive and Gram- negative bacteria resides in their
A)
Cell wall
done
clear
B)
Cell membrane
done
clear
C)
Cytoplasm
done
clear
D)
Flagella
done
clear
View Answer play_arrow
Plasmodium, the malaria parasite, belongs to class
A)
Sarcodina
done
clear
B)
Ciliata
done
clear
C)
Sporozoa
done
clear
D)
Dinophyceae
done
clear
View Answer play_arrow
Two bacteria found to be very useful in genetic engineering experiments are
A)
Escherichia and Agrobacterium
done
clear
B)
Nitrobacter and Azotobacter
done
clear
C)
Rhizobium and Diplococcus
done
clear
D)
Nitrosomonas and Klebsiella
done
clear
View Answer play_arrow
Viruses that infect bacteria, multiply and cause their lysis are called
A)
Lysozymes
done
clear
B)
Lipolytic
done
clear
C)
Lytic
done
clear
D)
Lysogenic
done
clear
View Answer play_arrow
White rust disease is caused by
A)
Claviceps
done
clear
B)
Alternaria
done
clear
C)
Phytophthora
done
clear
D)
Albugo Candida
done
clear
View Answer play_arrow
In fungi, stored food material is
A)
Glycogen
done
clear
B)
Starch
done
clear
C)
Sucrose
done
clear
D)
Glucose
done
clear
View Answer play_arrow
Which one of the following is a slime mould?
A)
Physarum
done
clear
B)
Thiobacillus
done
clear
C)
Anabaena
done
clear
D)
Rhizopus
done
clear
View Answer play_arrow
A plant in which sporophytic generation is represented by zygote
A)
Pinus
done
clear
B)
Selaginella
done
clear
C)
Chlamydomonas
done
clear
D)
Dryopteris
done
clear
View Answer play_arrow
A well-developed archegonium with a neck consisting of 4-6 rows of neck canal cells, characterizes
A)
Gymno sperms only
done
clear
B)
Bryophytes and pteridophytes
done
clear
C)
Pteridophytes and gymnosperms
done
clear
D)
Gymnosperms and flowering plants
done
clear
View Answer play_arrow
Which of the following propagates through leaf-tip?
A)
Walking fern
done
clear
B)
Sproux-leaf plant
done
clear
C)
Marchantia
done
clear
D)
Moss
done
clear
View Answer play_arrow
The latest model for plasma membrane is
A)
Lamellar model
done
clear
B)
Unit membrane model
done
clear
C)
Fluid mosaic model
done
clear
D)
Molecular lipid model
done
clear
View Answer play_arrow
Hammerling's experiments of Acetabularia involves exchanging
A)
Cytoplasm
done
clear
B)
Nucleus
done
clear
C)
Rhizoid and stalk
done
clear
D)
Gametes
done
clear
View Answer play_arrow
Segregation ofMendelian factor (Aa) occurs during
A)
Diplotene
done
clear
B)
Anaphase I
done
clear
C)
Zygotene/Pachytene
done
clear
D)
Anaphase II
done
clear
View Answer play_arrow
Mitochondrial cristae are sites of
A)
Breakdown of macromolecules
done
clear
B)
Protein synthesis
done
clear
C)
Phosphorylation of flavoproteins
done
clear
D)
Oxidation-reduction reactions
done
clear
View Answer play_arrow
ABO blood group system is due to
A)
Multifactor inheritance
done
clear
B)
Incomplete dominance
done
clear
C)
Multiple allelism
done
clear
D)
Epistasis
done
clear
View Answer play_arrow
Both husband and wife have normal vision though their fathers were colour-blind. The probability of their daughter becoming colour-blind is
A)
0%
done
clear
B)
25%
done
clear
C)
50%
done
clear
D)
75%
done
clear
View Answer play_arrow
Nucleosome core is made of
A)
\[\text{H1, H2A, H2B and H3}\]
done
clear
B)
\[\text{H1, H2A, H2B, H4}\]
done
clear
C)
\[\text{H1, H2A, H2B, H3}\,\text{and }\,\text{H4}\]
done
clear
D)
\[\text{ }\!\!~\!\!\text{ H2A, H2B, H3 and H4}\]
done
clear
View Answer play_arrow
Two non-allelic genes produce new phenotype when present together but fail to do so independently are called?
A)
Epistatsis
done
clear
B)
Polygene
done
clear
C)
Non-complimentary genes
done
clear
D)
Complimentary genes
done
clear
View Answer play_arrow
In pea plant, yellow seeds are dominant to green. If a heterozygous yellow seeded plant is crossed with a green seeded plant, what ration of yellow and green seeded plants would you expect in \[{{\text{F}}_{\text{1}}}\]generation?
A)
9:1
done
clear
B)
1 : 3
done
clear
C)
3:1
done
clear
D)
50 : 50
done
clear
View Answer play_arrow
Khorana first deciphered the triplet codons of
A)
Serine and isoleucine
done
clear
B)
Cysteine and valine
done
clear
C)
Tyrosine and tryptophan
done
clear
D)
Phenylalanine and methionine
done
clear
View Answer play_arrow
During DNA replication, the strands separate by
A)
DNA polymerase
done
clear
B)
Topoisomerase
done
clear
C)
Unwindase/Helicase
done
clear
D)
Gyrase
done
clear
View Answer play_arrow
Gene and cistron words are sometimes used synony mously because
A)
One cistron contains many genes
done
clear
B)
One gene contains many cistrons
done
clear
C)
One gene contains one cistron
done
clear
D)
One gene contains no cistron
done
clear
View Answer play_arrow
Out of 64 codons, 61 codons code for 20 types of amino acid. It is called
A)
Degeneracy of genetic code
done
clear
B)
Overlapping of gene
done
clear
C)
Wobbling of codon
done
clear
D)
Universality of codons
done
clear
View Answer play_arrow
During transcription, the RNA polymerase holoenzyme binds to a gene promoter and assumes a saddle-like structure, what is its DNA-binding sequence?
A)
AATT
done
clear
B)
CACC
done
clear
C)
TATA
done
clear
D)
TTAA
done
clear
View Answer play_arrow
Restriction endonucleases are
A)
Synthesized by bacteria as part of their defence mechanism.
done
clear
B)
Used for in vitro DNA synthesis.
done
clear
C)
Used in genetic engineering for ligating two DNA molecules.
done
clear
D)
Present in mammalian cells for degradation of DNA when the cell dies.
done
clear
View Answer play_arrow
The first successfully cloned mammal that gained worldwide publicity was
A)
Chance, a bull
done
clear
B)
Dolly, a sheep
done
clear
C)
Molly, a sheep
done
clear
D)
Polly, a sheep
done
clear
View Answer play_arrow
Telomere repetitive DNA sequences control the function of eukaryotic chromosomes because they
A)
Are RNA transcription initiator
done
clear
B)
Help chromosome pairing
done
clear
C)
Prevent chromosome loss
done
clear
D)
Act as replicons
done
clear
View Answer play_arrow
Vegetative reproduction of Agave occurs through
A)
Rhizome
done
clear
B)
Stolon
done
clear
C)
Bulbils
done
clear
D)
Sucker
done
clear
View Answer play_arrow
What is eye of potato?
A)
Axillary bud
done
clear
B)
Accessory bud
done
clear
C)
Adventitious bud
done
clear
D)
Apical bud
done
clear
View Answer play_arrow
Which of the following is a correct pair?
A)
Cuscuta - Parasite
done
clear
B)
Dischidia - Insectivorous
done
clear
C)
Opuntia - Predator
done
clear
D)
Capsella - Hydrophyte
done
clear
View Answer play_arrow
is the floral formula of
A)
Liliaceae
done
clear
B)
Solanaceae
done
clear
C)
Asteraceae
done
clear
D)
Fabaceae
done
clear
View Answer play_arrow
The aleurone layer in maize grain is especially rich in
A)
Auxins
done
clear
B)
Proteins
done
clear
C)
Starch
done
clear
D)
Lipids
done
clear
View Answer play_arrow
Monocot leaves possess
A)
Intercalary meristem
done
clear
B)
Lateral meristem
done
clear
C)
Apical meristem
done
clear
D)
Mass meristem
done
clear
View Answer play_arrow
Bordered pits are found in
A)
Sieve cells
done
clear
B)
Vessel wall
done
clear
C)
Companion cells
done
clear
D)
Sieve tube wall
done
clear
View Answer play_arrow
The main function of lenticel is
A)
Transpiration
done
clear
B)
Guttation
done
clear
C)
Gaseous exchange
done
clear
D)
Bleeding
done
clear
View Answer play_arrow
Guttation is mainly due to
A)
Root pressure
done
clear
B)
Osmosis
done
clear
C)
Transpiration
done
clear
D)
Imbibition
done
clear
View Answer play_arrow
Which one of the following is not an essential element for plants?
A)
Potassium
done
clear
B)
Iron
done
clear
C)
Iodine
done
clear
D)
Zinc
done
clear
View Answer play_arrow
A free living nitrogen-fixing cyanobacterium which can also form symbiotic association with the water fern Azolla is
A)
Tolypothrix
done
clear
B)
Chlorella
done
clear
C)
Nostoc
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
In \[{{C}_{4}}\] plants, Calvin cycle operates in the
A)
Stroma of bundle sheath chloroplasts
done
clear
B)
Grana of bundle sheath chloroplasts
done
clear
C)
Grana of mesophyll chloroplasts
done
clear
D)
Stroma of mesophyll chloroplasts
done
clear
View Answer play_arrow
Ferredoxin is a constituent of
A)
PS I
done
clear
B)
PS II
done
clear
C)
Hill reaction
done
clear
D)
\[{{P}_{680}}\]
done
clear
View Answer play_arrow
A photosynthesizing plant is releasing \[^{18}O\] more than the normal. The plant must have been supplied with
A)
\[{{O}_{3}}\]
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
\[C{{O}_{2}}\,with{{\,}^{18}}O\]
done
clear
D)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\,with{{\,}^{18}}O\]
done
clear
View Answer play_arrow
Terminal cytochrome of respiratory chain which donates electrons to oxygen is
A)
Cyt b
done
clear
B)
Cyt c
done
clear
C)
Cyt \[{{a}_{1}}\]
done
clear
D)
Cyt \[{{a}_{3}}\]
done
clear
View Answer play_arrow
Double fertilisation and triple fusion were discovered by
A)
Hofmeister
done
clear
B)
Nawaschin and Guignard
done
clear
C)
Leeuwenhoek
done
clear
D)
Strasburger
done
clear
View Answer play_arrow
Eight nucleate embryo sacs are
A)
Always tetrasporic
done
clear
B)
Always monosporic
done
clear
C)
Always bisporic
done
clear
D)
Sometimes monosporic, sometimes bisporic and sometimes tetrasporic.
done
clear
View Answer play_arrow
The arrangement of the nuclei in a normal embryo sac in the dicot plants is
A)
3+2 +3
done
clear
B)
2+3 + 3
done
clear
C)
3 + 3 + 2
done
clear
D)
2 + 4 + 2
done
clear
View Answer play_arrow
The hormone which is primarily connected with the cell division is
A)
IAA
done
clear
B)
NAA
done
clear
C)
Cytokinin/Zeatin
done
clear
D)
Gibberellic acid
done
clear
View Answer play_arrow
Which of the following breaks bud dormancy of potato tuber?
A)
Gibberellin
done
clear
B)
IAA
done
clear
C)
ABA
done
clear
D)
Zeatin
done
clear
View Answer play_arrow
Differentiation of shoot is controlled by
A)
High gibberellins: Cytokinin ratio
done
clear
B)
High auxin: Cytokinin ratio
done
clear
C)
High cytokinin: Auxin ratio
done
clear
D)
High gibberellins: Auxin ratio
done
clear
View Answer play_arrow
Which of the following is true about ecosystem?
A)
Primary consumers are least dependent upon producers.
done
clear
B)
Primary consumers out-number producers.
done
clear
C)
Producers are more than primary consumers.
done
clear
D)
Secondary consumers are the largest and most powerful.
done
clear
View Answer play_arrow
A group of individuals of the same kind of a phenotype characters and can interbreed is terms as
A)
Colony
done
clear
B)
Genus
done
clear
C)
Community
done
clear
D)
Species
done
clear
View Answer play_arrow
In grass-deer-tiger food chain, grass biomass is one tonne. The tiger biomass shall be
A)
100 kg
done
clear
B)
10 kg
done
clear
C)
200 kg
done
clear
D)
1 kg
done
clear
View Answer play_arrow
The transfer of energy from one trophic level to another is governed by the 2nd law of thermodynamics. The average efficiency of energy transfer from herbivores to carnivores is
A)
5 %
done
clear
B)
10 %
done
clear
C)
25 %
done
clear
D)
50 %
done
clear
View Answer play_arrow
Identify the odd combination of the habitat and the particular animal concerned.
A)
Sunderbans - Bengal Tiger
done
clear
B)
Periyar - Elephant
done
clear
C)
Rann of kutch - Wild Ass
done
clear
D)
Dachigam National - Snow Leopard park
done
clear
View Answer play_arrow
The highest DDT deposition shall occur in
A)
Phytoplankton
done
clear
B)
Seagull/Birds
done
clear
C)
Crab
done
clear
D)
Eel
done
clear
View Answer play_arrow
Which gas contributes the most to greenhouse effect?
A)
CFC
done
clear
B)
Freon
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
The most common indicator organism that represents polluted water is
A)
E. coli
done
clear
B)
S. typhi
done
clear
C)
V. cholerae
done
clear
D)
Entamoeba
done
clear
View Answer play_arrow
Which of the following is absent in polluted water?
A)
Hydrilla
done
clear
B)
Water hyacinth
done
clear
C)
Larva of stone fly
done
clear
D)
Blue-green algae
done
clear
View Answer play_arrow
The first transgenic crop was
A)
Pea
done
clear
B)
Flax
done
clear
C)
Tobacco
done
clear
D)
Cotton
done
clear
View Answer play_arrow
Probiotics are
A)
Cancer inducing microbes
done
clear
B)
New kind of food allergens
done
clear
C)
Live microbial food supplement
done
clear
D)
Safe antibiotics
done
clear
View Answer play_arrow
Sarcoma is a type cancer of that develops in
A)
Epithelial tissue
done
clear
B)
Mesodermal tissues
done
clear
C)
Blood
done
clear
D)
Endodermal tissues
done
clear
View Answer play_arrow
Farmers have reported over 50 per cent higher yields of rice by using the biofertilizer
A)
Azolla pinnata
done
clear
B)
Cyanobacteria
done
clear
C)
Legume-Rhizobium symbiosis
done
clear
D)
My corrhiza
done
clear
View Answer play_arrow
Hybridoma cells are
A)
Nervous cells of frog
done
clear
B)
Hybrid cells resulting from myeloma cells
done
clear
C)
Only cells having oncogenes
done
clear
D)
Product of spore formation in bacteria
done
clear
View Answer play_arrow
Biological organisation starts with
A)
Cellular level
done
clear
B)
Organismic level
done
clear
C)
Atomic level
done
clear
D)
Submicroscopic molecular level
done
clear
View Answer play_arrow
Which is not consistent with the double helical structure of DNA?
A)
A = T, C = G
done
clear
B)
Density of DNA decreases on heating
done
clear
C)
A + T/C + G is not constant
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
Protein synthesis in an animal cell takes place
A)
Only in the cytoplasm
done
clear
B)
In the nucleolus as well as in cytoplasm
done
clear
C)
In cytoplasm as well as in mitochondria
done
clear
D)
Only on ribosomes attached to the nuclear envelope.
done
clear
View Answer play_arrow
The transfer RNA molecule in 3D appears
A)
L-shaped
done
clear
B)
E-shaped
done
clear
C)
Y-shaped
done
clear
D)
S-shaped
done
clear
View Answer play_arrow
Transfer of Taenia to secondary host occurs as
A)
Oncosphere
done
clear
B)
Cysticercus
done
clear
C)
Morula
done
clear
D)
Egg
done
clear
View Answer play_arrow
A chordate character is
A)
Gills
done
clear
B)
Spiracles
done
clear
C)
Postanal tail
done
clear
D)
Chitinous exoskeleton
done
clear
View Answer play_arrow
An insect regarded as the greatest mechanical carrier of diseases is
A)
Pediculus
done
clear
B)
Cimex
done
clear
C)
Musca
done
clear
D)
Xenopsylla
done
clear
View Answer play_arrow
Sound box of birds is called
A)
Pygostyle
done
clear
B)
Larynx
done
clear
C)
Syrinx
done
clear
D)
Synsacrum
done
clear
View Answer play_arrow
Which one of the following is a matching set of a phylum and its three examples?
A)
Platyhelminthes - Planaria, Schistosoma, Enterobius
done
clear
B)
Mollusca - Loligo, Teredo, Octopus
done
clear
C)
Porifera - Spongilla, Euplectella, Pennatula
done
clear
D)
Cnidaria - Bonellia, Physalia, Aurelia
done
clear
View Answer play_arrow
Water is a resource which is
A)
Non-degradable and non-maintainable
done
clear
B)
Degradable and maintainable
done
clear
C)
Renewable
done
clear
D)
Non-renewable
done
clear
View Answer play_arrow
A number of natural reserves have been created to conserve specific wild life species. Identify the correct combination from the following
A)
Gir Forest - Lion
done
clear
B)
Kaziranga - Elephants
done
clear
C)
Rann of Kutch - Wild Ass
done
clear
D)
Manas wildlife Sanctuary - Musk deer
done
clear
View Answer play_arrow
Characteristics of smooth muscle fibres are
A)
Spindle-shaped, unbranched, unstriated, uninucleate and involuntary.
done
clear
B)
Spindle-shaped, unbranched, unstriped, multinucleate and involuntary.
done
clear
C)
Cylindrical, unbranched, unstriped, multinucleate and involuntary.
done
clear
D)
Cylindrical, unbranched, striated, multinucleate and voluntary.
done
clear
View Answer play_arrow
The polysaccharide present in the matrix of cartilage is known as
A)
Cartilagin
done
clear
B)
Ossein
done
clear
C)
Chondroitin
done
clear
D)
Casein
done
clear
View Answer play_arrow
Hydrolytic enzymes which act on low pH are known as
A)
Proteases
done
clear
B)
\[\alpha -\] Amylases
done
clear
C)
Hydrolases
done
clear
D)
Peroxidases
done
clear
View Answer play_arrow
Which one of the following is a fat-soluble vitamin and its related deficiency diseases?
A)
Retinol - Xerophthalmia
done
clear
B)
Cobalamin - Beriberi
done
clear
C)
Calciferol - Pellagra
done
clear
D)
Ascorbic acid - Scurvy
done
clear
View Answer play_arrow
When
concentration in blood increases, breathing becomes
A)
Shallower and slow
done
clear
B)
There is no effect on breathing
done
clear
C)
Slow and deep
done
clear
D)
Faster and deeper
done
clear
View Answer play_arrow
People living at sea level have around 5 million RBC per cubic millimetre of their blood whereas those living at an altitude of 5400 metres have around 8 million. This is because at high altitudes
A)
Atmospheric
level is less and hence more RBC's are needed to absorb the required amount of
to survive.
done
clear
B)
There is more UV radiation which enhances the RBC production.
done
clear
C)
People eat more nutritive food; therefore more RBC's are formed.
done
clear
D)
People get pollution-free air to breathe and more oxygen is available.
done
clear
View Answer play_arrow
Closed circulatory system occurs in
A)
Cockroach
done
clear
B)
Tadpole/fish
done
clear
C)
Mosquito
done
clear
D)
Housefly
done
clear
View Answer play_arrow
Contraction of the ventricle in the heart begins by the command from
A)
Chordae tendineae
done
clear
B)
Sinoatrial node
done
clear
C)
Purkinje fibres
done
clear
D)
Atrioventricular node
done
clear
View Answer play_arrow
In Ornithine cycle, which one pair of the following wastes is removed from the blood?
A)
and urea
done
clear
B)
and ammonia
done
clear
C)
Ammonia and urea
done
clear
D)
Urea and sodium salts
done
clear
View Answer play_arrow
Bowman's glands are located in the
A)
Anterior pituitary region
done
clear
B)
Female reproductive system
done
clear
C)
Olfactory epithelium of our nose
done
clear
D)
Proximal end of uriniferous tubules
done
clear
View Answer play_arrow
Tendon is made up of
A)
Adipose tissue
done
clear
B)
Modified white fibrous tissue
done
clear
C)
Areolar tissue
done
clear
D)
Yellow fibrous connective tissue
done
clear
View Answer play_arrow
Which of the following pairs is correctly matched?
A)
Hinge joint - Between vertebrae
done
clear
B)
Gliding joint - Between zygapophyses of the successive vertebrae
done
clear
C)
Cartilaginous - Skull bones joint
done
clear
D)
Fibrous joint - Between phalanges
done
clear
View Answer play_arrow
Which of the following is an example of negative feedback loop in humans?
A)
Secretion of tears after falling of sand particles into the eye.
done
clear
B)
Salivation of mouth at the sight of delicious food.
done
clear
C)
Secretion of sweat glands and constriction of skin blood vessels when it is too hot.
done
clear
D)
Constriction of skin blood vessels and contraction of skeletal muscles when it is too cold.
done
clear
View Answer play_arrow
Which one of the following hormone stimulates the 'let-down' (release) of milk from the mother's breasts when the baby is sucking?
A)
Progesterone
done
clear
B)
Oxytocin
done
clear
C)
Prolactin
done
clear
D)
Relaxin
done
clear
View Answer play_arrow
Acromegaly is caused by
A)
Excess of GH
done
clear
B)
Excess of thyroxin
done
clear
C)
Deficiency of thyroxin
done
clear
D)
Excess of adrenaline
done
clear
View Answer play_arrow
Which of the following hormones is not a secretion product of human placenta?
A)
Human chorionic gonadotropin
done
clear
B)
Prolactin
done
clear
C)
Estrogen
done
clear
D)
Progesterone
done
clear
View Answer play_arrow