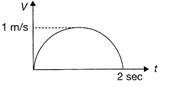
The velocity- time graph of a car is a semi-circle as shown in the figure. Which of the following statement is correct?
A)
The car must move in circular path.
done
clear
B)
Acceleration of the car is never zero.
done
clear
C)
The mean speed of the car is\[\frac{\pi }{4}\,m/s.\]
done
clear
D)
The car makes a turn once during its motion.
done
clear
View Answer play_arrow
A particle is moving eastward with a velocity 10 m/s. In 10 s the velocity changes to 10 m/s northwards. The average acceleration during this time is:
A)
\[\sqrt{2}\,m/{{s}^{2}}N-W\]
done
clear
B)
\[\frac{1}{2}\,m/{{s}^{2}}\,N-W\]
done
clear
C)
\[\sqrt{2}\,m/{{s}^{2}}\,N-E\]
done
clear
D)
0
done
clear
View Answer play_arrow
The point from where a ball is projected is taken as the origin of the coordinate axes. The\[x\]and y components of its displacement are given by\[x=6t\]and \[y=8t-5{{t}^{2}}.\]What is the velocity of projection?
A)
\[~6\text{ }m{{s}^{-1}}\]
done
clear
B)
\[~8\text{ }m{{s}^{-1}}\]
done
clear
C)
\[10\text{ }m{{s}^{-1}}\]
done
clear
D)
\[~14\text{ }m{{s}^{-1}}\]
done
clear
View Answer play_arrow
A ball is projected upwards from the top of a tower with a velocity of 50 m/s making in angle of \[30{}^\circ \] with the horizontal. The height of the tower is 70 m. After how many seconds from the instant of throwing, will the ball reach the ground? (Take\[g=10\,m/{{s}^{2}}\])
A)
2s
done
clear
B)
5 s
done
clear
C)
7s
done
clear
D)
9 s
done
clear
View Answer play_arrow
A particle is projected from the ground with an initial speed of v at an angle\[\theta \] with horizontal. The magnitude of the average velocity of the particle between its point of projection and highest point of trajectory is:
A)
\[\frac{v}{2}\sqrt{1+2{{\cos }^{2}}\theta }\]
done
clear
B)
\[\frac{v}{2}\sqrt{1+2{{\cos }^{2}}\theta }\]
done
clear
C)
\[\frac{v}{2}\sqrt{1+3{{\cos }^{2}}\theta }\]
done
clear
D)
\[v\cos \theta \]
done
clear
View Answer play_arrow
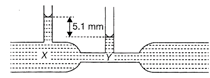
The diagram shows a venturimeter through which water is flowing. The speed of water at X is\[2\,cm/{{s}^{-1}}.\]. The speed of water at Y (taking\[g=1000\,cm/{{s}^{-2}}\]) is:
A)
\[23\,cm/{{s}^{-1}}\]
done
clear
B)
\[32\,cm/{{s}^{-1}}\]
done
clear
C)
\[101\,cm/{{s}^{-1}}\]
done
clear
D)
\[1024\,cm/{{s}^{-1}}\]
done
clear
View Answer play_arrow
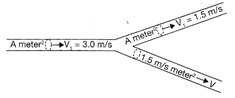
An incompressible liquid flows through a horizontal tube as shown in the adjoining figure. Then the velocity v of the fluid is:
A)
3.0 m/s
done
clear
B)
1.5 m/s
done
clear
C)
1.0 m/s
done
clear
D)
2.25 m/s
done
clear
View Answer play_arrow
A message signal of frequency \[{{\omega }_{m}}\]is superposed or a carrier wave of frequency \[{{\omega }_{c}}\]to get an amplitude modulated wave (AM), The frequency of the AM wave will be
A)
\[{{\omega }_{m}}\]
done
clear
B)
\[{{\omega }_{c}}\]
done
clear
C)
\[\frac{{{\omega }_{c}}+{{\omega }_{m}}}{2}\]
done
clear
D)
\[\frac{{{\omega }_{c}}-{{\omega }_{m}}}{2}\]
done
clear
View Answer play_arrow
Two concentric spheres of radii R and r have similar charges with equal surface densities \[(\sigma ).\]What is the electric potential at their common center?
A)
\[\frac{\sigma }{{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{\sigma }{{{\varepsilon }_{0}}}(R-r)\]
done
clear
C)
\[\frac{\sigma }{{{\varepsilon }_{0}}}(R+r)\]
done
clear
D)
None of the above.
done
clear
View Answer play_arrow
A solid conducting sphere having a charge Q is surrounded by an uncharged concentric conducting spherical shell. The potential difference between the surface of solid sphere and shell is V. The shell is now given a charge -3Q. The new potential difference between the same surfaces will be
A)
V
done
clear
B)
2V
done
clear
C)
4V
done
clear
D)
-IV
done
clear
View Answer play_arrow
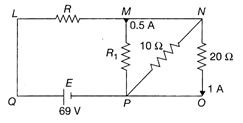
In the circuit shown in the figure
A)
The current through NP is 0.5 A
done
clear
B)
The value of \[{{R}_{1}}=20\Omega \]
done
clear
C)
The value of \[R=14\Omega \]
done
clear
D)
The potential difference across R = 49 V
done
clear
View Answer play_arrow
A charged particles a uniform magnetic field with velocity vector making an angle of \[30{}^\circ \] with the magnetic field. The particle describes a helical trajectory of pitch \[x.\]The radius of the helix is
A)
\[\frac{x}{2\pi }\]
done
clear
B)
\[\frac{x}{2\sqrt{2}\pi }\]
done
clear
C)
\[\frac{x}{2\sqrt{3}\pi }\]
done
clear
D)
\[\frac{\sqrt{3}x}{2\pi }\]
done
clear
View Answer play_arrow
A ball is projected horizontally from the top of a tower with a velocity of \[4\text{ }m{{s}^{-1}}.\] The velocity of I the ball after\[0.7\text{ }s\,(g=10\,m{{s}^{-2}})\]
A)
\[11\text{ }m{{s}^{-1}}\]
done
clear
B)
\[~10\text{ }m{{s}^{-1}}\]
done
clear
C)
\[~8\text{ }m{{s}^{-1}}\]
done
clear
D)
\[~3\text{ }m{{s}^{-1}}\]
done
clear
View Answer play_arrow
A particle of mass m strikes a wall at an angle of incidence \[60{}^\circ \] with velocity b elastically. The impulse is
A)
\[mv\]
done
clear
B)
\[m\frac{v}{2}\]
done
clear
C)
\[-2\,m\,v\]
done
clear
D)
0
done
clear
View Answer play_arrow
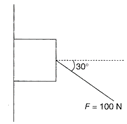
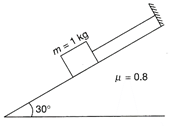
A force of 100 N is applied on a block of mass 3 kg as shown in the figure. The coefficient of friction between the wall and block is\[1/4\] .The frictional force acting on the block is
A)
20 N downwards
done
clear
B)
25 N upwards
done
clear
C)
15 N downwards
done
clear
D)
20 N upwards
done
clear
View Answer play_arrow
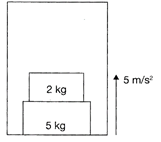
Find the force exerted by 5 kg block on floor of lift, as shown in the figure. (Take\[g=10\text{ }m/{{s}^{2}}\])
A)
100 N
done
clear
B)
200 N
done
clear
C)
105 N
done
clear
D)
210 N
done
clear
View Answer play_arrow
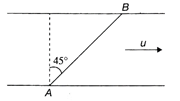
A man wants to reach point B on the opposite bank of a river flowing at a speed as shown in the figure. What minimum speed relative to water should the man have so that he can reach point B?
A)
\[u\sqrt{2}\]
done
clear
B)
\[\frac{u}{\sqrt{2}}\]
done
clear
C)
\[2u\]
done
clear
D)
\[\frac{u}{2}\]
done
clear
View Answer play_arrow
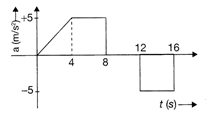
The acceleration of a train between two stations 2 km apart is shown in the figure. The maximum speed of the train is:
A)
60 m/s
done
clear
B)
30 m/s
done
clear
C)
120 m/s
done
clear
D)
90 m/s
done
clear
View Answer play_arrow
A force \[\vec{F}=3\hat{i}-2\hat{j}+4\hat{k}\]displaces an object from a point P (1, 1, 1) to another point Q (2, 0, 3). The work done by force is:
A)
10 J
done
clear
B)
12 J
done
clear
C)
13 J
done
clear
D)
None of these
done
clear
View Answer play_arrow
The figure shows a block of mass m kept on inclined plane with inclination \[\theta .\] The tension in the string is:
A)
8 N
done
clear
B)
10 N
done
clear
C)
0.8 N
done
clear
D)
0
done
clear
View Answer play_arrow
The efficiency of a Carnot engine operating between temperatures of \[100{}^\circ C\] and \[-23{}^\circ C\] will be
A)
\[\frac{100-23}{273}\]
done
clear
B)
\[\frac{100+23}{273}\]
done
clear
C)
\[\frac{100+23}{100}\]
done
clear
D)
\[\frac{100-23}{100}\]
done
clear
View Answer play_arrow
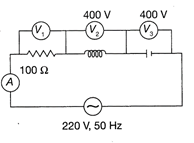
In the given circuit, the readings of the voltmeter \[{{V}_{1}}\] and the ammeter A are
A)
220 V, 2.2 A
done
clear
B)
110 V, 1.1 A
done
clear
C)
220 V, 1.1 A
done
clear
D)
110 V, 2.2 A
done
clear
View Answer play_arrow
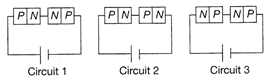
Two identical p-n junctions may be connected in series with a battery in three ways as shown. The potential drops across the two p-n junctions are equal in
A)
circuit 1 and circuit 2.
done
clear
B)
circuit 2 and circuit 3.
done
clear
C)
circuit 3 and circuit 1.
done
clear
D)
circuit 1 only.
done
clear
View Answer play_arrow
A circular coil of radius 0.1 m has 80 turns of wire. If the magnetic field through the coil increases from 0 to 2 Tesla in 0.4 sec and the coil is connected to a\[11\,\Omega \] resistor, what is the current (in A) through the resistor during the 0.4 sec interval?
A)
\[\left( \frac{8}{7} \right)A\]
done
clear
B)
\[\left( \frac{7}{8} \right)A\]
done
clear
C)
\[8\,A\]
done
clear
D)
\[7\,A\]
done
clear
View Answer play_arrow
In a uniform magnetic field of induction B, a wire in the form of a semi-circle of radius r rotates about the diameter of the circle with angular frequency\[\omega .\]The axis of rotation is perpendicular to the field. If the total resistance of the circuit is R, the mean power generated per period of rotation is:
A)
\[\frac{{{(B\pi r\omega )}^{2}}}{2R}\]
done
clear
B)
\[\frac{B\pi {{r}^{2}}\omega }{2R}\]
done
clear
C)
\[\frac{B\pi {{r}^{2}}\omega }{8R}\]
done
clear
D)
\[\frac{{{(B\pi r{{\omega }^{2}})}^{2}}}{8R}\]
done
clear
View Answer play_arrow
A galvanometer is connected to the secondary coil. The galvanometer shows an instantaneous maximum deflection of 7 divisions when a current is started in the primary coil of the solenoid. Now, if the primary coil is rotated through \[180{}^\circ C\], then the new instantaneous maximum deflection will be:
A)
7 divisions,
done
clear
B)
14 divisions.
done
clear
C)
0 divisions,
done
clear
D)
21 divisions.
done
clear
View Answer play_arrow
In Young's double experiment carried out with light of wavelength \[\lambda =5000\,\overset{\text{o}}{\mathop{\text{A}}}\,,\]the distance between the slits is 0.2 mm and the screen is at 200 cm from the plane of slits. The central maximum is at \[x=0.\]The third maximum will be at x equal to:
A)
1.67 cm
done
clear
B)
1.5 cm
done
clear
C)
0.5 cm
done
clear
D)
5.0 cm
done
clear
View Answer play_arrow
Two uniform strings A and B made of steel an made to vibrate under the same tension. If the firs overtone of A is equal to the second overtone of I. and if the radius of A is twice that of B, the ratio of the lengths of the strings is:
A)
2:1
done
clear
B)
3:2
done
clear
C)
3:4
done
clear
D)
1:3
done
clear
View Answer play_arrow
A cylindrical tube partially filled with water is in resonance with a tuning fork when height of air column is 0.1 m. When the level of water is lowered, the resonance is again observed at 0.35 m. The end correction is:
A)
0.025 m
done
clear
B)
0.015 m
done
clear
C)
0.001 m
done
clear
D)
0.002 m
done
clear
View Answer play_arrow
The angular velocities of three bodies in simple harmonic motion are \[{{\omega }_{1}},{{\omega }_{2}},{{\omega }_{3}}\]with respective amplitudes as\[{{A}_{1}},{{A}_{2}},{{A}_{3}}\]If all the three bodies have the same mass and maximum speeds, then:
A)
\[{{A}_{1}}{{\omega }_{1}}={{A}_{2}}{{\omega }_{2}}={{A}_{3}}{{\omega }_{3}}\]
done
clear
B)
\[{{A}_{1}}\omega _{1}^{2}={{A}_{2}}\omega _{2}^{2}={{A}_{3}}\omega _{3}^{2}\]
done
clear
C)
\[A_{1}^{2}{{\omega }_{1}}=A_{2}^{2}{{\omega }_{2}}=A_{3}^{2}{{\omega }_{3}}\]
done
clear
D)
\[A_{1}^{2}\sqrt{{{\omega }_{1}}}=A_{2}^{2}\sqrt{{{\omega }_{2}}}=A_{3}^{2}\sqrt{{{\omega }_{3}}}\]
done
clear
View Answer play_arrow
The root mean square speed of the gas molecules is 300 m/s. What will be the root mean square speed of the molecules if the atomic weight is doubled and absolute temperature is halved?
A)
300 m/s
done
clear
B)
150 m/s
done
clear
C)
600 m/s
done
clear
D)
75 m/s
done
clear
View Answer play_arrow
A gas is compressed at a constant pressure of \[50\text{ }N/{{m}^{2}}\] from volume of 10 m3 to a volume of \[4\text{ }{{m}^{3}}.\] Energy of 100 J is then added to the gas by heating. Its internal energy is
A)
increased by 400 J
done
clear
B)
increased by 200 J
done
clear
C)
increased by 100 J
done
clear
D)
decreased by 200 J
done
clear
View Answer play_arrow
When the temperature of a black body increases, it is observed that the wavelength corresponding to maximum energy changes from \[0.26\,\mu m\]to \[0.13\,\mu m.\] The ratio of the emissive powers of the body at the respective temperatures is:
A)
\[\frac{16}{1}\]
done
clear
B)
\[\frac{4}{1}\]
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
\[\frac{1}{16}\]
done
clear
View Answer play_arrow
70 calories of heat is required to raise the temperature of 2 moles of an ideal gas at constant pressure from \[40{}^\circ C\] to \[45{}^\circ C\] (R = 2\[cal/mol{{\,}^{-0}}C\]). The gas may be
A)
\[{{H}_{2}}\]
done
clear
B)
\[He\]
done
clear
C)
\[C{{O}_{2}}\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
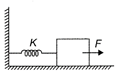
A block attached to a spring, pulled by a constant horizontal force, is kept on a smooth surface as shown in the figure. Initially, the spring is in the natural state. Then the maximum positive work that the applied force F can do is (given that string does not break)
A)
\[\frac{{{F}^{2}}}{K}\]
done
clear
B)
\[\frac{2{{F}^{2}}}{K}\]
done
clear
C)
\[\infty \]
done
clear
D)
\[\frac{{{F}^{2}}}{2K}\]
done
clear
View Answer play_arrow
Two particles A and B initially at rest, move towards each other under mutual force of attraction. At the instant when the speed of A is V and the speed of B is 2F, the speed of the center of mass of the system is
A)
3V
done
clear
B)
V
done
clear
C)
1.5 V
done
clear
D)
0
done
clear
View Answer play_arrow
The dimension of K in the wave equation given by\[y=A\sin \omega \left( \frac{x}{V}-K \right)\]are
A)
LT
done
clear
B)
T
done
clear
C)
\[{{T}^{-1}}\]
done
clear
D)
\[{{T}^{2}}\]
done
clear
View Answer play_arrow
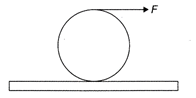
A force F is applied at the top of a ring of mass M and radius R placed on a rough horizontal surface as shown in the figure. Friction is sufficient to prevent slipping. The frictional force acting on the ring is
A)
\[\frac{F}{2}\]towards right
done
clear
B)
\[\frac{F}{3}\]towards left
done
clear
C)
\[\frac{2F}{3}\] towards right
done
clear
D)
0
done
clear
View Answer play_arrow
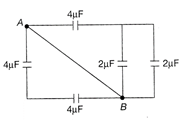
In the circuit shown in the figure. The effective capacitance between A and B is:
A)
\[3\,\mu F\]
done
clear
B)
\[2\,\mu F\]
done
clear
C)
\[4\,\mu F\]
done
clear
D)
\[8\,\mu F\]
done
clear
View Answer play_arrow
The maximum wavelength of a beam of light that can be used to produce photoelectric effect on a metal is 250 nm. The energy of the electrons in joule emitted from the surface of the metal when a beam of light of wavelength 200 nm is used, is
A)
\[89.61\times {{10}^{-22}}\]
done
clear
B)
\[69.81\times {{10}^{-22}}\]
done
clear
C)
\[18.96\times {{10}^{-20}}\]
done
clear
D)
\[19.86\times {{10}^{-20}}\]
done
clear
View Answer play_arrow
If\[{{\lambda }_{1}}\]and\[{{\lambda }_{2}}\]denote the wavelengths of de Broglie waves for electrons in Bohr's first and second orbits in the hydrogen atom, then\[{{\lambda }_{1}}/{{\lambda }_{2}}\]will be:
A)
2
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
4
done
clear
D)
\[\frac{1}{4}\]
done
clear
View Answer play_arrow
If the series limit wavelength of the Lyman series for hydrogen atom is \[912\overset{\text{o}}{\mathop{\text{A}}}\,\text{,}\] then the series limit wave- length for the Balmer series for the hydrogen atom is:
A)
\[912\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[912\,\times 2\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[912\,\times 4\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[\frac{912}{2}\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
The threshold frequency for a certain photosensitive metal is \[{{V}_{0}}.\]When it is illuminated by light of frequency \[v=2{{V}_{0}},\]the maximum velocity of photoelectrons is \[{{\upsilon }_{0}}.\]What will be the maximum velocity of the photoelectrons when the same metal is illuminated by light of frequency\[v=5{{v}_{0}}?\]
A)
\[\sqrt{2}{{v}_{0}}\]
done
clear
B)
\[2{{v}_{0}}\]
done
clear
C)
\[2\sqrt{2}{{v}_{0}}\]
done
clear
D)
\[4{{v}_{0}}\]
done
clear
View Answer play_arrow
Which energy state of doubly ionized lithium \[(L{{i}^{++}})\]has the same energy as that of the ground state of hydrogen? Given Z for lithium = 3.
A)
\[~n=1\]
done
clear
B)
\[~n=2\]
done
clear
C)
\[~n=3\]
done
clear
D)
\[~n=4\]
done
clear
View Answer play_arrow
A current of \[(1/4\pi )\]ampere is flowing in a long straight conductor. The line integral of magnetic field around a closed path enclosing the current carrying conductor is:
A)
\[{{10}^{-7}}\] weber per meter
done
clear
B)
\[4\pi \times {{10}^{-7}}\] weber per meter
done
clear
C)
\[16{{\pi }^{2}}\times {{10}^{-7}}\]weber per meter
done
clear
D)
0
done
clear
View Answer play_arrow
One mole of an ideal gas is allowed to expand reversibly and adiabatically from a temperature of \[27{}^\circ C\]. The work done is 3 kJ. The final temperature of the gas is equal to\[[{{C}_{v}}=20k{{J}^{-1}}\,]:\]
A)
450 K
done
clear
B)
150 K
done
clear
C)
600 K
done
clear
D)
300 K
done
clear
View Answer play_arrow
The compound used as an anti-freeze is
A)
Propanol
done
clear
B)
Ethanol
done
clear
C)
Methanol
done
clear
D)
Glycerol
done
clear
View Answer play_arrow
The main component of green oil fraction of coal tar is
A)
Pyrrole
done
clear
B)
Phenol
done
clear
C)
Naphthalene
done
clear
D)
Benzene
done
clear
View Answer play_arrow
Which of the following is not a polyamide?
A)
Protein
done
clear
B)
Glyptal
done
clear
C)
Nylon 6
done
clear
D)
Nylon 66
done
clear
View Answer play_arrow
The sulphide ore of copper is concentrated by
A)
Magnetic method
done
clear
B)
Froth floating method
done
clear
C)
Chemical method
done
clear
D)
None of these
done
clear
View Answer play_arrow
Le Chatelier's principle is applicable to
A)
Homogeneous reaction
done
clear
B)
Heterogeneous reaction
done
clear
C)
Irreversible reaction
done
clear
D)
System in equilibrium
done
clear
View Answer play_arrow
Normality of \[\text{0}\text{.3 M }{{\text{H}}_{\text{3}}}\text{P}{{\text{O}}_{\text{4}}}\]solution is
A)
0.6 N
done
clear
B)
0.1 N
done
clear
C)
0.3 N
done
clear
D)
0.9 N
done
clear
View Answer play_arrow
Physical adsorption is inversely proportional to
A)
Temperature
done
clear
B)
Concentration
done
clear
C)
Volume
done
clear
D)
All of these
done
clear
View Answer play_arrow
One gram of oxygen at NTP occupies a volume of
A)
0.7 L
done
clear
B)
2.8 L
done
clear
C)
1.4 L
done
clear
D)
3.5 L
done
clear
View Answer play_arrow
Bauxite is concentrated by
A)
Chemical method
done
clear
B)
Magnetic method
done
clear
C)
Froth floating process
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[{{\text{P}}_{2}}{{O}_{5}}\]is an anhydride of
A)
\[HP{{O}_{3}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
C)
\[{{H}_{2}}{{P}_{2}}{{O}_{7}}\]
done
clear
D)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
View Answer play_arrow
Which of the following shows electrical conduction?
A)
Cl
done
clear
B)
Na
done
clear
C)
P
done
clear
D)
Diamond
done
clear
View Answer play_arrow
\[{{H}_{2}}+C{{l}_{2}}\to 2HCl,\Delta H=-194\,kJ\]. In the above reaction the heat formation of HC1 is
A)
+97 kJ
done
clear
B)
-97 kJ
done
clear
C)
+10 kJ
done
clear
D)
-10 kJ
done
clear
View Answer play_arrow
The number of unpaired electron in \[1{{s}^{2}},\,2{{s}^{2}}\,2{{p}^{3}}\]is
A)
2
done
clear
B)
1
done
clear
C)
3
done
clear
D)
0
done
clear
View Answer play_arrow
Ammonia and sodium hypochlorite reacts to produce
A)
NO
done
clear
B)
\[N{{H}_{2}}N{{H}_{2}}\]
done
clear
C)
\[N{{H}_{2}}OH\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
Oleum is a mixture of
A)
\[{{H}_{2}}{{S}_{2}}{{O}_{3}}+S{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}+S{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}+S{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{4}}+S{{O}_{3}}\]
done
clear
View Answer play_arrow
When primary amine is heated with \[C{{S}_{2}}\]in the presence of excess of mercuric chloride, it produces isothiocyanate. This reaction is known as
A)
Carbylamine reaction
done
clear
B)
Hofmann bromamide reaction
done
clear
C)
Hofmann mustard oil reaction
done
clear
D)
Hinsberg's reaction
done
clear
View Answer play_arrow
The law of equilibrium was first of all given by
A)
Boyle
done
clear
B)
Guldberg
done
clear
C)
Waage
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
Internal energy does not include
A)
Rotational energy
done
clear
B)
Vibrational energy
done
clear
C)
Nuclear energy
done
clear
D)
Energy due to gravitational pull
done
clear
View Answer play_arrow
On heating one end of a piece of metal, the other end becomes hot because of
A)
Resistance of the metal.
done
clear
B)
Mobility of atoms in the metal.
done
clear
C)
Minor perturbation in the energy of the metal.
done
clear
D)
Energized electrons moving to the other end.
done
clear
View Answer play_arrow
Nessler's reagent is used for the test of
A)
\[S{{O}_{4}}\]
done
clear
B)
\[C{{O}_{3}}\]
done
clear
C)
\[N{{H}^{+}}_{4}\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
Benzene when oxidized with\[{{V}_{2}}{{O}_{5}}\]produces
A)
Benzoic acid
done
clear
B)
Toluene
done
clear
C)
Benzaldehyde
done
clear
D)
Maleic anhydride
done
clear
View Answer play_arrow
The metal that does not displace hydrogen from an acid is
A)
Zn
done
clear
B)
Ca
done
clear
C)
Al
done
clear
D)
Hg
done
clear
View Answer play_arrow
During electrolysis the species that migrate to cathode are
A)
Cation
done
clear
B)
Anion
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The number of a and n bonds present in l-buten-3-yne are
A)
\[7\sigma \]and\[5\pi \]
done
clear
B)
\[6\sigma \] and\[4\pi \]
done
clear
C)
\[6\sigma \]and\[6\pi \]
done
clear
D)
\[7\sigma \]and \[3\pi \]
done
clear
View Answer play_arrow
A solution with pH of 2 has higher acidity than a solution with a pH of 6 is
A)
1000
done
clear
B)
100
done
clear
C)
10000
done
clear
D)
100000
done
clear
View Answer play_arrow
The most stable carbonium ion is
A)
\[{{C}_{6}}{{H}_{5}}\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
B)
\[{{(C{{H}_{3}})}_{3}}\overset{+}{\mathop{C}}\,\]
done
clear
C)
\[C{{H}_{3}}-\underset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
D)
\[{{({{C}_{6}}{{H}_{5}})}_{3}}\overset{+}{\mathop{C}}\,\]
done
clear
View Answer play_arrow
Dehydration of alcohol is an example of
A)
Redox reaction
done
clear
B)
Elimination reaction
done
clear
C)
Addition reaction
done
clear
D)
Substitution reaction
done
clear
View Answer play_arrow
Total number of electrons in all the p-orbitals of bromine will be
A)
5
done
clear
B)
7
done
clear
C)
15
done
clear
D)
17
done
clear
View Answer play_arrow
Duralumin is an alloy of
A)
Mg and Cu
done
clear
B)
Al and Cu
done
clear
C)
Al and Mg
done
clear
D)
Al, Mg, Mn and Cu
done
clear
View Answer play_arrow
The most abundant metal in the earth's crust is
A)
Al
done
clear
B)
Fe
done
clear
C)
Na
done
clear
D)
Ca
done
clear
View Answer play_arrow
The number of moles of oxygen obtained by the electrolytic decomposition of 90 g water is
A)
2.5
done
clear
B)
5
done
clear
C)
6.5
done
clear
D)
1.25
done
clear
View Answer play_arrow
A closed container contains equal number of oxygen and hydrogen molecules at a total pressure of 740 mm. If oxygen is removed from the system then the pressure will
A)
Become 1/9 of 740 mm
done
clear
B)
Become half of 740 mm
done
clear
C)
Remains unchanged
done
clear
D)
Become double of 740 mm
done
clear
View Answer play_arrow
The most acidic oxide is
A)
CaO
done
clear
B)
MgO
done
clear
C)
\[N{{a}_{2}}O\]
done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
View Answer play_arrow
Flux is used to remove
A)
Metal oxide
done
clear
B)
Silica
done
clear
C)
Silica and metal oxide
done
clear
D)
Impurities form ore
done
clear
View Answer play_arrow
The first law of thermodynamics is represented by the equation
A)
\[\Delta E =Q-W\]
done
clear
B)
\[\Delta E =O+W\]
done
clear
C)
\[Q=W+\Delta E \]
done
clear
D)
\[W=Q+\Delta E \]
done
clear
View Answer play_arrow
Ionization constant of acetic acid is \[1.8\times {{10}^{-5}}.\]The concentration of\[{{H}^{+}}\]ions in 0.1 M solution is
A)
\[1.8\times {{10}^{-3}}M\]
done
clear
B)
\[1.6\times {{10}^{-5}}M\]
done
clear
C)
\[CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
D)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
View Answer play_arrow
Epsom salt is
A)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[BaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
C)
\[CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
D)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
View Answer play_arrow
Paracetamol is an
A)
Analgesic
done
clear
B)
Antimalarial
done
clear
C)
Antipyretics
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
Acetone and acetaldehyde can be identified by
A)
Bromoform test
done
clear
B)
Molisch's test
done
clear
C)
Schiff's test
done
clear
D)
Iodoform test
done
clear
View Answer play_arrow
The difference in \[{{\,}_{17}}C{{l}^{35}}\]and \[{{\,}_{17}}C{{l}^{37}}\]is of
A)
Atomic number
done
clear
B)
Number of neutron
done
clear
C)
Number of proton
done
clear
D)
Number of electron
done
clear
View Answer play_arrow
The enthalpy of combustion of \[{{C}_{6}}{{H}_{6}}\]is 3250 kJ when 0.39 g of \[{{C}_{6}}{{H}_{6}}\]is burnt in excess of oxygen in an open vessel, the amount of heat evolved is
A)
8.32 kJ
done
clear
B)
12.34 kJ
done
clear
C)
16.25 kJ
done
clear
D)
24.74 kJ
done
clear
View Answer play_arrow
\[{{C}_{6}}{{H}_{6}}\]on treatment with KOH produces
A)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}OH\]
done
clear
C)
\[{{C}_{6}}{{H}_{3}}C{{l}_{3}}\]
done
clear
D)
\[~{{C}_{6}}{{H}_{6}}C{{l}_{4}}\]
done
clear
View Answer play_arrow
The movement of colloidal particles towards oppositely charged electrodes of passing electricity is known as
A)
Tyndall effect
done
clear
B)
Cataphoresis
done
clear
C)
Brownian movement
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Which is the colligative property?
A)
Refractive index
done
clear
B)
Osmotic pressure
done
clear
C)
Surface tension
done
clear
D)
Viscosity
done
clear
View Answer play_arrow
The common oxidation state of the elements of lanthanide series is
A)
+1
done
clear
B)
+3
done
clear
C)
+2
done
clear
D)
+4
done
clear
View Answer play_arrow
The pH of pure water at \[80{}^\circ C\] will be
A)
=7
done
clear
B)
< 7
done
clear
C)
> 7
done
clear
D)
None of these
done
clear
View Answer play_arrow
Producer gas is a mixture of
A)
\[{{H}_{2}}+{{N}_{2}}\]
done
clear
B)
\[CO+{{N}_{2}}\]
done
clear
C)
\[CO+{{H}_{2}}\]
done
clear
D)
\[CO+C{{O}_{2}}\]
done
clear
View Answer play_arrow
A compound has C = 40%, H =13.33% and N = 46.67. The empirical formula is
A)
\[C{{H}_{4}}N\]
done
clear
B)
\[C{{H}_{4}}{{H}_{4}}\]
done
clear
C)
\[C{{H}_{2}}N\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}N\]
done
clear
View Answer play_arrow
This alcohol manufactured from water gas is
A)
Isobutanol
done
clear
B)
Methanol
done
clear
C)
Ethanol
done
clear
D)
Butanol
done
clear
View Answer play_arrow
In his classic experiments on pea plants, Mendel did not use.
A)
Pod colour
done
clear
B)
Pod length
done
clear
C)
Flower position
done
clear
D)
Flower colour
done
clear
View Answer play_arrow
Which one of the following enzyme is responsible for transcription of snRNA in eukaryotes?
A)
RNA polymerase I
done
clear
B)
RNA polymerase II
done
clear
C)
RNA polymerase III
done
clear
D)
DNA polymerase
done
clear
View Answer play_arrow
Perisperm is
A)
Persistent nucellus
done
clear
B)
Persistent endosperm
done
clear
C)
Persistent sepals
done
clear
D)
Persistent synergids
done
clear
View Answer play_arrow
The membrane bound vesicular structure formed from Golgi apparatus and contain hydrolases.
A)
Ribosomes
done
clear
B)
Lysosomes
done
clear
C)
Mesosomes
done
clear
D)
Vacuoles
done
clear
View Answer play_arrow
The chitinous exoskeleton of arthropods is formed by the polymerization of
A)
D-Glucosamine
done
clear
B)
N-Acetylglucosamine
done
clear
C)
Lipoglycans
done
clear
D)
Keratin sulphate and chondroitin sulphate
done
clear
View Answer play_arrow
Among china rose, mustard, brinjal, potato, guava, cucumber, onion and tulip, how many plants have inferior ovary?
A)
Six
done
clear
B)
Three
done
clear
C)
Two
done
clear
D)
Four
done
clear
View Answer play_arrow
The cell junctions called tight, adhering and gap junctions are found in
A)
Connective tissue
done
clear
B)
Epithelial tissue
done
clear
C)
Neural tissue
done
clear
D)
Muscular tissue
done
clear
View Answer play_arrow
Which of the following immunoglobulins are abundantly found in colostrum?
A)
IgM
done
clear
B)
IgA
done
clear
C)
IgG
done
clear
D)
IgD
done
clear
View Answer play_arrow
In mammalian eye, the 'fovea9 is the centre of the visual field, where
A)
The optic nerve leaves the eye
done
clear
B)
Only rods are present
done
clear
C)
More rods than cones are found
done
clear
D)
Visual acuity is maximum
done
clear
View Answer play_arrow
Doctors use stethoscope to hear the sounds produced during each cardiac cycle. The second sound is heard when
A)
Ventricular walls vibrate due to gushing of blood from atria.
done
clear
B)
Semilunar valves close down after the blood flows into vessels from ventricles.
done
clear
C)
AV node receives signal from SA node.
done
clear
D)
AV valves open up
done
clear
View Answer play_arrow
What is ploidy of coconut water
A)
n
done
clear
B)
2n
done
clear
C)
3n
done
clear
D)
4n
done
clear
View Answer play_arrow
The following are restriction enzymes except
A)
BamHI
done
clear
B)
PvuII
done
clear
C)
PstI
done
clear
D)
pBR322
done
clear
View Answer play_arrow
Which of the following structures is not found in a plant cell?
A)
Ribosome
done
clear
B)
Mesosome
done
clear
C)
Plasma membrane
done
clear
D)
Nuclear envelope
done
clear
View Answer play_arrow
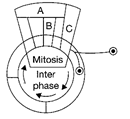
Given below is a schematic break-up of the phases/ stages of cell cycle. Which one of the following is the correct indication of the stage/phase in the cell cycle?
A)
C: Karyokinesis
done
clear
B)
D: Synthetic phase
done
clear
C)
A: Cytokinesis
done
clear
D)
B: Metaphase
done
clear
View Answer play_arrow
Transpiration is affected by
A)
Temperature
done
clear
B)
Light
done
clear
C)
Wind speed
done
clear
D)
All of the above
done
clear
View Answer play_arrow
The imperfect fungi which are decomposers of litter and helps in mineral cycling belongs to
A)
Basidiomycetes
done
clear
B)
Phycomycetes
done
clear
C)
Ascomycetes
done
clear
D)
Deuteromycetes
done
clear
View Answer play_arrow
Which of the following is not a bacterial disease?
A)
Tetanus
done
clear
B)
Cholera
done
clear
C)
Typhoid
done
clear
D)
Cysticercosis
done
clear
View Answer play_arrow
The segment of DNA which participates in crossing over is
A)
Recon
done
clear
B)
Operon
done
clear
C)
Muton
done
clear
D)
Cistron
done
clear
View Answer play_arrow
Pick up the wrong statement:
A)
Protista has photo synthetic and heterotrophic modes of nutrition.
done
clear
B)
Some fungi are edible.
done
clear
C)
Nuclear membrane is present in Monera.
done
clear
D)
Cell wall is absent in Animalia.
done
clear
View Answer play_arrow
Peripatus is a connecting link between
A)
Mollusca and Echinodermata
done
clear
B)
Annelida and Arthropoda
done
clear
C)
Coelenterata and Porifera
done
clear
D)
Ctenophora and Platyhelminthes
done
clear
View Answer play_arrow
Which of the following events is not associated with ovulation in human female?
A)
Full development of Graafian follicle
done
clear
B)
Release of secondary oocyte
done
clear
C)
LH surge
done
clear
D)
Decrease in estradiol
done
clear
View Answer play_arrow
Which one of the following is the correct matching of three items and their grouping category?
A)
Items Group ilium, ischium, pubis coxal bones of pelvic girdle
done
clear
B)
Items Group actin, myosin, rhodopsin muscle proteins
done
clear
C)
Items Group cytosine, uracil, thiamine pyrimidines
done
clear
D)
Items Group malleus, incus, cochlea ear ossicles
done
clear
View Answer play_arrow
Match the following list of microbes and their importance:
[A] Saccharomyes (i) Production of cerevisiae immunosuppressive agents [B] Monascus Purpureus (ii) Ripening of Swiss cheese [C] Trichoderma polysporum (iii) Commercial production of ethanol [D] Propionibacterium shermanii (iv) Production of blood-cholesterol lowering agents
A)
A-iv, B-iii, C-ii, D-i
done
clear
B)
A-iv, B-ii, C-i, D-iii
done
clear
C)
A-iii, B-i, C-iv, D-ii
done
clear
D)
A-iii, B-iv, C-i, D-ii
done
clear
View Answer play_arrow
The UN conference of Parties on climate change in the year 2012 was held at
A)
Doha
done
clear
B)
Lima
done
clear
C)
Warsaw
done
clear
D)
Durban
done
clear
View Answer play_arrow
If you suspect major deficiency of antibodies in a person, to which of the following would you look for confirmatory evidence?
A)
Serum albumins
done
clear
B)
Haemocytes
done
clear
C)
Serum globulins
done
clear
D)
Fibrinogen in plasma
done
clear
View Answer play_arrow
Chromatophores take part in
A)
Growth
done
clear
B)
Movement
done
clear
C)
Respiration
done
clear
D)
Photosynthesis
done
clear
View Answer play_arrow
A terminator codon which is called Amber is
A)
UGA
done
clear
B)
UAG
done
clear
C)
UAA
done
clear
D)
UUU
done
clear
View Answer play_arrow
During ecological succession which of the following takes place?
A)
The establishment of a new biotic community is very fast in its primary phase.
done
clear
B)
The numbers and types of animals remain constant.
done
clear
C)
The changes lead to a community that is in near equilibrium with the environment and is called pioneer community.
done
clear
D)
The gradual and predictable change in species composition occurs in a given area.
done
clear
View Answer play_arrow
In photorespiration the substrate is
A)
PEP
done
clear
B)
ATP
done
clear
C)
RuBP
done
clear
D)
RuBisCO
done
clear
View Answer play_arrow
Which of the following pairs is not correctly matched?
A)
Mode of reproduction Example Rhizome Banana
done
clear
B)
Mode of reproduction Example Binary fission Sargassum
done
clear
C)
Mode of reproduction Example Conidia Penicillium
done
clear
D)
Mode of reproduction Example Offset Water hyacinth
done
clear
View Answer play_arrow
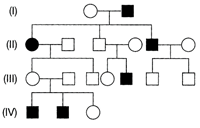
In the following human pedigree, the filled symbols represent the affected individual. Identify the type of given pedigree.
A)
X-linked recessive
done
clear
B)
Autosomal recessive
done
clear
C)
X-linked dominant
done
clear
D)
Autosomal dominant
done
clear
View Answer play_arrow
Which one of the following animals has two separate circulatory pathways?
A)
Lizard
done
clear
B)
Dolphin
done
clear
C)
Shark
done
clear
D)
Frog
done
clear
View Answer play_arrow
A staminode does not possess
A)
Filament
done
clear
B)
Anther
done
clear
C)
Male gametes
done
clear
D)
Pollen grains
done
clear
View Answer play_arrow
The phenomenon which shortens the vegetative period and hastens the flowering is known as
A)
Etiolation
done
clear
B)
Vernalization
done
clear
C)
Photoperiodism
done
clear
D)
Parthenocarpy
done
clear
View Answer play_arrow
A pleiotropic gene
A)
Is a gene evolved during Pliocene
done
clear
B)
Controls a trait only in combination with another gene
done
clear
C)
Controls multiple traits in an individual
done
clear
D)
Is expressed only in primitive plants
done
clear
View Answer play_arrow
Which of the following is not a function of the skeletal system?
A)
Storage of minerals
done
clear
B)
Production of body heat
done
clear
C)
Locomotion
done
clear
D)
Production of erythrocytes
done
clear
View Answer play_arrow
A jawless fish, which lays eggs in fresh water and whose 'ammocoetes larvae' after attaining metamorphosis and returns to the ocean is
A)
Myxine
done
clear
B)
Neomyxine
done
clear
C)
Petromyzon
done
clear
D)
Eptatretus
done
clear
View Answer play_arrow
Filiform apparatus is a characteristic feature of
A)
Nucellar embryo
done
clear
B)
Aleurone cell
done
clear
C)
Synergids
done
clear
D)
Generative cell
done
clear
View Answer play_arrow
Reduction in vascular tissue, mechanical tissue and cuticle is a characteristic of
A)
Mesophytes
done
clear
B)
Epiphytes
done
clear
C)
Hydrophytes
done
clear
D)
Xerophytes
done
clear
View Answer play_arrow
A temporary endocrine gland in human is
A)
Islets of Langerhans
done
clear
B)
Pineal body
done
clear
C)
Corpora allata
done
clear
D)
Corpus luteum
done
clear
View Answer play_arrow
Which of the following is not GMO?
A)
Golden rice
done
clear
B)
Tracy
done
clear
C)
Dolly
done
clear
D)
Bt brinjal
done
clear
View Answer play_arrow
Blue revolution is
A)
Increased exploitation of aquatic product
done
clear
B)
Increased grain production
done
clear
C)
Increased beef production
done
clear
D)
Increased milk production
done
clear
View Answer play_arrow
Point mutation involves
A)
Change in single base pair
done
clear
B)
Duplication
done
clear
C)
Deletion
done
clear
D)
Insertion
done
clear
View Answer play_arrow
Which one of the following hormones though synthesized elsewhere, is stored and released by the master gland?
A)
Luteinizing hormone
done
clear
B)
Prolactin
done
clear
C)
Melanocyte stimulating hormone
done
clear
D)
Antidiuretic hormone
done
clear
View Answer play_arrow
In a population that is growing as described by the logistic growth model.
A)
The number of individuals added per unit time is greatest when N is close to zero. (The key equation is \[\frac{dn}{dt}={{r}_{\max }}N\left( \frac{K-N}{K} \right)\])
done
clear
B)
The per capital growth rate (r) increases as N approaches K.
done
clear
C)
Population growth is zero when N equals K.
done
clear
D)
The population grows exponentially when K is small.
done
clear
View Answer play_arrow
Which of these ecosystems has the lowest net primary production per square metre?
A)
A salt marsh
done
clear
B)
An open ocean
done
clear
C)
A grassland
done
clear
D)
A tropical rain forest
done
clear
View Answer play_arrow
The primary dentition in human differs from permanent dentition in not having one of the following types of teeth
A)
Premolars
done
clear
B)
Molars
done
clear
C)
Incisors
done
clear
D)
Canine
done
clear
View Answer play_arrow
The colour of flower petal is due to
A)
Xanthophyll
done
clear
B)
Anthocyanin
done
clear
C)
Carotene
done
clear
D)
Phycoerythrin
done
clear
View Answer play_arrow
The body cells in cockroach discharge their nitrogenous waste in the haemolymph mainly in the form of
A)
Potassium urate
done
clear
B)
Urea
done
clear
C)
Calcium carbonate
done
clear
D)
Ammonia
done
clear
View Answer play_arrow
Which of the following biomolecules does have a phosphodiester bond?
A)
Monosaccharide in a polysaccharide
done
clear
B)
Amino acids in a polypeptide
done
clear
C)
Nucleic acids in a nucleotide
done
clear
D)
Fatty acids in a diglyceride
done
clear
View Answer play_arrow
The term 'linkage' was coined by
A)
T. Boveri
done
clear
B)
G. Mendel
done
clear
C)
W. Sutton
done
clear
D)
T. H. Morgan
done
clear
View Answer play_arrow
Which one is a wrong statement?
A)
Mucor has biflagellate zoospores.
done
clear
B)
Haploid endosperm is a typical feature of gymno sperms.
done
clear
C)
Brown algae have chlorophyll a and c, and fucoxanthin.
done
clear
D)
Archegonia are found in Bryophyta, Pteridophyta and Gymno sperms.
done
clear
View Answer play_arrow
Ectopic pregnancies are referred to as
A)
Implantation of embryo side other than uterus.
done
clear
B)
Implantation of defective embryo in the uterus.
done
clear
C)
Pregnancies terminated due to hormonal imbalance.
done
clear
D)
Pregnancies with genetic abnormality.
done
clear
View Answer play_arrow
Most animals that live in deep oceanic waters are
A)
Secondary consumers
done
clear
B)
Tertiary consumers
done
clear
C)
Detritivores
done
clear
D)
Primary consumers
done
clear
View Answer play_arrow
Which of the following disease is caused by a protozoan?
A)
Influenza
done
clear
B)
Babesiosis
done
clear
C)
Blastomycosis
done
clear
D)
Syphilis
done
clear
View Answer play_arrow
In which of the following interactions both partners are adversely affected?
A)
Predation
done
clear
B)
Parasitism
done
clear
C)
Mutualism
done
clear
D)
Competition
done
clear
View Answer play_arrow
Most cells cannot harness heat to perform work because
A)
Heat is not a form of energy.
done
clear
B)
Cells do not have much heat; they are relatively cool.
done
clear
C)
Heat can never be used to do work.
done
clear
D)
Heat denatures enzymes.
done
clear
View Answer play_arrow
A colour blind man marries a woman with normal sight who has no history of colour blindness in her family. What is the probability of their grandson being colour blind?
A)
1
done
clear
B)
Nil
done
clear
C)
0.25
done
clear
D)
0.5
done
clear
View Answer play_arrow
Most \[C{{O}_{2}}\]from catabolism is released during
A)
Glycolysis
done
clear
B)
The citric acid cycle
done
clear
C)
Lactate fermentation
done
clear
D)
Electron transport
done
clear
View Answer play_arrow
In which of the following both pairs have correct combination?
A)
Gaseous nutrient cycle Carbon and sulphur Sedimentary nutrient cycle Nitrogen and phosphorus
done
clear
B)
Gaseous nutrient cycle Nitrogen and sulphur Sedimentary nutrient cycle Carbon and phosphorus
done
clear
C)
Gaseous nutrient cycle Sulphur and phosphorus Sedimentary nutrient cycle Carbon and nitrogen
done
clear
D)
Gaseous nutrient cycle Carbon and nitrogen Sedimentary nutrient cycle Sulphur and phosphorus
done
clear
View Answer play_arrow
The introduction of T-DNA into plants involves
A)
Altering the pH of soil, then heat-socking the plants.
done
clear
B)
Exposing the plants to cold for a brief period
done
clear
C)
Allowing the plant roots to stand in water.
done
clear
D)
Infection of the plant by Agrobacterium tumefaciens.
done
clear
View Answer play_arrow
The wings of a bird and the wings of an insect are
A)
Analogous structures and represent convergent evolution.
done
clear
B)
Phylogenetic structures and represent divergent evolution.
done
clear
C)
Homologous structures and represent convergent evolution.
done
clear
D)
Homologous structures and represent divergent evolution.
done
clear
View Answer play_arrow
Which of the following re-establish the continuous chain of water molecule in the xylem which often breaks under the enormous tension created by transpiration.
A)
Imbibition
done
clear
B)
Passive absorption
done
clear
C)
Root pressure
done
clear
D)
Capillary force
done
clear
View Answer play_arrow
Human urine is usually acidic because
A)
Excreted plasma proteins are acidic.
done
clear
B)
Potassium and sodium exchange generates acidity.
done
clear
C)
Hydrogen ions are actively secreted into the filtrate.
done
clear
D)
The sodium transporter exchange one hydrogen ion for each sodium ion, in peritubular capillaries.
done
clear
View Answer play_arrow
Which one of the following types of organisms occupies more than one trophic level in a pond ecosystem?
A)
Fish
done
clear
B)
Zooplankton
done
clear
C)
Frog
done
clear
D)
Phytoplankton
done
clear
View Answer play_arrow
The species that drive major ecosystem function is termed as
A)
Alien
done
clear
B)
Endemic
done
clear
C)
Rare
done
clear
D)
Keystone
done
clear
View Answer play_arrow
Select the wrong statement
A)
W. M. Stanley showed that viruses could be crystallized.
done
clear
B)
The term 'contagium vivumfluidum' was coined by M. W. Beijerinck.
done
clear
C)
Mosaic disease in tobacco and AIDS in human beings are caused by viruses.
done
clear
D)
The viroids were discovered by D. J. Ivanowsky.
done
clear
View Answer play_arrow
Axile placentation is present in all except
A)
Lemon
done
clear
B)
Tomato
done
clear
C)
China rose
done
clear
D)
Dianthus
done
clear
View Answer play_arrow
Select the correct matching:
A)
IUI - Intra Uterine Insemination
done
clear
B)
ICSI - Intra Cellular Sperm Insemination
done
clear
C)
ZIFT - Zygote Intra Fusion Transfer
done
clear
D)
GIFT - Gamete Inseminated Fallopian Transfer
done
clear
View Answer play_arrow
Destruction of the anterior horn cells of the spinal cord would result in the loss of
A)
Voluntary motor impulse
done
clear
B)
Commissural impulses
done
clear
C)
Integrating impulses
done
clear
D)
Sensory impulses
done
clear
View Answer play_arrow
Denitrification is carried out by
A)
Nitrobacter
done
clear
B)
Nitrococcus
done
clear
C)
Thiobacillus
done
clear
D)
Rhizobium
done
clear
View Answer play_arrow
An association of individuals of different species living in the same habitat and having functional interactions is
A)
Biotic community
done
clear
B)
Ecosystem
done
clear
C)
Population
done
clear
D)
Ecological niche
done
clear
View Answer play_arrow
Neural signals from which part of brain can alter respiratory mechanism
A)
Pneumotaxic centre
done
clear
B)
Chemosensitive area in medulla
done
clear
C)
Both of these
done
clear
D)
None of these
done
clear
View Answer play_arrow
Balbiani rings are sites of
A)
Nucleotide synthesis
done
clear
B)
Polysaccharide synthesis
done
clear
C)
RNA and protein synthesis
done
clear
D)
Carbohydrate synthesis
done
clear
View Answer play_arrow
Match the columns and identify the correct option.
Column I Column II [a] Thylakoids (i) Disc-shaped sacs in Golgi apparatus [b] Cristae (ii) Condensed structure of DNA [c] Cisternae (iii) Flat membranous sacs in stroma [d] Chromatic (iv) Infoldings in mitochondria
A)
A-(iii), B-(iv), C-(i), D-(ii)
done
clear
B)
A-(iii), B-(i), C-(iv), D-(ii)
done
clear
C)
A-(iii), B-(iv), C-(ii), D-(i)
done
clear
D)
A-(iv), B-(iii), C-(i), D-(ii)
done
clear
View Answer play_arrow
Steroidal hormones are formed in
A)
SER
done
clear
B)
RER
done
clear
C)
Lysosomes
done
clear
D)
Golgi body
done
clear
View Answer play_arrow
Auxin can be bioassayed by
A)
Hydroponics
done
clear
B)
Potometer
done
clear
C)
Lettuce hypocotyl elongation
done
clear
D)
Cress root inhibition test
done
clear
View Answer play_arrow
Which of the following layers in an antral follicle is acellular?
A)
Theca internet
done
clear
B)
Stroma
done
clear
C)
Zona pellucida
done
clear
D)
Granulosa
done
clear
View Answer play_arrow
Satellite DNA is important because it
A)
Shows high degree of polymorphism in population and also the same degree of polymorphism in an individual, which is heritable from parents to children.
done
clear
B)
Does not code for proteins and is same in all members of the population.
done
clear
C)
Codes for enzymes needed for DNA
done
clear
D)
Codes for proteins needed in cell
done
clear
View Answer play_arrow
Motile bacteria moves with the help of
A)
Pili
done
clear
B)
Fimbriae
done
clear
C)
Flagella
done
clear
D)
Cilia
done
clear
View Answer play_arrow
In angiosperms, microsporogenesis and genesis
A)
From gametes without further divisions
done
clear
B)
Involve meiosis
done
clear
C)
Occur in ovule
done
clear
D)
Occur in anther
done
clear
View Answer play_arrow
Roots play insignificant role in absorption of water in
A)
Eichhornia
done
clear
B)
Pea
done
clear
C)
Wheat
done
clear
D)
Sunflower
done
clear
View Answer play_arrow
Which of the following are most suitable indicators of \[S{{O}_{2}}\]pollution in the environment?
A)
Conifers
done
clear
B)
Algae
done
clear
C)
Fungi
done
clear
D)
Lichens
done
clear
View Answer play_arrow
An example of a seed with endosperm, perisperm and caruncle is
A)
Coffee
done
clear
B)
Lily
done
clear
C)
Castor
done
clear
D)
Cotton
done
clear
View Answer play_arrow
Radula, a rasping organ is found in
A)
Salpa
done
clear
B)
Ophiura
done
clear
C)
Sea Hare
done
clear
D)
Aptenodytes
done
clear
View Answer play_arrow
Diatomaceous earth is formed by
A)
Euglenoids
done
clear
B)
Dinoflagellates
done
clear
C)
Slime moulds
done
clear
D)
Chrysophytes
done
clear
View Answer play_arrow
Choose the wrong statement from the following
A)
Neurospora is used in the study of biochemical genetics.
done
clear
B)
Morels and truffles are poisonous mushrooms
done
clear
C)
Yeast is unicellular and useful in fermentation.
done
clear
D)
Penicillium is multicellular and produces antibiotics.
done
clear
View Answer play_arrow
Which is characterised by afluid filled cavity called antrum?
A)
Primary follicle
done
clear
B)
Secondary follicle
done
clear
C)
Tertiary follicle
done
clear
D)
Oogonia
done
clear
View Answer play_arrow
Natural aging of a lake by nutrient enrichment of its water is known as
A)
Cultural eutrophication
done
clear
B)
Accelerated eutrophication
done
clear
C)
Eutrophication
done
clear
D)
Biomagnification
done
clear
View Answer play_arrow
The physiologic values of carbohydtares, protiens and fats are respectively (in Kcal/g).
A)
4, 9, 4
done
clear
B)
9, 9, 4
done
clear
C)
4, 4, 9
done
clear
D)
9, 4, 4
done
clear
View Answer play_arrow