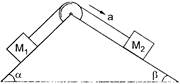
Two masses \[{{M}_{1}}\] and \[{{M}_{2}}\] are attached to the ends of a string which pass over a pulley attached to the top of a double inclined plane of angles of inclination \[\alpha \] and \[\beta \]. If \[{{M}_{2}}>{{M}_{1}},\] the acceleration a of the system is given by
A)
\[\frac{{{M}_{2}}(\sin \beta )g}{{{M}_{1}}+{{M}_{2}}}\]
done
clear
B)
\[\frac{{{M}_{1}}(\sin \alpha )g}{{{M}_{1}}+{{M}_{2}}}\]
done
clear
C)
\[\left( \frac{{{M}_{2}}\sin \beta -{{M}_{1}}\sin \alpha }{{{M}_{1}}+{{M}_{2}}} \right)g\]
done
clear
D)
zero
done
clear
View Answer play_arrow
Two simple harmonic motions are represented by the following equations \[{{Y}_{1}}-40\,\sin \omega \] and \[{{Y}_{2}}-100\,(\sin \omega t+c\,\cos \omega )\]. If their diplacement amplitudes are equal then the value of \[cl\] in appropriate units) is:
A)
\[\sqrt{13}\]
done
clear
B)
\[\sqrt{15}\]
done
clear
C)
\[\sqrt{17}\]
done
clear
D)
4
done
clear
View Answer play_arrow
Time period of a disc about a tangent parallel to the diameter is same as the time period of a simple pendulum. The ratio of radius of disc to the length of pendulum is:
A)
\[\frac{1}{4}\]
done
clear
B)
\[\frac{4}{5}\]
done
clear
C)
\[\frac{2}{3}\]
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
If a body is to be projected vertically upwards from the earths surface to reach a height of 10R from the surface of the earth (where R is the radius of earth). The velocity required to do so is:
A)
\[\sqrt{\frac{24}{11}g\,\,R}\]
done
clear
B)
\[\sqrt{\frac{22}{11}g\,\,R}\]
done
clear
C)
\[\sqrt{\frac{20}{11}g\,\,R}\]
done
clear
D)
\[\sqrt{\frac{18}{11}g\,\,R}\]
done
clear
View Answer play_arrow
An object is acted upon by the forces, \[{{F}_{1}}=4\,\hat{i}\,N\] and \[{{F}_{2}}=(\,\hat{i}-j)\,N\]. If the displacement of the object is \[D=(\hat{i}+6\hat{j}-6\hat{k})m.\]. The kinetic energy of the object.
A)
remains constant
done
clear
B)
increased by 1 J
done
clear
C)
decreases by 1 J
done
clear
D)
decreases by 2 J
done
clear
View Answer play_arrow
A ball of mass m approaches a wall fo mass m \[(>>m)\]with speed 4 m/s. along the normal to the wall. The speed of wall is 1m/s towards the ball. The speed of the ball after an elastin collision with the wall is:
A)
5m/s away from the wall
done
clear
B)
9m/s away from the wall
done
clear
C)
3m/s away from the wall
done
clear
D)
6m/s away from the wall
done
clear
View Answer play_arrow
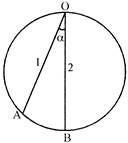
Two particles 1 and 2 are allowed to descend on the two friction less chord OH and OB of a vertical circel, at the same instant from point O. The ratio of the velocities of the particles 1 and 2 respectively, when they reach on the circumference will be (OB is the diameter):
A)
\[\sin a\]
done
clear
B)
\[tan\text{ }a\]
done
clear
C)
\[cos\text{ }a\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
The diameter of a wire is measured with a screw gauge having 50 divisions an circular scale and by one complete rotation of circular scale, main scale moves\[0.5\text{ }mm\]. If reading of screw gauge is\[0.250\text{ }c.m\]. The minimum percentage error in the reading will be
A)
\[0.4\]
done
clear
B)
\[0.8\]
done
clear
C)
\[4\]
done
clear
D)
\[5\]
done
clear
View Answer play_arrow
The equation of state of one mole of some gases can be expressed as \[\left( P+\frac{a}{{{V}^{2}}} \right)\,(V-b)=RT\]. Here, P is the pressure, V is the volume, T is the absolute temperature and a, b, R are the constants. The dimensions of ?a? are:
A)
\[[M{{L}^{5}}{{T}^{-2}}]\]
done
clear
B)
\[[M{{L}^{-1}}{{T}^{-2}}]\]
done
clear
C)
\[[M{{L}^{3}}T]\]
done
clear
D)
\[[M{{L}^{6}}T]\]
done
clear
View Answer play_arrow
A particle is projected vertically upward with initial velocity \[25m{{s}^{-1}}\]. During third second of its motion, which of the following statements is correct?
A)
Displacement of the particle is 30m.
done
clear
B)
Distance covered by the particle is 30m.
done
clear
C)
Distance covered by the particle is \[2.5m\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A simple pendulum of lenght \[l\], has a bob of mass m with a charge q on it. A verticle sheet of charge with surface charge density Q, posses through the point of suspension. At equilibrium, the string makes an angle Q with the verticle then:
A)
\[\tan \theta =\frac{Qq}{2{{\varepsilon }_{0}}mg}\]
done
clear
B)
\[\tan \theta =\frac{Qq}{{{\varepsilon }_{0}}mg}\]
done
clear
C)
\[\cot \theta =\frac{Qq}{2{{\varepsilon }_{0}}mg}\]
done
clear
D)
\[\cot \theta =\frac{Qq}{{{\varepsilon }_{0}}mg}\]
done
clear
View Answer play_arrow
A body weights w in air and it loses its weight by 25% in water. The relative density of the body is:
A)
4
done
clear
B)
\[1/4\]
done
clear
C)
\[3/4\]
done
clear
D)
\[4/3\]
done
clear
View Answer play_arrow
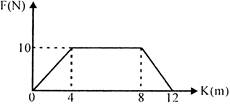
A particle of mass \[0.1\,kg\] is subjected to a force which varies with distance as shown in figure. If it starts its journey from rest at \[x=0,\] its velocity at \[X=12cm\] is:
A)
\[0\,m/s\]
done
clear
B)
\[20\,\sqrt{2}\,m/s\]
done
clear
C)
\[20\,\sqrt{3}\,m/s\]
done
clear
D)
\[40\,m/s\]
done
clear
View Answer play_arrow
A projectile has the same range R for two angles of projection. If \[{{T}_{1}}\] and \[{{T}_{2}}\] be the times of flight in the two cases, then R is:
A)
\[{{T}_{1}}{{T}_{2}}g\]
done
clear
B)
\[\frac{{{T}_{1}}{{T}_{2}}g}{2}\]
done
clear
C)
\[\left( T_{1}^{2}+T_{2}^{2} \right)g\]
done
clear
D)
\[\frac{\left( T_{1}^{2}+T_{2}^{2} \right)}{2}g\]
done
clear
View Answer play_arrow
Two springs A and B \[({{k}_{B}}=2{{k}_{A}})\] are stretched by same suspended weight, then ratio of work done in stretching is
A)
\[1:2\]
done
clear
B)
\[2:1\]
done
clear
C)
\[1:1\]
done
clear
D)
\[1:4\]
done
clear
View Answer play_arrow
The unit vector perpendicular to both, \[\hat{i}+\hat{j}\]and \[\hat{j}-\hat{k}\] is:
A)
\[\frac{1}{\sqrt{3}}(\hat{i}+\hat{j}-\hat{k})\]
done
clear
B)
\[\frac{1}{\sqrt{3}}(-\hat{i}+\hat{j}+\hat{k})\]
done
clear
C)
\[\frac{1}{\sqrt{3}}(\hat{i}-\hat{j}+\hat{k})\]
done
clear
D)
\[\frac{1}{\sqrt{3}}(\hat{i}+\hat{j}+\hat{k})\]
done
clear
View Answer play_arrow
At what temperature will the resistance of a copper wire become three times its value at \[{{0}^{o}}C\]? (Temperature coefficient of resistance for copper \[=4\times {{10}^{-3}}pe{{r}^{o}}C\])?
A)
\[{{400}^{o}}C\]
done
clear
B)
\[{{450}^{o}}C\]
done
clear
C)
\[{{500}^{o}}C\]
done
clear
D)
\[{{550}^{o}}C\]
done
clear
View Answer play_arrow
Four bodies of masses 2, 3, 5, and 8 kg are placed at the four corners of a square of side 2 m. The position of CM will be
A)
\[\left( \frac{8}{9},\frac{13}{9} \right)\]
done
clear
B)
\[\left( \frac{7}{9},\frac{11}{9} \right)\]
done
clear
C)
\[\left( \frac{11}{9},\frac{13}{9} \right)\]
done
clear
D)
\[\left( \frac{11}{9},\frac{8}{9} \right)\]
done
clear
View Answer play_arrow
Two waves are represented by the equations, \[{{Y}_{1}}=A\sin \left( 10\pi X-15\pi t+\frac{\pi }{2} \right)\] and \[{{Y}_{2}}=2A\,(30\pi X+45\pi t):\] Which of the following statement is correct?
A)
The maximum particle velocity of the second wave is twice that of first.
done
clear
B)
Their super position will produce a standing wave
done
clear
C)
Maximum particle acceleration for the second wave is eighteen times that of the first wave.
done
clear
D)
Their wave velocities are different
done
clear
View Answer play_arrow
A whistle giving out 450Hz, approaches a stationary observer at a speed of\[33m/s\]. The frequency heard by the observer in Hz is (speed of sound\[=330m/s\])
A)
409
done
clear
B)
429
done
clear
C)
517
done
clear
D)
500
done
clear
View Answer play_arrow
Two particles X and Y having equal charges, after being accelerated through the same potential difference, enters a region of an uniform magnetic field and describe circular paths of radii \[{{R}_{1}}\] and \[{{R}_{2}}\]respectively. The ratio of masses of X and Y is:
A)
\[{{\left( \frac{{{R}_{1}}}{{{R}_{2}}} \right)}^{1/2}}\]
done
clear
B)
\[\frac{{{R}_{2}}}{{{R}_{1}}}\]
done
clear
C)
\[{{\left( \frac{{{R}_{1}}}{{{R}_{2}}} \right)}^{2}}\]
done
clear
D)
\[\left( \frac{{{R}_{1}}}{{{R}_{2}}} \right)\]
done
clear
View Answer play_arrow
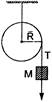
A mass M is supported by a massless string wound round a uniform cylinder of mass M and radius R as shown. The mass is released from rest. The acceleration with which it falls down is
A)
\[\frac{g}{3}\]
done
clear
B)
\[\frac{g}{2}\]
done
clear
C)
\[\frac{2g}{3}\]
done
clear
D)
\[\frac{3g}{3}\]
done
clear
View Answer play_arrow
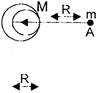
A solid sphere of uniform density and radius R applies a gravitational force of attraction equal to \[{{F}_{1}}\] on a particle placed at A, distant 2R from the centre of the sphere. A spherical cavity of radius R/2 is now made on the sphere as shown in the figure. The sphere with cavity now applies a gravitational force \[\frac{{{F}_{2}}}{{{F}_{1}}}\] will be
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{3}{4}\]
done
clear
C)
\[\frac{7}{8}\]
done
clear
D)
\[\frac{14}{9}\]
done
clear
View Answer play_arrow
The period of revolution of planet A around the sun is 8 times that of B. The distance of A from the sun is how many times greater than that of B from the sun?
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
A S.H.M. oscillator has period of\[0.1\text{ }sec\]. and amplitude of \[0.2\text{ }m\]. The maximum velocity is given by
A)
\[100m{{s}^{-1}}\]
done
clear
B)
\[100\pi \,m{{s}^{-1}}\]
done
clear
C)
\[4\pi \,m{{s}^{-1}}\]
done
clear
D)
\[20\pi \,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
The resistance of a wire is 10W its length is increased by 10% by stretching. The new resistance will now be nearly:
A)
\[12\,\Omega \]
done
clear
B)
\[1.2\,\Omega \]
done
clear
C)
\[13\,\,\Omega \]
done
clear
D)
\[11\,\,\Omega \]
done
clear
View Answer play_arrow
The same mass of copper is drawn into two wires 1mm, 2mm thick two wires connected in series and current is passed through them. Heat produced in the wire is in the ratio:
A)
\[2:1\]
done
clear
B)
\[1:16\]
done
clear
C)
\[4:1\]
done
clear
D)
\[16:1\]
done
clear
View Answer play_arrow
A particle is fastened at the end of a string and whirled in a vertical circle with the other end of the string being fixed. The motion of the particle is
A)
periodic
done
clear
B)
oscillatory
done
clear
C)
simple harmonic
done
clear
D)
angular harmonic
done
clear
View Answer play_arrow
If the tension and diameter of sonometer wire of fundamental frequency n is doubled and density is halved, then its fundamental frequency will become
A)
\[\frac{n}{4}\]
done
clear
B)
\[\sqrt{2}n\]
done
clear
C)
\[n\]
done
clear
D)
\[\frac{n}{\sqrt{2}}\]
done
clear
View Answer play_arrow
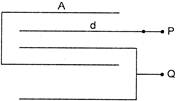
The equivalent capacitance between P and Q on the figure is;
A)
\[\frac{A\,{{\varepsilon }_{0}}}{2d}\]
done
clear
B)
\[\frac{3A\,{{\varepsilon }_{0}}}{2d}\]
done
clear
C)
\[\frac{2A\,{{\varepsilon }_{0}}}{2d}\]
done
clear
D)
\[\frac{5A\,{{\varepsilon }_{0}}}{3d}\]
done
clear
View Answer play_arrow
The speed of sound in pure hydrogen is v. The speed of sound in a mixture of hydrogen and oxygen as \[5:1\] by volume at the same temperature is
A)
\[\upsilon \sqrt{\frac{6}{5}}\]
done
clear
B)
\[\upsilon \sqrt{\frac{5}{6}}\]
done
clear
C)
\[\upsilon \sqrt{\frac{7}{2}}\]
done
clear
D)
\[\upsilon \sqrt{\frac{2}{7}}\]
done
clear
View Answer play_arrow
The half-life of a radioactive element, \[^{222}Ruis\,3.8h,\] mass of this element which has activity equal to \[{{10}^{16}}\] Rutherford is:
A)
\[0.37\,kg\]
done
clear
B)
\[0.37g\]
done
clear
C)
\[0.073g\]
done
clear
D)
\[0.07g\]
done
clear
View Answer play_arrow
An electromagnetic wave going through vacuum is decribed by \[\varepsilon ={{\varepsilon }_{0}}\,\sin \,(KX-\cot \theta );\] and \[B={{B}_{0}}\]\[B={{B}_{0}}\sin \]\[B={{B}_{0}}\sin \,(KX-\omega t)\]. Which of the following equations is true?
A)
\[{{\varepsilon }_{0}}K={{B}_{0}}\omega \]
done
clear
B)
\[{{\varepsilon }_{0}}\omega ={{B}_{0}}K\]
done
clear
C)
\[{{\varepsilon }_{0}}K={{B}_{0}}\omega K\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A common emitter amplifier is designed with n-p-n transistor \[(a=0.99)\]. The input impedance is \[1\,K\Omega \] and load is \[10\,R\Omega \] The voltage gain will be:
A)
\[9.9\]
done
clear
B)
\[99\]
done
clear
C)
\[990\]
done
clear
D)
\[9900\]
done
clear
View Answer play_arrow
The wavelength of the \[{{K}_{a}}\] line for the uranium atom \[(Z=92,\,R={{10}^{7}}{{m}^{-1}})\] is:
A)
\[1.6\overset{o}{\mathop{A}}\,\]
done
clear
B)
\[0.16\overset{o}{\mathop{A}}\,\]
done
clear
C)
\[0.5\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[2.0\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
In n-p-n transistor, the collection current is 10MA. If 90% of electrons emitted reach the collector then:
A)
emitter current will be \[9\text{ }mA\]
done
clear
B)
emitter current will be \[\text{11}\text{.1 }mA\]
done
clear
C)
base current will be \[\text{0}\text{.1 }mA\]
done
clear
D)
base current will be \[\text{0}\text{.0 }mA\]
done
clear
View Answer play_arrow
An ideal carnot engine, whose efficiency is 40% receives heat at \[{{5000}^{o}}K\]. If its efficiency is 50%, then the intake temperature for the same exhaust temperature is
A)
\[{{600}^{o}}K\]
done
clear
B)
\[{{700}^{o}}K\]
done
clear
C)
\[{{800}^{o}}K\]
done
clear
D)
\[{{900}^{o}}K\]
done
clear
View Answer play_arrow
In the hydrogen atom spectrum \[{{\lambda }_{3}}-1\] and \[{{\lambda }_{2}}-1\] represent wavelengths emitted due to transition from second and first excited status to the ground state respectively. The value of \[\frac{{{\lambda }_{3}}-1}{{{\lambda }_{2}}-1}\] is:
A)
\[\frac{27}{32}\]
done
clear
B)
\[\frac{32}{27}\]
done
clear
C)
\[\frac{4}{9}\]
done
clear
D)
\[\frac{9}{4}\]
done
clear
View Answer play_arrow
For a concave mirror of focal length 20cm, if the object is at a distance of 30cm from the pole, then the nature of the image and its magnification will be:
A)
real and \[-2\]
done
clear
B)
virtual and \[-2\]
done
clear
C)
real and \[+2\]
done
clear
D)
virtual and \[+2\]
done
clear
View Answer play_arrow
Focal length of an equiconvex lens is\[20cm\]. If we cut it once perpendicular to principal exis in and then along principal axis. Then, focal length of each part will be:
A)
\[20cm\]
done
clear
B)
\[10cm\]
done
clear
C)
\[40cm\]
done
clear
D)
\[5cm\]
done
clear
View Answer play_arrow
A bulb is placed at a depth of \[2\sqrt{7}\] in water and a floating opaque is placed over the bulb, so that the bulb is not visible from the surface. The radius of the disc should be at least: (\[\mu \] water\[=4/3\])
A)
\[42\,m\]
done
clear
B)
\[6\,m\]
done
clear
C)
\[2\sqrt{7m}\,\]
done
clear
D)
\[12m\]
done
clear
View Answer play_arrow
There is a current of \[1.344\text{ }A\] in a copper wire whose area of cross-section normal to the length the wire is \[1m{{m}^{2}}\]. If the number of free electrons per \[c{{m}^{3}}\]is \[8.4\times {{10}^{22}},\] then the drift velocity of elections will be:
A)
\[1.0\,mm/s\]
done
clear
B)
\[10m/s\]
done
clear
C)
\[0.1\,mm/s\]
done
clear
D)
\[0.01\,mm/s\]
done
clear
View Answer play_arrow
The equation of the state of a gas is defined as \[P+\frac{a}{{{V}^{2}}}=\frac{R\theta }{V},\]where \[P\to \] pressure, \[V\to \]volume, \[\theta \to \]absolute temperature and a, \[R\to \] constants, then the dimensional formula for a is
A)
\[{{M}^{-1}}{{L}^{5}}{{T}^{-2}}\]
done
clear
B)
\[M{{L}^{5}}{{T}^{-2}}\]
done
clear
C)
\[M{{L}^{-5}}{{T}^{2}}\]
done
clear
D)
\[M{{L}^{-1}}{{T}^{-2}}\]
done
clear
View Answer play_arrow
In an L-C-R series circuit, the AC voltage across, R, L and C come out as 10V, 10V and 20V respectively. The voltage across the entire combination will be:
A)
\[30\,V\]
done
clear
B)
\[10\sqrt{3}\,V\]
done
clear
C)
\[20\,V\]
done
clear
D)
\[10\sqrt{2}\,V\]
done
clear
View Answer play_arrow
The wire loop formed by join in two semi-circular sections of radii \[{{R}_{1}}\] and \[{{R}_{2}},\] carries a current I, as shown. The magnitude of magnetic field at the centre C is:
A)
\[\frac{{{H}_{o}}I}{2}\left( \frac{1}{{{R}_{1}}}+\frac{1}{{{R}_{2}}} \right)\]
done
clear
B)
\[\frac{{{H}_{o}}I}{4}\left( \frac{1}{{{R}_{1}}}+\frac{1}{{{R}_{2}}} \right)\]
done
clear
C)
\[\frac{{{H}_{o}}I}{2}\left( \frac{1}{{{R}_{1}}}-\frac{1}{{{R}_{2}}} \right)\]
done
clear
D)
\[\frac{{{H}_{o}}I}{4}\left( \frac{1}{{{R}_{1}}}-\frac{1}{{{R}_{2}}} \right)\]
done
clear
View Answer play_arrow
Which species has the maximum number of lone pair of electrons on the central atom?
A)
\[\left[ ClO_{3}^{-} \right]\]
done
clear
B)
\[Xe{{F}_{4}}\]
done
clear
C)
\[S{{F}_{4}}\]
done
clear
D)
\[\left[ I_{3}^{-} \right]\]
done
clear
View Answer play_arrow
Which one of the following statement for order of reaction is not correct?
A)
Order can be determined experimentally
done
clear
B)
Order of reaction is equal to sum of the power of concentration terms in differential rate law
done
clear
C)
It is not affected with stoichiometric coefficient of the reactants
done
clear
D)
Order can not be fractional
done
clear
View Answer play_arrow
Which blue liquid is obtained on reacting equimolar amounts of two gases at \[{{30}^{o}}C\]?
A)
\[{{N}_{2}}O\]
done
clear
B)
\[{{N}_{2}}{{O}_{3}}\]
done
clear
C)
\[{{N}_{2}}{{O}_{4}}\]
done
clear
D)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
Which are contains both iron and copper?
A)
Cuprite
done
clear
B)
Chalcocite
done
clear
C)
Chalcopyrite
done
clear
D)
Malachite
done
clear
View Answer play_arrow
Name of the structure of silicates in which three oxygen atoms of \[{{[Si{{O}_{4}}]}^{4-}}\] are shared is
A)
pyro silicate
done
clear
B)
sheet silicate
done
clear
C)
linear chain silicate
done
clear
D)
three dimensional silicate
done
clear
View Answer play_arrow
Which pair of compounds is expected to show similar colour in aqueous medium?
A)
\[FeC{{l}_{2}}\] and \[MnC{{l}_{2}}\]
done
clear
B)
\[VOC{{l}_{2}}\] and \[CuC{{l}_{2}}\]
done
clear
C)
\[FeC{{l}_{3}}\] and \[CuC{{l}_{2}}\]
done
clear
D)
\[VOC{{l}_{2}}\] and \[FeC{{l}_{2}}\]
done
clear
View Answer play_arrow
The number of radial nodes of 3s and 2p orbitals are respectively
A)
\[2,0\]
done
clear
B)
\[0,2\]
done
clear
C)
\[1,2\]
done
clear
D)
\[2,1\]
done
clear
View Answer play_arrow
A metal nitrate reacts with KI to give a black precipitate which on addition of excess of KI convert into orange colour solution. The cation of metal nitrate is
A)
\[H{{g}^{2+}}\]
done
clear
B)
\[B{{i}^{3+}}\]
done
clear
C)
\[P{{b}^{2+}}\]
done
clear
D)
\[C{{u}^{+}}\]
done
clear
View Answer play_arrow
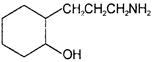
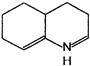
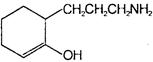
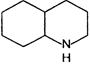
What would be the product formed when 1-bromo-3-chloro cyclobutane reacts with two equivalents of metallic sodium in ether?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
When benzene sulphonic acid and p-nitrophenol are treated with \[NaHC{{O}_{3}}\]the gases released respectively are:
A)
\[S{{O}_{2}},N{{O}_{2}}\]
done
clear
B)
\[S{{O}_{2}},NO\]
done
clear
C)
\[S{{O}_{2}},C{{O}_{2}}\]
done
clear
D)
\[C{{O}_{2}},C{{O}_{2}}\]
done
clear
View Answer play_arrow
Amonoatomic ideal gas undergoes a process in which the ratio of P to V at any instant is constant and equals to 1. What is the molar heat capacity of the gas?
A)
\[\frac{4R}{2}\]
done
clear
B)
\[\frac{3R}{2}\]
done
clear
C)
\[\frac{5R}{2}\]
done
clear
D)
\[0\]
done
clear
View Answer play_arrow
The orbital angular momentum of an electron in 3s orbital is
A)
\[\frac{h}{2\pi }\]
done
clear
B)
zero
done
clear
C)
\[\frac{1}{2}.\frac{h}{2\pi }\]
done
clear
D)
\[\frac{1}{3}.\frac{h}{2\pi }\]
done
clear
View Answer play_arrow
If the energy difference between the ground state of an atom and its excited state is \[4.4\times {{10}^{-14}}J,\] the wavelength of photon required to produce the transition is
A)
\[2.26\times {{10}^{-12}}m\]
done
clear
B)
\[1.13\times {{10}^{-12}}m\]
done
clear
C)
\[8.52\times {{10}^{-16}}m\]
done
clear
D)
\[4.52\times {{10}^{-12}}m\]
done
clear
View Answer play_arrow
A binary solid \[({{A}^{+}}{{B}^{-}})\]has a rock salt structure. If the edge length is 400 pm and radius of cation is 75 pm, the radius of anion is
A)
\[100\text{ }pm\]
done
clear
B)
\[\text{125 }pm\]
done
clear
C)
\[\text{250 }pm\]
done
clear
D)
\[\text{325 }pm\]
done
clear
View Answer play_arrow
Which of the following has \[s{{p}^{2}}\]hybridisation?
A)
\[S{{O}_{2}}\]
done
clear
B)
\[N{{H}_{3}}\]
done
clear
C)
\[SO_{4}^{2-}\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
Which of the following are isoelectronic and isostructural? \[NO_{3}^{-},CO_{3}^{2-},ClO_{3}^{-},S{{O}_{3}}\]
A)
\[NO_{3}^{-},CO_{3}^{2-}\]
done
clear
B)
\[S{{O}_{3}},NO_{3}^{-}\]
done
clear
C)
\[ClO_{3}^{-},CO_{3}^{2-}\]
done
clear
D)
\[CO_{3}^{2-},S{{O}_{3}}\]
done
clear
View Answer play_arrow
Acetic acid exists in benzene solution in the dimeric form. In an actual experiment the van't Hoff factor was found to the\[0.52\]. Then the degree of dissociation of acetic acid is
A)
\[0.48\]
done
clear
B)
\[0.88\]
done
clear
C)
\[0.96\]
done
clear
D)
\[0.52\]
done
clear
View Answer play_arrow
The pH value of the following half cell is _______ \[P{{t}_{{\scriptstyle{}^{{{H}_{2}}}/{}_{{{H}_{2}}S{{O}_{4}}}}}}\]. The electrode potential is\[-0.3V\].
A)
\[5.08\]
done
clear
B)
\[3\]
done
clear
C)
\[4\]
done
clear
D)
\[6\]
done
clear
View Answer play_arrow
Calcium carbonate dissociates in the following manner \[CaC{{O}_{3(s)}}Ca{{O}_{(s)}}+C{{O}_{2(g)}};\] \[\Delta H=110\,kJ\] in a closed vessel. The pressure of \[C{{O}_{2}}\]
A)
increases if temperature is raised
done
clear
B)
increases on adding a catalyst
done
clear
C)
decreases if temperature is raised
done
clear
D)
increases if an inert gas is pumped keeping the temperature constant
done
clear
View Answer play_arrow
How many \[\sigma \] and \[\pi \] bonds are there in the molecule of tetracyano ethylene \[{{(CN)}_{2}}C=C{{(CN)}_{2}}\]?
A)
\[9\sigma \] and \[9\pi \]
done
clear
B)
\[5\sigma \] and \[9\pi \]
done
clear
C)
\[9\sigma \] and \[7\pi \]
done
clear
D)
\[5\sigma \] and \[8\pi \]
done
clear
View Answer play_arrow
A gaseous mixture contains \[56g\,{{N}_{2}};\] \[44g\,C{{O}_{2}}\] and \[16g\,C{{H}_{4}}\]. The total pressure of the mixture is\[720\text{ }mm\text{ }Hg\]. What is the partial pressure of \[C{{H}_{4}}\]?
A)
\[220\,mm\]
done
clear
B)
\[540\text{ }mm\]
done
clear
C)
\[180\text{ }mm\]
done
clear
D)
\[100\text{ }mm\]
done
clear
View Answer play_arrow
A solution of \[NaCl\] in contact with atmosphere has a pH of about:
A)
\[3.5\]
done
clear
B)
\[5\]
done
clear
C)
\[7\]
done
clear
D)
\[14\]
done
clear
View Answer play_arrow
2-pentanone and 3-pentanone can be distinguished by
A)
Cannizzaro's reaction
done
clear
B)
Aldol condensation
done
clear
C)
lodoform reaction
done
clear
D)
Clemmensen's reduction
done
clear
View Answer play_arrow
An organic compound X is oxidised by using acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]. The product obtain reacts with phenyl hydrazine but does not answer silver mirror test. The possible structure of X is
A)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{3}}-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\]
done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CHOH\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
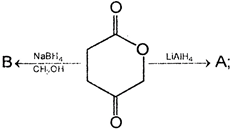
Adipic acid on heating gives
A)
done
clear
B)
done
clear
C)
done
clear
D)
\[\begin{align} & C{{H}_{2}}C{{H}_{2}}COOH \\ & | \\ & C{{H}_{2}}C{{H}_{2}}COOH \\ \end{align}\]
done
clear
View Answer play_arrow
Which of the following represents the correct order of the acidity in the given compounds?
A)
\[C{{H}_{3}}COOH>BrC{{H}_{2}}COOH>\]\[ClC{{H}_{2}}COOH>FC{{H}_{2}}COOH\]
done
clear
B)
\[FC{{H}_{2}}COOH>C{{H}_{3}}COOH>\]\[BrC{{H}_{2}}COOH>ClC{{H}_{2}}COOH\]
done
clear
C)
\[BrC{{H}_{2}}COOH>ClC{{H}_{2}}COOH>\]\[FC{{H}_{2}}COOH>C{{H}_{3}}COOH\]
done
clear
D)
\[FC{{H}_{2}}COOH>ClC{{H}_{2}}COOH>\]\[BrC{{H}_{2}}COOH>C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
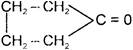
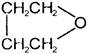
A and B are
A)
in both cases
done
clear
B)
in both cases
done
clear
C)
done
clear
D)
formation of A and B is not possible.
done
clear
View Answer play_arrow
compound A is,
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Oral contraceptive drugs contain
A)
Mestranol
done
clear
B)
Norethindrone
done
clear
C)
Both (A) and (B)
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which one of the following statements is incorrect?
A)
Salvarsan is an arsenic drug
done
clear
B)
N, N-Diethyl-m-toluamide is an insect repellant
done
clear
C)
Cationic detergents can be used as germicide
done
clear
D)
Aspirin is a narcotic drug
done
clear
View Answer play_arrow
Sanger's reagent is used for the identification of
A)
N-terminal of a peptide chain
done
clear
B)
C-terminal of a petide chain
done
clear
C)
Side chain of amino acids
done
clear
D)
number of amino acids in the peptide chain
done
clear
View Answer play_arrow
Which of the following polymers is easily damaged by accidental spillage of acid or base?
A)
Nylon
done
clear
B)
Orion
done
clear
C)
Polypropylene
done
clear
D)
Polyethylene
done
clear
View Answer play_arrow
The standard emf for the cell reaction \[2Cu_{(aq)}^{+}\xrightarrow{{}}Cu+Cu_{(aq)}^{2+}\] is +\[0.36V\]at 298K. The equilibrium constant for the reaction is
A)
\[5\times {{10}^{6}}\]
done
clear
B)
\[1.2\times {{10}^{6}}\]
done
clear
C)
\[1.4\times {{10}^{12}}\]
done
clear
D)
\[7.4\times {{10}^{12}}\]
done
clear
View Answer play_arrow
A factory produces 40 kg of calcium in two hours by electrolysis. How much aluminium can be produced by the same current in two hours?
A)
22kg
done
clear
B)
18 kg
done
clear
C)
9 kg
done
clear
D)
27 kg
done
clear
View Answer play_arrow
The radioisotope used for treatment of thyroid disorders is
A)
\[Na-24\]
done
clear
B)
\[P-32\]
done
clear
C)
\[Co-60\]
done
clear
D)
\[I-131\]
done
clear
View Answer play_arrow
The conjugate bases in the following reaction \[{{H}_{2}}S{{O}_{4}}+{{H}_{2}}O{{H}_{3}}{{O}^{+}}HSO_{4}^{-}\] are
A)
\[{{H}_{2}}O,{{H}_{3}}{{O}^{+}}\]
done
clear
B)
\[HSO_{4}^{-},{{H}_{2}}O\]
done
clear
C)
\[{{H}_{3}}{{O}^{+}},{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{4}},HSO_{4}^{-}\]
done
clear
View Answer play_arrow
How many \[m1\] of \[1\,M\,\,{{H}_{2}}S{{O}_{4}}\] is required to neutralise \[10\text{ }ml\] of \[1M\text{ }NaOH\] solution?
A)
\[2.5\]
done
clear
B)
\[5.0\]
done
clear
C)
\[10.0\]
done
clear
D)
\[20.0\]
done
clear
View Answer play_arrow
In the reaction \[2KMn{{O}_{4}}+16HCl\xrightarrow{{}}5C{{l}_{2}}+2MnC{{l}_{2}}+2KCl+8{{H}_{2}}O\] the reduced product
A)
\[C{{l}_{2}}\]
done
clear
B)
\[KCl\]
done
clear
C)
\[HCl\]
done
clear
D)
\[MnC{{l}_{2}}\]
done
clear
View Answer play_arrow
The name 'Blue John' is given to which of the following compounds.
A)
\[CaO\]
done
clear
B)
\[Ca{{H}_{2}}\]
done
clear
C)
\[Ca{{F}_{2}}\]
done
clear
D)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\]
done
clear
View Answer play_arrow
Which of the following statement is z about the ferrocene?
A)
It is a sandwiched complex
done
clear
B)
It is \[\pi \]-bonded complex
done
clear
C)
A complex in which all the five carbon atoms of cyclopentadiene anion are bonded to the metal.
done
clear
D)
All of these
done
clear
View Answer play_arrow
Yellow colour of chromates changes to orange on acidiflcation due to the formation of
A)
\[Cr{{O}_{3}}\]
done
clear
B)
\[CrO_{4}^{2-}\]
done
clear
C)
\[C{{r}_{2}}O_{7}^{2-}\]
done
clear
D)
\[C{{r}_{3}}O_{10}^{2-}\]
done
clear
View Answer play_arrow
How many cyclic isomers of \[{{C}_{5}}{{H}_{10}}\] are possible?
A)
4
done
clear
B)
3
done
clear
C)
6
done
clear
D)
5
done
clear
View Answer play_arrow
The most stable carbanion among the following is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A hypothetical reaction \[{{A}_{2}}+{{B}_{2}}\xrightarrow{{}}2AB\] follows the mechanism as given below:
\[{{A}_{2}}A+A\] (fast) \[A+{{B}_{2}}AB+B\] (slow) \[A+BAB\] (fast)
The order of the overall reaction is:
A)
2
done
clear
B)
1
done
clear
C)
\[1\frac{1}{2}\]
done
clear
D)
0
done
clear
View Answer play_arrow
A nitrate of an alkali metal M on heating gives \[{{O}_{2}},N{{O}_{2}}\] and \[{{M}_{2}}O\]. The metal M will be
A)
\[Na\]
done
clear
B)
\[K\]
done
clear
C)
\[Rb\]
done
clear
D)
\[Li\]
done
clear
View Answer play_arrow
For its activity, carboxy peptidase ?requires
A)
Zink
done
clear
B)
Copper
done
clear
C)
Niacin
done
clear
D)
Iron
done
clear
View Answer play_arrow
An organelle within an organelle is
A)
Meso some
done
clear
B)
ER
done
clear
C)
Ribosome
done
clear
D)
Peroxisome
done
clear
View Answer play_arrow
Select wrong with respect to mutations
A)
A single base pair change in DNA net results into mutation
done
clear
B)
Very high temperature is mutagenic
done
clear
C)
Frame shift mutation is caused due to deletion and insertion of base pair
done
clear
D)
Cancer cells commonly show chromosomal aberrations
done
clear
View Answer play_arrow
In five kingdom classification, Chlamydomonas and chlorella have been including
A)
Monera
done
clear
B)
Protista
done
clear
C)
Algae
done
clear
D)
Plantae
done
clear
View Answer play_arrow
Select the wrong match
A)
Archcgonia - Ginkgo
done
clear
B)
Prothallus - Salvinia
done
clear
C)
RNA-Viroids
done
clear
D)
Synergids - Mustard
done
clear
View Answer play_arrow
A test cross is carried out to
A)
determine whether two species or varieties will breed successfully
done
clear
B)
assess the number of allele of a gene
done
clear
C)
determine the genotype of \[{{f}_{2}}\] plant
done
clear
D)
predict whether two traits are linked
done
clear
View Answer play_arrow
Nitrosomonas and Nitrobacter are
A)
Photoautotrophs
done
clear
B)
Chemoautotrophs
done
clear
C)
Photoorganotrophy
done
clear
D)
Photolithotroph
done
clear
View Answer play_arrow
Porphyra is a
A)
Red alga
done
clear
B)
Brown alga
done
clear
C)
Green alga
done
clear
D)
Yellow alga
done
clear
View Answer play_arrow
Read the following five statements \[(A-E)\] and answer as next to them
A. In equisetum the female gametophyte is retained on parent sporophyte B. In Ginkgo male gametophyte is not independent C. The spores of slime moulds lack cell wall D. Sexual reproduction in volvox is isogramous E. The sporophyte in Riccia is more developed than that in polytrichum
How many of above statements are correct?
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
Wolfia and Ginkgo are
A)
Bryophytes
done
clear
B)
Spermatophytes
done
clear
C)
Angiosperms
done
clear
D)
Pteridophyte
done
clear
View Answer play_arrow
Perisperm differs from endosperm in
A)
being a triploid tissue
done
clear
B)
being a diploid tissue
done
clear
C)
having no reserve food
done
clear
D)
its formation by fusion of secondary nucleus with two sperms
done
clear
View Answer play_arrow
DNA \[\to \] mRNA \[\to \] Protein The scheme was proposed by
A)
F. Crick
done
clear
B)
J. Watson
done
clear
C)
R. Franklin
done
clear
D)
E. Chargaff
done
clear
View Answer play_arrow
In which of following symbiotic association with BGA is found
A)
Pinus
done
clear
B)
Psilotum
done
clear
C)
Equisctum
done
clear
D)
Cycas
done
clear
View Answer play_arrow
Megasporangium develops from
A)
Fruit
done
clear
B)
Nucellus
done
clear
C)
Ovule
done
clear
D)
Placenta
done
clear
View Answer play_arrow
If two persons with AB blood group marry and have sufficiently large number of children, these children could be classified as 'A' blood group: 'AB' blood group ='B' blood group in \[1:2:1\]ratio. Modern technique of protein electrophoresis reveals presence of both A and B types of proteins in AB blood group individuals. This is an example of
A)
Co-dominance
done
clear
B)
Complete dominance
done
clear
C)
Partial dominance
done
clear
D)
Incomplete dominance
done
clear
View Answer play_arrow
Read the following four statements (A - D)
A. Haemophilia is a sex linked recessive disease B. The human genome has about 50,000 genes C. Regulation of lac operon by its represser is referred as positive regulation D. Adenosine pairs with uracil in transcription.
How many of the above are right statements.
A)
Two
done
clear
B)
Four
done
clear
C)
One
done
clear
D)
Three
done
clear
View Answer play_arrow
\[{{C}_{3}}\] pathway is found in
A)
Pea
done
clear
B)
Maize
done
clear
C)
Onion
done
clear
D)
All above
done
clear
View Answer play_arrow
Axile placontation, bulb, imbricate aestivation are found in
A)
Pea
done
clear
B)
Tomato
done
clear
C)
Onion
done
clear
D)
Maize
done
clear
View Answer play_arrow
Marginal placentation is found in family of
A)
Pea and Lupin
done
clear
B)
Gram and Arhar
done
clear
C)
Moong and Sunhemp
done
clear
D)
More than one are correct
done
clear
View Answer play_arrow
A. Symbiotic Nitrogen fixers occur in free living state also in soil B. In dicot stem, a new cambium originates from cells of pericycle
A)
Both A and B are correct
done
clear
B)
Only A is correct
done
clear
C)
Only B is correct
done
clear
D)
Both A and B are incorrect
done
clear
View Answer play_arrow
Through their effect on plant growth regulators, what do the temperature and light control in plants?
A)
Fruit elongation
done
clear
B)
Closure of stomata
done
clear
C)
Apical dominance
done
clear
D)
Flowering
done
clear
View Answer play_arrow
The PGR that is antagonist to \[G{{A}_{3}}\]
A)
IAA
done
clear
B)
ABA
done
clear
C)
Zeatin
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
A monocot root has (as compared to Dicot root)
A)
thick periderm
done
clear
B)
many xylem bundles
done
clear
C)
inconspicuous annual rings
done
clear
D)
more abundant secondary xylem
done
clear
View Answer play_arrow
For its action nitrogen age requires
A)
Super oxygen radicals
done
clear
B)
Light
done
clear
C)
\[M{{n}^{2+}}\]
done
clear
D)
High input of energy
done
clear
View Answer play_arrow
Select odd with respect to elongation factor in E. coli
A)
Ef-Tu
done
clear
B)
Ef-TS
done
clear
C)
eEf
done
clear
D)
Ef-G
done
clear
View Answer play_arrow
In purine \[{{N}_{2}}\] is present at
A)
1 and 3 position
done
clear
B)
1, 3, 7 and 9 position
done
clear
C)
Only 7 and 9 position
done
clear
D)
2 and 4 position
done
clear
View Answer play_arrow
HGP was completed in year
A)
1990
done
clear
B)
2003
done
clear
C)
2000
done
clear
D)
2006
done
clear
View Answer play_arrow
Total number of \[{{N}_{2}}\] base and genes in drosophila are
A)
180 million, 18000 genes
done
clear
B)
103 million, 15000 genes
done
clear
C)
180 million, 15000 genes
done
clear
D)
3.2 million, 25000 genes
done
clear
View Answer play_arrow
Largest gene is made of _________ b» p.
A)
2400 K
done
clear
B)
231
done
clear
C)
14
done
clear
D)
\[2.4K\]
done
clear
View Answer play_arrow
Carrier female of haemophilia is married with normal male. The percentage of progeny without haemophilia allele will be
A)
\[0%\]
done
clear
B)
\[25%\]
done
clear
C)
\[50%\]
done
clear
D)
\[100%\]
done
clear
View Answer play_arrow
For incorporation of 1 ammo acid in polypeptide during translation, how many ATP are required?
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
Select odd one with respect to periderm
A)
Cork
done
clear
B)
Phellogen
done
clear
C)
Phelloderm
done
clear
D)
Lenticels
done
clear
View Answer play_arrow
Select odd one with respect to enzymes of lac-operon.
A)
\[\beta \] galactosidase
done
clear
B)
Permease
done
clear
C)
Transacetylase
done
clear
D)
Carboxy mutase
done
clear
View Answer play_arrow
Select odd with respect to Nucleoid of E'-coil.
A)
DS DNA
done
clear
B)
Circular linear DNA
done
clear
C)
SS RNA
done
clear
D)
No Histanes
done
clear
View Answer play_arrow
Bead is (with respect to chromosome)
A)
Nobody
done
clear
B)
Nucleosome
done
clear
C)
Chromatin
done
clear
D)
Chromonemata
done
clear
View Answer play_arrow
A chromosome, has not
A)
RNA
done
clear
B)
Acidic protein
done
clear
C)
Basic protein
done
clear
D)
Lipids
done
clear
View Answer play_arrow
A gene expresses
A)
to control a function
done
clear
B)
to control a set of function
done
clear
C)
to form enzymes
done
clear
D)
more than one are correct
done
clear
View Answer play_arrow
ATP dependent enzyme that unwinds DNA is
A)
Helicase
done
clear
B)
Gyrase
done
clear
C)
Primase
done
clear
D)
Topoisomerase
done
clear
View Answer play_arrow
In Gobar gas, the maximum amount is that of
A)
Butane
done
clear
B)
Propane
done
clear
C)
Methane
done
clear
D)
Carbon dioxide
done
clear
View Answer play_arrow
The second stage of hydrosere is occupied by plants like
A)
Typha
done
clear
B)
Salix
done
clear
C)
Azolla
done
clear
D)
Vallisneria
done
clear
View Answer play_arrow
Cuscuta is an example of
A)
End parasitism
done
clear
B)
Hemiparasitism
done
clear
C)
Total ectoparasitism
done
clear
D)
Partial ectoparasitism
done
clear
View Answer play_arrow
The rate of formation of new organic matter by rabbit in grassland, is called
A)
Gross primary productivity
done
clear
B)
Net primary productivity
done
clear
C)
Secondary productivity
done
clear
D)
Net productivity
done
clear
View Answer play_arrow
Sacred groves are specially useful in
A)
conservating rare and threatened species
done
clear
B)
flow of water in rivers throughout year
done
clear
C)
prevention of soil erosion
done
clear
D)
creating environmental awareness
done
clear
View Answer play_arrow
Germ pore's function is
A)
Release of male gamete
done
clear
B)
Absorption of water for seed-germination
done
clear
C)
formation of radicle
done
clear
D)
formation/initiation of pollen tube
done
clear
View Answer play_arrow
Select the wrong statements.
A)
Inline is made up of pectin and cellulose
done
clear
B)
Viability of pollen grain is variable in different plants
done
clear
C)
Vegetative cell is larger than generative cell
done
clear
D)
Pollen grains are released at 3 celled stage
done
clear
View Answer play_arrow
Select odd one out with respect to pest or insect resistance
A)
Pusa Swarnim
done
clear
B)
Pusa Sawani
done
clear
C)
Pusa Sem-2
done
clear
D)
Pusa Gaurav
done
clear
View Answer play_arrow
Select odd one with respect to flagellar movement
A)
Locomotion in Euglena
done
clear
B)
Swimming of spermatozoa
done
clear
C)
Movement of ova in fallopian tube
done
clear
D)
Maintenance of water current in canal system of sponges
done
clear
View Answer play_arrow
In a STP microbial floes are formed in
A)
Aeration tank
done
clear
B)
Primary treatment
done
clear
C)
Anaerobic sludge digester
done
clear
D)
Physiochemical treatment
done
clear
View Answer play_arrow
Select odd one with respect to protein energy malnutrition ? Marasmus
A)
It is found in infant less than a year
done
clear
B)
It is produced by a simultaneous deficiency of protein and calories
done
clear
C)
Some fat is left under the skin of patient extensive odema and swelling of body parts are seen
done
clear
D)
Patient shows wasting of muscles, thinning of limbs, failure of growth and brain development
done
clear
View Answer play_arrow
HapIoids are of great importance in crop improvements because
A)
Do not express mutation
done
clear
B)
Grow better in marshy area
done
clear
C)
Are useful in study of meiosis
done
clear
D)
Give homozygous lines after colchicine treatment
done
clear
View Answer play_arrow
The most important cause of biodiversity loss is
A)
Co-extinction
done
clear
B)
Alien species invasion
done
clear
C)
Habitat loss
done
clear
D)
Over exploitation
done
clear
View Answer play_arrow
High concentration in birds disturbs the metabolism of
A)
Iron
done
clear
B)
Calcium
done
clear
C)
Phosphorus
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
Mark the odd one with respect to origin from germinal layers
A)
Notochord
done
clear
B)
Pituitary gland
done
clear
C)
Sclera of eye
done
clear
D)
Adrenal cortex
done
clear
View Answer play_arrow
In which of the following animal tympanum represents the ear and skin is moist
A)
Testudo and Bungarus
done
clear
B)
Crocadilus and Salamandra
done
clear
C)
Rana and Salamandra
done
clear
D)
Chelone and Hemidactylus
done
clear
View Answer play_arrow
Select the odd one with respect to sexuality of organism.
A)
Waucheria
done
clear
B)
Ascaris
done
clear
C)
Neres
done
clear
D)
Hirudinarea
done
clear
View Answer play_arrow
Chaanocytes are the characteristics cells of
(A) Sycon (B) Euspongea (C) Pennatula (D) Ctenophora (E) Cliona
A)
A, B and E only
done
clear
B)
A, B and C
done
clear
C)
A and B only
done
clear
D)
A, B and D
done
clear
View Answer play_arrow
Select incorrect for Neuroglial cells
A)
It protects and supports neurons
done
clear
B)
When this cell is suitably stimulated, an electrical disturbance is generated which swiftly travels along its PM
done
clear
C)
Neuroglial makes up more than one half the volume of neural tissue in our body
done
clear
D)
All arc correct
done
clear
View Answer play_arrow
Which of the following is not present in mucosa of intestine.
A)
Intestinal gland
done
clear
B)
Brunner's gland
done
clear
C)
Crypts of lieber kuhn
done
clear
D)
All are present in mucosa of intestine
done
clear
View Answer play_arrow
Uridine is
A)
\[{{N}_{2}}\] base
done
clear
B)
Sugar + \[{{N}_{2}}\] base
done
clear
C)
\[P{{O}_{4}}\] + sugar +\[{{N}_{2}}\] base
done
clear
D)
\[P{{O}_{4}}\] + \[{{N}_{2}}\] base
done
clear
View Answer play_arrow
Trypsinogen is activated by an enzyme __________ secreted by intestinal mucosa.
A)
Erepsin
done
clear
B)
Enterokinase
done
clear
C)
Carboxy peptidase
done
clear
D)
Enterogastron
done
clear
View Answer play_arrow
This enzyme present in salvia acts as an antibacterial agent for preventing microbial infection
A)
Lysozyme
done
clear
B)
Chitinase
done
clear
C)
Thiocyanate
done
clear
D)
Salivary - amylase
done
clear
View Answer play_arrow
The hyper production of growth hormone during growing stage in mammals results in a condition
A)
Dwarfism
done
clear
B)
Nanism
done
clear
C)
Tanism
done
clear
D)
Gigantism
done
clear
View Answer play_arrow
Out of the following classes ofimmunoglobin which one is known to be effective in causing Rh-incompatibility reaction?
A)
IgD
done
clear
B)
IgE
done
clear
C)
IgG
done
clear
D)
IgA
done
clear
View Answer play_arrow
One among the following is not associated with pancreatic juice.
A)
Nucleotidase
done
clear
B)
Steapsin
done
clear
C)
Nucleases
done
clear
D)
Amylopsin
done
clear
View Answer play_arrow
Periplaneta can move on smooth surface with the help of
A)
Claws
done
clear
B)
Elytra
done
clear
C)
Plantulac and Tegmma
done
clear
D)
Arolium and plantulal.
done
clear
View Answer play_arrow
The vector used to transfer gene to produce pest resistant tobacco plant is
A)
X phase
done
clear
B)
PUC - 18
done
clear
C)
Ti-plasmid
done
clear
D)
PBR-322
done
clear
View Answer play_arrow
Which layer of uterus undergoes thickening under the effect of progesterone
A)
Peritoneum
done
clear
B)
Mymetrium
done
clear
C)
Perimetrium
done
clear
D)
Endometrium
done
clear
View Answer play_arrow
Select the venereal disease caused by a virus
A)
Syphilis
done
clear
B)
Gonorrhoea
done
clear
C)
Genital herpes
done
clear
D)
Trichomoniasis
done
clear
View Answer play_arrow
Which hormone is responsible to regulate calcium and phosphate balance in body?
A)
Thyrocalcitonin
done
clear
B)
Paratharmone
done
clear
C)
Thyroxine
done
clear
D)
Both (A) and (B)
done
clear
View Answer play_arrow
Select the objective of Animal breeding
A)
Increase production of wool milk
done
clear
B)
Increase resistance of various disease
done
clear
C)
Improve growth rate
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which phase of menstrual cycle remains fairly constant even if the cycle is of 3-1 days
A)
Secretory phase
done
clear
B)
Folicular phase
done
clear
C)
Bleeding phase
done
clear
D)
Proliferative phase
done
clear
View Answer play_arrow
Most abundant protein in animal world is
A)
Insulin
done
clear
B)
Keratin
done
clear
C)
Rubisco
done
clear
D)
Collagen
done
clear
View Answer play_arrow
How many mature eggs are typically produced by each ovary of non pregnant woman each year?
A)
52
done
clear
B)
24
done
clear
C)
12
done
clear
D)
6
done
clear
View Answer play_arrow
The part of neuron that receives nerve impulse and pass on graded electric potential towards the soma arc
A)
Dendrites
done
clear
B)
Axon
done
clear
C)
Axon hilock
done
clear
D)
Myelin sheath
done
clear
View Answer play_arrow
The extinct human who lived 1 lac to forty thousand years ago in Europe, Asia with short stature, Large eyebrows, retreating foreheads, large jaw with heavy teeth stocky body was
A)
Neanderthal human
done
clear
B)
Ramapithecus
done
clear
C)
Dryopithecus
done
clear
D)
Cromagnon man
done
clear
View Answer play_arrow
Menstruation is triggered by an abrupt decline in amount of a hormone secreted by
A)
Corpus luteum
done
clear
B)
Pituitary gland
done
clear
C)
Primary follicle
done
clear
D)
All of these
done
clear
View Answer play_arrow
Select technique that is odd with respect to diagnosis of AIDS.
A)
ELISA
done
clear
B)
Southern blotting
done
clear
C)
PCR
done
clear
D)
Western blotting
done
clear
View Answer play_arrow
The cranial nerve that is not related to movement of eyeball as it does not inewate muscles attaching eye ball to orbit
A)
Abducens
done
clear
B)
Trochlear
done
clear
C)
Optic
done
clear
D)
Oculomotar
done
clear
View Answer play_arrow
Select most common symptom of silicosis.
A)
Collapsing of Alveoli
done
clear
B)
Fibrosis of upper parts of lungs
done
clear
C)
Increase in thickening of mucus in lungs
done
clear
D)
Inflammation of bronchi!
done
clear
View Answer play_arrow
Interferons protect non-infected cells from infection by virus. They belong to which barrier?
A)
Physiological
done
clear
B)
Cytokine
done
clear
C)
Physical
done
clear
D)
Cellular
done
clear
View Answer play_arrow
Animals which possess cleidoic eggs exhibit
A)
external fertilization and internal development
done
clear
B)
internal fertilization and external development
done
clear
C)
internal fertilization and internal development
done
clear
D)
external fertilization and external development
done
clear
View Answer play_arrow
Which one of the following is categorized as a parasite in true sense?
A)
The cuckoo lays its egg in nest of crow
done
clear
B)
The female anopheles bites and sucks blood from hum an
done
clear
C)
Human foetus developing inside the uterus draws the nutrition from mother
done
clear
D)
Head louse living on the human scalp as well as laying eggs on human hairs
done
clear
View Answer play_arrow
Which one of the following group of animals is correctly matched with its characteristics feature without any exception?
A)
Mammalia give birth to young ones
done
clear
B)
Reptilia possess 3 chambered heart with an incompletely divided ventricles
done
clear
C)
Chordata possess a mouth with an upper and lower jaw
done
clear
D)
Chondrichthyes possess cartilaginous Endoskeleton
done
clear
View Answer play_arrow
Volkmanns canals connects
A)
Lacunae to lacunae
done
clear
B)
Haversian canal with matrix
done
clear
C)
Haversian canal with haversian canal
done
clear
D)
Haversian canal with marrow cavity
done
clear
View Answer play_arrow
Which one of the following is correct pairing of a body part and kind of muscle tissue that moves it?
A)
Heart wall - Involuntary unstriated muscle
done
clear
B)
Biceps of upper arm - smooth muscle fibre
done
clear
C)
Abdominal wall - smooth muscle
done
clear
D)
Iris - Involuntary smooth muscle
done
clear
View Answer play_arrow
Earthworms have no skeleton, but during burrowing, the anterior end becomes turgid and acts as a hydraulic skeleton. It is due to
A)
Blood
done
clear
B)
Gut peristalisis
done
clear
C)
Setae
done
clear
D)
Coelomic fluid
done
clear
View Answer play_arrow
Open circulatory system is found in
A)
Cockroach
done
clear
B)
Earthworm
done
clear
C)
Snail
done
clear
D)
Both (A) and (C)
done
clear
View Answer play_arrow
Which one precedes reformation of nuclear envelope during M. Phase of cell cycle?
A)
Transcription from chromosomes and reassembly of nuclear lamina
done
clear
B)
Formation of contractile ring and phragmoplast
done
clear
C)
Formation of contractile ring and transcription from chromosome
done
clear
D)
Deondcnsation of chromosome and appearance of Nucler lamina
done
clear
View Answer play_arrow
What is true about RBC in human?
A)
They carry about 25% of \[C{{O}_{2}}\]
done
clear
B)
They transport 99.5% of \[{{O}_{2}}\]
done
clear
C)
They transport about 80% of \[{{O}_{2}}\] only and rest 20% of its is transported in dissolved state in blood plasma
done
clear
D)
They never carry \[C{{O}_{2}}\]
done
clear
View Answer play_arrow
During the propagation of a nerve impulse, the action potential results from the movement of
A)
\[N{{a}^{+}}\] ions from intracellular fluid to extra-cellular fluid
done
clear
B)
\[{{K}^{+}}\] ions from extracellular fluid to intracellular fluid
done
clear
C)
\[{{K}^{+}}\] ions from intracellular fluid to extra- cellular fluid
done
clear
D)
\[N{{a}^{+}}\] ions from extracellular fluid to intracellular fluid
done
clear
View Answer play_arrow
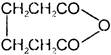
 A and B are
A and B are 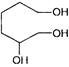 in both cases
in both cases 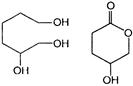
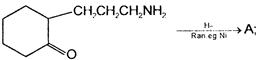 compound A is,
compound A is,