A bulb (100W, 200V) is attached to a voltage of 160 V. The power dissipation is :-
A)
64W
done
clear
B)
100W
done
clear
C)
32W
done
clear
D)
50W
done
clear
View Answer play_arrow
How much energy will be released when 10 Kg of \[{{U}^{235}}\]is completely converts into energy :-
A)
\[5\times {{10}^{27}}\text{ }MeV\]
done
clear
B)
\[5\times {{10}^{24}}\text{ }MeV\]
done
clear
C)
\[9\times {{10}^{17}}\,J\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
If the electron in hydrogen atom jumps from n orbit to first orbit, the wavelength of the emitted photon is \[\lambda \]. What is value of n - (R = Rydberg constant)
A)
\[\sqrt{\frac{\lambda R}{\lambda R-1}}\]
done
clear
B)
\[\frac{hc}{\lambda }\]
done
clear
C)
\[\frac{R}{\lambda R-1}\]
done
clear
D)
\[\frac{\lambda R}{R-1}\]
done
clear
View Answer play_arrow
A particle of charge q, mass m is moving on a circular track of radius R with angular speed \[\omega \]. The ratio of magnetic moment and angular momentum of particle depends on :-
A)
q, m
done
clear
B)
q, m, \[\omega \]
done
clear
C)
m, \[\omega \]
done
clear
D)
q, \[\omega \]
done
clear
View Answer play_arrow
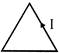
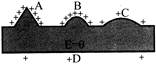
In the following figure an isolated charged conductor is shown. The correct statement will be.
A)
\[{{E}_{A}}>{{E}_{B}}>{{E}_{C}}>{{E}_{D}}\]
done
clear
B)
\[{{E}_{A}}<{{E}_{B}}<{{E}_{C}}<{{E}_{D}}\]
done
clear
C)
\[{{E}_{A}}={{E}_{B}}={{E}_{C}}={{E}_{D}}\]
done
clear
D)
\[{{E}_{A}}={{E}_{B}}\] and \[{{E}_{A}}>{{E}_{D}}\]
done
clear
View Answer play_arrow
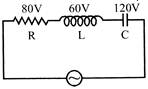
In the circuit shown the potential differences across R, L and C are as given, then the voltage of the A.C. source will be :-
A)
260 V
done
clear
B)
20 V
done
clear
C)
140V
done
clear
D)
100V
done
clear
View Answer play_arrow
If potential applied to an X-ray tube is doubled then value of minimum wavelength of K-series will become :-
A)
Double
done
clear
B)
Half
done
clear
C)
Unchanged
done
clear
D)
Zero
done
clear
View Answer play_arrow
Rutherford experiment of scattering of a-particles showed for the first time that atom has :-
A)
Nucleus
done
clear
B)
Electron
done
clear
C)
Proton
done
clear
D)
Neutron
done
clear
View Answer play_arrow
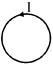
A conducting hollow spherical shell (Shown as in fig is given a charge q. then which is true for electric field at given three positions:-
A)
\[{{E}_{1}}={{E}_{2}}={{E}_{3}}=0\]
done
clear
B)
\[{{E}_{1}}={{E}_{2}}=0,\,{{E}_{3}}\ne 0\]
done
clear
C)
\[{{E}_{1}}=0,\,{{E}_{2}}\ne 0,{{E}_{3}}\ne 0\]
done
clear
D)
\[{{E}_{1}}\ne 0,\,{{E}_{2}}\ne 0,{{E}_{3}}\ne 0\]
done
clear
View Answer play_arrow
In an A.C. series L-C-R circuit \[R=8\Omega ,\,{{X}_{L}}=8\Omega ,\,{{X}_{C}}=2\Omega \]then impedence of circuit will be
A)
\[8\Omega \]
done
clear
B)
\[18\Omega \]
done
clear
C)
\[2\Omega \]
done
clear
D)
\[10\,\Omega \]
done
clear
View Answer play_arrow
Radius of which nucleus is half of radius of the nucleus\[F{{e}^{56}}\]:-
A)
\[{{S}^{32}}\]
done
clear
B)
\[L{{i}^{7}}\]
done
clear
C)
\[{{O}^{16}}\]
done
clear
D)
\[N{{a}^{23}}\]
done
clear
View Answer play_arrow
When will conductivity of a Ge semiconductor decrease :-
A)
On adding donor impurity
done
clear
B)
On adding acceptor impurity
done
clear
C)
On making UV light incident
done
clear
D)
On decreasing the temperature
done
clear
View Answer play_arrow
Which is not the vector form of Biot-Savart law:-
A)
\[\overrightarrow{\delta B}=\frac{KI\overrightarrow{\delta \ell }\times \vec{r}}{{{r}^{3}}}\]
done
clear
B)
\[\overrightarrow{\delta B}=\frac{KI\overrightarrow{\delta \ell }\times \hat{r}}{{{r}^{2}}}\]
done
clear
C)
\[\overrightarrow{\delta B}=\frac{KI\delta \ell \sin \theta }{{{r}^{2}}}\hat{n}\]
done
clear
D)
\[\overrightarrow{\delta B}=\frac{KI\delta \ell \sin \theta }{{{r}^{2}}}\hat{r}\]
done
clear
View Answer play_arrow
How many electrons are flowing per second from a filament of bulb of (220V, HOW)
A)
2
done
clear
B)
\[6.25\times {{10}^{18}}\]
done
clear
C)
\[31.25\times {{10}^{17}}\]
done
clear
D)
\[3.125\times {{10}^{17}}\]
done
clear
View Answer play_arrow
If \[E=200\]Volt, \[R=25\Omega ,\text{ }L=2H\]and \[C=2\mu F\]and the frequency is variable then the current at \[f=0\]and \[f=\infty \]will be respectively :-
A)
0A, 8A
done
clear
B)
8A, 0A
done
clear
C)
8A, 8A
done
clear
D)
0A, 0A
done
clear
View Answer play_arrow
When two different materials A and B having atomic number \[{{Z}_{1}}\] and \[{{Z}_{2}},\] are used as the target in coolidge x-ray tube at different operating voltages \[{{V}_{1}}\] and \[{{V}_{2}}\]respectively their spectrums are found as below:
The correct relation is:-
A)
\[{{V}_{1}}>{{V}_{2}}\]and \[{{Z}_{1}}>{{Z}_{2}}\]
done
clear
B)
\[{{V}_{1}}<{{V}_{2}}\] and \[{{Z}_{1}}<{{Z}_{2}}\]
done
clear
C)
\[{{V}_{1}}<{{V}_{2}}\] and \[{{Z}_{1}}>{{Z}_{2}}\]
done
clear
D)
\[{{V}_{1}}>{{V}_{2}}\] and \[{{Z}_{1}}<{{Z}_{2}}\]
done
clear
View Answer play_arrow
For detecting the light :-
A)
The photodiode has to be forward biased
done
clear
B)
The photo diode has to be reverse biased
done
clear
C)
The LED has to be connected in forward bias
done
clear
D)
The LED has to be connected in reverse bias
done
clear
View Answer play_arrow
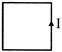
In fig. a network of currents is shown. The magnitude of currents is shown here. The current I will be :-
A)
\[-3\,A\]
done
clear
B)
3A
done
clear
C)
13A
done
clear
D)
20A
done
clear
View Answer play_arrow
In an A.C. circuit the instantaneous values of e.m.f. and current are \[e=10\text{ }sin\text{ }50\,\pi t\]volt and \[I=10\,\sin (50\pi t+\pi /3)\] A. The average power consumed in the circuit is :-
A)
100 watts
done
clear
B)
25 watts
done
clear
C)
50 watts
done
clear
D)
200 watts
done
clear
View Answer play_arrow
Half-life of two radioactive A and B samples are 5 min. and 30 min respectively. Initially decay rate of A is 64 times of B. At which instant they have equal decay rate:-
A)
t = 30 min.
done
clear
B)
t = 5 min.
done
clear
C)
t = 36 min.
done
clear
D)
t = 55 min.
done
clear
View Answer play_arrow
Which type of semiconductor device does not need any bias voltage:-
A)
Photo diode
done
clear
B)
Varactor diode
done
clear
C)
Solar cell
done
clear
D)
Transistor
done
clear
View Answer play_arrow
Magnetic susceptibility \[({{\chi }_{m}})\]of which material not depend upon temperature :-
A)
Paramagnetic
done
clear
B)
Diamagnetic
done
clear
C)
Ferromagnetic
done
clear
D)
None
done
clear
View Answer play_arrow
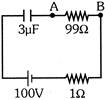
The potential difference between point A & B in steady state will be:-
A)
99 V
done
clear
B)
100 V
done
clear
C)
97 V
done
clear
D)
Zero
done
clear
View Answer play_arrow
A capacitor, an inductor and an electric bulb are connected in series to an a. c. supply of variable frequency. As the frequency of the supply is increased gradually, from zero then the electric bulb is found to
A)
Increase in brightness
done
clear
B)
Decrease in brightness
done
clear
C)
Increase, reach a maximum and then decrease in brightness
done
clear
D)
Show no change in brightness
done
clear
View Answer play_arrow
Which relation can not be apply for a moving electron:-
A)
\[\lambda =\frac{h}{p}\]
done
clear
B)
\[\lambda =\frac{hc}{K.E.}\]
done
clear
C)
\[\lambda =\frac{h}{\sqrt{2mK.E.}}\]
done
clear
D)
\[K.E.=qv\]
done
clear
View Answer play_arrow
The CE amplifier has voltage gain equal to 200 and its input signal is \[0.5\,\cos \,(313\,t)\,V\]the output signal will be equal to .......... volt:-
A)
\[100\,\cos \,(313t+90)\]
done
clear
B)
\[100\,\cos \,(313t+180)\]
done
clear
C)
\[100\,\cos \,\,(493\,t)\]
done
clear
D)
\[0.5\,\cos \,\,(313\,t+200)\]
done
clear
View Answer play_arrow
Two charged spheres having radii \[{{R}_{1}}\] and \[{{R}_{2}}=5\,{{R}_{1}}\] are joined with a wire, then the ratio of electric field on their surface is :-
A)
5
done
clear
B)
\[\frac{1}{5}\]
done
clear
C)
25
done
clear
D)
\[\frac{1}{25}\]
done
clear
View Answer play_arrow
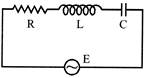
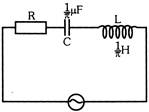
In the circuit shown in figure, the supply has constant rms value V but variable frequency f. Calculate the frequency at which the voltage drop across R is maximum -
A)
100 Hz
done
clear
B)
500 Hz
done
clear
C)
300 Hz
done
clear
D)
None
done
clear
View Answer play_arrow
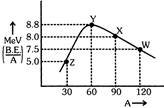
Binding energy per nucleon v/s mass number curve (for nuclei is given:
W, X, Y and Z are four nuclei indicated on the curve. The process that would release energy is:-
A)
\[Y\to 2Z\]
done
clear
B)
\[W\to X+Z\]
done
clear
C)
\[W\to 2Y\]
done
clear
D)
\[X\to Y+Z\]
done
clear
View Answer play_arrow
A short magnet of magnetic moment \[6.75\text{ }A-{{m}^{2}}\]is placed in horizontal plane in such a way that its north pole in south direction and south pole in north direction. If earth field is \[5\times {{10}^{-5}}\text{ }wb/{{m}^{2}}\]then the distance of neutral point from centre of magnet will be:-
A)
10cm
done
clear
B)
20 cm
done
clear
C)
30 cm
done
clear
D)
40 cm
done
clear
View Answer play_arrow
Electrical energy can be achieved :-
A)
Only chemical reactions
done
clear
B)
Only magnetic flux changes
done
clear
C)
Only by thermal energy
done
clear
D)
By all above three
done
clear
View Answer play_arrow
A metal sphere A of radius a charged to potential V. What will be its potential if it is enclosed by a spherical conducting shell B of radius b and the two are connected by a wire
A)
\[2V\]
done
clear
B)
V
done
clear
C)
\[\frac{b}{a}\,V\]
done
clear
D)
\[\frac{a}{b}\,V\]
done
clear
View Answer play_arrow
Balancing length for a standard cell of emf \[1.5\] volt is\[7.5\text{ }m\]. Balancing length for a \[3.5\]ohm resistance, through which a current of \[0.2\] ampere is flowing, will be :-
A)
\[3.5\,m\]
done
clear
B)
\[5.0\,\,m\]
done
clear
C)
\[5.7\text{ }m\]
done
clear
D)
\[6.5\text{ }m\]
done
clear
View Answer play_arrow
A proton and an \[\alpha -particle\] are accelerated through same potential difference. Then ratio of the de-Broglie wavelength \[{{\lambda }_{p}}\] and \[{{\lambda }_{a}}\] will be:-
A)
2
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[2\sqrt{2}\]
done
clear
D)
\[{{\left( \frac{1}{2} \right)}^{1/2}}\]
done
clear
View Answer play_arrow
Magnetic field produced by electrons in atoms and molecules is due to their:-
A)
Spin motion only
done
clear
B)
Orbital motion only
done
clear
C)
Spin and orbital motion both
done
clear
D)
None
done
clear
View Answer play_arrow
For a coil the value of rate of change of flux is \[(6{{t}^{2}}+2)\]in S.I. system. Then the induced emf inside the coil at \[t=2\text{ }sec\]will be:-
A)
24 V
done
clear
B)
26 V
done
clear
C)
12V
done
clear
D)
48 V
done
clear
View Answer play_arrow
The emf of a cell is balanced at 60 cm length of the potentiometer wire. If 3 ohm resistance is inserted from the resistance box connected with the cell, then balancing length obtained is 30 cm. The internal resistance of the cell will be :-
A)
\[1.5\]ohm
done
clear
B)
\[2.0\]ohm
done
clear
C)
\[1.8\]ohm
done
clear
D)
\[3.0\]ohm
done
clear
View Answer play_arrow
The radioactivity of a sample is \[{{R}_{1}}\] at instant \[{{T}_{1}}\] and \[{{R}_{2}}\] at instant \[{{T}_{2}}\]. If the mean life of the specimen is T, the number of atoms that have disintegrated in the time \[({{T}_{2}}-{{T}_{1}})\] is :-
A)
\[{{R}_{1}}{{T}_{1}}-{{R}_{2}}{{T}_{2}}\]
done
clear
B)
\[{{R}_{1}}-{{R}_{2}}\]
done
clear
C)
\[\frac{\left( {{R}_{1}}-{{R}_{2}} \right)}{T}\]
done
clear
D)
\[({{R}_{1}}-{{R}_{2}})\,T\]
done
clear
View Answer play_arrow
Half-life of a radioactive sample is T. At instant t, 60% amount is active. At which instant 30% amount will remain active :-
A)
T
done
clear
B)
\[t+T\]
done
clear
C)
\[{{2}^{t/T}}\]
done
clear
D)
\[\frac{t}{T}\]
done
clear
View Answer play_arrow
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Electromagnetic waves are invented by:-
A)
Maxwell
done
clear
B)
Hertz
done
clear
C)
Marconi
done
clear
D)
None
done
clear
View Answer play_arrow
Thermocouple is based on the principle of-
A)
Seebeck effect
done
clear
B)
Peltier effect
done
clear
C)
Thomson effect
done
clear
D)
Joule effect
done
clear
View Answer play_arrow
A nuclear fusion reaction is given below \[_{1}{{H}^{2}}{{+}_{1}}{{H}^{2}}{{\to }_{2}}H{{e}^{3}}+n+3.2MeV\] How much energy will be generated when 2kg of deuterons are fused.
A)
\[{{10}^{30}}\,eV\]
done
clear
B)
\[2\times {{10}^{23}}\,MeV\]
done
clear
C)
\[{{10}^{22}}\,MeV\]
done
clear
D)
\[{{10}^{23}}\,eV\]
done
clear
View Answer play_arrow
An electron is accelerated by \[200\,kV\] potential. Its energy is:-
A)
\[200\,keV\]
done
clear
B)
\[0.51\,MeV\]
done
clear
C)
\[0.71\,MeV\]
done
clear
D)
\[0.91\,MeV\]
done
clear
View Answer play_arrow
Two long wire which are carrying same current I along x-axis and y-axis what is the value of magnetic field due to both wire at point \[P(3,4)\]:-
A)
\[\frac{{{\mu }_{0}}I}{12\pi }\]
done
clear
B)
\[\frac{{{\mu }_{0}}I}{24\pi }\]
done
clear
C)
\[\frac{{{\mu }_{0}}I}{48\pi }\]
done
clear
D)
\[\frac{{{\mu }_{0}}I}{6\pi }\]
done
clear
View Answer play_arrow
The value of\[{{E}_{red}}\]. of a hydrogen electrode at \[pH=0\]and \[298\,K,\]1 atm is:-
A)
\[0.59\,V\]
done
clear
B)
zero volt
done
clear
C)
\[-0.59\,V\]
done
clear
D)
\[-0.059\,V\]
done
clear
View Answer play_arrow
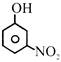
Which of the following is most acidic:-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Consider following two configurations :-
\[X:\,[Ne]\,\,3{{s}^{2}},\,3p_{x}^{1},\,\,3p_{y}^{1},\,3p_{z}^{1},\,3d_{{{z}^{2}}}^{1}\] \[Y:\,[Ne]\,\,3{{s}^{2}},\,3p_{x}^{2},\,\,3p_{y}^{1},\,3p_{z}^{1}\]
what is true about them ;-
A)
IP of \[X>Y\]
done
clear
B)
'X' belongs to d-block, and 'Y to p -block
done
clear
C)
covalence of X is four and that of Y is two
done
clear
D)
Molecules of X and Y obeys octet rule
done
clear
View Answer play_arrow
Wh.ch of the following statement is correct.
A)
\[P{{b}^{+4}}\]is good reducing agent
done
clear
B)
\[G{{e}^{+2}}\] is good oxidising agent
done
clear
C)
\[S{{n}^{+4}}\] is good oxidising agent
done
clear
D)
\[P{{b}^{+4}}\] is good oxidising agent
done
clear
View Answer play_arrow
\[{{N}_{2}}+3{{H}_{2}}2N{{H}_{3}}\]is a gaseous phase equilibrium reaction taking place in a 5 litre flask at 400 K. For this:-
A)
\[{{K}_{C}}={{K}_{X}}\]
done
clear
B)
\[{{K}_{c}}=25{{K}_{X}}\]
done
clear
C)
\[{{K}_{X}}=25{{K}_{C}}\]
done
clear
D)
\[{{K}_{c}}=5{{K}_{X}}\]
done
clear
View Answer play_arrow
When a lead storage battery is charged, it acts as
A)
a fuel cell
done
clear
B)
an electrolytic cell
done
clear
C)
a galvanic cell
done
clear
D)
a concentration cell
done
clear
View Answer play_arrow
A)
\[I<II<III<IV\]
done
clear
B)
\[I<IV<II<III\]
done
clear
C)
\[IV<III<II<I\]
done
clear
D)
\[I<II<IV<III\]
done
clear
View Answer play_arrow
Which of the following condition is in favour of formation of ionic bond:-
A)
Total \[\Delta H=+ve\]
done
clear
B)
high electrical charge on cation and. anion
done
clear
C)
high IP of non metal and less EA of metal
done
clear
D)
less electrical charge on cation and anion
done
clear
View Answer play_arrow
The value of equilibrium constant for self ionisation of water is :-
A)
\[55.5\times {{10}^{-18}}\]
done
clear
B)
\[5.55\times {{10}^{-18}}\]
done
clear
C)
\[3.25\times {{10}^{-18}}\]
done
clear
D)
\[1\times {{10}^{-18}}\]
done
clear
View Answer play_arrow
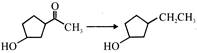
The IUPAC name of the following compound is \[{{(C{{H}_{3}})}_{3}}CC{{H}_{2}}CH=C{{H}_{2}}\]
A)
2,2-Dimethyl-4-pentene
done
clear
B)
4,4-Dimethyl-l-pentene
done
clear
C)
1,1,1 -Trimethyl-3-butene
done
clear
D)
4,4,4-Trimethyl-l-butene
done
clear
View Answer play_arrow
A mixture of salicylic acid and (3-naphthol can be separated by :-
A)
\[NaOH\]Solution
done
clear
B)
\[NaHC{{O}_{3}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
Dil. \[HCl\]
done
clear
View Answer play_arrow
What is wrong about" Ozone" molecule:-
A)
\[O-O\] bond order is \[1.5\]
done
clear
B)
\[O-O\] bond parameters are same due to resonance
done
clear
C)
It is bent molecule due to \[\ell p-\ell p\] and \[\ell p-bp\]repulsion
done
clear
D)
It is a polar molecule
done
clear
View Answer play_arrow
Al dissolves in molten \[NaOH\]and forms \[{{H}_{2}}\] and.
A)
\[N{{a}_{3}}Al{{O}_{3}}\]
done
clear
B)
\[NaAl{{O}_{2}}\]
done
clear
C)
\[Al{{(OH)}_{3}}\]
done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
View Answer play_arrow
Addition of which of the following solutions will have no effect on the pH of the 100 ml of \[0.1\text{ }M\text{ }HCl\]
A)
\[100\text{ }ml\]of \[0.1\text{ }M\text{ }NaOH\]
done
clear
B)
\[100\text{ }ml\]of Hp
done
clear
C)
\[100\text{ }ml\] of \[0.1\text{ }M\,\,HCl\]
done
clear
D)
\[100\text{ }ml\]of \[~NaCl\]
done
clear
View Answer play_arrow
Which of the following has the lowest dipole moment:-
A)
done
clear
B)
\[C{{H}_{3}}-C\equiv C-C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C\equiv CH\]
done
clear
D)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Which of the following compounds does not represent a meso form :-
A)
Cis-1, 2-Dimethyl cyclopropane
done
clear
B)
Cis-1, 3-Dimethyl cyclobutane
done
clear
C)
Cis-1, 3-Dimethyl cyclopentane
done
clear
D)
Cis-1, 2-Dimethyl cyclobutane
done
clear
View Answer play_arrow
A diatomic homonuclear molecule having molecular electronic configuration:- \[{{X}_{2}}:\,\,{{(\sigma 1s)}^{2}}\,{{(\sigma *1s)}^{2}}\,{{(\sigma 2s)}^{2}}{{(\sigma *2s)}^{2}}(\pi 2p_{x}^{2}=\pi 2p_{y}^{2})\]pick out the true statement about it :-
A)
Its bond order is \[3.0\]
done
clear
B)
On addition of an electron bond order decrease
done
clear
C)
It has a double bond consist of only two pi bonds
done
clear
D)
It has four electron pairs in ABMO
done
clear
View Answer play_arrow
What is the molality of aq. solution of \[{{C}_{2}}{{H}_{5}}OH\] in which mole fraction of \[{{C}_{2}}{{H}_{5}}OH\]is\[0.04\]:-
A)
\[2.13\text{ }m\]
done
clear
B)
\[0.213\text{ }m\]
done
clear
C)
\[41.66\text{ }m\]
done
clear
D)
\[0.041\,\,m\]
done
clear
View Answer play_arrow
The number of possible enantiomeric pairs that can be produced during monochlorination of 2-methyl butane is :-
A)
3
done
clear
B)
1
done
clear
C)
4
done
clear
D)
2
done
clear
View Answer play_arrow
A mixture of benzaldehyde and formaldehyde on heating with aqueous \[NaOH\]solution gives :-
A)
Benzyl alcohol and sodium formate
done
clear
B)
Sodium benzoate and methyl alcohol
done
clear
C)
Sodium benzoate and sodium formate
done
clear
D)
Benzyl alcohol and methyl alcohol
done
clear
View Answer play_arrow
In the reaction \[K{{I}_{(aq)}}+{{I}_{2}}\to K{{I}_{3}}\] (water soluble)
A)
The solution of product possess 4 ions/mole
done
clear
B)
In the product Lewis acid is \[{{k}^{+}}\]
done
clear
C)
In the product Lewis base Is \[{{H}_{2}}{{O}_{2}}+N{{a}_{2}}C{{O}_{3}}\xrightarrow{{}}N{{a}_{2}}{{O}_{2}}+C{{O}_{2}}+{{H}_{2}}O\]
done
clear
D)
In the solution Lewis acid is \[{{I}_{2}}\]
done
clear
View Answer play_arrow
For the gaseous reaction: \[2A(g)+B(g)\xrightarrow{{}}\] Product. The rate of reaction will be increased........ Times if the pressure is increased three times:-
A)
27
done
clear
B)
9
done
clear
C)
3
done
clear
D)
12
done
clear
View Answer play_arrow
How much electricity in terms of Faraday is required to produce \[20.0\text{ }g\]of Ca from molten \[CaC{{l}_{2}}\].
A)
\[\text{1 }F\]
done
clear
B)
\[2\text{ }F\]
done
clear
C)
\[20\text{ }F\]
done
clear
D)
\[0.1\text{ }F\]
done
clear
View Answer play_arrow
Which of the following is not aromatic compound
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The appropriate reagent for the following conversion:-
A)
\[Zn-Hg/HCl\]
done
clear
B)
\[N{{H}_{2}}N{{H}_{2}}/\overset{O-}{\mathop{OH}}\,\]
done
clear
C)
\[{{H}_{2}}/Ni\]
done
clear
D)
Red \[P\text{ }/HI\]
done
clear
View Answer play_arrow
A complex of Pt, \[N{{H}_{3}}\] and \[Cl\]produces four ions per molecule in the aqueous solution. The structure pertaining to above observation is:-
A)
Optically active
done
clear
B)
Outer orbital complex
done
clear
C)
Pentaamminechloroplatinum (IV) chloride
done
clear
D)
Having three ionisation isomers
done
clear
View Answer play_arrow
If potassium chlorate is 80% pure then 48g of oxygen would be produced from (atomic mass of k = 39)
A)
\[153.12g\] of \[KC1{{O}_{3}}\]
done
clear
B)
\[122.5\text{ }g\]of \[KC1{{O}_{3}}\]
done
clear
C)
\[245\text{ }g\]of \[KC1{{O}_{3}}\]
done
clear
D)
\[98.0\text{ }g\]of \[KC1{{O}_{3}}\]
done
clear
View Answer play_arrow
The most stable carbonium ion is :-
A)
\[C{{H}_{3}}\,\overset{\oplus }{\mathop{C{{H}_{2}}}}\,\]
done
clear
B)
\[{{(C{{H}_{3}})}_{3}}\overset{\oplus }{\mathop{C}}\,\]
done
clear
C)
\[Ph\,\overset{\oplus }{\mathop{C{{H}_{2}}}}\,\]
done
clear
D)
\[P{{h}_{3}}\,\overset{\oplus }{\mathop{C}}\,\]
done
clear
View Answer play_arrow
Among the following, the weakest base is :-
A)
\[PhC{{H}_{2}}-N{{H}_{2}}\]
done
clear
B)
\[PhN{{H}_{2}}\]
done
clear
C)
\[PhC{{H}_{2}}-NH-C{{H}_{3}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}N\]
done
clear
View Answer play_arrow
The mixing of which of the following will not produce blue colouration :-
A)
\[N{{H}_{4}}O{{H}_{(aq)}}\] and \[CuS{{O}_{4(aq)}}\]
done
clear
B)
Iron (III) chloride and KSCN
done
clear
C)
Iron (III) chloride and \[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
D)
adding anhyd. \[CuS{{O}_{4}}\]to water
done
clear
View Answer play_arrow
In which of the following transition will the wavelength be minimum :-
A)
\[n=6\]to \[n=4\]
done
clear
B)
\[n=4\] to \[n=2\]
done
clear
C)
\[n=3\]to\[n=1\]
done
clear
D)
\[n=2\]to \[n=1\]
done
clear
View Answer play_arrow
XL of \[{{N}_{2}}\] at NTP contains \[3\times {{10}^{22}}\]molecules the number of molecules in \[\frac{X}{2}L\] of ozone at NTP will be :-
A)
\[3\times {{10}^{22}}\]
done
clear
B)
\[1.5\times {{10}^{22}}\]
done
clear
C)
\[1.5\times {{10}^{21}}\]
done
clear
D)
\[1.5\times {{10}^{11}}\]
done
clear
View Answer play_arrow
A)
\[III>I>II>IV\]
done
clear
B)
\[IV>I>II>III\]
done
clear
C)
\[I>II>III>IV\]
done
clear
D)
\[II>I>III>IV\]
done
clear
View Answer play_arrow
A condensation polymer among the following is
A)
Dacron
done
clear
B)
PVC
done
clear
C)
Polystyrene
done
clear
D)
Teflon
done
clear
View Answer play_arrow
Which of the following system has maximum magnetic moment:-
A)
\[{{d}^{7}}({{\Delta }_{0}}<PE)\]
done
clear
B)
\[{{d}^{6}}({{\Delta }_{0}}>PE)\]
done
clear
C)
\[{{d}^{5}}\] (tetrahedral)
done
clear
D)
\[{{d}^{4}}\] (low spin)
done
clear
View Answer play_arrow
The heat of combustion of sucrose \[({{C}_{12}}{{H}_{22}}{{O}_{11}})\] is\[-5.65\times {{10}^{3}}\,kJ.\]If complete combustion of \[1.0\text{ }kg\]sucrose is done, heat evolve will be :-
A)
\[1.65\times {{10}^{4}}\text{ }kJ\]
done
clear
B)
\[5.25\times {{10}^{3}}\text{ }kJ\]
done
clear
C)
\[7.38\times {{10}^{5}}\text{ }kJ\]
done
clear
D)
\[3.51\times {{10}^{5}}\text{ }kJ\]
done
clear
View Answer play_arrow
When Mercuric iodide is added to the aqueous solution of \[KI,\]then :-
A)
Freezing point is raised
done
clear
B)
Freezing point is lowered
done
clear
C)
boiling point does not change
done
clear
D)
freezing point does not change
done
clear
View Answer play_arrow
The treatment of benzene with isobutene in the presence of sulphuric acid gives :-
A)
Isobutyl benzene
done
clear
B)
t-butyl benzene
done
clear
C)
n-butyl benzene
done
clear
D)
No reaction
done
clear
View Answer play_arrow
Cobalt is present in :-
A)
Vitamin \[{{B}_{2}}\]
done
clear
B)
Vitamin \[{{B}_{1}}\]
done
clear
C)
Vitamin \[{{B}_{6}}\]
done
clear
D)
Vitamin \[{{B}_{12}}\]
done
clear
View Answer play_arrow
Hg container are made of:-
A)
\[Fe\]
done
clear
B)
\[Pb\]
done
clear
C)
\[Al\]
done
clear
D)
\[Zn\]
done
clear
View Answer play_arrow
One mole of an ideal gas at 500 K is expended isothermally from an initial volume of 1 litre to 10 litre. The AE for this process is:-
A)
\[163.7\text{ }cal\]
done
clear
B)
Zero
done
clear
C)
\[1381.1\text{ }cal\]
done
clear
D)
\[9\,L\,atm\]
done
clear
View Answer play_arrow
In the reaction \[{{H}_{2}}{{O}_{2}}+N{{a}_{2}}C{{O}_{3}}\xrightarrow{{}}N{{a}_{2}}{{O}_{2}}+C{{O}_{2}}+{{H}_{2}}O\] the substance undergoing oxidation is :-
A)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
B)
None
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
View Answer play_arrow
The order of reactivity of the following alcohols towards cone. \[HCl\]is :- (I)
(II)
(III)
(IV)
A)
\[I>II>III>IV\]
done
clear
B)
\[~I>II>II>IV\]
done
clear
C)
\[\text{IV}>III>II>I\]
done
clear
D)
\[\text{IV}>III>I>II\]
done
clear
View Answer play_arrow
An element Unq, Americans named is Ruther fordium (Rf) and Soviets named is Kurchatovium (Ku) is :-
A)
a p-block element, placed in IV A group
done
clear
B)
a f-block, first post lanthanoide elements
done
clear
C)
a lanthanoid, placed in III-B group
done
clear
D)
a d-block, first post actinoide elements
done
clear
View Answer play_arrow
Which of the following is not Thermite process:-
A)
\[F{{e}_{2}}{{O}_{3}}+2Al\xrightarrow{{}}A{{l}_{2}}{{O}_{3}}+2Fe\]
done
clear
B)
\[2Fe+A{{l}_{2}}{{O}_{3}}\xrightarrow{{}}F{{e}_{2}}{{O}_{3}}+2Al\]
done
clear
C)
\[ZnO+C\xrightarrow{{}}CO+Zn\]
done
clear
D)
Both &
done
clear
View Answer play_arrow
For a critical study of secondary growth in plants, which one of the following pairs is suitable?
A)
Wheat and maiden hair fem
done
clear
B)
Sugarcane and sunflower
done
clear
C)
Teak and pine
done
clear
D)
Deodar and fem
done
clear
View Answer play_arrow
Spore dissemination in some liverworts is aided by:
A)
Peristome teeth
done
clear
B)
Elaters
done
clear
C)
Indusium
done
clear
D)
Calyptra
done
clear
View Answer play_arrow
Adaptive radiation refers to :-
A)
Power of adaptation in an individual to a variety of environments
done
clear
B)
Adaptations due to Geographical isolation
done
clear
C)
Evolution of different species from a common ancestor
done
clear
D)
Migration of members of a species to different geographical areas
done
clear
View Answer play_arrow
One of the important consequences of geographical isolation is :-
A)
Random creation of new species
done
clear
B)
No change in the isolated fauna
done
clear
C)
Preventing Speciation
done
clear
D)
Speciation through reproductive isolation
done
clear
View Answer play_arrow
Which one of the following is a matching pair of a body feature and the animal possessing it
A)
Ventral heart - Scorpion
done
clear
B)
Post-anal tail - Octopus
done
clear
C)
Ventral central - Leech nervous system
done
clear
D)
Pharyngeal gill slits - Chameleon absent in embryo
done
clear
View Answer play_arrow
All enzymes of TCA cycle are located in the mitochondrial martix except one which is located in inner mitochondrial membranes in eukaryotes and in cytosol in prokaryotes. This enzyme is:-
A)
Succinate dehydrogenase
done
clear
B)
Lactate dehydrogenase
done
clear
C)
Isocitrate dehydrogenase
done
clear
D)
Malate dehydrogenase
done
clear
View Answer play_arrow
Which one of the following elements is not an essential micronutrient for plant growth?
A)
Ca
done
clear
B)
Mn
done
clear
C)
Zn
done
clear
D)
Cu
done
clear
View Answer play_arrow
In the human female, menstruation can be deferred by the administration of:-
A)
FSH only
done
clear
B)
LH only
done
clear
C)
Combination of FSH and LH
done
clear
D)
Combination of estrogen and progesterone
done
clear
View Answer play_arrow
A high density of elephant population in an area can result in :-
A)
Predation on one another
done
clear
B)
Mutualism
done
clear
C)
Intra specific competition
done
clear
D)
Inter specific competition
done
clear
View Answer play_arrow
In the hexaploid wheat, the haploid (n) and basic(x) numbers of chromosomes are :
A)
\[n=21\]and \[x=7\]
done
clear
B)
n = 7 and \[x=21\]
done
clear
C)
\[n=21\]and \[x=21\]
done
clear
D)
\[n=21\]and\[~x=14\]
done
clear
View Answer play_arrow
The two polynucleotide chain in DNA are :-
A)
Semiconservative
done
clear
B)
Parallel
done
clear
C)
Discontinuous
done
clear
D)
Antiparallel
done
clear
View Answer play_arrow
Praying mantis is a good example of -
A)
Mullerian mimicry
done
clear
B)
Warning colouration
done
clear
C)
Social insects
done
clear
D)
Camouflage
done
clear
View Answer play_arrow
Which one of the following is not included under in-situ conservation?
A)
Sanctuary
done
clear
B)
Botanical garden
done
clear
C)
Biosphere reserve
done
clear
D)
National park
done
clear
View Answer play_arrow
The bacterium (Clostridium botulinum) that causes botulism is -
A)
A facultative anaerobe
done
clear
B)
An obligate anaerobe
done
clear
C)
A facultative aerobe
done
clear
D)
An obligate aerobe
done
clear
View Answer play_arrow
Which of the following pairs of an animal and a plant represents endangered organisms in India -
A)
Bentinckia nicobarica and Red Panda
done
clear
B)
Tamarind and Rhesus monkey
done
clear
C)
Cinchona and Liopard
done
clear
D)
Banyan and Black buck
done
clear
View Answer play_arrow
Which one of the following does not act as a neurotransmitter?
A)
Acetylcholine
done
clear
B)
Epinephrine
done
clear
C)
Norepinephrine
done
clear
D)
Cortisone
done
clear
View Answer play_arrow
Limit of BOD prescribed by Central Pollution Control Board for the discharge of industrial and municipal waste waters into natural surface waters, is -
A)
\[<3-0\text{ }ppm~\]
done
clear
B)
\[<10\text{ }ppm\]
done
clear
C)
\[<100\text{ }ppm\]
done
clear
D)
\[<30\text{ }ppm\]
done
clear
View Answer play_arrow
In order to obtain virus-free plants through tissue culture the best method is -
A)
Protoplast culture
done
clear
B)
Embryo rescue
done
clear
C)
Anther culture
done
clear
D)
Meristem culture
done
clear
View Answer play_arrow
Both sickle cell anemia and Huntington's chorea are-
A)
Bacteria-related diseases
done
clear
B)
Congenital disorders
done
clear
C)
Pollutant-induced disorders
done
clear
D)
Virus-related diseases
done
clear
View Answer play_arrow
In contrast to Annelids the Platyhelminths show
A)
Absence of body cavity
done
clear
B)
Presence of pseudocoel
done
clear
C)
Radial symmetry
done
clear
D)
Bilateral symmetry
done
clear
View Answer play_arrow
In ornithine cycle, which of the following wastes removed from the blood -
A)
\[C{{O}_{2}}\] and ammonia
done
clear
B)
Ammonia and urea
done
clear
C)
\[C{{O}_{2}}\] and urea
done
clear
D)
Urea and urine
done
clear
View Answer play_arrow
Which of the following is not used for disinfection of drinking water -
A)
Chlorine
done
clear
B)
Phenyl
done
clear
C)
Chloramine
done
clear
D)
Ozone
done
clear
View Answer play_arrow
Epithelial cells of the intestine involved in food absorption have on their surface -
A)
Zymogen granules
done
clear
B)
Pinocytic vesicles
done
clear
C)
Phagocytic vesicles
done
clear
D)
Microvilli
done
clear
View Answer play_arrow
Centromere is required for -
A)
Movement of chromosomes towards poles
done
clear
B)
Cytoplasmic cleavage
done
clear
C)
Crossing over
done
clear
D)
Transcription
done
clear
View Answer play_arrow
During transcription holoenzyme RNA polymerase binds to a DNA sequence and the DNA assumes a saddle like structure at that point. What is that sequence called -
A)
CAAT box
done
clear
B)
GGTT box
done
clear
C)
AAAT box
done
clear
D)
TATA box
done
clear
View Answer play_arrow
Chlorophyll in chloroplasts is located in -
A)
Grana
done
clear
B)
Pyrenoid
done
clear
C)
Stroma
done
clear
D)
Both grana and stroma
done
clear
View Answer play_arrow
What is a keystone species?
A)
A common species that has plenty of biomass, yet has a fairly low impact on the community's organization
done
clear
B)
A rare species that has minimal impact on the biomass and on other species in the community
done
clear
C)
A dominant species that constitutes a large proportion of the biomass and which affects many other species
done
clear
D)
A species which makes up only a small proportion of the total biomass of a community, yet has a huge impact on the community's organization and survival
done
clear
View Answer play_arrow
In Arthropoda, head and thorax are often fused to form cephalothorax, but in which one of the following classes, is the body divided into head, thorax and abdomen?
A)
Myriapoda
done
clear
B)
Crustacea
done
clear
C)
Arachnida and Crustacea
done
clear
D)
Insecta
done
clear
View Answer play_arrow
Viruses that infect bacteria, multiply and cause their lysis are called -
A)
Lipolytic
done
clear
B)
Lydc
done
clear
C)
Lysogenic
done
clear
D)
Lysozymes
done
clear
View Answer play_arrow
Edible part of mango is:-
A)
Receptacle
done
clear
B)
Epicarp
done
clear
C)
Mesocarp
done
clear
D)
Endocarp
done
clear
View Answer play_arrow
In your opinion, which is the most effective way to conserve the plant diversity of an area.
A)
By creating biosphere reserve
done
clear
B)
By creating botanical garden
done
clear
C)
By developing seed bank
done
clear
D)
By tissue culture method
done
clear
View Answer play_arrow
Duodenum has characteristic Brunner's glands which secrete two hormones called-
A)
Secretin, Cholecystokinin
done
clear
B)
Prolactin, parathormone
done
clear
C)
Extradiol, progesterone
done
clear
D)
Kinase, estrogen
done
clear
View Answer play_arrow
In the ABO system of blood groups if both antigens are present but no antibody, the blood group of the individual would be :-
A)
O
done
clear
B)
AB
done
clear
C)
A
done
clear
D)
B
done
clear
View Answer play_arrow
One of the following is a very unique feature of the mammalian body -
A)
Presence of diaphragm
done
clear
B)
Four chambered heart
done
clear
C)
Rib cage
done
clear
D)
Homeothermy
done
clear
View Answer play_arrow
Recently Govt. of India has allowed mixing of alcohol in petrol. What is the amount of alcohol permitted for mixing in petrol :-
A)
10-15%
done
clear
B)
10%
done
clear
C)
5%
done
clear
D)
2.5%
done
clear
View Answer play_arrow
When a diploid female plant is crossed with a tetraploid male, the ploidy of endosperm cells in the resulting seed is :-
A)
Pentaploidy
done
clear
B)
Diploidy
done
clear
C)
Triploidy
done
clear
D)
Tetraploidy
done
clear
View Answer play_arrow
Which one of the following sequences was proposed by Darwin and Wallace for organic evolution :-
A)
Overproduction, variations, constancy of population size, natural selection
done
clear
B)
Variations, constancy of population size, overproduction, natural selection
done
clear
C)
Overproduction, constancy of population size, variations, natural selection
done
clear
D)
Variations, natural selection, overproduction, constancy of population size
done
clear
View Answer play_arrow
If Henle's loop were absent from mammalian nephron, which of the following is to be expected
A)
There will be no urine formation
done
clear
B)
There will be hardly any change in the quality and quantity of urine formed
done
clear
C)
The urine will be more concentrated
done
clear
D)
The urine will be more dilute
done
clear
View Answer play_arrow
Escherichia coli is used as an indicator organism to determine pollution of water with.
A)
Heavy metals
done
clear
B)
Faecal matter
done
clear
C)
Industrial effluents
done
clear
D)
Pollen of aquatic plants
done
clear
View Answer play_arrow
Industrial melanism is an example of :-
A)
Drug resistance
done
clear
B)
Darkening of skin due to smoke from industries
done
clear
C)
Protective resemblance with the surroundings
done
clear
D)
Defensive adaptation of skin against ultraviolet radiations
done
clear
View Answer play_arrow
In the genetic code dictionary, how many codons are used to code for all the 20 essential amino acids :-
A)
20
done
clear
B)
64
done
clear
C)
61
done
clear
D)
60
done
clear
View Answer play_arrow
During transcription, the DNA site at which RNA polymerase binds is called :-
A)
Promoter
done
clear
B)
Regulator
done
clear
C)
Receptor
done
clear
D)
Enhancer
done
clear
View Answer play_arrow
Which one of the following mineral elements plays an important role in biological nitrogen fixation :-
A)
Copper
done
clear
B)
Manganese
done
clear
C)
Zinc
done
clear
D)
Molybdenum
done
clear
View Answer play_arrow
The apical meristem of the root is present
A)
Only in radicals
done
clear
B)
Only in tap roots
done
clear
C)
Only in adventitious roots
done
clear
D)
In all the roots
done
clear
View Answer play_arrow
Which one of the following pairs of plants are not seed producers :-
A)
Fern and Funaria
done
clear
B)
Funaria and Ficus
done
clear
C)
Ficus and Chlamydomonas
done
clear
D)
Punica and Pinus
done
clear
View Answer play_arrow
In a population, unrestricted reproductive capacity is called as -
A)
Biotic potential
done
clear
B)
Fertility
done
clear
C)
Carring capacity
done
clear
D)
Birth rate
done
clear
View Answer play_arrow
Genetic drift operates in :-
A)
Small isolated population
done
clear
B)
Large isolated population
done
clear
C)
Fast reproductive population
done
clear
D)
Slow reproductive population
done
clear
View Answer play_arrow
Which of the following is used in the treatment of Thyroid cancer :-
A)
\[{{I}_{131}}\]
done
clear
B)
\[{{U}_{238}}\]
done
clear
C)
\[R{{a}_{224}}\]
done
clear
D)
\[{{C}_{14}}\]
done
clear
View Answer play_arrow
In five kingdom system, the main basis of classification:-
A)
Structure of nucleus
done
clear
B)
Nutrition
done
clear
C)
Structure of cell wall
done
clear
D)
Asexual reproduction
done
clear
View Answer play_arrow
Number wild life is continuously decreasing. What is the main reason of this :-
A)
Predation
done
clear
B)
cutting down of forest
done
clear
C)
Destruction of habitat
done
clear
D)
Hunting
done
clear
View Answer play_arrow
Which pigment absorbs the red and far-red light
A)
Cytochrome
done
clear
B)
Phytochrome
done
clear
C)
Carotenoids
done
clear
D)
Chlorophyll
done
clear
View Answer play_arrow
In which animal nerve cell is present but brain is absent:-
A)
Sponge
done
clear
B)
Earthworm
done
clear
C)
Cockroach
done
clear
D)
Hydra
done
clear
View Answer play_arrow
What is the reason of formation of embryoid from pollen grain in tissue culture medium
A)
Cellular totipotency
done
clear
B)
Organogenesis
done
clear
C)
Double fertilization
done
clear
D)
Test tube culture
done
clear
View Answer play_arrow
There are three genes a, b and c. The percentage of crossing over between a and b is 20%, b and c is 28% and a and c is 8%. What is the sequence of genes on chromosome
A)
b, a, c
done
clear
B)
a, b, c
done
clear
C)
a, c, b
done
clear
D)
None
done
clear
View Answer play_arrow
Independent assortment of genes does not takes place when :-
A)
Genes are located on homologous chromosomes
done
clear
B)
Genes are linked and located on same chromosome
done
clear
C)
Genes are located on non-homologous chromosome
done
clear
D)
All the above
done
clear
View Answer play_arrow
When dominant and recessive allels express itself together it is called :-
A)
Co-dominance
done
clear
B)
Dominance
done
clear
C)
Amphidominance
done
clear
D)
Pseudo dominance
done
clear
View Answer play_arrow
Bicarpellary gyanoecium and oblique ovary occurs in -
A)
Mustard
done
clear
B)
Banana
done
clear
C)
Pisum
done
clear
D)
Brinjal
done
clear
View Answer play_arrow
Which breaks dormancy of potato tuber :-
A)
Gibberellin
done
clear
B)
IAA
done
clear
C)
ABA
done
clear
D)
Zeatin
done
clear
View Answer play_arrow
Which fish selectively feed on larva of mosquito
A)
Gambusia
done
clear
B)
Rohu
done
clear
C)
Clarias
done
clear
D)
Exocoetus
done
clear
View Answer play_arrow
Difference in gram © and gram 0 bacteria is due to-
A)
Cell wall
done
clear
B)
Cell membrane
done
clear
C)
Ribosome
done
clear
D)
Cytoplasm
done
clear
View Answer play_arrow
Sl. Proteinaceous pigment which control the activities concerned with light :-
A)
Phytochrome
done
clear
B)
Chlorophyll
done
clear
C)
Anthocyanin
done
clear
D)
Carotenoids
done
clear
View Answer play_arrow
Most abundant organic compound on earth is
A)
Protein
done
clear
B)
Cellulose
done
clear
C)
Lipids
done
clear
D)
Steroids
done
clear
View Answer play_arrow
m-RNA is synthesised on DNA template in which direction:-
A)
\[5'\to 3'\]
done
clear
B)
\[3\to 5\]
done
clear
C)
Both
done
clear
D)
Any
done
clear
View Answer play_arrow
A mutant strain of \[{{T}_{4}}\] - Bacteriophage, R-II, fails to lyse the E-Coli but when two strains \[R-I{{I}^{X}}\]and \[R-I{{I}^{y}}\]are mixed then they lyse the E. Coli What may be the possible reason:-
A)
Bacteriophage transforms in wild
done
clear
B)
It is not mutated
done
clear
C)
Both strains have simillar cistrons
done
clear
D)
Both strains have different cistrons
done
clear
View Answer play_arrow
First life on earth was :-
A)
Cyanobacteria
done
clear
B)
Chemohetrotrophs
done
clear
C)
Autotrophs
done
clear
D)
Photoautotrophs
done
clear
View Answer play_arrow
Which is the first \[C{{O}_{2}}\] Acceptor enzyme in \[{{C}_{4}}\] plants
A)
RuDP Carboxylase
done
clear
B)
Phosphoric acid
done
clear
C)
RUBISCO
done
clear
D)
PEP- Carboxylase
done
clear
View Answer play_arrow
Endosperm is formed during the double fertilization by:-
A)
Two polar nuclei & one male gamete
done
clear
B)
One polar nuclei & One male gamete
done
clear
C)
Ovum and male gamete
done
clear
D)
Two polar nuclei & two male gametes
done
clear
View Answer play_arrow
What is name of joint between ribs and stemum-
A)
Cartilaginous joint
done
clear
B)
Angular joint
done
clear
C)
Gliding joint
done
clear
D)
Fibrous joints
done
clear
View Answer play_arrow
Which pair is correct-
A)
Sweat = temperature regulation
done
clear
B)
Saliva = sense of food taste
done
clear
C)
Sebum = sexual attraction
done
clear
D)
Humerus = Hind leg
done
clear
View Answer play_arrow
A giant rat is formed in the laboratory, what is the reason-
A)
Gene mutation
done
clear
B)
Gene synthesis
done
clear
C)
Gene manipulation
done
clear
D)
Gene replication
done
clear
View Answer play_arrow
ATP is-
A)
Nucleotide
done
clear
B)
Nucleoside
done
clear
C)
Nucliec acid
done
clear
D)
Vitamin
done
clear
View Answer play_arrow
Which of the following is used to manufacture ethanol from starch:-
A)
Penicilline
done
clear
B)
Saccharomyces
done
clear
C)
Azotobactor
done
clear
D)
Lactobacillus
done
clear
View Answer play_arrow
Most stable pesticides:-
A)
Organophosphates
done
clear
B)
Organ chlorines
done
clear
C)
Bordeaux mix.
done
clear
D)
Azederectin
done
clear
View Answer play_arrow
Which induces the development of corpus Luteum:-
A)
LH
done
clear
B)
Oestrogen
done
clear
C)
FSH
done
clear
D)
LTH
done
clear
View Answer play_arrow
What change occurs by changing one base in DNA:-
A)
Always a change of one amino acid in protein.
done
clear
B)
Change in complex sequence of amino acid.
done
clear
C)
Always a change in property of protein
done
clear
D)
Does not necessarily change the phenotype.
done
clear
View Answer play_arrow
Which hormone is concerned with the concentration of urine:-
A)
Oxytocin
done
clear
B)
Vassopressin
done
clear
C)
Prolactin
done
clear
D)
Cortisol
done
clear
View Answer play_arrow
In stomach after physical and chemical digestion food is called:-
A)
Chyme
done
clear
B)
Chyle
done
clear
C)
Amino acid
done
clear
D)
Bolus
done
clear
View Answer play_arrow
Insectivorous plants grow in the soil which is deficient in:-
A)
\[Mg\]
done
clear
B)
\[Ca\]
done
clear
C)
P
done
clear
D)
N
done
clear
View Answer play_arrow
Maximum photosynthesis takes place by:-
A)
Phytoplankton
done
clear
B)
Zooplankton
done
clear
C)
Marsh plants
done
clear
D)
Woody plants
done
clear
View Answer play_arrow
Casparian bands are found in:-
A)
Endodermis
done
clear
B)
Pericycle
done
clear
C)
Periderm
done
clear
D)
Cortex
done
clear
View Answer play_arrow
Recently extinct animal from India is:-
A)
Acinonyx
done
clear
B)
Rhino ceros unicomieus
done
clear
C)
Panthera leo
done
clear
D)
Panthera tigris
done
clear
View Answer play_arrow
Indicator of water pollution:-
A)
E. Coli
done
clear
B)
Chlorella
done
clear
C)
Beggiatoa
done
clear
D)
Ulothrix
done
clear
View Answer play_arrow
Species separated by geographical barriers are called:-
A)
Allopatric
done
clear
B)
Sympatric
done
clear
C)
Sibling
done
clear
D)
Endemic
done
clear
View Answer play_arrow
Vitamin which induces maturation of R.B.C.:-
A)
\[{{B}_{1}}\]
done
clear
B)
\[A\]
done
clear
C)
\[{{B}_{12}}\]
done
clear
D)
\[D\]
done
clear
View Answer play_arrow
Which of the following induces morphogenesis in tissue culture:-
A)
Gibberellin
done
clear
B)
Cytokinin
done
clear
C)
IAA
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Largest sperm of :-
A)
Pinus
done
clear
B)
Cycas
done
clear
C)
Ephedra
done
clear
D)
Sequoia
done
clear
View Answer play_arrow
In angiosperm, characters of flowers are used in classification because :-
A)
Characters of flowers are conservative
done
clear
B)
Flowers are large
done
clear
C)
Flowers are attractive
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Oxidation of palmitic acid yield:-
A)
129 ATP
done
clear
B)
132 ATP
done
clear
C)
38 ATP
done
clear
D)
76 ATP
done
clear
View Answer play_arrow
Mental retardness in man occur due to:-
A)
Loss of one X chromosome
done
clear
B)
Addition of one X chromosome
done
clear
C)
Slight growth in Y
done
clear
D)
Overgrowth in Y
done
clear
View Answer play_arrow
Effect of light and dark rhythm on plants:-
A)
Photonasty
done
clear
B)
Phototropism
done
clear
C)
Photoperiodism
done
clear
D)
Photo morphogenesis
done
clear
View Answer play_arrow
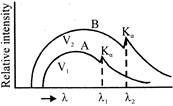 The correct relation is:-
The correct relation is:- 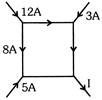
 W, X, Y and Z are four nuclei indicated on the curve. The process that would release energy is:-
W, X, Y and Z are four nuclei indicated on the curve. The process that would release energy is:-