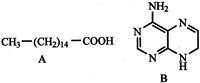A physical quantity is determined P\[=\frac{\sqrt{ab{{c}^{2}}}}{{{d}^{3}}{{e}^{1/3}}}\] measuring a, b, c, d and e separately with the percentage error of 2%, 3%, 2%, 1% and 6% respectively. Minimum amount of error is contributed by the measurement:
A)
b
done
clear
B)
a
done
clear
C)
d
done
clear
D)
e
done
clear
View Answer play_arrow
A particle of mass M, attached to one end of a spiral spring describes S.H.M. When the particle is passing through the mean position another particle of mass m is fixed on mass M. The ratio of frequency of the system in the two cases is:
A)
\[\frac{M}{m+M}\]
done
clear
B)
\[\sqrt{\frac{M}{m+M}}\]
done
clear
C)
\[\frac{m+M}{M}\]
done
clear
D)
\[\sqrt{\frac{m+M}{M}}\]
done
clear
View Answer play_arrow
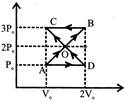
A ball rolls off the top of a stairway with horizontal velocity \[{{v}_{o}}\text{ }m/s.\]If the steps are h metres high and w metres wide, the ball will hit the edge of n111 step, if:
A)
\[n=\frac{2h{{v}_{o}}}{g{{w}^{2}}}\]
done
clear
B)
\[n=\frac{2h{{v}^{2}}_{o}}{gw}\]
done
clear
C)
\[n=\frac{h{{v}^{2}}_{o}}{g{{w}^{2}}}\]
done
clear
D)
\[n=\frac{2h{{v}^{2}}_{o}}{g{{w}^{2}}}\]
done
clear
View Answer play_arrow
A steel wire is suspended vertically from a rigid support. When loaded with a weight in air, it extends by \[{{l}_{a}}\] and when the weight is immersed completely in water, the extension is reduced to\[{{l}_{w}}\]. Then the relative density of:
A)
\[\frac{{{l}_{a}}}{{{l}_{w}}}\]
done
clear
B)
\[\frac{{{l}_{a}}}{{{l}_{a}}-{{l}_{w}}}\]
done
clear
C)
\[\frac{{{l}_{w}}}{{{l}_{a}}-{{l}_{w}}}\]
done
clear
D)
\[\frac{{{l}_{w}}}{{{l}_{a}}}\]
done
clear
View Answer play_arrow
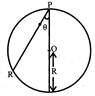
A frictionless wire is fixed between P and R inside a sphere of radius R. A small ball slips along the wire. The time taken by the ball to slip from P to R will be:
A)
\[\frac{2\sqrt{\mathrm{R}}}{\sqrt{\mathrm{g}}\,\mathrm{cos}\,\theta }\]
done
clear
B)
\[\frac{2\sqrt{\mathrm{R}}\,\mathrm{cos}\,\theta }{\mathrm{g}\,}\]
done
clear
C)
\[2\sqrt{\frac{\mathrm{R}}{\mathrm{g}\,}}\]
done
clear
D)
\[\frac{\sqrt{\operatorname{gR}}}{\cos \,\theta \,}\]
done
clear
View Answer play_arrow
A particle gets displaced through \[\hat{i}+2\hat{j}+4\hat{k}\]due to force\[\left( 2\hat{i}-3\hat{j}+16\hat{k} \right)\] N in 3 seconds. The power in watts is:
A)
18
done
clear
B)
6
done
clear
C)
54
done
clear
D)
20
done
clear
View Answer play_arrow
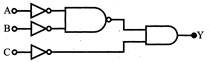
The output Y of the logic circuit as shown in figure is:
A)
\[\left( A+B \right).\overline{C}\]
done
clear
B)
\[\left( A+C \right).\overline{B}\]
done
clear
C)
\[\left( B+C \right).\bar{A}\]
done
clear
D)
\[\operatorname{A}+B+\bar{C}\]
done
clear
View Answer play_arrow
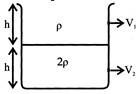
Equal volume of two immiscible liquids of densities p and 2 p are filled in a vessel as shown in figure. Two small holes are punched at depths \[\frac{h}{2}\] and\[\frac{3h}{2}\] from the surface of lighter liquid. If \[{{\operatorname{v}}_{1}}\]and\[{{\operatorname{v}}_{2}}\]are the velocities of efflux at these two holes.
A)
\[\frac{1}{2\sqrt{2}}\]
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
\[\frac{1}{\sqrt{2}}\]
done
clear
View Answer play_arrow
De-Broglie wavelength of a neutron at \[927{}^\circ C\]is \[\lambda \]. What will be its wavelength at\[27{}^\circ C\]?
A)
\[\frac{\lambda }{9}\]
done
clear
B)
\[\lambda \]
done
clear
C)
\[2\lambda \]
done
clear
D)
\[4\lambda \]
done
clear
View Answer play_arrow
A magnetic needle is kept in a non-uniform magnetic field. If experiences:
A)
A force only but not a torque
done
clear
B)
A force and torque both
done
clear
C)
A torque only but not a force
done
clear
D)
Neither a torque nor a force
done
clear
View Answer play_arrow
If \[{{\operatorname{f}}_{1}}\]is the frequency of the series limit of Lyman series, \[{{\operatorname{f}}_{\,2}}\]is the frequency of the first line of Lyman series and \[{{\operatorname{f}}_{\,3}}\] is the frequency of the series limit of the Balmer series then:
A)
\[{{\operatorname{f}}_{\,1}}-{{\operatorname{f}}_{\,2}}={{\operatorname{f}}_{\,3}}\]
done
clear
B)
\[{{\operatorname{f}}_{\,1}}={{\operatorname{f}}_{\,2}}-{{\operatorname{f}}_{\,3}}\]
done
clear
C)
\[\frac{1}{{{\operatorname{f}}_{\,2}}}=\frac{1}{{{\operatorname{f}}_{\,1}}}+\frac{1}{{{\operatorname{f}}_{\,3}}}\]
done
clear
D)
\[\frac{1}{{{\operatorname{f}}_{\,1}}}=\frac{1}{{{\operatorname{f}}_{\,2}}}+\frac{1}{{{\operatorname{f}}_{\,3}}}\]
done
clear
View Answer play_arrow
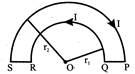
A wire loop PQRSP is constructed by joining two semi-circular coils of radius\[{{\operatorname{r}}_{\,1}}\]and\[{{\operatorname{r}}_{\,2}}\] respectively, as shown in the figure. If the current flowing in the loop is I, then the magnetic induction at the point o is:
A)
\[\frac{{{\mu }_{\operatorname{o}}}\operatorname{I}}{4}\left[ \frac{1}{{{r}_{1}}}-\frac{1}{{{r}_{2}}} \right]\]
done
clear
B)
\[\frac{{{\mu }_{\operatorname{o}}}\operatorname{I}}{4}\left[ \frac{1}{{{r}_{1}}}+\frac{1}{{{r}_{2}}} \right]\]
done
clear
C)
\[\frac{{{\mu }_{\operatorname{o}}}\operatorname{I}}{2}\left[ \frac{1}{{{r}_{1}}}-\frac{1}{{{r}_{2}}} \right]\]
done
clear
D)
\[\frac{{{\mu }_{\operatorname{o}}}\operatorname{I}}{2}\left[ \frac{1}{{{r}_{1}}}+\frac{1}{{{r}_{2}}} \right]\]
done
clear
View Answer play_arrow
A particle of mass 100g is thrown vertically upwards with a speed of 5w/s. The work done by the force of gravity during the time, the particle goes up:
A)
-0.5J
done
clear
B)
-1.25J
done
clear
C)
1.25J
done
clear
D)
0.5J
done
clear
View Answer play_arrow
The photoelectric threshold wavelength for Ag (silver) is \. The \[{{\lambda }_{\operatorname{o}}}\]energy of the electron ejected from the surface of silver by an incident wavelength \[\lambda (\lambda <{{\lambda }_{\operatorname{o}}})\] will be:
A)
\[\operatorname{hc}({{\lambda }_{\operatorname{o}}}-\lambda )\]
done
clear
B)
\[\frac{\operatorname{hc}}{{{\lambda }_{\operatorname{o}}}-\lambda }\]
done
clear
C)
\[\frac{h}{c}\left[ \frac{{{\lambda }_{\operatorname{o}}}-\lambda }{\lambda {{\lambda }_{\operatorname{o}}}} \right]\]
done
clear
D)
\[\operatorname{hc}\left[ \frac{{{\lambda }_{\operatorname{o}}}-\lambda }{\lambda {{\lambda }_{\operatorname{o}}}} \right]\]
done
clear
View Answer play_arrow
A npn transistor is connected in common emitter configuration in a given amplifier. A load resistance of \[800\Omega \]is connected in the collector circuit and the voltage drop across it is 0.8V. If the current amplification factor is 0.96 and the input resistance of the circuit is \[192\Omega \], the voltage gain and the power gain of the amplifier will respectively by:
A)
4, 3.84
done
clear
B)
3.69, 3.84
done
clear
C)
4, 4
done
clear
D)
4, 3.69
done
clear
View Answer play_arrow
The shear modulus of a liquid is:
A)
Zero
done
clear
B)
Unity
done
clear
C)
Infinity
done
clear
D)
None
done
clear
View Answer play_arrow
Imagine a new planet having the same density as that of earth but it is 3times bigger than the earth in size. If the acceleration due to gravity on the surface of earth is g and that on the surface of new plant is g1 then:
A)
g1 = 3g
done
clear
B)
g1=9g
done
clear
C)
\[\operatorname{g}1\frac{g}{9}\]
done
clear
D)
g1=27g
done
clear
View Answer play_arrow
A transformer of 100% efficiency has 200 turns in the primary and 40,000 turns in secondary. It is connected to a 220V main supply and secondary feeds to a\[100k\Omega \]resistance. The potential difference per turn is:
A)
1.1V
done
clear
B)
25V
done
clear
C)
18V
done
clear
D)
11V
done
clear
View Answer play_arrow
Two coherent sources of different intensities sound waves which interfere. The ratio of maximum intensity to the minimum intensity is 25. The intensities of the sources are in the ratio:
A)
25:1
done
clear
B)
5:l
done
clear
C)
9:4
done
clear
D)
25:16
done
clear
View Answer play_arrow
A potentiometer wire is 100 cm long and a constant potential difference is maintained across it. Two cells are connected in series? first to support one another and then in opposite direction. The balance points are obtained at 50cm and 10cm from the positive end of the wire in the two cases. The ratio of emf' s is:
A)
5:1
done
clear
B)
5:4
done
clear
C)
3:4
done
clear
D)
3:2
done
clear
View Answer play_arrow
A radio isotope \[X\]with a half-life \[1.4\times {{10}^{9}}\] yr of Y which is stable. A sample of the rock from a cave was found to contain \[X\]and \[Y\]in the ratio 1: 7. The age of the rock is:
A)
\[1.96\times {{10}^{9}}\,\operatorname{yr}\]
done
clear
B)
\[3.92\times {{10}^{9}}\,\operatorname{yr}\]
done
clear
C)
\[4.2\times {{10}^{9}}\,\operatorname{yr}\]
done
clear
D)
\[8.2\times {{10}^{9}}\,\operatorname{yr}\]
done
clear
View Answer play_arrow
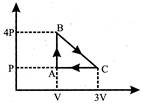
An ideal gas is taken through the cycle ABCA as shown in the P-V diagram. The total work done by the gas during the cycle is:
A)
pV
done
clear
B)
2pV
done
clear
C)
3pV
done
clear
D)
6Pv
done
clear
View Answer play_arrow
The motion of a particle along a straight line is described by equation x=8+12t-t3, where, x is in meter and t is in second. The retardation of the particle when its velocity becomes zero, is:
A)
\[24 m/{{s}^{2}}\]
done
clear
B)
Zero
done
clear
C)
\[6 m/{{s}^{2}}\]
done
clear
D)
\[12 m/{{s}^{2}}\]
done
clear
View Answer play_arrow
The change in the gravitational potential energy when a body of mass m is raised to a height nR above the surface of the earth is:
A)
\[\frac{n}{n+1}(mgR)\]
done
clear
B)
\[\frac{n}{n-1}(mgR)\]
done
clear
C)
\[nmgR\]
done
clear
D)
\[\frac{(mgR)}{n}\]
done
clear
View Answer play_arrow
A machine which is 75% efficient, uses \[12\mathrm{J}\] of energy in lifting 1Kg mass through a certain distance. The mass is then allowed to fall through the same distance. The velocity at the energy of its fall is:
A)
\[\sqrt{12}\,m/s\]
done
clear
B)
\[\sqrt{18}\,m/s\]
done
clear
C)
\[\sqrt{24}\,m/s\]
done
clear
D)
\[\sqrt{32}\,m/s\]
done
clear
View Answer play_arrow
A closed pipe and an open pipe of same length produce 2 beats, when they are set into vibration simultaneously in their fundamental mode. If the length of the open pipe is halved and that of closed pipe is doubled, and if they are vibrating in the fundamental mode, then the number of beats produced is:
A)
4
done
clear
B)
7
done
clear
C)
2
done
clear
D)
8
done
clear
View Answer play_arrow
A steady current is set up in a metallic wire of non - uniform cross section how is the rate of flow of free electrons related to the 8. A body starts from rest and moves with uniform acceleration. Which of the following graphs represent its motion:
A)
\[\operatorname{K}\propto \,{{\operatorname{A}}^{o}}\]
done
clear
B)
\[\operatorname{K}\propto \,{{\operatorname{A}}^{-1}}\]
done
clear
C)
\[\operatorname{K}\propto \,\operatorname{A}\]
done
clear
D)
\[\operatorname{K}\propto \,{{\operatorname{A}}^{2}}\]
done
clear
View Answer play_arrow
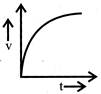
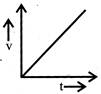
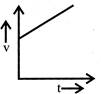
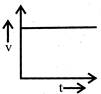
A body starts from rest and moves with uniform acceleration. Which of the following graph represent its motion:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The coefficient of performance of a refrigerator is 5. It the temperature inside freezer is \[-20{}^\circ C\], the temperature of the surroundings to which it rejects heat is:
A)
\[11{}^\circ C\]
done
clear
B)
\[21{}^\circ C\]
done
clear
C)
\[31{}^\circ C\]
done
clear
D)
\[41{}^\circ C\]
done
clear
View Answer play_arrow
W. A beam of light of wavelength 600nm from a distant source falls on a single slit 1mm wide and the resulting diffraction pattern is observed on a screen 2m away. The distance between the first dark fringes on either side of the central bright fringe is:
A)
1.2 cm
done
clear
B)
1.2 mm
done
clear
C)
2.4 cm
done
clear
D)
2.4 mm
done
clear
View Answer play_arrow
M. A convex lens of focal length f is placed somewhere in between an object and a screen. The distance between object and screen is x. if numerical value of magnification produced by lens is m, focal length of lens is:
A)
\[\frac{mx}{{{\left( m+1 \right)}^{2}}}\]
done
clear
B)
\[\frac{mx}{{{\left( m-1 \right)}^{2}}}\]
done
clear
C)
\[\frac{{{\left( m+1 \right)}^{2}}}{m}x\]
done
clear
D)
\[\frac{{{\left( m-1 \right)}^{2}}}{m}x\]
done
clear
View Answer play_arrow
A resistor R has power dissipation P with cell voltage E. The resistor is cut in n equal parts and all parts are connected in parallel with same cell. The new power dissipation is:
A)
nP
done
clear
B)
\[{{\operatorname{nP}}^{2}}\]
done
clear
C)
\[{{\operatorname{n}}^{2}}P\]
done
clear
D)
\[\operatorname{n}/P\]
done
clear
View Answer play_arrow
The angle of a prism is 'A'. One of its refracting surfaces is silvered. Light rays falling at an angle of incidence 2A on the first surface returns back through the same path after suffering reflection at the silvered surface. The refractive index u, of the prism is:
A)
2 sin A
done
clear
B)
2 cos A
done
clear
C)
\[\frac{\cos A}{2}\]
done
clear
D)
tan A
done
clear
View Answer play_arrow
The upper half of an inclined plane of inclination 0 is perfectly smooth while lower half is rough. A block starting from rest at the top of the plane will again come to rest at the bottom, if the coefficient of friction between block and lower half of the plane is given by:
A)
\[\mu =\frac{1}{\tan \,\theta }\]
done
clear
B)
\[\mu =\frac{2}{\tan \,\theta }\]
done
clear
C)
\[\mu =\,2\,\tan \,\theta \]
done
clear
D)
\[\mu =\,\,\tan \,\theta \]
done
clear
View Answer play_arrow
A capacitor of capacity\[{{\operatorname{C}}_{1}}\], is charged by connected it across a battery of emf\[{{\operatorname{V}}_{o}}\]. The battery is then removed and the capacitor is connected in parallel with an uncharged capacitor of capacites\[{{\operatorname{C}}_{2}}\]. The potential difference across this combination is
A)
\[\frac{{{C}_{2}}}{{{C}_{1}}+{{C}_{2}}}{{V}_{o}}\]
done
clear
B)
\[\frac{{{C}_{1}}}{{{C}_{1}}+{{C}_{2}}}{{V}_{o}}\]
done
clear
C)
\[\frac{{{C}_{1}}+{{C}_{2}}}{{{C}_{2}}}{{V}_{o}}\]
done
clear
D)
\[\frac{{{C}_{1}}+{{C}_{2}}}{{{C}_{1}}}{{V}_{o}}\]
done
clear
View Answer play_arrow
In an LCR circuit, the potential difference between the terminals of the inductance is 60V, between the terminals of the capacitor is 30V and that across the resistance is 40V. Then, supply voltage will be:
A)
50V
done
clear
B)
70V
done
clear
C)
130V
done
clear
D)
10V
done
clear
View Answer play_arrow
Moment of inertia of a uniform rod of length L and mass M, about an axis passing through L/4 from one end and perpendicular to its length is:
A)
\[\frac{7}{36}{{\operatorname{ML}}^{2}}\]
done
clear
B)
\[\frac{7}{48}{{\operatorname{ML}}^{2}}\]
done
clear
C)
\[\frac{11}{48}{{\operatorname{ML}}^{2}}\]
done
clear
D)
\[\frac{{{\operatorname{ML}}^{2}}}{12}\]
done
clear
View Answer play_arrow
A thermo dynamical system undergo cyclic process ABCDA as shown in figure. Work done by the system is:
A)
Zero
done
clear
B)
\[2{{P}_{0}}{{V}_{0}}\]
done
clear
C)
\[{{P}_{0}}V\]
done
clear
D)
\[\frac{3}{2}{{P}_{0}}{{V}_{0}}\]
done
clear
View Answer play_arrow
A double convex lens made of glass \[\left( \mu =1.5 \right)\] has both radii of curvature of magnitude 20 cm incident rays parallel to the axis of the lens will converge at a distance L such that:
A)
L=20 cm
done
clear
B)
L=10cm
done
clear
C)
L=40 cm
done
clear
D)
\[\operatorname{L}=\frac{20}{3}cm\]
done
clear
View Answer play_arrow
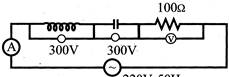
In the circuit shown below what will be the reading of the voltmeter and ammeter?
A)
200V, 1A
done
clear
B)
800V, 2A
done
clear
C)
100V, 2A
done
clear
D)
220V, 2.2A
done
clear
View Answer play_arrow
The figure shows the path of a positively charged particle 1 through a rectangular region of uniform electric field as shown in the figure. What is the direction of electric field and the direction of deflection of particles 1, 2, 3,4?
A)
Top, down, top, down
done
clear
B)
Top, down, down, top
done
clear
C)
Down, top, top, down
done
clear
D)
Down, top, down, down
done
clear
View Answer play_arrow
If a 2Kg mass is rotating on a circular path of radius 0.8m with angular velocity of 44 rad/ sec. If radius of the path becomes 1m, then what will be the value of angular velocity?
A)
28.16 rad/sec
done
clear
B)
19.28 rad/sec
done
clear
C)
8.12 rad/sec
done
clear
D)
35.26 rad/sec
done
clear
View Answer play_arrow
A particle is moving on a circular path of 10 m radius. At any instant of time, its speed is 5 m/s and the speed is increasing at a rate of 2 nVs2. The magnitude of net acceleration at this instant is:
A)
\[5 m/{{s}^{2}}\]
done
clear
B)
\[2 m/{{s}^{2}}\]
done
clear
C)
\[3.2 m/{{s}^{2}}\]
done
clear
D)
\[4.3 m/{{s}^{2}}\]
done
clear
View Answer play_arrow
A liquid wets glass. Its angle of contact with glass is:
A)
Less than\[\frac{\pi }{2}\]
done
clear
B)
\[\frac{\pi }{2}\]
done
clear
C)
\[\frac{3\pi }{2}\]
done
clear
D)
\[\pi \]
done
clear
View Answer play_arrow
Four wires of identical length, diameters and of the same material are stretched on a sonometre wire. If the ratio of their tensions is 1:4 : 9 :16, then the ratio of their fundamental frequencies are:
A)
16:9:4:1
done
clear
B)
4:3:2:1
done
clear
C)
1:4:2:16
done
clear
D)
1:2:3:4
done
clear
View Answer play_arrow
The no. of o and n bonds in allyl isocyanides are:
A)
\[9\sigma ,\,\,3\pi \]
done
clear
B)
\[9\sigma ,\,\,9\pi \]
done
clear
C)
\[3\sigma ,\,\,4\pi \]
done
clear
D)
\[5\sigma ,\,\,7\pi \]
done
clear
View Answer play_arrow
A \[0{}^\circ C\]and one atom pressure, a gas occupies 100cc it the pressure is increased to one and half-lime and temperature is increased by one - third of absolute temperature, then final volume of gas will be:
A)
80c
done
clear
B)
88.9c
done
clear
C)
66.7c
done
clear
D)
100cc
done
clear
View Answer play_arrow
The decreasing order of boiling points of following hybrids is:
(i) \[{{\operatorname{NH}}_{3}}\]
(ii) \[{{\operatorname{PH}}_{3}}\]
(iii) \[{{\operatorname{AsH}}_{3}}\]
(iv) \[{{\operatorname{SbH}}_{3}}\]
(v) \[{{\operatorname{H}}_{2}}0\]
A)
(v) > (iv) > (i) > (iii) > (ii)
done
clear
B)
(v) > (i) > (ii) > (iii) > (iv)
done
clear
C)
(ii) > (iv) > (iii) > (i) > (v)
done
clear
D)
(iv) > (iii) > (i) > (ii) > (v)
done
clear
View Answer play_arrow
An ideal gas expands in volume from \[1\times 1{{0}^{-3}}{{m}^{3}}\,\,\,to\,\,1{{0}^{-2}}{{m}^{3}}\] at 300K against a constant pressure of \[1\times 1{{0}^{5}}N{{m}^{-2}}\]. The work done is:
A)
-900J
done
clear
B)
-900KJ
done
clear
C)
270J
done
clear
D)
900KJ
done
clear
View Answer play_arrow
Which of the following pair of molecules will have permanent dipole moment for both members:
A)
\[{{\operatorname{NO}}_{2}} and C{{O}_{2}}\]
done
clear
B)
\[{{\operatorname{NO}}_{2}} and {{O}_{3}}\]
done
clear
C)
\[{{\operatorname{SiF}}_{2}} and C{{O}_{2}}\]
done
clear
D)
\[{{\operatorname{SiF}}_{4}} and N{{O}_{2}}\]
done
clear
View Answer play_arrow
The number of atoms in 4.25g of \[{{\operatorname{NH}}_{3}}\]is approximately:
A)
\[1\times {{10}^{23}}\]
done
clear
B)
\[2\times {{10}^{23}}\]
done
clear
C)
\[4\times {{10}^{23}}\]
done
clear
D)
\[6\times {{10}^{23}}\]
done
clear
View Answer play_arrow
The correct order of decreasing first ionisation energy is:
A)
C>B>Be>Li
done
clear
B)
C > Be > B > Li
done
clear
C)
B>C>Be>Li
done
clear
D)
Be > Li > B > C
done
clear
View Answer play_arrow
The total number of orbitals in a shell having principle quantum number n is:
A)
2n
done
clear
B)
\[{{\operatorname{n}}^{2}}\]
done
clear
C)
\[2{{n}^{2}}\]
done
clear
D)
n+1
done
clear
View Answer play_arrow
The maximum number of \[90{}^\circ \] angles between bond pair-bond pair of electrons is observed in:
A)
\[{{\operatorname{dsp}}^{3}}\]hybridization
done
clear
B)
\[{{\operatorname{sp}}^{3}}d\]hybridization
done
clear
C)
\[{{\operatorname{dsp}}^{2}}\]hybridization
done
clear
D)
\[{{\operatorname{sp}}^{3}}{{d}^{2}}\]hybridization
done
clear
View Answer play_arrow
The orbital diagram in which the Aufbau principle is violated is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
High concentration of fluoride is poisonous and harmful to bones and teeth at levels over:
A)
1ppm
done
clear
B)
3ppm
done
clear
C)
5ppm
done
clear
D)
10ppm
done
clear
View Answer play_arrow
The compounds of alkaline earth metals have the following magnetic nature:
A)
Diamagnetic
done
clear
B)
Paramagnetic
done
clear
C)
Ferromagnetic
done
clear
D)
Antiferromagnetic
done
clear
View Answer play_arrow
Heating an aqueous solution of aluminum chloride to dryness will give:
A)
\[{{\operatorname{AlCl}}_{3}}\]
done
clear
B)
\[{{\operatorname{Al}}_{2}}C{{l}_{6}}\]
done
clear
C)
\[{{\operatorname{Al}}_{2}}{{O}_{3}}\]
done
clear
D)
\[\operatorname{Al}(OH)C{{l}_{2}}\]
done
clear
View Answer play_arrow
The highest boiling point is expected for:
A)
Iso-octane
done
clear
B)
n - octane
done
clear
C)
2, 2, 3, 3 - tetramethylbutane
done
clear
D)
n - butane
done
clear
View Answer play_arrow
A compound x\[\left( {{C}_{5}}{{H}_{8}} \right)\] reacts with ammonical \[{{\operatorname{AgNO}}_{3}}\]to give a white precipitate, and an oxidation with hot alkaline \[{{\operatorname{KMnO}}_{4}}\]gives the acid, \[{{(C{{H}_{3}})}_{2}}\]CHCOOH. Therefore X is:
A)
\[{{\operatorname{CH}}_{2}}=CHCH=CHC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}{{\left( C{{H}_{2}} \right)}_{2}}C=CH\]
done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{2}}CH-C=C{{H}_{2}}\]
done
clear
D)
\[{{\left( C{{H}_{2}} \right)}_{2}}CH-C=CH\]
done
clear
View Answer play_arrow
For the reaction \[{{\operatorname{H}}_{2}}\left( g \right)+{{I}_{2}} \left( g \right)\rightleftharpoons 2HI\left( g \right)\], the equilibrium constant \[{{\operatorname{K}}_{P}}\]changes with:
A)
Temperature
done
clear
B)
Total pressure
done
clear
C)
Catalyst
done
clear
D)
Amount of K, and I,
done
clear
View Answer play_arrow
\[{{\operatorname{K}}_{P}}\]for reaction:
\[3Fe\left( s \right)+4{{H}_{2}}O\left( g \right).=F{{e}_{3}}{{O}_{4}}, \left( s \right)+4{{H}_{2}} \left( g \right)\]is equal:
A)
\[{{\left( P{{H}_{2}} \right)}^{4}}\left( PE{{e}_{3}}{{O}_{4}} \right)\]
done
clear
B)
\[\frac{P{{H}_{2}}}{P{{H}_{2}}O}\]
done
clear
C)
\[\frac{{{\left( P{{H}_{2}} \right)}^{4}}}{{{\left( P{{H}_{2}}O \right)}^{4}}}\]
done
clear
D)
\[\frac{\left( P{{H}_{2}} \right)\left( P\,F{{e}_{3}}\,{{O}_{4}} \right)}{P\,Fe}\]
done
clear
View Answer play_arrow
The oxidation state of chromium in Cr(CO) is:
A)
0
done
clear
B)
+2
done
clear
C)
-2
done
clear
D)
+6
done
clear
View Answer play_arrow
2 - Acetoxybenzoic acid is called:
A)
Antiseptic
done
clear
B)
Aspirin
done
clear
C)
Antibiotic
done
clear
D)
Mordant dye
done
clear
View Answer play_arrow
Which is not a polymer?
A)
Sucrose
done
clear
B)
Enzyme
done
clear
C)
Starch
done
clear
D)
Teflon
done
clear
View Answer play_arrow
Complete hydrolysis of cellulose gives:
A)
L - glucose
done
clear
B)
D - fructose
done
clear
C)
D - ribose
done
clear
D)
D - glucose
done
clear
View Answer play_arrow
Among the following, weakest base is:
A)
\[{{\operatorname{H}}_{2}}{{C}_{6}}{{H}_{5}}CN{{H}_{2}}\]
done
clear
B)
\[{{\operatorname{C}}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\]
done
clear
C)
\[{{\operatorname{O}}_{2}}N-C{{H}_{2}}N{{H}_{2}}\]
done
clear
D)
\[{{\operatorname{CH}}_{3}}NHCHO\]
done
clear
View Answer play_arrow
Strongest base among the following is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The liquefied metal expanding on solidification is:
A)
Ga
done
clear
B)
Al
done
clear
C)
Zn
done
clear
D)
Cu
done
clear
View Answer play_arrow
The crystal system of a compound with unit cell dimensions a = 0.387nm, b = 0.387nm, and c = 0.504 nm and \[\alpha =\beta =90{}^\circ and \gamma =120{}^\circ \]is:
A)
Cubic
done
clear
B)
Hexagonal
done
clear
C)
Orthorhombic
done
clear
D)
Rhombohedral
done
clear
View Answer play_arrow
When ethylamine is treated with \[C{{H}_{3}}MgBr,\]the product is:
A)
\[C{{H}_{3}}C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{4}}\]
done
clear
C)
\[C{{H}_{3}}H{{C}_{2}}C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\]
done
clear
View Answer play_arrow
A 5% solution of cane sugar (mol. wt. = 342) is isotonic with 1% solution of substance X. the molecular weight of X is:
A)
34.2
done
clear
B)
171.2
done
clear
C)
68.4
done
clear
D)
136.8
done
clear
View Answer play_arrow
In the following reaction, X is:
A)
Benzoic acid
done
clear
B)
Salicylic acid
done
clear
C)
Phenol
done
clear
D)
Aniline
done
clear
View Answer play_arrow
Camphor is often used in molecular mass determination because:
A)
It is readily available
done
clear
B)
It has very high cryoscopic constant
done
clear
C)
It is volatile
done
clear
D)
It is solvent for organic substances
done
clear
View Answer play_arrow
Standard electrode potentials are:
\[F{{e}^{2+}}/Fe\] \[E{}^\circ =-0.44V\] \[F{{e}^{3+}}/F{{e}^{2+}}\], \[E{}^\circ =+0.77V\] \[F{{e}^{2+}},F{{e}^{3+}}\]and Fe bloks are kept together, then:
A)
\[F{{e}^{3+}}increases\]
done
clear
B)
\[F{{e}^{3+}}decreases\]
done
clear
C)
\[F{{e}^{2+}}/F{{e}^{3+}}remains\text{ }unchanged\]
done
clear
D)
\[F{{e}^{2+}}\ decreases\]
done
clear
View Answer play_arrow
Product of the reaction:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The half life of a reaction is halved as the initial concentration of the reactant is doubled. The order of reaction is:
A)
0.5
done
clear
B)
1
done
clear
C)
2
done
clear
D)
O
done
clear
View Answer play_arrow
Colloidal silver is obtained by reduction of silver nitrate \[\left( AgN{{O}_{3}} \right)\] with:
A)
Acetic acid
done
clear
B)
Glucose
done
clear
C)
Molecular hydrogen
done
clear
D)
Fructose
done
clear
View Answer play_arrow
The rate constant K, for the reaction \[{{N}_{2}}{{O}_{5}}\text{ }\left( g \right)\to 2N{{O}_{2}}\left( g \right)+~{{0}_{2}}\left( g \right)\text{ }is\text{ }1.2\times l{{0}^{-2}}\text{ }{{S}^{-1}}.\]Which equation given below describes the change of \[\left[ {{N}_{2}}{{O}_{5}} \right]\] with time? \[{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{0}}\] and \[{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{t}}\] correspond to the concentration of \[{{N}_{2}}{{O}_{5}}\]cinitially and at time t:
A)
\[{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{t}}={{\left[ {{N}_{2}}{{O}_{5}} \right]}_{0}}+kt\]
done
clear
B)
\[\left[ {{N}_{2}}{{O}_{5}} \right]o={{\left[ {{N}_{2}}{{O}_{5}} \right]}_{t}}{{e}^{kt}}\]
done
clear
C)
\[log{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{t}}=log{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{o}}+kt\]
done
clear
D)
\[In\,\,\frac{{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{o}}}{{{\left[ {{N}_{2}}{{O}_{5}} \right]}_{t}}}=kt\]
done
clear
View Answer play_arrow
Smelting involve reduction of metal oxide with:
A)
Carbon
done
clear
B)
Carbon monoxide
done
clear
C)
Magnesium
done
clear
D)
Aluminium
done
clear
View Answer play_arrow
The compound which has molecular nature in gas phase but ionic in solid state is:
A)
\[~PC{{l}_{5}}\]
done
clear
B)
\[CC{{I}_{4}}\]
done
clear
C)
\[PC{{l}_{3}}\]
done
clear
D)
\[POC{{l}_{3}}\]
done
clear
View Answer play_arrow
Which of the following does not have optical isomer?
A)
\[\left[ Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}} \right]\]
done
clear
B)
\[\left[ Co{{\left( en \right)}_{3}} \right]C{{l}_{3}}\]
done
clear
C)
\[\left[ Co{{\left( en \right)}_{2}}C{{l}_{2}} \right]Cl\]
done
clear
D)
\[\left[ Co\left( en \right){{\left( N{{H}_{3}} \right)}_{2}}C{{l}_{2}} \right]Cl\]
done
clear
View Answer play_arrow
In which of following pairs, the two species are isostructural?
A)
\[BrO\overline{_{3}}\text{ }and\text{ }Xe{{O}_{3}}\]
done
clear
B)
\[S{{F}_{4}}\text{ }and\text{ }Xe{{F}_{4}}\]
done
clear
C)
\[S{{O}^{2-}}_{\text{3}}and\text{ }NO\overline{_{3}}\]
done
clear
D)
\[B{{F}_{3}}\text{ }and\text{ }N{{F}_{3}}\]
done
clear
View Answer play_arrow
The correct structure of Fe(CO),; is:
A)
Octahedral
done
clear
B)
Tetrahedral
done
clear
C)
Square pyramidal
done
clear
D)
Trigonal bipyramidal
done
clear
View Answer play_arrow
1-Chlorobutane on reaction with alcoholic potash gives:
A)
1 - Butene
done
clear
B)
1 - Butanol
done
clear
C)
2-Butene
done
clear
D)
2-Butanol
done
clear
View Answer play_arrow
The compound used for gravimetric estimation of Cu(II) is:
A)
\[C{{u}_{2}}{{\left( SCN \right)}_{2}}\]
done
clear
B)
Cup
done
clear
C)
\[C{{u}_{2}}{{I}_{2}}\]
done
clear
D)
\[C{{u}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
The dipole moment is the highest for:
A)
Trans-2-butene
done
clear
B)
2-dimethyl benzene
done
clear
C)
Acetophenone
done
clear
D)
Ethanol
done
clear
View Answer play_arrow
The compound X is:
A)
\[CC{{I}_{4}}\]
done
clear
B)
\[C{{o}_{2}}\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
Co
done
clear
View Answer play_arrow
Which is correct?
A)
Reduction of any aldehyde gives secondary alchol
done
clear
B)
Reaction of vegetable oil with \[{{H}_{2}}S{{O}_{4}}\]gives glycerine
done
clear
C)
Alcholic iodine with NaOH gives iodoform
done
clear
D)
Surcose on reaction with NaCI gives invert sugar
done
clear
View Answer play_arrow
One mole of an ideal gas at 300K is expanded isothermally from an initial velume of Il to 10l. The AE for the process \[\left( R=2\text{ }cal\text{ }mo{{l}^{-1}}\text{ }{{K}^{-1}} \right)\]
A)
163.7 cal
done
clear
B)
Zero
done
clear
C)
1381.1 cal
done
clear
D)
9 lit atm
done
clear
View Answer play_arrow
The term species was given by:
A)
Aristotle
done
clear
B)
Darwin
done
clear
C)
Hugo de vries
done
clear
D)
John ray
done
clear
View Answer play_arrow
R.H. Whittaker proposed a five kingdom classification in:
A)
1968
done
clear
B)
1979
done
clear
C)
1969
done
clear
D)
1959
done
clear
View Answer play_arrow
Biogas is produced by:
A)
Eubacteria
done
clear
B)
Archaebacteria
done
clear
C)
Mycoplasma
done
clear
D)
None of these
done
clear
View Answer play_arrow
Common phylum of unicellular animals and plants is:
A)
Protista
done
clear
B)
Plantae
done
clear
C)
Fungi
done
clear
D)
Monera
done
clear
View Answer play_arrow
Member of rhodophyceae are red in colour due to pigment:
A)
Fucoxanthin
done
clear
B)
r-phycoerythrin
done
clear
C)
Chlorophyll
done
clear
D)
Anthocyanin
done
clear
View Answer play_arrow
Sporangia bearing leaves in pteridophytes are called:
A)
Indusium
done
clear
B)
Sort
done
clear
C)
Ramenta
done
clear
D)
Sporophylls
done
clear
View Answer play_arrow
Haplontic life cycle involved in:
A)
Volvox
done
clear
B)
Mustard
done
clear
C)
Bryophytes
done
clear
D)
Eucalyptus
done
clear
View Answer play_arrow
Radial symmetry occurs in:
A)
Molluscs
done
clear
B)
Tapeworm
done
clear
C)
Starfish
done
clear
D)
Annelida
done
clear
View Answer play_arrow
Drones are formed in Honey-Bee by:
A)
Fasting Larvae
done
clear
B)
Fertilized eggs
done
clear
C)
Unfertilised egg
done
clear
D)
None of these
done
clear
View Answer play_arrow
Actinomorphic flowers are found in:
A)
Pea
done
clear
B)
Mustard
done
clear
C)
Bean
done
clear
D)
Cassia
done
clear
View Answer play_arrow
Water and turgid are absorbed in grasses by:
A)
Mesophyll
done
clear
B)
Bundle sheath cells
done
clear
C)
Bulliform cells
done
clear
D)
Epidermis
done
clear
View Answer play_arrow
Nucleus is absent in:
A)
WBCs
done
clear
B)
RBCs
done
clear
C)
Macrophage
done
clear
D)
Monocytes
done
clear
View Answer play_arrow
In animal cells lipid like steroidal hormones are synthesised in:
A)
RER
done
clear
B)
SER
done
clear
C)
Golgi apparatus
done
clear
D)
Cell-wall
done
clear
View Answer play_arrow
Floral formula of tomato is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which one of the following structure formulae of two organic compounds is correctly identified along with its related function:
A)
Triglyceride: Major source of Energy
done
clear
B)
Adenine: Nitrogen base of DNA
done
clear
C)
Glycerol: Components of alcohol
done
clear
D)
Uracil: Components of DNA
done
clear
View Answer play_arrow
Alternate phyllotaxy observed in:
A)
Mustard
done
clear
B)
Calotropis
done
clear
C)
Alstonia
done
clear
D)
Guava plants
done
clear
View Answer play_arrow
Centromeres split and chromatids separate in:
A)
Metaphase
done
clear
B)
Anaphase
done
clear
C)
Telophase
done
clear
D)
Prophase
done
clear
View Answer play_arrow
Opening and closing of the stomata caused by:
A)
Bulliform cells
done
clear
B)
Epidermis
done
clear
C)
Guard cell
done
clear
D)
Mesophyll
done
clear
View Answer play_arrow
Which force does a plant use to move water molecule into the leaf parenchyma cell:
A)
Root pressure
done
clear
B)
Diffusion
done
clear
C)
Transpiration pull
done
clear
D)
Osmosis
done
clear
View Answer play_arrow
How many carbon atom present in PGA:
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
2
done
clear
View Answer play_arrow
Vegetative propagules in water hyancinth are:
A)
Bulbil
done
clear
B)
Eye buds
done
clear
C)
Root
done
clear
D)
Offset
done
clear
View Answer play_arrow
Ovule: embryo sac: Anther:
A)
Androceium
done
clear
B)
Pollen grain
done
clear
C)
Stamen
done
clear
D)
Pistil
done
clear
View Answer play_arrow
Seminal plasma rich in:
A)
Glucose
done
clear
B)
Sucrose
done
clear
C)
Fructose
done
clear
D)
Galactose
done
clear
View Answer play_arrow
Closest part of the ovary is:
A)
Ampulla
done
clear
B)
Isthmus
done
clear
C)
Fimbriae
done
clear
D)
Fundus
done
clear
View Answer play_arrow
The function of leg haemoglobin in the root nodules of legume is:
A)
Oxygen removal
done
clear
B)
Oxidise ammonia to nitrate
done
clear
C)
Convert nitrogen to nitrogen compound
done
clear
D)
Convert proteins into ammonia
done
clear
View Answer play_arrow
What is leg-Haemoglobin:
A)
Inhibition of nitrogenase activity
done
clear
B)
Oxygen scavenger
done
clear
C)
Nodule differentiation
done
clear
D)
Expression of nit gene
done
clear
View Answer play_arrow
The ovary is half - inferior in flowers of:
A)
Rose
done
clear
B)
Guava
done
clear
C)
Cucumber
done
clear
D)
Mustard
done
clear
View Answer play_arrow
Which organism contain 8 chromosomes in meiocyte (2n):
A)
Dog
done
clear
B)
Rice
done
clear
C)
Fruit fly
done
clear
D)
Cat
done
clear
View Answer play_arrow
Pollen grains are well reserved as fossils because of:
A)
Exine
done
clear
B)
Intine
done
clear
C)
Sporopollenin
done
clear
D)
Chitin
done
clear
View Answer play_arrow
Each trophic level has a certain mass of living material at a particular time called as:
A)
Standing crop
done
clear
B)
Detritus
done
clear
C)
Decomposers
done
clear
D)
Saprotrophs
done
clear
View Answer play_arrow
Name the mixture which is mixed with bitumen to lay roads:
A)
Plastic
done
clear
B)
Polyblend
done
clear
C)
Nylon 6,6
done
clear
D)
Charcoal
done
clear
View Answer play_arrow
The sewage water is treated till the B, O.D is:
A)
Increase
done
clear
B)
Reduce
done
clear
C)
Remain same
done
clear
D)
Oxidised
done
clear
View Answer play_arrow
The prime contaminants of cultural is:
A)
Nitrate and phosphate
done
clear
B)
Mercury
done
clear
C)
Lead
done
clear
D)
Nitrate and Sulphate
done
clear
View Answer play_arrow
Which forest has greatest diversity on earth:
A)
Temperate forest
done
clear
B)
Tropical forest
done
clear
C)
Coniferous forest
done
clear
D)
Arctic and Alpine tundra
done
clear
View Answer play_arrow
Which one of the following expanded forms of the following acronyms is correct?
A)
IUCN \[\to \] Internation Union Code for Bo- tanical Nomenclature
done
clear
B)
AIDS \[\to \] Acquired Immuno Deficiency Viroids
done
clear
C)
ELISA \[\to \] Enzyme Linked Immuno Sorbent Assay
done
clear
D)
UNEP \[\to \] United Nations Environment Policy
done
clear
View Answer play_arrow
Which one of the following is a condition of primary succession:
A)
It begins on a bare rock
done
clear
B)
It occurs on a deforested site
done
clear
C)
It follows secondary succession
done
clear
D)
It is relatively faster process than second- ary succession
done
clear
View Answer play_arrow
Which crop is resistance to leaf and stripe rust hill bunt:
A)
Brassica
done
clear
B)
Cauliflower
done
clear
C)
Wheat
done
clear
D)
Cow pea
done
clear
View Answer play_arrow
Which one of the following is not a biofertilizer:
A)
Rhizobium
done
clear
B)
Nostoc
done
clear
C)
Agrobacterium
done
clear
D)
Mycorrhiza
done
clear
View Answer play_arrow
Agaros gel used in:
A)
PCR
done
clear
B)
Tissue culture
done
clear
C)
Gelelectrophoresis
done
clear
D)
Northern blotting
done
clear
View Answer play_arrow
The first transgenic cow name is:
A)
Dolly
done
clear
B)
Rosie
done
clear
C)
Roumy
done
clear
D)
None of these
done
clear
View Answer play_arrow
Bt toxin genes were isolated from:
A)
Agrobacterium
done
clear
B)
Bacillus thuringiensis
done
clear
C)
Nematode
done
clear
D)
Fungi
done
clear
View Answer play_arrow
Sonalika & Kalyan sona are hybrid variety of:
A)
Rice
done
clear
B)
Wheat
done
clear
C)
Maize
done
clear
D)
Sugarcane
done
clear
View Answer play_arrow
Which one of the following organism is not an example of eukaryotic cells:
A)
Euglena
done
clear
B)
E-coli
done
clear
C)
Amoeba
done
clear
D)
Parmecium
done
clear
View Answer play_arrow
Which one of the following animals is correctly matched with it particular named taxonomic category:
A)
Human \[\to \] sapiens, the species
done
clear
B)
Cuttlefish \[\to \] Mollusca, a class
done
clear
C)
Lion \[\to \] tigris, the species
done
clear
D)
Housefly \[\to \] Domestica, the order
done
clear
View Answer play_arrow
Which cells occur in fallopian tubes and bronchioles?
A)
Ciliated columnar cell
done
clear
B)
Simple squamous cell
done
clear
C)
Glandular cell
done
clear
D)
Blood cell
done
clear
View Answer play_arrow
Most significant trend in evolution of modern man:
A)
Upright posture
done
clear
B)
Brain capacity
done
clear
C)
Shortening of jaws
done
clear
D)
Binocular vision
done
clear
View Answer play_arrow
Maximum number of genes are found in:
A)
Y- Chromosome
done
clear
B)
Chromosome - 1
done
clear
C)
X - Chromosome
done
clear
D)
Chromosome - 2
done
clear
View Answer play_arrow
Human blood group is an example of:
A)
Incomplete dominance
done
clear
B)
Codominance
done
clear
C)
Independent assortement
done
clear
D)
Dominance
done
clear
View Answer play_arrow
Basic amino acid found in histones is:
A)
Lysines
done
clear
B)
Valine
done
clear
C)
Glutamic acid
done
clear
D)
Histidine
done
clear
View Answer play_arrow
Purkinje fibre present in:
A)
Brain
done
clear
B)
Spleen
done
clear
C)
Heart
done
clear
D)
Liver
done
clear
View Answer play_arrow
Organ of corti present in cochlea is a charactristics of:
A)
Eye
done
clear
B)
Ear
done
clear
C)
Nose
done
clear
D)
Liver
done
clear
View Answer play_arrow
YelIowish pigmented spot called macula lutea with a central pit called:
A)
Cornea
done
clear
B)
Pupil
done
clear
C)
Fovea
done
clear
D)
Blind spot
done
clear
View Answer play_arrow
____ is a fibrous, non-globular protein involved in the clotting of blood:
A)
Thrombin
done
clear
B)
Albumin
done
clear
C)
Globulin
done
clear
D)
Fibrin
done
clear
View Answer play_arrow
Uricotelic mode of passing out nitrogenous wastes found in:
A)
Birds and annelids
done
clear
B)
Reptiles and birds
done
clear
C)
Amphibians
done
clear
D)
Insects and amphibians
done
clear
View Answer play_arrow
Most widely accepted method for contraception in India:
A)
Tubectomy
done
clear
B)
Cervical caps
done
clear
C)
IDUs
done
clear
D)
Diaphragms
done
clear
View Answer play_arrow
Medical termination of pregnancy is considered safe upto how many weeks:
A)
Six weeks
done
clear
B)
Ten weeks
done
clear
C)
Twelve weeks
done
clear
D)
Eighteen weeks
done
clear
View Answer play_arrow
Which type of human population is represented by the followingpyramis:
A)
Expanding population
done
clear
B)
Declining population
done
clear
C)
Stable population
done
clear
D)
Expansive population
done
clear
View Answer play_arrow
Cyclosporin A, that is used as an immune suppressive agent in organ transplant patient, is produced by:
A)
Fungus
done
clear
B)
Bacteria
done
clear
C)
Protozoa
done
clear
D)
Yeast
done
clear
View Answer play_arrow
Which one of the following pair of gases are the major cause of 'Greenhouse effect':
A)
\[C{{O}_{2}}\And {{\text{O}}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\And {{\text{O}}_{4}}\]
done
clear
C)
\[CFCs\And \,\,S{{\text{O}}_{2}}\]
done
clear
D)
\[C{{O}_{2}}\,\,\And CO\]
done
clear
View Answer play_arrow
A certain patient is suspected to be suffering from intestinal perforation. Which diagnostic technique will you recommend for its detection:
A)
ELISA
done
clear
B)
Ultrasound
done
clear
C)
WIDAL
done
clear
D)
MRI
done
clear
View Answer play_arrow
When two unrelated individuals or linea are crossed the performance of\[{{F}_{1}}\]hybrid is often superior to both its parents this is called:
A)
Spheing
done
clear
B)
Transformation
done
clear
C)
Heterosis
done
clear
D)
Inbreeding
done
clear
View Answer play_arrow
The yellowish fluid colostrum contain abundant antibodies i.e.:
A)
IgM
done
clear
B)
IgE
done
clear
C)
IgA
done
clear
D)
IgD
done
clear
View Answer play_arrow
EcoRl comes from
A)
Escherichia coli RY12
done
clear
B)
Escherichia coli RY13
done
clear
C)
Escherichia coli RY14
done
clear
D)
Escherichia coli RY2
done
clear
View Answer play_arrow
An organism used as biofertilizer for raising soyabean crop is:
A)
Nostoc
done
clear
B)
Rhizobium
done
clear
C)
Azotobacter
done
clear
D)
Azospirillum
done
clear
View Answer play_arrow
Symptoms of AIDS can be seen when:
A)
No. of \[{{T}_{H}}\]cell increases
done
clear
B)
No. of \[{{T}_{C}}\] cell decreases
done
clear
C)
No. of \[{{T}_{H}}\] cell decreases
done
clear
D)
No. of \[{{T}_{C}}\] cell Increases
done
clear
View Answer play_arrow
The government of India introduced the concept of Joint Forest Management (JFM) in which year:
A)
1970
done
clear
B)
1980
done
clear
C)
1960
done
clear
D)
1985
done
clear
View Answer play_arrow
Parbhani kranti is a new variety of:
A)
Rice
done
clear
B)
Bhindi
done
clear
C)
Wheat
done
clear
D)
Maize
done
clear
View Answer play_arrow
Drugs that are normally used as medicine to help patients cope with mental illness are:
A)
Lysergic acid diethylamide
done
clear
B)
Morphine
done
clear
C)
Cocaine
done
clear
D)
Cannabinoids
done
clear
View Answer play_arrow
Which peptide is not present in mature insulin:
A)
A -peptide
done
clear
B)
B -peptide
done
clear
C)
C -peptide
done
clear
D)
Both a & b
done
clear
View Answer play_arrow
Light saturation occurs at:
A)
10% of the full sunlight
done
clear
B)
5 % of the full sunlight
done
clear
C)
8% of the full sunlight
done
clear
D)
15% of the full sunlight
done
clear
View Answer play_arrow
Chief pigment associated with photosynthesis is:
A)
Xanthophylls
done
clear
B)
Carotenoids
done
clear
C)
Chlorophylla
done
clear
D)
Chlorophyll
done
clear
View Answer play_arrow
Bombay phenotype is a characteristics of:
A)
Blood group
done
clear
B)
Dominance
done
clear
C)
Incomplete dominance
done
clear
D)
Huntington disorder
done
clear
View Answer play_arrow
Growth activities of plants is inhibited by which plant growth regulators:
A)
Auxin
done
clear
B)
Gibberellins
done
clear
C)
Cytokiniri
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Ringworms is caused by:
A)
Ascaris
done
clear
B)
W, malayi
done
clear
C)
Epidermophyton
done
clear
D)
Entamoeba histolytica
done
clear
View Answer play_arrow
Red rot of sugarcane caused by:
A)
Bacteria
done
clear
B)
Viruses
done
clear
C)
Fungi
done
clear
D)
Worms
done
clear
View Answer play_arrow
Who discovered the vaccine for Small Pox:
A)
Louis pasteur
done
clear
B)
Roentgen
done
clear
C)
Alexander
done
clear
D)
Edward
done
clear
View Answer play_arrow
The first clinical gene therapy was given in:
A)
1980
done
clear
B)
1970
done
clear
C)
1960
done
clear
D)
1990
done
clear
View Answer play_arrow
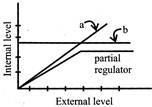
Find a & b in the following figure:
A)
b - conformer, a - regulator
done
clear
B)
a - conformer, b - regulator
done
clear
C)
a - conformer, b - suspensor
done
clear
D)
b - conformer, a - suspensor
done
clear
View Answer play_arrow
Which type of interaction found in Cusucta and hedge plants:
A)
Mutualism
done
clear
B)
Commensalism
done
clear
C)
Amensalism
done
clear
D)
Parasitism
done
clear
View Answer play_arrow
Excessive growth of planktonic algae is called:
A)
Eutrophication
done
clear
B)
Accelerated Eutrophication
done
clear
C)
Algal bloom
done
clear
D)
Biomagnification
done
clear
View Answer play_arrow
Relative contribution of various greenhouse gases to total global warming:
A)
\[a~\to C{{O}_{2}},\text{ }b\to \text{ }Methane,\text{ }c\to CFCs,\text{ }d\to \]\[{{N}_{2}}O\]
done
clear
B)
\[a\to C{{H}_{4}},b\to C{{O}_{2}},c\to {{N}_{2}}0,\,\,d\to CFCs\]
done
clear
C)
\[a\to CFCs,\text{ }b\to C{{O}_{2}},\text{ }c\to C{{O}_{2}},\text{ }d\to {{N}_{2}}0\]
done
clear
D)
\[a\to {{N}_{2}}0,\text{ }b\to CFCs,\text{ }c\to C{{H}_{2}},\text{ }d\to C{{O}_{2}}\]
done
clear
View Answer play_arrow
Blood - cholesterol lowering agents are:
A)
Trichoderma polysporum
done
clear
B)
SaccHaromyces cerevisiae
done
clear
C)
Penicillium notatum
done
clear
D)
Monascus purpureus
done
clear
View Answer play_arrow
The first acceptor of electrons from an excited chlorophyll molecule of photosystem II is:
A)
Quinone
done
clear
B)
Cytochrome
done
clear
C)
Ferredpxin
done
clear
D)
Iron-sulphur protein
done
clear
View Answer play_arrow
Our lungs remove large amount of CO which is:
A)
20 Litres/day
done
clear
B)
24 Litres/day
done
clear
C)
15 Litres/day
done
clear
D)
18 Litres/day
done
clear
View Answer play_arrow
Muscle contraction is initiated by a signal sent by:
A)
[A] N.S via sensory neuron
done
clear
B)
[C] N.S vis sensory neuron
done
clear
C)
[A] N.S via motor neuron
done
clear
D)
[C] N.S via motro neuron
done
clear
View Answer play_arrow
Vasopressin acts mainly at the:
A)
Heart
done
clear
B)
Liver
done
clear
C)
Lungs
done
clear
D)
Kidney
done
clear
View Answer play_arrow
Among the human ancestors the brain size was more than 1000 cc in:
A)
Ramapithecus
done
clear
B)
Homo habilis
done
clear
C)
Homo erectus
done
clear
D)
Homo neanderthalensis
done
clear
View Answer play_arrow
Which one of the following elements is not an essential micronutrient for plant growth:
A)
Ca
done
clear
B)
Zn
done
clear
C)
Cl
done
clear
D)
Ni
done
clear
View Answer play_arrow
What is common to whale seal and shark:
A)
Homeothermic
done
clear
B)
Seasonal migration
done
clear
C)
Convergent evolution
done
clear
D)
Thick subcutaneous fat
done
clear
View Answer play_arrow
Probiotics are:
A)
Safe antibiotics
done
clear
B)
Live microbial food supplement
done
clear
C)
Cancer inducing microbes
done
clear
D)
New kind of food allergens
done
clear
View Answer play_arrow

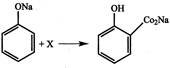 The compound X is:
The compound X is: