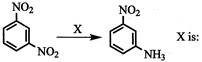
If the dimensions of a physical quantity are given by\[{{\operatorname{M}}^{a}}{{T}^{b}}{{T}^{c}}\], then the physical quantity will be:
A)
Force if a = 0, b = -1, c = -2
done
clear
B)
Pressure if a = 1, b = -1, c = -2
done
clear
C)
Velocity if a = 1, b = 0, c = -1
done
clear
D)
Acceleration if a = 1, b = 1, c = -2
done
clear
View Answer play_arrow
A block rests on a horizontal table which is executing an SHM in the horizontal plane with an amplitude a. If the coefficient of friction is |li, the block just starts to slip when the frequency of oscillation is:
A)
\[\sqrt{\frac{\mu g}{a}}\]
done
clear
B)
\[\frac{1}{2\pi }\sqrt{\frac{a}{\mu \operatorname{g}}}\]
done
clear
C)
\[2\pi \sqrt{\frac{\mu g}{a}}\]
done
clear
D)
\[\frac{1}{2\pi }\sqrt{\frac{\mu \operatorname{g}}{\operatorname{a}}}\]
done
clear
View Answer play_arrow
If h and h be the greatest heights in the two path of a projectile for a given range, then the range is given by:
A)
\[\sqrt{{{\operatorname{h}}_{1}}{{h}_{2}}}\]
done
clear
B)
\[\frac{{{h}_{1}}+{{h}_{2}}}{2}\]
done
clear
C)
\[4\sqrt{{{\operatorname{h}}_{1}}{{h}_{2}}}\]
done
clear
D)
\[\frac{2\sqrt{{{h}_{1}}{{h}_{2}}}}{{{h}_{1}}+{{h}_{2}}}\]
done
clear
View Answer play_arrow
Young's modulus of a wire of length L and radius r is Y(N/m2). If the length is reduced to\[\frac{L}{2}\,\,and \frac{r}{2}\]radius, its young modulus will be:
A)
X
done
clear
B)
Y
done
clear
C)
2Y
done
clear
D)
4Y
done
clear
View Answer play_arrow
A point moves along an arc of a circle of radius R. Its velocity depends upon the distance covered V as v=aVs, where a is constant. The angle e between the vector of total acceleration and tangential acceleration is:
A)
\[\tan \,\theta =\sqrt{\frac{s}{R}}\]
done
clear
B)
\[\tan \,\theta =\sqrt{\frac{s}{2R}}\]
done
clear
C)
\[\tan \,\theta =\frac{s}{2R}\]
done
clear
D)
\[\tan \,\theta =\frac{2s}{2R}\]
done
clear
View Answer play_arrow
If \[\vec{A} = \vec{i} A cos\theta +\vec{j} A sin\theta ,\]then another vector \[\vec{B}\]which is orthogonal to \[\vec{A}\]can be expressed as:
A)
\[\vec{i}\,B cos\theta -\vec{j}\,B\,sin\theta \]
done
clear
B)
\[\vec{i}\,B sin\theta +\vec{j}\,B cos\theta \]
done
clear
C)
\[\vec{i}\,B cos\theta +6\vec{j}\,B\,sin\theta \]
done
clear
D)
\[\vec{i}\,B sin\theta +\vec{j}\,B cos\theta \]
done
clear
View Answer play_arrow
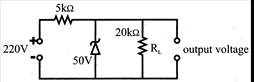
From the zener diode circuit shown in figure, the current through the zener is:
A)
34mA
done
clear
B)
31.5mA
done
clear
C)
36.5mA
done
clear
D)
2.5mA
done
clear
View Answer play_arrow
Water flows steadily through a horizontal pipe of variable cross section. If the pressure of water is P at a point where flow speed is v, the pressure at another point where the flow speed is 2v, is:
A)
\[P-\frac{3\rho {{v}^{2}}}{2}\]
done
clear
B)
\[P-\frac{\rho {{v}^{2}}}{2}\]
done
clear
C)
\[P-\frac{3\rho {{v}^{2}}}{4}\]
done
clear
D)
\[P-\rho {{v}^{2}}\]
done
clear
View Answer play_arrow
The de-droglie wavelength of an electron in 4th orbit is:
A)
\[2\pi r\]
done
clear
B)
\[4\pi r\]
done
clear
C)
\[8\pi r\]
done
clear
D)
\[16\pi r\]
done
clear
View Answer play_arrow
When photons of energy hu fall on an aluminium plate photoelectrons of maximum kinetic energy K are ejected If the frequency of the radiation is doubled, the maximum kinetic energy of the ejected photoelectrons will be:
A)
\[\operatorname{K} + {{E}_{o}}\]
done
clear
B)
2K
done
clear
C)
K
done
clear
D)
\[\operatorname{K}+hv\]
done
clear
View Answer play_arrow
The work done in turning a magnet of magnetic moment M by an angle of \[90{}^\circ \] from the meridian is n times the corresponding work done to turn it through an angle of\[60{}^\circ \].
A)
\[\operatorname{n}=\frac{1}{2}\]
done
clear
B)
n=2
done
clear
C)
\[\operatorname{n}=\frac{1}{4}\]
done
clear
D)
n = 1
done
clear
View Answer play_arrow
The transition from the state n = 3 to n = 1 in a hydrogen like atom results in ultraviolet radiation. Infrared radiation will be obtained in the transition.
A)
\[2\to 1\]
done
clear
B)
\[3\to 2\]
done
clear
C)
\[4\to 2\]
done
clear
D)
\[4\to 3\]
done
clear
View Answer play_arrow
Two long parallel wires P and Q are both perpendicular to the plane of the paper with distance 5m between them. If P and Q carry current of 2.5A and 5A respectively in the same direction, then the magnetic field at point halfway between the wires is:
A)
\[\frac{\sqrt{3}{{\mu }_{\operatorname{o}}}}{2\pi }\]
done
clear
B)
\[\frac{{{\mu }_{\operatorname{o}}}}{\pi }\]
done
clear
C)
\[\frac{3{{\mu }_{\operatorname{o}}}}{2\pi }\]
done
clear
D)
\[\frac{{{\mu }_{\operatorname{o}}}}{2\pi }\]
done
clear
View Answer play_arrow
In old age arteries carrying blood in the human body become narrow resulting in an increase in the blood pressure. This follows from:
A)
Pascal's law
done
clear
B)
Stoke' slaw
done
clear
C)
Bernoulli's principle
done
clear
D)
Archimedes principle
done
clear
View Answer play_arrow
A uniform force of \[\left( 3\hat{i}+\hat{j} \right)N\]acts on a particle of mass 2kg. Hence, the particle is displaced from position \[\left( 2\hat{i}+\hat{k} \right)m\]to position\[\left( 4\hat{i}+3\hat{j}-\hat{k} \right) m\]. The work done by the force on the particle is:
A)
9J
done
clear
B)
6J
done
clear
C)
13J
done
clear
D)
l5J
done
clear
View Answer play_arrow
A coil of resistance \[400\Omega \] is placed in a magnetic field If the magnetic \[\operatorname{flu}x\text{ }\phi \left( Wb \right)\] linked with the coil varies with time t(s) as \[\phi \]\[=50{{t}^{2}}+4\], then current at 2s is:
A)
0.5A
done
clear
B)
0.1A
done
clear
C)
2A
done
clear
D)
1A
done
clear
View Answer play_arrow
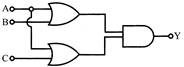
The output of given logic circuit is:
A)
A. (B + C)
done
clear
B)
A. (B .C)
done
clear
C)
(A + B).(A + C)
done
clear
D)
A + B + C
done
clear
View Answer play_arrow
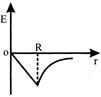
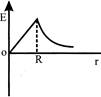
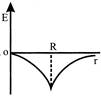
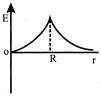
Dependence of intensity of gravitational field (E) of earth with distance (r) from centre of earth is correctly represented by:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Three sound waves of equal amplitudes have frequencies (v - 1), v, (v + 1). They superimpose to produce beats. The number of beats produced per second will be:
A)
\[\nu \]
done
clear
B)
\[\frac{\nu }{2}\]
done
clear
C)
2v
done
clear
D)
1
done
clear
View Answer play_arrow
A potentiometer having the potential gradient of 2mV/cm is used to measure the difference of potential across a resistance of\[10\phi \]. If a length of 50cm of the potentiometer wire is required to get the null points, the current passing through \[10\phi \] resistor is: (in mA)
A)
1
done
clear
B)
2
done
clear
C)
5
done
clear
D)
10
done
clear
View Answer play_arrow
Half lives of two radioactive substances A and B are respectively 20(min) and 40(min). Initially the samples of A and B have equal number of nuclei. After 80min, the ratio of remaining number of A and B nuclei is:
A)
1:16
done
clear
B)
4:1
done
clear
C)
1: 4
done
clear
D)
1:1
done
clear
View Answer play_arrow
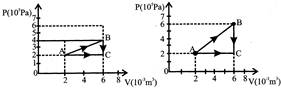
A gas is taken through the cycle \[\operatorname{A}\to B\to C\]\[\to A\]as shown. What is the net work done by the gas:
A)
2000J
done
clear
B)
1000J
done
clear
C)
Zero
done
clear
D)
1-2000J
done
clear
View Answer play_arrow
A particle is moving such that its position coordinates (x, y) are (2m, 3m) at time t = 0, (6m, 7m) at t = 2sec and (13m, 14m) at time t = 5 seAverage velocity vector V from t = 0 to t = 5 sec is:
A)
\[\frac{13\hat{i}+14\hat{j}}{4}\]
done
clear
B)
\[\frac{7}{3}\left( \hat{i}+\hat{j} \right)\]
done
clear
C)
\[2\left( \hat{i}+\hat{j} \right)\]
done
clear
D)
\[\frac{11}{5}\left( \hat{i}+\hat{j} \right)\]
done
clear
View Answer play_arrow
If the angle of incidence is twice the angle of refraction in a medium of refractive index u, then angle of incidence:
A)
\[2\,Co{{s}^{-1}}\frac{\mu }{2}\]
done
clear
B)
\[2\,Si{{n}^{-1}}\frac{\mu }{2}\]
done
clear
C)
\[2\,Co{{s}^{-1}}\mu \]
done
clear
D)
\[2\,Si{{n}^{-1}}\mu \]
done
clear
View Answer play_arrow
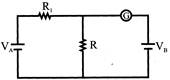
In the circuit shown below, the cells A and B have negligible resistance. For\[{{\operatorname{V}}_{A}}=12V\], \[{{\operatorname{R}}_{1}} = 500\Omega and r = 100\Omega ,\]the galvanometer (G) shows no deflection. The value of V,, is:
A)
4V
done
clear
B)
2V
done
clear
C)
12V
done
clear
D)
6V
done
clear
View Answer play_arrow
Infinite number of bodies, each of mass 2kg are situated on X-axis at distance 1m, 2m, 4m, 8m, respectively from the origin. The resulting gravitational potential due to this system at the origin will be:
A)
-G
done
clear
B)
\[-\frac{8G}{3}\]
done
clear
C)
\[-\frac{4G}{3}\]
done
clear
D)
-4G
done
clear
View Answer play_arrow
A block C of mass m is moving with velocity \[{{\operatorname{v}}_{o}}\]and collides elastically with block A of mass m and connected to another block B of mass 2m through spring with spring constant K. What is the value of K, if \[{{\operatorname{x}}_{o}}\] is compression of spring when, velocity of A and B is same?
A)
\[\frac{m{{v}^{2}}_{o}}{{{x}^{2}}_{o}}\]
done
clear
B)
\[\frac{m{{v}^{2}}_{o}}{2{{x}^{2}}_{o}}\]
done
clear
C)
\[\frac{3m{{v}^{2}}_{o}}{2{{x}^{2}}_{o}}\]
done
clear
D)
\[\frac{2m{{v}^{2}}_{o}}{3{{x}^{2}}_{o}}\]
done
clear
View Answer play_arrow
A cylindrical tube closed at one end contain air. It produces the fundamental note of frequency 512Hz. If the tube is opened at both ends, then the fundamental frequency that can be excited is:
A)
256 Hz
done
clear
B)
512 Hz
done
clear
C)
1024 Hz
done
clear
D)
128 Hz
done
clear
View Answer play_arrow
Charge passes through a conductor of cross section area \[\operatorname{A} = 0.3 {{m}^{2}}\]is given by \[\operatorname{q} = 3{{t}^{2}} + 5t\]\[+2\]in coulomb, where t is in second. What is the value of drift velocity at \[\operatorname{t}= 2 sec \]\[\left( given, n=2\times 1025/{{m}^{2}} \right)\]
A)
\[0.77\times 1{{0}^{-5}}m/s\]
done
clear
B)
\[1.77\times 1{{0}^{-5}}m/s\]
done
clear
C)
\[2.08\times 1{{0}^{-5}}m/s\]
done
clear
D)
\[0.55\times 1{{0}^{-5}}m/s\]
done
clear
View Answer play_arrow
A body is released from a great height falls freely towards the earth. Another body is released from the same height exactly a second later. Then, the separation between two bodies, 2 sec after the release of the second body is nearly:
A)
15m
done
clear
B)
20m
done
clear
C)
25m
done
clear
D)
30m
done
clear
View Answer play_arrow
During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of \[\frac{{{C}_{p}}}{{{C}_{v}}}\]for the gas is:
A)
4
done
clear
B)
2
done
clear
C)
5
done
clear
D)
3
done
clear
View Answer play_arrow
A young's double slit experiment. If the set up brought into the water from air, then fringe width:
A)
Becomes infinite
done
clear
B)
Decreases
done
clear
C)
Increases
done
clear
D)
Remains unchanged
done
clear
View Answer play_arrow
A ray of light travelling in transparent medium of refractive index p falls on a surface separating the medium from air at an angle of incidence of \[45{}^\circ \] for which of the following value of\[\mu \]the ray can undergo TIR?
A)
\[\mu >1.33\]
done
clear
B)
\[\mu >1.4\]
done
clear
C)
\[\mu >1.5\]
done
clear
D)
\[\mu >1.25\]
done
clear
View Answer play_arrow
The force required to just move a body up an inclined plane is double the force required to just prevent it from sliding down. If \[\theta \] is the angle of friction and \[\phi \] is the angle which the plane makes with horizontal, then:
A)
\[\tan \phi = 2 tan\theta \]
done
clear
B)
\[\tan \phi = 3 tan\theta \]
done
clear
C)
\[\tan \phi = tan\theta \]
done
clear
D)
\[\tan \theta = 3tan\phi \]
done
clear
View Answer play_arrow
Two condenses of capacity 0.3uF and 0.6uF respectively are connected in series. The combination is connected across a potential of 6V. The ratio of energies stored by the condenser will be:
A)
\[\frac{1}{2}\]
done
clear
B)
2
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
4
done
clear
View Answer play_arrow
A circuit draws 330W from a 110V, 60Hz AC line. The power factor is 0.6 and the current lags the voltage. The capacitance of a series capacitor that will result in a power factor of unity is equal to:
A)
\[31\,\mu F\]
done
clear
B)
54\[54\,\mu F\]
done
clear
C)
\[151\,\mu F\]
done
clear
D)
\[201\,\mu F\]
done
clear
View Answer play_arrow
The value of coefficient of volume expansion of glycerine is \[5\times {{10}^{-4}}{{K}^{-1}}.\]The fractional change in the density of glycerine for a rise of \[40{}^\circ C\]in its temperature, is:
A)
0.025
done
clear
B)
0.010
done
clear
C)
0.015
done
clear
D)
0.020
done
clear
View Answer play_arrow
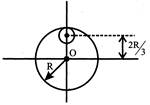
From a circular disc of radius R and mass 9M , a small disc of radius \[\frac{R}{3}\] is removeThe moment of inertia of the remaining disc about an axis perpendicular to the plane of the disc and passing through 0 is:
A)
\[4M{{R}^{2}}\]
done
clear
B)
\[\frac{40}{9}M{{R}^{2}}\]
done
clear
C)
\[10M{{R}^{2}}\]
done
clear
D)
\[\frac{37}{9}M{{R}^{2}}\]
done
clear
View Answer play_arrow
\[\gamma \]represent of ratio of the specific heat of a gas. For a given mass of the gas, the change in internal energy when the volume expands from V to 3V at constant pressure P is:
A)
\[\frac{3PV}{\gamma -1}\]
done
clear
B)
\[\frac{3PV}{\gamma +1}\]
done
clear
C)
\[\frac{2PV}{\gamma +1}\]
done
clear
D)
s \[\frac{2PV}{\gamma -1}\]
done
clear
View Answer play_arrow
A piano convex lens fits exactly into a piano concave lens. Their plane surfaces are parallel to each other. If lenses are made of different materials of refractive index \[{{\mu }_{1}}\operatorname{and}{{\mu }_{2}}\] and R is the radius of curvature of the curved surfaces of the lenses, then the focal length of the combination is:
A)
\[\frac{R}{2\left( {{\mu }_{1}}+{{\mu }_{2}} \right)}\]
done
clear
B)
\[\frac{R}{2\left( {{\mu }_{1}}-{{\mu }_{2}} \right)}\]
done
clear
C)
\[\frac{R}{{{\mu }_{1}}-{{\mu }_{2}}}\]
done
clear
D)
\[\frac{2R}{{{\mu }_{1}}-{{\mu }_{2}}}\]
done
clear
View Answer play_arrow
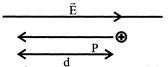
In the figure, a proton moves a distance d in a uniform electric field E as shown in the figure. Does the electric field do a positive or negative work on the proton? Does the electric potential energy of the proton increases or decreases:
A)
-ve, increase
done
clear
B)
+ve, decrease
done
clear
C)
-ve, decrease
done
clear
D)
+ve, increase
done
clear
View Answer play_arrow
A motor car is moving with speed 30m/s on a circular path of radius 500m. Its speed is increasing at the rate of\[2m/{{s}^{2}}\]. What will be its resultant acceleration?
A)
\[2.5m/{{s}^{2}}\]
done
clear
B)
\[2.7m/{{s}^{2}}\]
done
clear
C)
\[2m/{{s}^{2}}\]
done
clear
D)
\[4.5m/{{s}^{2}}\]
done
clear
View Answer play_arrow
A square gate of size 1m x 1m is hinged at its mid point. A fluid of density p fills space to the left to the gate. The force F required to hold the gate stationary:
A)
\[\frac{\rho \operatorname{g}}{3}\]
done
clear
B)
\[\frac{\rho \operatorname{g}}{2}\]
done
clear
C)
\[\frac{\rho \operatorname{g}}{6}\]
done
clear
D)
None
done
clear
View Answer play_arrow
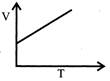
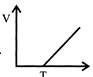
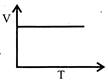
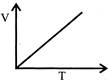
Volume - temperature graph at constant pressure for a monotomic gas is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The power factor of an R-L circuit is\[\frac{1}{\sqrt{2}}\]. If the frequency of AC is doubled, what will be the power factor?
A)
\[\frac{1}{\sqrt{3}}\]
done
clear
B)
\[\frac{1}{\sqrt{5}}\]
done
clear
C)
\[\frac{1}{\sqrt{7}}\]
done
clear
D)
\[\frac{1}{\sqrt{11}}\]
done
clear
View Answer play_arrow
How many moles of magnesium phosphate, \[{{\operatorname{Mg}}_{3}}{{\left( P{{O}_{4}} \right)}_{2}}\]will contain 0.25 mole of oxygen atoms?
A)
\[2.5\times {{10}^{-2}}\]
done
clear
B)
0.02
done
clear
C)
\[3.125\times {{10}^{-2}}\]
done
clear
D)
\[1.25\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
The total volume of atoms present in a face centred cubic unit cell of a metal is Cr is atomic radius?
A)
\[\frac{16}{3}\pi {{\operatorname{r}}^{3}}\]
done
clear
B)
\[\frac{20}{3}\pi {{\operatorname{r}}^{3}}\]
done
clear
C)
\[\frac{24}{3}\pi {{\operatorname{r}}^{3}}\]
done
clear
D)
\[\frac{12}{3}\pi {{\operatorname{r}}^{3}}\]
done
clear
View Answer play_arrow
According to Bohr's theory, the angular momentum of an electron in 5th orbit is:
A)
\[\frac{2.5\operatorname{h}}{\pi }\]
done
clear
B)
\[\frac{25\operatorname{h}}{\pi }\]
done
clear
C)
\[\frac{1.0\operatorname{h}}{\pi }\]
done
clear
D)
\[\frac{10\operatorname{h}}{\pi }\]
done
clear
View Answer play_arrow
The maximum no. of electrons in an orbit with 1 = 2, n = 3 is:
A)
2
done
clear
B)
6
done
clear
C)
12
done
clear
D)
10
done
clear
View Answer play_arrow
A semiconductor of Ge can be made p-type by adding:
A)
Trivalent impurity
done
clear
B)
Tetravalent impurity
done
clear
C)
Pentavalent impurity
done
clear
D)
Divalent impurity
done
clear
View Answer play_arrow
Best way to prevent runting of iron by:
A)
Making iron cathode
done
clear
B)
Putting it in saline water
done
clear
C)
Both
done
clear
D)
None
done
clear
View Answer play_arrow
A plot of log Vm versus log p for the adsorption of a gas on a solid gives a straight line with slope equal to:
A)
\[\frac{1}{n}\]
done
clear
B)
log k
done
clear
C)
-log k
done
clear
D)
n
done
clear
View Answer play_arrow
The magnetite ore is:
A)
Ferrous oxide
done
clear
B)
Ferric oxide
done
clear
C)
Ferric hydroxide
done
clear
D)
Ferroso - ferric oxide
done
clear
View Answer play_arrow
Which of the following compounds has the highest boiling point?
A)
\[{{\operatorname{CH}}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
B)
\[{{\operatorname{CH}}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
C)
\[{{\operatorname{CH}}_{3}}CH\left( C{{H}_{3}} \right)C{{H}_{2}}Cl\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}CCl\]
done
clear
View Answer play_arrow
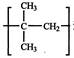
Monomer of
is:
A)
2-Methlpropene
done
clear
B)
Styrene
done
clear
C)
Propylene
done
clear
D)
Ethene
done
clear
View Answer play_arrow
The hormone that helps in the conversion of glucose into glycogen is:
A)
Cortisone
done
clear
B)
Bile acids
done
clear
C)
Adrenaline
done
clear
D)
Insulin
done
clear
View Answer play_arrow
The reaction,
example of:
A)
Wurtz reaction
done
clear
B)
Witting reaction
done
clear
C)
Ullmann reaction
done
clear
D)
Williamson reaction
done
clear
View Answer play_arrow
The correct structure of drug paracetamol is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The smog is caused by the presence of mostly:
A)
\[{{\operatorname{O}}_{2}}\,and\,{{O}_{3}}\]
done
clear
B)
Oxides of sulphur and nitrogen
done
clear
C)
\[{{\operatorname{O}}_{2}} and {{N}_{2}}\]
done
clear
D)
\[{{\operatorname{O}}_{3}} and {{N}_{2}}\]
done
clear
View Answer play_arrow
Oxidation state of osmium \[\left( {{O}_{s}} \right) in {{O}_{s}}{{O}_{4}}\]is:
A)
+7
done
clear
B)
+6
done
clear
C)
+4
done
clear
D)
+8
done
clear
View Answer play_arrow
Which is the correct order of increasing enthalpy of vaporisation?
A)
\[{{\operatorname{NH}}_{3}}<P{{H}_{3}}<AS{{H}_{3}}\]
done
clear
B)
\[{{\operatorname{ASH}}_{3}}<P{{H}_{3}}<N{{H}_{3}}\]
done
clear
C)
\[{{\operatorname{PH}}_{3}}<AS{{H}_{3}}<N{{H}_{3}}\]
done
clear
D)
\[{{\operatorname{NH}}_{3}}<AS{{H}_{3}}<P{{H}_{3}}\]
done
clear
View Answer play_arrow
Pressure of a mixture of 4g of \[{{\operatorname{O}}_{2}} and 2g of {{H}_{2}}\] confined in a bulb of 1 litre at \[\operatorname{O}{}^\circ C\] is:
A)
25.215 atm
done
clear
B)
31.205 atm
done
clear
C)
45.215 atm
done
clear
D)
15.210 atm
done
clear
View Answer play_arrow
For the reaction of one mole of zinc dust with one mole of sulphuric acid in a bomb caloriemeter, Au and w correspond to:
A)
\[\Delta u < 0, w = 0\]
done
clear
B)
\[\Delta u < 0, w < 0\]
done
clear
C)
\[\Delta u > 0, w = 0\]
done
clear
D)
\[\Delta u > 0, w > 0\]
done
clear
View Answer play_arrow
The radii of F, F , 0 and O2 are in the order of:
A)
\[{{\operatorname{O}}^{2}} >{{F}^{-}} >F>O\]
done
clear
B)
\[{{\operatorname{F}}^{-}} >{{O}^{2-}} >F>\operatorname{O}\]
done
clear
C)
\[{{\operatorname{O}}^{2-}}>O>r>F\]
done
clear
D)
\[{{\operatorname{O}}^{2-}} >{{F}^{2}} >O>F\]
done
clear
View Answer play_arrow
Which of the following colligative properties can provide molar mass of proteins (polymers or colloids) with greatest precision?
A)
Depression in freezing point
done
clear
B)
Osmotic pressure
done
clear
C)
Relative lowering of vapour pressure
done
clear
D)
Elevation of boiling point
done
clear
View Answer play_arrow
75% of the first order reaction was completed in 32 min. 50% of the reaction was completed in:
A)
24 min
done
clear
B)
8min
done
clear
C)
16 min
done
clear
D)
4min
done
clear
View Answer play_arrow
The freezing point of equimolal aqueous solution will be highest for:
A)
\[{{\operatorname{C}}_{6}}{{H}_{5}}N{{H}_{3}}C{{l}^{-}}\]
done
clear
B)
\[\operatorname{Ca}{{\left( N{{O}_{3}} \right)}_{2}}\]
done
clear
C)
\[La{{\left( N{{O}_{3}} \right)}_{2}}\]
done
clear
D)
\[{{\operatorname{C}}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
View Answer play_arrow
The reaction \[2FeC{{l}_{3}}+SnC{{l}_{2}}\to 2FeC{{l}_{2}}+SnC{{l}_{4}}\] is an example of:
A)
First order
done
clear
B)
Second order
done
clear
C)
Third order
done
clear
D)
None of these
done
clear
View Answer play_arrow
Intermolecular hydrogen bonding is strongest in:
A)
Methylamine
done
clear
B)
Phenol
done
clear
C)
Formaldehyde
done
clear
D)
Methanol
done
clear
View Answer play_arrow
Which of the following is planar?
A)
\[{{\operatorname{XeO}}_{4}}\]
done
clear
B)
\[{{\operatorname{XeO}}_{3}}{{F}_{2}}\]
done
clear
C)
\[{{\operatorname{XeO}}_{2}}{{F}_{2}}\]
done
clear
D)
\[{{\operatorname{XeF}}_{4}}\]
done
clear
View Answer play_arrow
\[{{\operatorname{C}}_{6}}{{H}_{5}}CONHC{{H}_{3}}\]can be converted into \[{{\operatorname{C}}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\]by:
A)
\[{{\operatorname{NaBH}}_{4}}\]
done
clear
B)
\[{{\operatorname{H}}_{2}}-Pd/C\]
done
clear
C)
\[{{\operatorname{LiAlH}}_{4}}\]
done
clear
D)
\[\operatorname{Zn}-Hg/HCl\]
done
clear
View Answer play_arrow
Which of the following has \[\operatorname{p}\pi - d\pi \]bonding?
A)
\[{{\operatorname{NO}}_{3}}^{-}\]
done
clear
B)
\[{{\operatorname{SO}}_{3}}^{2-}\]
done
clear
C)
\[{{\operatorname{BO}}^{3-}}_{3}\]
done
clear
D)
\[{{\operatorname{CO}}^{2-}}_{3}\]
done
clear
View Answer play_arrow
\[{{\operatorname{CrO}}_{3}}\]dissolves in aqueous NaOH to give:
A)
\[{{\operatorname{CrO}}^{2-}}_{4}\]
done
clear
B)
\[\operatorname{Cr}{{\left( OH \right)}_{3}}\]
done
clear
C)
\[\operatorname{Cr},{{\operatorname{O}}^{2-}}_{3}\]
done
clear
D)
\[\operatorname{Cr}{{\left( OH \right)}_{2}}\]
done
clear
View Answer play_arrow
Dimethyl glyoxime gives a red precipitate with\[{{\operatorname{Ni}}^{2+}}\]. Which is used for its detection. To get this precipitate readily, the best Ph range is:
A)
>l
done
clear
B)
2-3
done
clear
C)
3-4
done
clear
D)
9-11
done
clear
View Answer play_arrow
\[\left[ Fe\left( N{{O}_{2}} \right)C{{l}_{3}} \right]and\left[ Fe{{\left( O-NO \right)}_{3}}C{{l}_{3}} \right]\] shown:
A)
Linkage isomerism
done
clear
B)
Geometrical isomerism
done
clear
C)
Optical isomerism
done
clear
D)
None of these
done
clear
View Answer play_arrow
The product obtained is/are:
A)
o - product,
done
clear
B)
m - product
done
clear
C)
o - and p - products
done
clear
D)
o -, m - and p - products
done
clear
View Answer play_arrow
Which has highest \[{{\operatorname{pK}}_{b}}\]value?
A)
\[{{\operatorname{R}}_{3}}C-N{{H}_{2}}\]
done
clear
B)
\[{{\operatorname{R}}_{2}}NH\]
done
clear
C)
\[\operatorname{R}-N{{H}_{2}}\]
done
clear
D)
\[{{\operatorname{NH}}_{3}}\]
done
clear
View Answer play_arrow
It the enolate ion combines with carbonyl group of an ester, we get:
A)
Aldol
done
clear
B)
\[\alpha -,\beta - unsaturated ester\]
done
clear
C)
\[\beta - ketoaldehyde\]
done
clear
D)
Acid
done
clear
View Answer play_arrow
A)
\[{{\operatorname{Na}}_{2}}S\]
done
clear
B)
\[\operatorname{Sn}/HCl\]
done
clear
C)
\[{{\operatorname{LiAlH}}_{4}}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
For the reaction \[{{\operatorname{H}}_{2}} \left( g \right)+{{I}_{2}} \left( g \right)\rightleftharpoons 2HI\left( g \right)\] the equilibrium constant \[{{\operatorname{k}}_{p}}\]changes with:
A)
Temperature
done
clear
B)
Total pressure
done
clear
C)
Catalyst
done
clear
D)
Amount of \[{{\operatorname{H}}_{2}} and {{I}_{2}}\]
done
clear
View Answer play_arrow
The strongest conjugate base is:
A)
\[{{\operatorname{NO}}_{3}}^{-}\]
done
clear
B)
\[{{\operatorname{Cl}}^{-}}\]
done
clear
C)
\[{{\operatorname{SO}}^{2-}}_{4}\]
done
clear
D)
\[C{{H}_{3}}CO{{O}^{-}}\]
done
clear
View Answer play_arrow
How many sigma and pi bonds are present in toluene?
A)
\[3\pi + 8\sigma \]
done
clear
B)
\[3\pi +6\sigma \]
done
clear
C)
\[3\pi +15\sigma \]
done
clear
D)
\[6\pi +2\sigma \]
done
clear
View Answer play_arrow
What is false about \[{{\operatorname{H}}_{2}}{{O}_{2}}\]?
A)
Act as both oxidizing and reducing agent
done
clear
B)
Two OH bonds lie in same plane
done
clear
C)
Pale blue liquid
done
clear
D)
Can be oxidised by \[{{\operatorname{O}}_{3}}\]
done
clear
View Answer play_arrow
The correct order in which 0-0 bond length increases in the following is:
A)
\[{{O}_{2}} < {{H}_{2}}{{O}_{2}} < {{O}_{2}}\]
done
clear
B)
\[\operatorname{O}{{ }_{2}}< {{0}_{3}} < {{H}_{2}}{{O}_{2}}\]
done
clear
C)
\[{{O}_{2}} < {{H}_{2}}{{O}_{2}} < {{O}_{3}}\]
done
clear
D)
\[{{H}_{2}}{{O}_{2}} <{{O}_{2}} < {{O}_{3}}\]
done
clear
View Answer play_arrow
An oxide which gives\[{{H}_{2}}{{O}_{2}}\]on treatment with dilute acid is:
A)
\[{{\operatorname{PbO}}_{2}}\]
done
clear
B)
\[{{\operatorname{Na}}_{2}}{{O}_{2}}\]
done
clear
C)
\[{{\operatorname{MnO}}_{2}}\]
done
clear
D)
\[{{\operatorname{TiO}}_{2}}\]
done
clear
View Answer play_arrow
Hydrogen bonding is strongest in:
A)
S-H......O
done
clear
B)
O-H......S
done
clear
C)
f-h.......O
done
clear
D)
O-H......N
done
clear
View Answer play_arrow
The number of lone pairs of electrons present on the central atom of \[{{\operatorname{CIF}}_{3}}\]is:
A)
O
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
Which of the following does not exist in free state:
A)
\[{{\operatorname{BF}}_{3}}\]
done
clear
B)
\[{{\operatorname{BCL}}_{3}}\]
done
clear
C)
\[{{\operatorname{BBr}}_{3}}\]
done
clear
D)
\[{{\operatorname{BH}}_{3}}\]
done
clear
View Answer play_arrow
The IUPAC name of the compound
is:
A)
1, 1-dimethyl-3-cyclohexanol
done
clear
B)
1, 1-dimethyl-3-hydroxycyclohexane
done
clear
C)
3, 3-dimethyl-1-cydohexanol
done
clear
D)
3, 3-dimethyl-1-hydroxycyclohexane
done
clear
View Answer play_arrow
The compounds of alkaline earth metals have the following magnetic nature:
A)
Diamagnetic
done
clear
B)
Paramagnetic
done
clear
C)
Ferromagnetic
done
clear
D)
Anti ferromagnetic
done
clear
View Answer play_arrow
In the pitcher plant, the pitcher are modified:
A)
Fruits
done
clear
B)
Branches
done
clear
C)
Petioles
done
clear
D)
Leaves
done
clear
View Answer play_arrow
Where are sugar polymer produce in the RBC's?
A)
Ribosome
done
clear
B)
Mitochondria
done
clear
C)
Plasma membrane
done
clear
D)
Golgi apparatus
done
clear
View Answer play_arrow
During meiosis the replication of DNA takes place in:
A)
Interphase I
done
clear
B)
Interphase II
done
clear
C)
Both
done
clear
D)
None
done
clear
View Answer play_arrow
Seedless fruit can be obtained by spraying:
A)
Gibberellins
done
clear
B)
Auxins
done
clear
C)
Cytokinins
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
When did environment protection act passed and which country passed it?
A)
Govt. of America, 1982
done
clear
B)
Govt. of India, 1984
done
clear
C)
Govt. of Colombia, 1986
done
clear
D)
Govt. of India, 1986
done
clear
View Answer play_arrow
The filtrate in PCT is:
A)
Isotonic
done
clear
B)
Hypotonic
done
clear
C)
Hypertonic
done
clear
D)
Watery
done
clear
View Answer play_arrow
Which species of plasmodium is founded in India?
A)
P. vivax
done
clear
B)
P. malavia
done
clear
C)
P. falciparum
done
clear
D)
All of these
done
clear
View Answer play_arrow
The related families are placed together in the same:
A)
Phylum
done
clear
B)
Class
done
clear
C)
Genera
done
clear
D)
Order
done
clear
View Answer play_arrow
Why RNA act as catalyst or unstable?
A)
Presence of-CHO
done
clear
B)
Presence of Z-OH
done
clear
C)
Presence of 2'-C=0
done
clear
D)
All of these
done
clear
View Answer play_arrow
Cork cells are:
A)
Lignified
done
clear
B)
Suberized
done
clear
C)
Cutmised
done
clear
D)
Pectirdsed
done
clear
View Answer play_arrow
When more than one pistil fused together called:
A)
Apocarpous
done
clear
B)
Multipistillate
done
clear
C)
Syncarpous
done
clear
D)
Monocarpous
done
clear
View Answer play_arrow
Apoplast movement of water occur through:
A)
Cell walls
done
clear
B)
Plasmodesmata
done
clear
C)
Cytoplasm
done
clear
D)
Plasma membrane
done
clear
View Answer play_arrow
Which hormone is released by ILJDs?
A)
Progestasert
done
clear
B)
Oxytostate
done
clear
C)
LNG-20
done
clear
D)
Both a & c
done
clear
View Answer play_arrow
Gall bladder store:
A)
Lipase
done
clear
B)
Amylase
done
clear
C)
Bile
done
clear
D)
Protease
done
clear
View Answer play_arrow
What is false for ecological pyramids?
A)
Relationship explained in turms of number
done
clear
B)
Energy flow from particular trophic level
done
clear
C)
Account for same species belonging different levels
done
clear
D)
Pyramid either inverted or uplight except some exceptional
done
clear
View Answer play_arrow
Synovial joints are found at the:
A)
Elbow
done
clear
B)
Skull
done
clear
C)
Symphysis pubis
done
clear
D)
Vertebrae
done
clear
View Answer play_arrow
50% of vertebrates are:
A)
Mammals
done
clear
B)
Fish
done
clear
C)
Birds
done
clear
D)
Reptiles
done
clear
View Answer play_arrow
The holozoic kind of nutrition is found in:
A)
Protozoans
done
clear
B)
Phytoplanctons
done
clear
C)
Cynobacteria
done
clear
D)
Fungi
done
clear
View Answer play_arrow
In which organismes both type of gametes are motile:
A)
Seaweeds
done
clear
B)
Cycas
done
clear
C)
Ulothrix
done
clear
D)
Ulva
done
clear
View Answer play_arrow
Blood platelets are formed from:
A)
Leucocytes
done
clear
B)
Megakaryocytes
done
clear
C)
Thrombocytes
done
clear
D)
Monocytes
done
clear
View Answer play_arrow
Which yeast inhibit the enzyme, responsible for synthesize of cholesterol:
A)
Trichoderma polysporum
done
clear
B)
Closteridium butylicum
done
clear
C)
Monascus purpureus
done
clear
D)
Saccharomyces cerevisiae
done
clear
View Answer play_arrow
Which of the following is a component of IAA:
A)
Zinc
done
clear
B)
Copper
done
clear
C)
Boron
done
clear
D)
Molybdenum
done
clear
View Answer play_arrow
Which is a palindromic sequence in these:
A)
AGATTC
done
clear
B)
GAAAAG
done
clear
C)
GAATTG
done
clear
D)
CTTAAG
done
clear
View Answer play_arrow
Earthworm respires through:
A)
Spiraeles
done
clear
B)
Gills
done
clear
C)
Trachea
done
clear
D)
Skin
done
clear
View Answer play_arrow
Number of cranial nerves in man are:
A)
31 pairs
done
clear
B)
10 pairs
done
clear
C)
13 pairs
done
clear
D)
12 pairs
done
clear
View Answer play_arrow
How can we say that amphibians evolved into reptiles on the basis of their egg shell:
A)
Egg shell are thin & rough
done
clear
B)
Egg shell are thick & dry
done
clear
C)
Thick egg shelled which do not dry up in sun
done
clear
D)
Thin egg shelled which do not dry up in sun
done
clear
View Answer play_arrow
Kelp is a kind of:
A)
An aquatic plant
done
clear
B)
A moss
done
clear
C)
An algae
done
clear
D)
A fungus
done
clear
View Answer play_arrow
What is wrong about desert lizard:
A)
It lack of physiological ability that a normal mammals have
done
clear
B)
Deal with high temperature
done
clear
C)
Keep body temperature constant.
done
clear
D)
Bask in the sun above the comfort zone
done
clear
View Answer play_arrow
The cell drinking is also termed as:
A)
Exocytosis
done
clear
B)
Pinocytosis
done
clear
C)
Phagocytosis
done
clear
D)
endocytosis
done
clear
View Answer play_arrow
Which hormone induce super ovulation in MOET technique:
A)
Progesterone
done
clear
B)
FSH
done
clear
C)
LH
done
clear
D)
Estrogen
done
clear
View Answer play_arrow
The first electron donor in cyclic phosphorylation is:
A)
Water
done
clear
B)
\[{{\operatorname{P}}_{700}}\]
done
clear
C)
\[{{\operatorname{P}}_{680}}\]
done
clear
D)
\[{{\operatorname{O}}_{2}}\]
done
clear
View Answer play_arrow
Which bond is present in between humar insulin:
A)
\[{{\operatorname{H}}_{2}}\]
done
clear
B)
\[{{\operatorname{S}}_{2}}\]
done
clear
C)
\[{{\operatorname{N}}_{2}}\]
done
clear
D)
\[N{{i}_{2}}\]
done
clear
View Answer play_arrow
The circulatory system of cockroach consisi of:
A)
Canals
done
clear
B)
Haemocoel
done
clear
C)
Coelentron
done
clear
D)
Capillaries
done
clear
View Answer play_arrow
How many genotype of the human ABO blood type give?
A)
8
done
clear
B)
6
done
clear
C)
10
done
clear
D)
4
done
clear
View Answer play_arrow
The enzymes for krebs cycle are present in:
A)
Cytoplasm
done
clear
B)
Outer membrane of mitochondria
done
clear
C)
Matrix of mitochondria
done
clear
D)
\[{{\operatorname{F}}_{1}}\] particles
done
clear
View Answer play_arrow
Earth's surface re-emits heat in the form of:
A)
Ultra violet rays
done
clear
B)
Infrared rays
done
clear
C)
\[\alpha -rays\]
done
clear
D)
\[ \beta -rays\]
done
clear
View Answer play_arrow
Rodents are also known as:
A)
Hoofed mammals
done
clear
B)
Toothless mammals
done
clear
C)
Gnawing mammals
done
clear
D)
Carnivores
done
clear
View Answer play_arrow
Hallucination is caused due to the drug:
A)
Opiods
done
clear
B)
Cocaine
done
clear
C)
Cannabinoids
done
clear
D)
All of these
done
clear
View Answer play_arrow
The primary level of protein is due to the presence of:
A)
Hydrogen bond
done
clear
B)
Peptide bond
done
clear
C)
-S-S-linkage
done
clear
D)
Ionic bonds
done
clear
View Answer play_arrow
Which sequences normally do not code for any protein, but form large portion of human genome:
A)
UGA
done
clear
B)
Satellite DNA.
done
clear
C)
Micro satellite
done
clear
D)
Mini satellite
done
clear
View Answer play_arrow
Acromegaly is due to over secretion of:
A)
GH is children
done
clear
B)
GH after adolescence
done
clear
C)
ACTH
done
clear
D)
Thyroid in adults
done
clear
View Answer play_arrow
Name the parasitic species of fruit which contain very large no. of seeds:
A)
Watermelon
done
clear
B)
Papaya
done
clear
C)
Orobanche
done
clear
D)
All of these
done
clear
View Answer play_arrow
When the leaflets are joined together at a common point at the petiole, the leaf is:
A)
Simple leaf
done
clear
B)
Pinnately compound leaf
done
clear
C)
Palmately compound leaf
done
clear
D)
A branch
done
clear
View Answer play_arrow
CDRI stand for:
A)
Common Dairy Research Institute
done
clear
B)
Common Drug Resolve Institute
done
clear
C)
Common Drug Research Institute
done
clear
D)
Community Drug Research Institute
done
clear
View Answer play_arrow
How many cells will be produced if a cell divide mitotically 6 times:
A)
12
done
clear
B)
24
done
clear
C)
32
done
clear
D)
64
done
clear
View Answer play_arrow
1ndole acetic acid belongs to the group of hormone called:
A)
Auxins
done
clear
B)
Gibberlins
done
clear
C)
Cytokinins
done
clear
D)
Abscisic acid
done
clear
View Answer play_arrow
Who put price tag on nature's life support services:
A)
Kaul pranti
done
clear
B)
George benthanm
done
clear
C)
Robert constanza
done
clear
D)
Adolf engler
done
clear
View Answer play_arrow
The excreatory organ of prawn are:
A)
Flame cells
done
clear
B)
Malphighin tubules
done
clear
C)
Nephridia
done
clear
D)
Green gland
done
clear
View Answer play_arrow
Find the odd one:
A)
Phylum
done
clear
B)
Taxon
done
clear
C)
Genera
done
clear
D)
Species
done
clear
View Answer play_arrow
Which medicinal plant shown genetic variation in Himalaya:
A)
Rauwolfia tuberosum
done
clear
B)
Solanum vomitoria
done
clear
C)
Rauwolfia vomitoria
done
clear
D)
Solanum tuberosum
done
clear
View Answer play_arrow
Pith is small & absent in:
A)
Monocot root
done
clear
B)
Dicot root
done
clear
C)
Monocot stem
done
clear
D)
Dicot stem
done
clear
View Answer play_arrow
Antibiotics used to control leprosy:
A)
Dapsone
done
clear
B)
Eitampin
done
clear
C)
Minocycline
done
clear
D)
All of these
done
clear
View Answer play_arrow
The water loss during transpiration:
A)
Contain minerals
done
clear
B)
Contain salts
done
clear
C)
contain pure water
done
clear
D)
contain dissolved nutrients
done
clear
View Answer play_arrow
Primer are chemically synthesised:
A)
Ribonucleotide
done
clear
B)
Deoxyribonucleotide
done
clear
C)
Oligonucleotide
done
clear
D)
All of these
done
clear
View Answer play_arrow
Chymotrypsin acts on:
A)
Carbohydrates
done
clear
B)
Proteins
done
clear
C)
Fats
done
clear
D)
Starch
done
clear
View Answer play_arrow
FSH help in the process:
A)
Spermatogenesis
done
clear
B)
Spermiogenesis
done
clear
C)
Spermiation
done
clear
D)
All of above
done
clear
View Answer play_arrow
Appendicular skeleton is made up of:
A)
Pectoral and pelvic girdle
done
clear
B)
Fore and hindlimbs
done
clear
C)
Girdles and limbs
done
clear
D)
Skull and vertebral column
done
clear
View Answer play_arrow
Few fossils of man bones have been discovered in:
A)
Ethiopia
done
clear
B)
South America
done
clear
C)
Africa
done
clear
D)
North America
done
clear
View Answer play_arrow
The nitrifying bacteria nitrosomonas is a:
A)
Photoautograph
done
clear
B)
Chemautograph
done
clear
C)
Anaerobic
done
clear
D)
Symbiotic
done
clear
View Answer play_arrow
Enzyme thromboplastin converts:
A)
Thrombin to prothrombin
done
clear
B)
Prothrombin to thrombin
done
clear
C)
Thrombin to fibrinogen
done
clear
D)
Fibrinogen to fibrin
done
clear
View Answer play_arrow
Hisardale is new breed of sheep developed form:
A)
Marino arms and Shameria ewes
done
clear
B)
Bikaner arms and Pahadi ewes
done
clear
C)
Bikaner ewes and Marino rams
done
clear
D)
Bikaner ewes and Marino
done
clear
View Answer play_arrow
Which macro-element is required in the middle lamellae if the cell wall:
A)
Potassium
done
clear
B)
Calcium
done
clear
C)
Phosphorous
done
clear
D)
Nitrogen
done
clear
View Answer play_arrow
Lepidopterans killed by protein of:
A)
Meloidegyne incognita
done
clear
B)
Bacillus thuringiensis
done
clear
C)
Thermos aquatics
done
clear
D)
All of these
done
clear
View Answer play_arrow
A slit like openings to larynx is called:
A)
Glottis
done
clear
B)
Gullel
done
clear
C)
Epiglottis
done
clear
D)
Trachea
done
clear
View Answer play_arrow
Who search X-body in organisms:
A)
Henking
done
clear
B)
Morgan
done
clear
C)
Mendel
done
clear
D)
De vries
done
clear
View Answer play_arrow
Gustatory receptous cells are present in:
A)
Nose epithelium
done
clear
B)
Taste bud
done
clear
C)
Retina
done
clear
D)
Inner car
done
clear
View Answer play_arrow
Which one of them is auto immune disease?
A)
Plague
done
clear
B)
Whooping cough
done
clear
C)
Rheumatid authritis
done
clear
D)
Diphtheria
done
clear
View Answer play_arrow
Which one of the following is dominant sporophytic generation:
A)
Dryopteris
done
clear
B)
Funaria
done
clear
C)
Spirogyra
done
clear
D)
Liverworts
done
clear
View Answer play_arrow
Repetitive DNA sequence known as:
A)
Micro-satellite
done
clear
B)
Mini-Satellite
done
clear
C)
VNTR-probe
done
clear
D)
All of these
done
clear
View Answer play_arrow
The unit membrane concept was given by:
A)
Danielli Davson
done
clear
B)
Roberston
done
clear
C)
Singer
done
clear
D)
Nicolson
done
clear
View Answer play_arrow
Acid rain is produced by:
A)
Excess \[{{\operatorname{NO}}_{2}} and S{{O}_{2}}\]from burning fossil fuels
done
clear
B)
Excess production of \[{{\operatorname{NO}}_{3}}\] by industry
done
clear
C)
Excess production of \[{{\operatorname{CO}}_{2}}\]combustion and animal respiration
done
clear
D)
Excess release of CO by incomplete combustion
done
clear
View Answer play_arrow
Domestic waste constitutes:
A)
Non-biodegradable l ubstance
done
clear
B)
Biodegradable substance
done
clear
C)
Effluents
done
clear
D)
Air pollution.
done
clear
View Answer play_arrow
Name the plant whom seed is viable, largely known:
A)
Orabanche
done
clear
B)
Striga
done
clear
C)
Phoenix daetylifera
done
clear
D)
Lupinus areticus
done
clear
View Answer play_arrow
Chlorophyll molecule appear green because it:
A)
Absorbs blue and green colour
done
clear
B)
Absorbs green colour
done
clear
C)
Reflect blue and green colour
done
clear
D)
Transmit blue & green colour
done
clear
View Answer play_arrow
Fossil fuels are reservoir of:
A)
Carbon
done
clear
B)
Phosphorous
done
clear
C)
Nitrogen
done
clear
D)
Calcium
done
clear
View Answer play_arrow
Coronary sulcus is present between:
A)
Two atria
done
clear
B)
Two vent rides
done
clear
C)
Atria and ventricle
done
clear
D)
All of these
done
clear
View Answer play_arrow
Give the equation of species-area relationship:
A)
Log C = log S - Z log A
done
clear
B)
Log Z = log C - Z log A
done
clear
C)
Log S = log C + Z log A
done
clear
D)
Log A = log C - Z log S
done
clear
View Answer play_arrow
An enzyme ETC are present in:
A)
Cytoplasm
done
clear
B)
Outer mitochondrial membrane
done
clear
C)
Materix
done
clear
D)
Inner mitochondrial membrane
done
clear
View Answer play_arrow
In biolistics method bombarded particles is of metal:
A)
Silver
done
clear
B)
Gold
done
clear
C)
Aluminium
done
clear
D)
Nickel
done
clear
View Answer play_arrow
Which one is not a feature of Agnatha?
A)
Parasise
done
clear
B)
Jawless mouth
done
clear
C)
Paired fins
done
clear
D)
Wormy teeth
done
clear
View Answer play_arrow
During luteal phase level of ovarian hormone is:
A)
High
done
clear
B)
Low
done
clear
C)
Attained peak level
done
clear
D)
Medium
done
clear
View Answer play_arrow
Glycogen differs from starch in:
A)
Kinds of bonds
done
clear
B)
Kinds of molecules
done
clear
C)
Being present in animals only
done
clear
D)
Structure of molecules
done
clear
View Answer play_arrow
What is the two key concept of Darwinian theory of evolution?
A)
Saltation & natural selection
done
clear
B)
Founders effect & industrialization
done
clear
C)
Natural selection & branching descent
done
clear
D)
Natural selection & founders effect
done
clear
View Answer play_arrow
Cushing's disease is due to over secretion of:
A)
Pituitary
done
clear
B)
Adrenal cortex
done
clear
C)
Thyroid
done
clear
D)
Adrenal medulla
done
clear
View Answer play_arrow
Who is the first transgenic cow?
A)
Curie
done
clear
B)
Rosie
done
clear
C)
Liksy
done
clear
D)
Piku
done
clear
View Answer play_arrow
Cystic fibrosis is:
A)
Medelliansex linked disorder
done
clear
B)
Autosomal exclssive
done
clear
C)
Mendellian recessive
done
clear
D)
Mendellian dominant
done
clear
View Answer play_arrow
PMNL stand for:
A)
Polymorpho nucleus lymphocyles
done
clear
B)
Plymorpho nucleus leukocytes
done
clear
C)
Polymorpho nuclease leukocytes
done
clear
D)
Polymorpho nuclear lymphocytes
done
clear
View Answer play_arrow
Severo Ochoa enzyme was helpful in:
A)
Polymerising mRNA
done
clear
B)
Enzymatic synthesis of RNA
done
clear
C)
Polymerise nucleotide
done
clear
D)
To checking the sequences of RNA
done
clear
View Answer play_arrow
The third tank where sludge is transferred during biological treatment is:
A)
Aeration tank
done
clear
B)
Filtration tank
done
clear
C)
Settling tank
done
clear
D)
none of these
done
clear
View Answer play_arrow
Find the wrong relation:
A)
\[{{\operatorname{N}}_{t+1}}={{N}_{t}}\left( B+I \right)-\left( D+E \right)\]
done
clear
B)
\[{{\operatorname{N}}_{t+1}}={{N}_{t}}+\left( B-D \right)-\left( I+E \right)\]
done
clear
C)
\[{{\operatorname{N}}_{t+1}}={{N}_{t}}+\left( B-E \right)+\left( I+D \right)\]
done
clear
D)
\[{{\operatorname{N}}_{t+1}}={{N}_{t}}\left( B+I \right)-\left( I-D \right)\]
done
clear
View Answer play_arrow

 is:
is: