If dimensions of length are expressed as \[{{\operatorname{G}}^{x}}{{C}^{y}}{{Y}^{z}}\]where G, C and h are universal gravitional constants, speed of light and Planck's constant respectively then:
A)
\[\operatorname{x}=\frac{1}{2}, y=\frac{1}{2}\]
done
clear
B)
\[\operatorname{x}=\frac{1}{2}, z=\frac{1}{2}\]
done
clear
C)
\[y=\frac{3}{2}, z=\frac{1}{2}\]
done
clear
D)
\[y=\frac{1}{2}, z=\frac{3}{2}\]
done
clear
View Answer play_arrow
A heavy brass sphere is hung from a spiral and it executes vertical vibrations with period T. The sphere is now immersed in a non-viscous liquid with a density \[\frac{1}{10}\]th that of brass. When set into vertical vibrations with the sphere remaining inside liquid all the time, the time period will be:
A)
\[\sqrt{\frac{9}{10}}\]
done
clear
B)
\[\sqrt{\frac{10}{9}}T\]
done
clear
C)
\[\frac{9}{10}T\]
done
clear
D)
Unchanged
done
clear
View Answer play_arrow
The height y and distance x along the horizontal plane of a projectile on a certain planet are given\[\operatorname{y} = 8t - 5{{t}^{2}} \left( mtr \right) and x = 6t \left( mtr \right)\]. The acceleration d due to gravity is:
A)
\[10m/{{s}^{2}}\]
done
clear
B)
\[5m/{{s}^{2}}\]
done
clear
C)
\[20m/{{s}^{2}}\]
done
clear
D)
\[2.5m/{{s}^{2}}\]
done
clear
View Answer play_arrow
The length of a metal wire is \[{{\ell }_{2}}\]when the tension in it is \[{{\operatorname{T}}_{1}}\]and \[{{\ell }_{2}}\] when the tension in it is\[{{\operatorname{T}}_{2}}\]. The natural length of the wire:
A)
\[\frac{{{\ell }_{1}}+{{\ell }_{2}}}{2}\]
done
clear
B)
\[\frac{{{\ell }_{1}}{{\operatorname{T}}_{2}}+{{\operatorname{T}}_{1}}{{\ell }_{2}}}{2}\]
done
clear
C)
\[\frac{{{\ell }_{1}}{{\operatorname{T}}_{2}}-{{\operatorname{T}}_{1}}{{\ell }_{2}}}{{{\operatorname{T}}_{1}}+{{\operatorname{T}}_{2}}}\]
done
clear
D)
\[\sqrt{{{\ell }_{1}}{{\ell }_{2}}}\]
done
clear
View Answer play_arrow
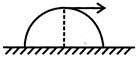
A small disc is placed at the top of a hemisphere of radius R as shown in figure. What is the smallest velocity that should be given to the disc for it to leave the hemisphere and not slide down it?
A)
\[\operatorname{v}=\sqrt{2gR}\]
done
clear
B)
\[\operatorname{v}=\sqrt{gR}\]
done
clear
C)
\[\operatorname{v}=2\sqrt{gR}\]
done
clear
D)
\[\operatorname{v}=\sqrt{\frac{gR}{2}}\]
done
clear
View Answer play_arrow
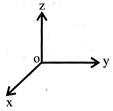
A force of \[\left( -F \right)\hat{k}\]acts on 0, the origin of the coordinate system. The torque about the point (1, -1) is:
A)
\[\operatorname{F}\left( \hat{i}+\hat{J} \right)\]
done
clear
B)
\[-\operatorname{F}\left( \hat{i}-\hat{j} \right)\]
done
clear
C)
\[\operatorname{F}\left( \hat{i}-\hat{j} \right)\]
done
clear
D)
\[-\operatorname{F}\left( \hat{i}+\hat{j} \right)\]
done
clear
View Answer play_arrow
In the given circuit for ideal diode, the current through the battery is:
A)
0.5A
done
clear
B)
1.5A
done
clear
C)
1A
Voltage = 10V Diode is forward biased \[{{\operatorname{R}}_{eq}}=5\Omega +5\Omega =10\Omega \] \[\operatorname{I}=\frac{10}{10}1A\Rightarrow \frac{10}{10}=1A\]
done
clear
D)
2A
done
clear
View Answer play_arrow
A metallic sphere of mass M falls through glycerine with a terminal velocity v. If the we drop a ball of mass 8M of same metal into a column of glycerine, the terminal velocity of the ball will be:
A)
2v
done
clear
B)
4v
done
clear
C)
8v
done
clear
D)
16v
done
clear
View Answer play_arrow
Light of two different frequencies whose photons have energy leV and 2eV respectively, illuminate a metallic surface whose work function is 0.5eV successfully. Ratio of maximum speeds of emitted electrons will be:
A)
1:2
done
clear
B)
1:1
done
clear
C)
1: 5
done
clear
D)
1:4
done
clear
View Answer play_arrow
A bar magnet is placed in the position of stable equilibrium in a uniform magnetic field of induction B. If is rotated through an angle\[180{}^\circ \], then the work is:
A)
MB
done
clear
B)
2MB
done
clear
C)
\[\frac{MB}{2}\]
done
clear
D)
Zero
done
clear
View Answer play_arrow
An electron of a stationary hy4rogen atom passes from the 5th energy level to the ground level. The velocity that the atom acquired as a result of photon emission will be:
A)
\[\frac{24Rh}{25m}\]
done
clear
B)
\[\frac{25hR}{24m}\]
done
clear
C)
\[\frac{25m}{24hR}\]
done
clear
D)
\[\frac{24m}{25hR}\]
done
clear
View Answer play_arrow
Two particles A and B having equal charges +6C, after being accelerated through the same potential difference, enter a region of uniform magnetic field and describe circular paths of radii 2cm and 3cm, respectively. The ratio of mass of A to that of B is:
A)
\[\frac{4}{9}\]
done
clear
B)
\[\frac{9}{5}\]
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\frac{1}{3}\]
done
clear
View Answer play_arrow
Blood is flowing at the rate of 200cmys in a capillary of cross sectional area 0.5m2. The velocity of flow in mm/s is:
A)
0.1
done
clear
B)
0.2
done
clear
C)
0.3
done
clear
D)
0.4
done
clear
View Answer play_arrow
The threshold wavelength for a metal having work function\[{{\operatorname{W}}_{o}} is {{\lambda }_{o}}\]. What is the threshold wavelength for a metal whose work function\[{}^{{{\operatorname{W}}_{o}}}/{}_{2}\]?
A)
\[4{{\lambda }_{\operatorname{o}}}\]
done
clear
B)
\[2{{\lambda }_{\operatorname{o}}}\]
done
clear
C)
\[\frac{{{\lambda }_{\operatorname{o}}}}{2}\]
done
clear
D)
\[\frac{{{\lambda }_{\operatorname{o}}}}{4}\]
done
clear
View Answer play_arrow
A string of length L and force constant K is stretched to obtain extension I. It is further stretched to obtain extension\[{{l}_{1}}\]. The work done in second stretching is:
A)
\[\frac{1}{2}k{{l}_{1}}\left( 2l+{{l}_{1}} \right)\]
done
clear
B)
\[\frac{1}{2}k{{l}_{1}}^{2}\]
done
clear
C)
\[\frac{1}{2}k\left( {{l}^{2}}+{{l}_{1}}^{2} \right)\]
done
clear
D)
\[\frac{1}{2}k\left( {{l}^{2}}_{1}-{{l}_{1}}^{2} \right)\]
done
clear
View Answer play_arrow
A coil having 500 turns of square shape each of side 10 cm is placed normal to a magnetic field which is increasing at IT/s. The induced emf is:
A)
0.1V
done
clear
B)
0.5V
done
clear
C)
1V
done
clear
D)
5V
done
clear
View Answer play_arrow
The output Y of the logic circuit shown in figure is best represented as ;
A)
\[\overline{\operatorname{A}}+\overline{B-C}\]
done
clear
B)
\[\operatorname{A}+\overline{B}.C\]
done
clear
C)
\[\overline{\operatorname{A}+B.C}\]
done
clear
D)
\[\overline{\operatorname{A}+\overline{B}.C}\]
done
clear
View Answer play_arrow
The ratio of acceleration due to gravity at a height 3R above earth's surface to the acceleration due to the gravity on the surface of the earth is:
A)
\[\frac{l}{9}\]
done
clear
B)
\[\frac{\operatorname{l}}{4}\]
done
clear
C)
\[\frac{\operatorname{l}}{16}\]
done
clear
D)
\[\frac{\operatorname{l}}{3}\]
done
clear
View Answer play_arrow
A longitudinal waves is represented by \[\operatorname{x} = {{x}_{o}}Sin2\pi \left( nt-\frac{x}{\lambda } \right)\]. The maximum particle velocity will be four times the wave's velocity if:
A)
\[\lambda =\frac{\pi {{x}_{o}}}{4}\]
done
clear
B)
\[\lambda =2\pi {{x}_{o}}\]
done
clear
C)
\[\lambda =\frac{\pi {{x}_{o}}}{2}\]
done
clear
D)
\[\lambda =4\pi {{x}_{o}}\]
done
clear
View Answer play_arrow
A 2V battery, a \[990\Omega \] resistor and a potentiometer of 2m length, all are connected in series the resistance of potentiometer wire is 1\[10\Omega \], then the potential gradient of potentiometer wire is:
A)
0.05V/m
done
clear
B)
0.5V/m
done
clear
C)
0.01V/m
done
clear
D)
0.1V/m
done
clear
View Answer play_arrow
The half-life of a radioactive substance is 30 minutes. The tim taken between 40% decay and 85% decay of the same radioactive substance is:
A)
60
done
clear
B)
15
done
clear
C)
30
done
clear
D)
45
done
clear
View Answer play_arrow
A cylinder of fixed capacity (of 44.8L) contains 2 moles of He gas at STF. What is the amount of heat needed to raise the temperature of the gas in the cylinder by \[20{}^\circ C \left[ R = 8.31 J/molK \right]\]
A)
996J
done
clear
B)
831J
done
clear
C)
499J
done
clear
D)
374J
done
clear
View Answer play_arrow
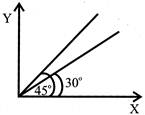
The displacement time graph of two moving particles make angles of \[30{}^\circ and 45{}^\circ \] with the X - axis. The ratio of their velocities is:
A)
\[\frac{\sqrt{3}}{2}\]
done
clear
B)
1
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\frac{1}{\sqrt{3}}\]
done
clear
View Answer play_arrow
A square of side 3cm is located at a distance 25 cm from a concave mirror of focal length 10cm. The centre of square is at the axis of the mirror and the plane is normal to axis of mirror. The area enclosed by the image of the square is:
A)
\[4 c{{m}^{2}}\]
done
clear
B)
\[6 c{{m}^{2}}\]
done
clear
C)
\[16 c{{m}^{2}}\]
done
clear
D)
\[36 c{{m}^{2}}\]
done
clear
View Answer play_arrow
At what height from the surf ace of earth the gravitational potential and the value of g are\[-5.4\times 1{{0}^{7}}\,J/k{{g}^{2}}\,\,and\,\,6m/{{s}^{2}}\]respectively take the radius of earth as 6400 km:
A)
2600 km
done
clear
B)
1600 km
done
clear
C)
1400 km
done
clear
D)
2000 km
done
clear
View Answer play_arrow
A bread gives a boy of mass 40kg an energy of 21kJ. If the efficiency is 28%, then the height that can be climbed by him using this energy, is:
A)
22.5m
done
clear
B)
15m
done
clear
C)
10m
done
clear
D)
5m
done
clear
View Answer play_arrow
A stretched wire of length 110cm is divided into three segments whose frequencies are in the ratio 1:2: 3. Their length must be:
A)
20cm, 30cm, 60cm
done
clear
B)
60cm, 30cm, 20cm
done
clear
C)
60cm, 20cm, 30cm
done
clear
D)
30cm, 60cm, 20cm
done
clear
View Answer play_arrow
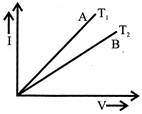
The current - voltage graph for a given metallic wire at two different temperatures \[{{\operatorname{T}}_{1}} and {{T}_{2}}\]is shown in the figure. The temperature \[{{\operatorname{T}}_{1}} and {{T}_{2}}\] are related as:
A)
\[{{\operatorname{T}}_{1}} >{{T}_{2}}\]
done
clear
B)
\[{{\operatorname{T}}_{1}} <{{T}_{2}}\]
done
clear
C)
\[{{\operatorname{T}}_{1}} ={{T}_{2}}\]
done
clear
D)
\[{{\operatorname{T}}_{1}} >\,\,2{{T}_{2}}\]
done
clear
View Answer play_arrow
A man is 45m behind the bus, when the bus start accelerating from rest with acceleration\[2.5m/{{s}^{2}}\]. With what minimum velocity should the man start running to catch the bus?
A)
12m/s
done
clear
B)
14m/s
done
clear
C)
15m/s
done
clear
D)
16m/s
done
clear
View Answer play_arrow
A perfect gas is found to obey the relation \[{{\operatorname{pV}}^{{}^{3}/{}_{2}}}=\]constant, during an adiabatic pro- cess, if such a gas initially at a temperature T, is compressed to half of its initial volume, then its final temperature will be:
A)
2T
done
clear
B)
4T
done
clear
C)
\[\sqrt{2}T\]
done
clear
D)
\[2\sqrt{2}T\]
done
clear
View Answer play_arrow
In young's double slit interference experiment, using two coherent waves of different amplitudes, the intensities ratio between bright and dark fringes is 3. Then, the value of the ratio of the amplitudes of the wave that arrive there is:
A)
\[\frac{\sqrt{3}+1}{\sqrt{3}-1}\]
done
clear
B)
\[\frac{\sqrt{3}-1}{\sqrt{3}+1}\]
done
clear
C)
\[\sqrt{3}:1\]
done
clear
D)
\[1:\sqrt{3}\]
done
clear
View Answer play_arrow
In young's double slit experiment, the ratio of maximum and minimum intensity in the fringe system is 9:1. The ratio of amplitudes of coherent sources is:
A)
9 :1
done
clear
B)
3 :1
done
clear
C)
2:1
done
clear
D)
1:1
done
clear
View Answer play_arrow
Two cells with the same emf E and different internal resistance \[{{\operatorname{r}}_{1}} and {{r}_{2}},\]are connected in series to an external resistance R. The value of R so that the pd across the first cell is zero, is:
A)
\[\sqrt{{{\operatorname{r}}_{1}}\,\,{{r}_{2}}}\]
done
clear
B)
\[{{\operatorname{r}}_{1}}+\,{{r}_{2}}\]
done
clear
C)
\[{{\operatorname{r}}_{1}}-\,{{r}_{2}}\]
done
clear
D)
\[\frac{{{\operatorname{r}}_{1}}+\,{{r}_{2}}}{2}\]
done
clear
View Answer play_arrow
Light enters at an angle of incidence in a transparent rod of refractive index of the material of the rod. The light once entered into it will not leave it through its lateral face what, so ever be the angle of incidence:
A)
\[\operatorname{n}>\sqrt{2}\]
done
clear
B)
n=1
done
clear
C)
n = 1.1
done
clear
D)
n=1.3
done
clear
View Answer play_arrow
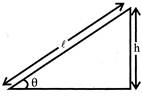
A smooth inclined plane is inclined at an angle with the horizontal. A body starts from rest and slides down the inclined surface, then the time taken by the body to reach the bottom is:
A)
\[\sqrt{\frac{2h}{g}}\]
done
clear
B)
\[\sqrt{\frac{2\ell }{g}}\]
done
clear
C)
\[\frac{1}{\sin \theta }\sqrt{\frac{2h}{g}}\]
done
clear
D)
\[\sin \theta \sqrt{\frac{2h}{g}}\]
done
clear
View Answer play_arrow
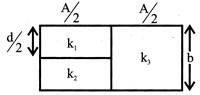
In the figure a capacitor is filled with dielectrics. The resultant capacitance is:
A)
\[\frac{2A{{\varepsilon }_{o}}}{\operatorname{d}}\left[ \frac{1}{{{k}_{1}}}+\frac{1}{{{k}_{2}}}+\frac{1}{{{k}_{3}}} \right]\]
done
clear
B)
\[\frac{A{{\varepsilon }_{o}}}{\operatorname{d}}\left[ \frac{1}{{{k}_{1}}}+\frac{1}{{{k}_{2}}}+\frac{1}{{{k}_{3}}} \right]\]
done
clear
C)
\[\frac{2A{{\varepsilon }_{o}}}{\operatorname{d}}\left[ {{k}_{1}}+{{k}_{2}}+{{k}_{3}} \right]\]
done
clear
D)
\[\frac{A{{\varepsilon }_{o}}}{\operatorname{d}}\left[ \frac{{{k}_{3}}}{2}+\frac{{{k}_{1}}{{k}_{2}}}{{{k}_{1}}+{{k}_{3}}} \right]\]
done
clear
View Answer play_arrow
If a circuit made up of a resistance 10, and inductance 0.01H, and alternating emf 200V at 50Hz is connected, then the phase difference between the current and the emf in the circuit is:
A)
\[{{\tan }^{-1}} \left( \pi \right)\]
done
clear
B)
\[{{\tan }^{-1}} \left( \frac{\pi }{2} \right)\]
done
clear
C)
\[{{\tan }^{-1}} \left( \frac{\pi }{4} \right)\]
done
clear
D)
\[{{\tan }^{-1}} \left( \frac{\pi }{3} \right)\]
done
clear
View Answer play_arrow
The moment of inertia of a body about a given axis is\[1.2kg{{m}^{2}}\]. Initially the body is at rest. In order to produce a rotational kinetic energy of 1500J, an angular acceleration of \[25rad/se{{c}^{2}}\]must be applied about that axis for a duration of:
A)
4s
done
clear
B)
2s
done
clear
C)
8s
done
clear
D)
10s
done
clear
View Answer play_arrow
A ray of light is incident at an angle of incidence Y on one face of a prism of angle A (assumed to be small) and emerges normally from the opposite face. If the refractive index of the prism is u, the angle of incidence \[\mu \], is nearly equal to:
A)
\[\mu \operatorname{A}\]
done
clear
B)
\[\frac{\mu \operatorname{A}}{2}\]
done
clear
C)
\[\frac{\operatorname{A}}{\mu }\]
done
clear
D)
\[\frac{\operatorname{A}}{2\mu }\]
done
clear
View Answer play_arrow
Turn ratio in a step-up transformer is 1: 2, if a Leclanche cell of 1.5V is connected across the input, then the voltage across the output will be:
A)
0.1V
done
clear
B)
1.5V
done
clear
C)
0.75V
done
clear
D)
zero
done
clear
View Answer play_arrow
A block of mass 200kg is being pulled up by men on an inclined plane at angle of \[45{}^\circ \] as shown. The coefficient of static friction is 0.5. Each man can only apply a maximum force of 500N. Calculation the number of men required for the block to just start moving up the plane:
A)
10
done
clear
B)
15
done
clear
C)
5
done
clear
D)
3
done
clear
View Answer play_arrow
Light of frequency \[v\] falls on material of threshold frequency\[{{v}_{\operatorname{o}}}\]. Maximum kinetic energy of emitted electron is proportional to:
A)
\[v-{{v}_{o}}\]
done
clear
B)
\[v\]
done
clear
C)
\[\sqrt{v-{{v}_{o}}}\]
done
clear
D)
\[{{v}_{o}}\]
done
clear
View Answer play_arrow
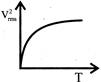
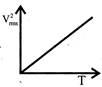
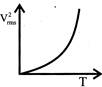
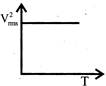
The curve between absolute temperature & \[{{\operatorname{V}}^{2}}_{rms}\]is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A car of mass 1000kg moves on a circular track of radius 20m. If the coefficient of friction is 0.64, then the maximum velocity with which the car move is:
A)
22.4 m/s
done
clear
B)
5.6 m/s
done
clear
C)
11.2m/s
done
clear
D)
6.5 m/s
done
clear
View Answer play_arrow
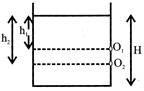
There are two holes\[{{\operatorname{O}}_{1}} and {{O}_{2}}\]in a tank of height H. The water emerging from \[{{\operatorname{O}}_{1}} and {{O}_{2}}\] strikes the ground at the same points, as shown in figure. Then:
A)
\[\operatorname{H}={{h}_{1}}+{{h}_{2}}\]
done
clear
B)
\[\operatorname{H}={{h}_{1}}-{{h}_{2}}\]
done
clear
C)
\[\operatorname{H}=\sqrt{{{h}_{1}}{{h}_{2}}}\]
done
clear
D)
None
done
clear
View Answer play_arrow
The specific heat of a metal is 0.16. Its approximate atomic weight would be:
A)
32
done
clear
B)
16
done
clear
C)
40
done
clear
D)
64
done
clear
View Answer play_arrow
Depletion of ozone layer causes:
A)
Blood cancer
done
clear
B)
Lung cancer
done
clear
C)
Skin cancer
done
clear
D)
Breast cancer
done
clear
View Answer play_arrow
Rutherford's scattering experiment is related to the size of the:
A)
Nucleus
done
clear
B)
Atom
done
clear
C)
Electron
done
clear
D)
Neutron
done
clear
View Answer play_arrow
Which of the following is most electronegative?
A)
Carbon
done
clear
B)
Silicon
done
clear
C)
Lead
done
clear
D)
Tin
done
clear
View Answer play_arrow
Which of the following has maximum unpaired d-electrons?
A)
\[{{\operatorname{Zn}}^{2+}}\]
done
clear
B)
\[{{\operatorname{Fe}}^{2+}}\]
done
clear
C)
\[{{\operatorname{Ni}}^{2+}}\]
done
clear
D)
\[{{\operatorname{Cu}}^{2+}}\]
done
clear
View Answer play_arrow
Paramagnetic species are:
A)
\[{{\operatorname{O}}^{+}}_{2}\]
done
clear
B)
\[{{\operatorname{O}}^{-}}_{2}\]
done
clear
C)
\[{{\operatorname{N}}^{+}}_{2}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
The density of air is 0.00130g/ml. The vapour density of air will be:
A)
0.00065
done
clear
B)
0.65
done
clear
C)
14.4816
done
clear
D)
14.56
done
clear
View Answer play_arrow
The energy of hydrogen bond is of the order of:
A)
\[4KJ mo{{l}^{-1}}\]
done
clear
B)
\[40KJ mo{{l}^{-1}}\]
done
clear
C)
\[400KJmo{{l}^{-1}}\]
done
clear
D)
\[4000 KJ mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
Given that
\[\mathbf{C+O2}\to \mathbf{C}{{\mathbf{O}}_{\mathbf{2}}}~\,\,\,\,\,\,\,\,\,,\Delta \mathbf{H}{}^\circ =-\mathbf{xKJ}\] \[\mathbf{2CO}+{{\mathbf{O}}_{\mathbf{2}}}\to \mathbf{2C}{{\mathbf{O}}_{\mathbf{2}}}\mathbf{ }\,\,\,\,\mathbf{,}\Delta \mathbf{H{}^\circ =}-\mathbf{yKJ}\] The enthalpy of carbon monoxide will be:
A)
\[\operatorname{y}-2x\]
done
clear
B)
\[\frac{2x-\operatorname{y}}{2}\]
done
clear
C)
\[\frac{\operatorname{y}-2x}{2}\]
done
clear
D)
\[2x-y\]
done
clear
View Answer play_arrow
The number and type of bonds between two carbon atoms in calcium carbide are:
A)
One sigma, one pi
done
clear
B)
One sigma, two pi
done
clear
C)
Two sigma, one pi
done
clear
D)
Two sigma, two pi
done
clear
View Answer play_arrow
Of the following which change will shift the reaction towards the product? \[{{\operatorname{I}}_{2}}\left( g \right)\rightleftharpoons 2I\left( g \right), \Delta {{H}^{o}}_{r}\left( 298k \right)=+150KJ\]
A)
Increase in concentration if I
done
clear
B)
Decrease in concentration of \[{{\operatorname{I}}_{2}}\]
done
clear
C)
Increase in temperature
done
clear
D)
Increase in total pressure
done
clear
View Answer play_arrow
Which is the about\[{{\operatorname{N}}_{3}}^{-}\]:
A)
It has a non - linear structure
done
clear
B)
It is called pseudohalogen
done
clear
C)
The formal oxidation state of nitrogen in this anion is -1
done
clear
D)
It is isoelectronic with \[{{\operatorname{NO}}_{2}}\]
done
clear
View Answer play_arrow
Which one is true for any diprotic acid\[{{\operatorname{H}}_{2}}X\]?
A)
\[{{\operatorname{K}}_{{{a}_{2}}}}>{{K}_{{{a}_{1}}}}\]
done
clear
B)
\[{{\operatorname{K}}_{{{a}_{1}}}}>{{K}_{{{a}_{2}}}}\]
done
clear
C)
\[{{\operatorname{K}}_{{{a}_{2}}}}>\frac{1}{{{K}_{{{a}_{1}}}}}\]
done
clear
D)
\[{{\operatorname{K}}_{{{a}_{2}}}}={{K}_{{{a}_{1}}}}\]
done
clear
View Answer play_arrow
Which of the following is a redox reaction?
A)
\[\operatorname{NaCl} + KN{{O}_{3}} \to NaN{{O}_{3}} + KCl\]
done
clear
B)
\[{{\operatorname{CaCO}}_{2}}+ 2HCl \to CaC{{I}_{2}} + {{H}_{2}}{{C}_{2}}{{O}_{4}}\]
done
clear
C)
\[\operatorname{Mg} {{\left( OH \right)}_{2}} + 2N{{H}_{4}}Cl \to MgC{{l}_{2}} + 2N{{H}_{4}}OH\]
done
clear
D)
\[\operatorname{Zn} +2AgCN \to 2Ag + Zn {{\left( CN \right)}_{2}}\]
done
clear
View Answer play_arrow
The high density of water as compared to ice is due to:
A)
Hydrogen bonding interactions
done
clear
B)
Dipole - Dipole interactions
done
clear
C)
Dipole - induced dipole interactions
done
clear
D)
Induced dipole induced dipole interactions
done
clear
View Answer play_arrow
Gypsum on heating to 390 k gives:
A)
\[{{\operatorname{CaSO}}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[{{\operatorname{CaSO}}_{4}}\]
done
clear
C)
\[{{\operatorname{CaSO}}_{4}}.1/2{{H}_{2}}O\]
done
clear
D)
\[{{\operatorname{SC}}_{3}}\,and\,\,CaO\]
done
clear
View Answer play_arrow
The IUPAC name of
is:
A)
3-methylcyclohexane
done
clear
B)
1-methylcyclohex-2-ene
done
clear
C)
6-methylcyclohexane
done
clear
D)
l-methylcyclohex-5-ene
done
clear
View Answer play_arrow
Which of the following is only acidic in nature:
A)
\[\operatorname{Be}{{\left( OH \right)}_{2}}\]
done
clear
B)
\[\operatorname{Mg}{{\left( OH \right)}_{2}}\]
done
clear
C)
\[\operatorname{B}{{\left( OH \right)}_{3}}\]
done
clear
D)
\[\operatorname{Al}{{\left( OH \right)}_{3}}\]
done
clear
View Answer play_arrow
An organic compound which produces a bluish green colored flame on heating in presence of copper is:
A)
Chlorobenzene
done
clear
B)
Benz aldehyde
done
clear
C)
Aniline
done
clear
D)
Benzoic acid
done
clear
View Answer play_arrow
The o/p-directing group among the following is:
A)
COOH
done
clear
B)
CN
done
clear
C)
\[{{\operatorname{COCH}}_{3}}\]
done
clear
D)
\[{{\operatorname{NHCOCH}}_{3}}\]
done
clear
View Answer play_arrow
Inductive effect involves:
A)
Displacement of \[\sigma -electrons\]
done
clear
B)
Delocalisation of \[\pi -electrons\]
done
clear
C)
Delocalisation of \[\sigma -electrons\]
done
clear
D)
Displacement of \[\pi -electrons\]
done
clear
View Answer play_arrow
With which one of the following elements silicon should be doped so as to give p-type of semiconductor?
A)
Germanium
done
clear
B)
Arsenic
done
clear
C)
Selenium
done
clear
D)
Boron
done
clear
View Answer play_arrow
A liquid is in equilibrium with its vapour as its boiling point on the average, the molecules in the two phases have equal:
A)
Intermolecular forces
done
clear
B)
Potential energy
done
clear
C)
Total energy
done
clear
D)
Kinetic energy
done
clear
View Answer play_arrow
Empty space in ccp lattice is:
A)
26%
done
clear
B)
45%
done
clear
C)
90%
done
clear
D)
30%
done
clear
View Answer play_arrow
If two substances A and B have \[{{\operatorname{P}}^{o}}_{A} : {{P}^{o}}_{B}\]and have mole fraction in solution 1 : 2, then mole fraction of A in vapours is:
A)
0.33
done
clear
B)
0.25
done
clear
C)
0.52
done
clear
D)
0.2
done
clear
View Answer play_arrow
Which of the following reactions ends in finite time?
A)
0 order
done
clear
B)
\[{{1}^{st}} order\]
done
clear
C)
\[{{2}^{nd}} order\]
done
clear
D)
\[{{3}^{rd}} order\]
done
clear
View Answer play_arrow
The volume of \[{{\operatorname{H}}_{2}}\]gas at NTP obtained by passing 4 amperes through acidified \[{{\operatorname{H}}_{2}}O\]for 30 minutes is:
A)
0.0836 L
done
clear
B)
0.0432 L
done
clear
C)
0.1672 L
done
clear
D)
0.836 L
done
clear
View Answer play_arrow
1/[A] vs. time is a straight line. The order of the reactions is:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
0
done
clear
View Answer play_arrow
Which of the following forms acid halides?
A)
HF
done
clear
B)
HCl
done
clear
C)
HBr
done
clear
D)
HI
done
clear
View Answer play_arrow
In coagulating the colloidal solution of A, 83 which has the minimum coagulating value?
A)
\[\operatorname{NaCl}\]
done
clear
B)
\[\operatorname{KCl}\]
done
clear
C)
\[{{\operatorname{BaCl}}_{2}}\]
done
clear
D)
\[{{\operatorname{AlCl}}_{3}}\]
done
clear
View Answer play_arrow
Among following molecules, \[{{\operatorname{XeO}}_{3}}, Xe{{O}_{4}}, Xe{{F}_{6}}\] those having same number of lone pairs on Xe are:
A)
\[{{\operatorname{XeO}}_{3}},Xe{{O}_{4}}\]
done
clear
B)
\[{{\operatorname{XeO}}_{3}},Xe{{F}_{6}}\]
done
clear
C)
\[{{\operatorname{XeO}}_{4}},XeF\]
done
clear
D)
All
done
clear
View Answer play_arrow
Which metal has a greater tendency to form metal oxide?
A)
Al
done
clear
B)
Ca
done
clear
C)
Cr
done
clear
D)
Fe
done
clear
View Answer play_arrow
In the reaction
\[\left( N{{H}_{4}} \right)C{{r}_{2}}{{O}_{2}} \xrightarrow{\Delta }{{N}_{2}}+{{H}_{2}}O+C{{r}_{2}}{{O}_{0}}\] The coefficient of \[{{\operatorname{H}}_{2}}O\]is:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
The colourless species is:
A)
\[{{\operatorname{VCl}}_{3}}\]
done
clear
B)
\[{{\operatorname{VOSO}}_{4}}\]
done
clear
C)
\[{{\operatorname{Na}}_{3}}V{{0}_{4}}\]
done
clear
D)
\[\left[ v{{\left( {{H}_{2}}O \right)}_{6}} S{{O}_{4}} \right].{{H}_{2}}O\]
done
clear
View Answer play_arrow
The formula of ferrocene is:
A)
\[{{\left[ Fe{{\left( CN \right)}_{6}} \right]}^{3-}}\]
done
clear
B)
\[{{\left[ Fe{{\left( CN \right)}_{6}} \right]}^{4-}}\]
done
clear
C)
\[\left[ Fe{{\left( CN \right)}_{5}} \right]\]
done
clear
D)
\[\left[ {{\left( {{C}_{5}}{{H}_{5}} \right)}_{2}}Fe \right]\]
done
clear
View Answer play_arrow
The reagent used for separation of acetaldehyde and acetophenone is:
A)
\[{{\operatorname{NaHSO}}_{3}}\]
done
clear
B)
\[{{\operatorname{C}}_{6}}{{H}_{5}}NHN{{H}_{2}}\]
done
clear
C)
\[{{\operatorname{NH}}_{2}}OH\]
done
clear
D)
\[\operatorname{NaOH}-{{I}_{2}}\]
done
clear
View Answer play_arrow
The major product formed is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Acetone is mixed with bleaching power to give:
A)
Chloroform
done
clear
B)
Acetaldehyde
done
clear
C)
Ethanol
done
clear
D)
Phosgene
done
clear
View Answer play_arrow
Which of the following enzymes converts glucose into ethyl alcohol?
A)
Diastase
done
clear
B)
Invertase
done
clear
C)
Maltase
done
clear
D)
Zymase
done
clear
View Answer play_arrow
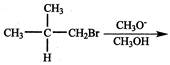
\[\xrightarrow[2.\,\,CuCN/{{H}_{3}}{{O}^{+}}]{1.\,\,NaN{{O}_{2}}/HCl}A\]A is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The human body does not produce:
A)
Enzymes
done
clear
B)
DNA
done
clear
C)
Vitamins
done
clear
D)
Hormones
done
clear
View Answer play_arrow
Which one is neither meant for the synthesis nor for separation of amines?
A)
Hinsberg method
done
clear
B)
Hofmann method
done
clear
C)
Wurtz reaction
done
clear
D)
Curtius reaction W
done
clear
View Answer play_arrow
hich of the following is used in paints?
A)
Terylene
done
clear
B)
Nylon
done
clear
C)
Glyptal
done
clear
D)
Chloroprene
done
clear
View Answer play_arrow
Tincture iodine is:
A)
Aqueous solution of I
done
clear
B)
Solution of \[{{\operatorname{I}}_{2}}\] in aqueous KI
done
clear
C)
Alcoholic solution of \[{{\operatorname{I}}_{2}}\]
done
clear
D)
Aqueous solution of KI
done
clear
View Answer play_arrow
The ion which is not tetrahedral in shape is:
A)
\[{{\operatorname{BF}}^{-}}_{4}\]
done
clear
B)
\[{{\operatorname{NH}}^{+}}_{4}\]
done
clear
C)
\[{{\left[ \operatorname{Cu}{{\left( N{{H}_{3}} \right)}_{4}} \right]}^{2+}}\]
done
clear
D)
\[{{\operatorname{NiCl}}^{2-}}_{4}\]
done
clear
View Answer play_arrow
Human eggs are:
A)
Mesolecithel
done
clear
B)
Microlecithel
done
clear
C)
Macrolecithel
done
clear
D)
Alecithel
done
clear
View Answer play_arrow
In five kingdom system, the main basis of classification is:
A)
Structure of all wall
done
clear
B)
Nutrition
done
clear
C)
Structure of nucleus
done
clear
D)
Asexual reproduction
done
clear
View Answer play_arrow
The dominant second trophic level, in a lake ecosysterms is:
A)
Benthos
done
clear
B)
Plankton
done
clear
C)
Zooplankton
done
clear
D)
Phytoplankton
done
clear
View Answer play_arrow
Histamine secreating cells are found in:
A)
Connective tissue
done
clear
B)
Nervous tissue
done
clear
C)
Lungs
done
clear
D)
Muscular tissue
done
clear
View Answer play_arrow
Which animal has the cylindrical form:
A)
Physalia
done
clear
B)
Admsia
done
clear
C)
Hydra
done
clear
D)
b & c
done
clear
View Answer play_arrow
Probiotics are:
A)
Cancer inducing microbe
done
clear
B)
Kind of food allergens
done
clear
C)
Live microbial food supplement
done
clear
D)
Safe antibiotics
done
clear
View Answer play_arrow
Inside the body, blood does not coagulate due to the presence of:
A)
Fibrin
done
clear
B)
Heparin
done
clear
C)
Haemoglobin
done
clear
D)
Thromoblastin
done
clear
View Answer play_arrow
The smallest unit of genetic material which when mutated produces a phenotypic effect is:
A)
Recon
done
clear
B)
Muton
done
clear
C)
Gene
done
clear
D)
Nucleic acid
done
clear
View Answer play_arrow
The first stable compound in Hatch and Slack cycle is:
A)
Fyruvic acid
done
clear
B)
Malic acid
done
clear
C)
Citric acid
done
clear
D)
Oxalo acetic acid
done
clear
View Answer play_arrow
Blastopore is the pore of:
A)
Coelom
done
clear
B)
Archentron
done
clear
C)
Blastocoel
done
clear
D)
Alimentary canal
done
clear
View Answer play_arrow
Collagen is:
A)
Lipid
done
clear
B)
Fibrous protein
done
clear
C)
Globular protein
done
clear
D)
Carbohydrates
done
clear
View Answer play_arrow
Haversians canals occur in:
A)
Humerus
done
clear
B)
Pubis
done
clear
C)
Scapula
done
clear
D)
Girdle
done
clear
View Answer play_arrow
In which kingdom would you classify the archea and nitrogen - fixing organism:
A)
Protista
done
clear
B)
Fungi
done
clear
C)
Plantae
done
clear
D)
Monera
done
clear
View Answer play_arrow
Blastopore is the pore of:
A)
Quantum recharge
done
clear
B)
Respiratory coefficient
done
clear
C)
A temperature coefficient
done
clear
D)
PAR coefficient
done
clear
View Answer play_arrow
Competition is severe in a population that has a distribution which is:
A)
Random
done
clear
B)
Uniform
done
clear
C)
Irregular
done
clear
D)
Non-uniform
done
clear
View Answer play_arrow
What is true for cleavage?
A)
Size of embryo increase
done
clear
B)
Size of cells decrease
done
clear
C)
Size of cells increase
done
clear
D)
Size of embryo decrease
done
clear
View Answer play_arrow
Areolar connective tissue joins:
A)
Haemoglobin
done
clear
B)
Hirrdin
done
clear
C)
Myoglobin
done
clear
D)
Histamine
done
clear
View Answer play_arrow
The respiratory organ of arthropoda:
A)
Gill
done
clear
B)
Book gill
done
clear
C)
Tracheal system
done
clear
D)
All of given
done
clear
View Answer play_arrow
The phylogenetic system of classification was put forth by:
A)
Thephrastus
done
clear
B)
George bentham & Joseph Dalton Hooker
done
clear
C)
Carolus Linnaeus
done
clear
D)
Adolf engler and karl pranti
done
clear
View Answer play_arrow
Short sightedness is due to:
A)
Weaker muscles
done
clear
B)
Shifting of the iris
done
clear
C)
Elongation of eye ball
done
clear
D)
Weakening of the retina
done
clear
View Answer play_arrow
The term genome is used for:
A)
Haploid set of chromosomes
done
clear
B)
Diploid set of chromosomes
done
clear
C)
Polyploid set of gene
done
clear
D)
Triploid set of chromosomes
done
clear
View Answer play_arrow
Ceolom derived from blastocoels is knowns as:
A)
Schizocoel
done
clear
B)
Pseudocoelom
done
clear
C)
Haemocoel
done
clear
D)
Enterocoelom
done
clear
View Answer play_arrow
Which one of the following is surrounded by a callose wall?
A)
Male gamete
done
clear
B)
Pollen grain
done
clear
C)
Egg
done
clear
D)
Microspore mother cell
done
clear
View Answer play_arrow
In ecological succession from pioneer to dimax community, the biomass shell:
A)
No relation
done
clear
B)
Increase continuously
done
clear
C)
Decrease
done
clear
D)
Increase and them decrease
done
clear
View Answer play_arrow
Which of the following is not the property of genetic code:
A)
Non overlapping
done
clear
B)
Almost universal
done
clear
C)
Four stop codons
done
clear
D)
Redundant
done
clear
View Answer play_arrow
The life spam of sperm is:
A)
2 days
done
clear
B)
2-3days
done
clear
C)
1-3days
done
clear
D)
7 days
done
clear
View Answer play_arrow
Common test to identify genotype of hybrid is by:
A)
Crossing of \[{{\operatorname{F}}_{2}}\]progeny with female parent
done
clear
B)
Studying of sexual behaviour of\[{{\operatorname{F}}_{1}}\] progeny
done
clear
C)
Crossing of one \[{{\operatorname{F}}_{1}}\] progeny with male parent
done
clear
D)
Crossing of one\[{{\operatorname{F}}_{2}}\] progeny with female parent
done
clear
View Answer play_arrow
A colour blind person has difficulty in distinguishing b/w
A)
Black & Blue
done
clear
B)
Blue & Red
done
clear
C)
Green & Red
done
clear
D)
Green & Blue
done
clear
View Answer play_arrow
Molecular formula of chlorophyll a is:
A)
\[{{\operatorname{C}}_{55}}{{H}_{70}}{{O}_{5}}{{N}_{4}}Mg\]
done
clear
B)
\[{{\operatorname{C}}_{55}}{{H}_{72}}{{O}_{6}}{{N}_{4}}Mg\]
done
clear
C)
\[{{\operatorname{C}}_{55}}{{H}_{70}}{{O}_{6}}{{N}_{4}}Mg\]
done
clear
D)
\[{{\operatorname{C}}_{55}}{{H}_{72}}{{O}_{5}}{{N}_{4}}Mg\]
done
clear
View Answer play_arrow
Pro proofs are found in:
A)
Mango trees
done
clear
B)
Tamarind trees
done
clear
C)
Banyan trees
done
clear
D)
Neem trees
done
clear
View Answer play_arrow
Lamina propria is connected with:
A)
Acini
done
clear
B)
Liver
done
clear
C)
Graffian follicle
done
clear
D)
Intestine
done
clear
View Answer play_arrow
Who is regarded as father of virology:
A)
Twort
done
clear
B)
Iwanowsky
done
clear
C)
Roux
done
clear
D)
Stanley
done
clear
View Answer play_arrow
Which mammalian have adapted for fly?
A)
Bats
done
clear
B)
Fox
done
clear
C)
Ostrich
done
clear
D)
Penguin
done
clear
View Answer play_arrow
Which series end with the cohort Umbel-lales in Bentham & Hooker's system of classification?
A)
Heteromerae
done
clear
B)
Discipflorae
done
clear
C)
Thalami florae
done
clear
D)
Calyciflorae
done
clear
View Answer play_arrow
In an ecosystem which can show one way passage:
A)
Nitrogen
done
clear
B)
Carbon
done
clear
C)
Potassium
done
clear
D)
Free energy
done
clear
View Answer play_arrow
Which among the following is not a model organization in genetic analysis?
A)
The zebra fish
done
clear
B)
The plant Arabidopsis thaliana
done
clear
C)
The mouse deer
done
clear
D)
The nematode caenorhabditis elegans
done
clear
View Answer play_arrow
The limbless amphibians is:
A)
Tree frog
done
clear
B)
Toad
done
clear
C)
Rana
done
clear
D)
Ichthyophis
done
clear
View Answer play_arrow
The number of thoraicic vertebrate in man is:
A)
9
done
clear
B)
10
done
clear
C)
7
done
clear
D)
12
done
clear
View Answer play_arrow
The first modern theory of evolution was explained by.
A)
Charles Darwin
done
clear
B)
Lamarck
done
clear
C)
Mendel
done
clear
D)
Devries
done
clear
View Answer play_arrow
Clear alternation of generation is seen in:
A)
Ferns
done
clear
B)
Monkeys
done
clear
C)
Mucor
done
clear
D)
Viruses 22
done
clear
View Answer play_arrow
Basement membrane is made up of:
A)
No cell product of epithelial cell.
done
clear
B)
Epidermal cell only
done
clear
C)
Endodermal cell
done
clear
D)
Both b & c
done
clear
View Answer play_arrow
An example of hermaphrodite is:
A)
Frog
done
clear
B)
Fish
done
clear
C)
Earthworm
done
clear
D)
Hydra
done
clear
View Answer play_arrow
Which of following belong to basidiomyces as:
A)
Puffballs & Claviceps
done
clear
B)
Peziza & stink borns
done
clear
C)
Morchella & mushroom
done
clear
D)
Birds nest fungi & puffballs
done
clear
View Answer play_arrow
Classification of organisms based on evolutionary as well as genetic relationship is called:
A)
Phenetics
done
clear
B)
Numerical taxonomy
done
clear
C)
Biosystematics
done
clear
D)
Cladistics
done
clear
View Answer play_arrow
A system of growing plant with their roots sapplied with moisture in the air:
A)
Hydroponics
done
clear
B)
Aeroponics
done
clear
C)
Bionomics
done
clear
D)
Genomics
done
clear
View Answer play_arrow
Collar cells are found in:
A)
Sponges
done
clear
B)
Roundworm
done
clear
C)
Earthworm
done
clear
D)
Spider
done
clear
View Answer play_arrow
One of endangered species of Indian medicinal plant is that of:
A)
Ocimum
done
clear
B)
Garlic
done
clear
C)
Nepenthers
done
clear
D)
Podophyllum
done
clear
View Answer play_arrow
Identify odd combination:
A)
Sunderban - Bangal tigers
done
clear
B)
Periyar - Elephant
done
clear
C)
Rann of kutch - Wild ass
done
clear
D)
Dachigam national park - Snow leopard
done
clear
View Answer play_arrow
Corpus callosum is present in the brain of:
A)
Fishes
done
clear
B)
Mammals
done
clear
C)
Birds
done
clear
D)
Snails
done
clear
View Answer play_arrow
Vegetative propagation in sweet potato is by:
A)
Stem
done
clear
B)
Root
done
clear
C)
Leaf
done
clear
D)
None of these
done
clear
View Answer play_arrow
Bacteriophages were studied in detail by:
A)
Twort
done
clear
B)
Iwanowsky
done
clear
C)
Roux
done
clear
D)
Herelle
done
clear
View Answer play_arrow
Coral have a skeleton composed of:
A)
\[{{\operatorname{CaCC}}_{3}}\]
done
clear
B)
\[{{\operatorname{CaPO}}_{4}}\]
done
clear
C)
\[{{\operatorname{CaCI}}_{2}}\]
done
clear
D)
\[{{\operatorname{CaSiO}}_{2}}\]
done
clear
View Answer play_arrow
The concept of chemical evolution based on:
A)
Interaction of water, soil & air under intense heat
done
clear
B)
Effect of solar radiation on chemical
done
clear
C)
Origin of life by combination of chemical under suitable environment condition
done
clear
D)
Crystallization of chemical
done
clear
View Answer play_arrow
The sum total of the population of the same kind of organisms constitute:
A)
Colony
done
clear
B)
Genus
done
clear
C)
Species
done
clear
D)
Community
done
clear
View Answer play_arrow
Most marsupial and all monotreme species today are found in:
A)
Australia
done
clear
B)
Asia
done
clear
C)
Europe
done
clear
D)
America
done
clear
View Answer play_arrow
The normal temperature of human body on the Kelvin scale is:
A)
280
done
clear
B)
290
done
clear
C)
300
done
clear
D)
310
done
clear
View Answer play_arrow
The closure of lid of pitcher in Nepenthesis is:
A)
A turgor Movement
done
clear
B)
A parotonic Movement
done
clear
C)
A tropic Movement
done
clear
D)
An autonomic Movement
done
clear
View Answer play_arrow
Find the odd:
A)
Sea fan
done
clear
B)
Sea urchin
done
clear
C)
Sea lily
done
clear
D)
Sea cucumber
done
clear
View Answer play_arrow
Gram staining was developed by:
A)
Louis Pasteur
done
clear
B)
Twort
done
clear
C)
Christian gram
done
clear
D)
Gerald gram
done
clear
View Answer play_arrow
In eukaryotes ATP-sythase enzyme involved in respiration is in the:
A)
Cytoplasm
done
clear
B)
Mitochondrial cell
done
clear
C)
Oxysome
done
clear
D)
Quantasomes
done
clear
View Answer play_arrow
Enzymes are made up of:
A)
Fats
done
clear
B)
Proteins
done
clear
C)
Vitamins
done
clear
D)
Starch
done
clear
View Answer play_arrow
Hypogynous flowers are:
A)
Flowers with superior ovary
done
clear
B)
Flowers with inferior ovary
done
clear
C)
Bisexual flower
done
clear
D)
Unisexual flower
done
clear
View Answer play_arrow
Escherichia colly a bacterium related to the:
A)
Streptococcus pneumonia
done
clear
B)
Salmonella typhimurium
done
clear
C)
Staphylco
done
clear
D)
Haemophus influenza
done
clear
View Answer play_arrow
Biological organisation start with:
A)
Cellular level
done
clear
B)
Atomic level
done
clear
C)
Organisation level
done
clear
D)
Microscopic level
done
clear
View Answer play_arrow
What would be the number of chromosome in the cells of the aleuronic layer in a plant species with 8 chromosome in its synergids?
A)
16
done
clear
B)
24
done
clear
C)
32
done
clear
D)
8
done
clear
View Answer play_arrow
Pineapple fruit develop form:
A)
A unilocular poly carpillary flower
done
clear
B)
A multistillate synearpour flower
done
clear
C)
A cluster of compactly bOrne flower on a common axis
done
clear
D)
A multilocular monocarpellary flower
done
clear
View Answer play_arrow
Golden rice is a promising transgenic crop. It help in:
A)
Vitamin-A deficiency alleviation
done
clear
B)
Pest resistance
done
clear
C)
Herbicide tolerance
done
clear
D)
Producing petrol-like fuel from rice
done
clear
View Answer play_arrow
The 'blue baby' syndrome results from:
A)
Excess of chloride
done
clear
B)
Methaemoglobin
done
clear
C)
Excess of dissolved oxygen
done
clear
D)
Total dissolved solid
done
clear
View Answer play_arrow
Praying mantis is a good example of:
A)
Mullerian mimicry
done
clear
B)
Warning colouration
done
clear
C)
Social insects
done
clear
D)
Camoudlage
done
clear
View Answer play_arrow
In which of the following fruit is edible part the aril?
A)
Custard apple
done
clear
B)
Pomegranate
done
clear
C)
Orange
done
clear
D)
Lichi
done
clear
View Answer play_arrow
Which one of the following amino acid was not found to be synthesized in Miller's experiment:
A)
Glycerine
done
clear
B)
Aspartic acid
done
clear
C)
Glutamic acid
done
clear
D)
Alanine
done
clear
View Answer play_arrow
Crop plant in monoculture are:
A)
Low in yield
done
clear
B)
Free from intraspecific competition
done
clear
C)
Characterised by poor root system
done
clear
D)
Highly prone to pests
done
clear
View Answer play_arrow
The formula for logistic population growth
A)
\[\frac{dN}{dt}=N\left( \frac{K-N}{K} \right)\]
done
clear
B)
\[\frac{rN}{dt}=rN\left( \frac{K-N}{K} \right)\]
done
clear
C)
\[\frac{dN}{dt}=rN\left( \frac{K-N}{K} \right)\]
done
clear
D)
\[\frac{dN}{dt}=rN\left( \frac{N-K}{K} \right)\]
done
clear
View Answer play_arrow
Which of following not used for construction of age pyramid?
A)
Dry weight
done
clear
B)
No. of individual
done
clear
C)
Rate of energy flow
done
clear
D)
Fresh weight
done
clear
View Answer play_arrow
In photosystem-I, the first \[{{e}^{-}}\] acceptor is:
A)
Ferrodoxin
done
clear
B)
Cytochrome
done
clear
C)
Plastocyanin
done
clear
D)
An iron - sulphur protein
done
clear
View Answer play_arrow
Treatment of seed at low temperature under moist condition to break its dormacy is:
A)
Scarification
done
clear
B)
Vernalization
done
clear
C)
Chelation
done
clear
D)
Stratification
done
clear
View Answer play_arrow
GEAC understand for:
A)
Genetic engineering authentic councelling
done
clear
B)
Genetic encoding assay committee
done
clear
C)
Genetic engineering approval committee
done
clear
D)
Genetic engineering approval councelling
done
clear
View Answer play_arrow
'Blue revolution' is responsible for:
A)
Food production
done
clear
B)
Milk production
done
clear
C)
Fish production
done
clear
D)
Honey productions
done
clear
View Answer play_arrow
In infra uterine insemination semen is collected from _ & transfer into__:
A)
Donor, vagina
done
clear
B)
Husband, sarogate mother
done
clear
C)
Husband, uterus
done
clear
D)
Donor or husband, vagina or uterus
done
clear
View Answer play_arrow
Chromosome no. in gamete of onion:
A)
8
done
clear
B)
16
done
clear
C)
24
done
clear
D)
19
done
clear
View Answer play_arrow
Calotropis plant produce __ for avoiding predators:
A)
Calcium glycosides
done
clear
B)
Cardiac glycosides
done
clear
C)
Opium
done
clear
D)
Strychinie
done
clear
View Answer play_arrow
Council's elegant field experiment based on which interaction:
A)
Predation
done
clear
B)
Commensalism
done
clear
C)
Competition
done
clear
D)
Anmensalism
done
clear
View Answer play_arrow
In oviparous animal fertilised egg covered by:
A)
Calcium containing shell
done
clear
B)
Calcareous shell
done
clear
C)
Phosphorous containing
done
clear
D)
All of these
done
clear
View Answer play_arrow
In which case tissue, blood & bone marrow for increase cell is tested:
A)
Leukemias
done
clear
B)
Tomography
done
clear
C)
Biopsy
done
clear
D)
Both a and c
done
clear
View Answer play_arrow
Nicotine stimulates on:
A)
Heart
done
clear
B)
Brain
done
clear
C)
Lung
done
clear
D)
Adrenal gland
done
clear
View Answer play_arrow
Which of these technique are used for early diagnosis:
A)
rDNA technology
done
clear
B)
PCR
done
clear
C)
ELISA
done
clear
D)
All of these
done
clear
View Answer play_arrow
In Insertinal inactivation technique DNA is inserted within the coding sequence of:
A)
\[\hat{\alpha } - glicosidase\]
done
clear
B)
\[\hat{\beta } - galactosidase\]
done
clear
C)
\[\hat{\alpha } - galactosidase\]
done
clear
D)
\[\hat{\beta }-Glicosidase\]
done
clear
View Answer play_arrow
The first restriction endonuclease is:
A)
Hind I
done
clear
B)
Hind III
done
clear
C)
EcoRI
done
clear
D)
Hind II
done
clear
View Answer play_arrow
'Meiocytes' represent:
A)
Megaspore mother cell
done
clear
B)
Microspore mother cell
done
clear
C)
Gametes mother cell
done
clear
D)
All of these
done
clear
View Answer play_arrow
Groundnut is the example of:
A)
Non - albuminous
done
clear
B)
Albuminous
done
clear
C)
Perisperm
done
clear
D)
Pericarp
done
clear
View Answer play_arrow
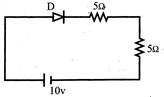
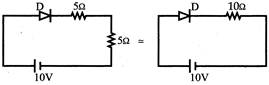 Voltage = 10V Diode is forward biased \[{{\operatorname{R}}_{eq}}=5\Omega +5\Omega =10\Omega \] \[\operatorname{I}=\frac{10}{10}1A\Rightarrow \frac{10}{10}=1A\]
Voltage = 10V Diode is forward biased \[{{\operatorname{R}}_{eq}}=5\Omega +5\Omega =10\Omega \] \[\operatorname{I}=\frac{10}{10}1A\Rightarrow \frac{10}{10}=1A\]