A and B are two metals with threshold frequencies \[1.8\,\,\times \,\,{{10}^{14}}\] Hz and \[2.2\,\,\times \,\,{{10}^{14}}\] Hz. Two identical photons of energy 0.825 eV each are incident on them. Then photoelectrons are emitted from -
A)
B alone
done
clear
B)
A alone
done
clear
C)
Neither A nor B
done
clear
D)
Both A & B
done
clear
View Answer play_arrow
An electron with speed v and a photon with speed c have the same de-Broglie wavelength. If the kinetic energy and momentum of electron is \[{{E}_{e}}\] and \[{{P}_{e}}\] and that of photon is \[{{E}_{ph}}\] and \[{{P}_{ph}}\] respectively, then correct statement is-
A)
\[\frac{{{E}_{e}}}{{{E}_{ph}}}\,\,=\,\,\frac{2c}{v}\]
done
clear
B)
\[\frac{{{E}_{e}}}{{{E}_{ph}}}\,\,=\,\,\frac{v}{2c}\]
done
clear
C)
\[\frac{{{P}_{e}}}{{{P}_{ph}}}\,\,=\,\,\frac{2c}{v}\]
done
clear
D)
\[\frac{{{P}_{e}}}{{{P}_{ph}}}\,\,=\,\,\frac{v}{2c}\]
done
clear
View Answer play_arrow
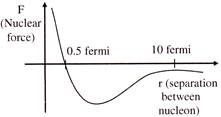
The dependence of nuclear force on distance between nucleons is not known precisely but approximate variation is shown graphically-
From the above graph it can be concluded that-
[a] Nuclear force is repulsive for separation less than 0.5 fermi [b] Nuclear force is attractive for separation less than 0.5 fermi [c] Nuclear force is attractive for separation more than 0.5 fermi [d] Nuclear force is negligible when the separation between nucleons is more than 10 fermi
A)
a, c, d
done
clear
B)
b, c, d
done
clear
C)
a
done
clear
D)
b, c
done
clear
View Answer play_arrow
A radioactive sample at any instant has its disintegration rate 5000 disintegration per minute. After 5 minutes, the rate is 1250 disintegrations per minute. Then, the decay constant (per minute) is-
A)
0.8 ln 2
done
clear
B)
0.4 ln 2
done
clear
C)
0.2 ln 2
done
clear
D)
0.1 ln 2
done
clear
View Answer play_arrow
A NPN transistor is connected in common emitter configuration in which collector supply is 8V and the voltage drop across the load resistance of 800 ohm connected in the collector circuit is 0.8 V. If current 25 amplification factor is \[\frac{25}{26}\] , then power gain is - (given \[\to \] input resistance = \[200\,\Omega \])
A)
6.93
done
clear
B)
3.69
done
clear
C)
9.63
done
clear
D)
None of these
done
clear
View Answer play_arrow
If for a gas \[R/{{C}_{v}}\,\,=\,\,2/3\], then the gas is-
A)
monoatomic
done
clear
B)
diatomic
done
clear
C)
triatomic
done
clear
D)
polyatomic
done
clear
View Answer play_arrow
A gas undergoes process ABCDAC. Work done by gas is-
A)
5 kJ
done
clear
B)
2.5 kJ
done
clear
C)
- 5 kJ
done
clear
D)
7.5 kJ
done
clear
View Answer play_arrow
A bucket full of hot water cools from \[75{}^\circ C\] to \[70{}^\circ \,C\] in time \[{{T}_{1}}\]), from \[70{}^\circ \,C\] to \[65{}^\circ \,C\] in time \[{{T}_{2}}\] and from \[65{}^\circ C\text{ }to\text{ }60{}^\circ C\] in time T.3, then -
A)
\[{{T}_{1}}={{T}_{2}}={{T}_{3}}~\]
done
clear
B)
\[{{T}_{1}}>{{T}_{2}}>{{T}_{3}}\]
done
clear
C)
\[{{T}_{1}}<{{T}_{2}}<{{T}_{3}}\]
done
clear
D)
\[{{T}_{1}}>{{T}_{2}}<{{T}_{3}}\]
done
clear
View Answer play_arrow
If the length of a cylinder on heating increases by \[2%\], the area of its base will increases by-
A)
\[0.5\,%\]
done
clear
B)
\[1\,%\]
done
clear
C)
\[2\,%\]
done
clear
D)
\[4\,%\]
done
clear
View Answer play_arrow
Velocity of sound in He at certain temperature is ?\[{{v}_{0}}\]?. Velocity of sound in \[{{N}_{2}}\] at that temperature will be-
A)
\[\frac{\sqrt{3}}{5}\,\,{{v}_{0}}\]
done
clear
B)
\[\frac{\sqrt{3}}{7}\,\,{{v}_{0}}\]
done
clear
C)
\[\frac{1}{\sqrt{7}}\,\,{{v}_{0}}\]
done
clear
D)
\[\sqrt{\frac{3}{7}}\,\,{{v}_{0}}\]
done
clear
View Answer play_arrow
Two closed end pipes when sounded together produce 5 beat per second. If their length are in the ratio \[100\,\,:\,\,101\], then fundamental notes produced by them, are-
A)
245, 250
done
clear
B)
250, 255
done
clear
C)
495, 500
done
clear
D)
500, 505
done
clear
View Answer play_arrow
Two strings of copper are stretched to the same tension. If their cross-section area are in the ratio 1 : 4, then the respective wave velocities will be-
A)
4 : 1
done
clear
B)
2 : 1
done
clear
C)
1 : 2
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
The period of rotation of the sun at its equator is T and its radius is R. Then the Doppler wavelength shift expected for light with wavelength \[\lambda \], emitted from the edge of the sun?s disc is- [\[c\,\,=\,\,speed\text{ }of\text{ }light\]]
A)
\[\pm \,\frac{R\lambda }{cT}\]
done
clear
B)
\[\pm \,\frac{T\lambda }{2\pi Rc}\]
done
clear
C)
\[\pm \,\frac{2\pi Rc}{T\lambda }\]
done
clear
D)
\[\pm \,\frac{2\pi R\lambda }{cT}\]
done
clear
View Answer play_arrow
The magnitude of the X and Y components of \[\overrightarrow{A}\] are 7 and 6. Also the magnitudes of the X and Y components of \[\overrightarrow{A}\,\,+\,\,\overrightarrow{B}\] are 11 and 9 respectively. What is the magnitude of\[\overrightarrow{B}\]?
A)
5
done
clear
B)
6
done
clear
C)
8
done
clear
D)
9
done
clear
View Answer play_arrow
A very large number of balls are thrown vertically upwards in quick succession in such a way that the next ball is thrown when the previous one is at the maximum height. If the maximum height is 5 m, then the number of balls thrown per minute is- (\[g=10\,m\,{{s}^{-2}}\])
A)
120
done
clear
B)
80
done
clear
C)
60
done
clear
D)
40
done
clear
View Answer play_arrow
Two stones are projected with the same speed but making different angles with the horizontal. Their horizontal ranges are equal. The angle of projection of one is \[\pi /3\] and the maximum height reached by it is 102 m. Then the maximum height reached by the other in metre is-
A)
336
done
clear
B)
224
done
clear
C)
56
done
clear
D)
34
done
clear
View Answer play_arrow
Two small balls of same size and masses \[{{m}_{1}}\] and \[{{m}_{2}}\] \[\left( {{m}_{1}}\,\,>\,\,{{m}_{2}} \right)\] are tied by a thin weightless thread and dropped from a certain height. Taking upward buoyancy force F into account the tension T of the thread during the flight after the motion of the balls becomes uniform will be-
A)
\[\left( {{m}_{1}}-{{m}_{2}} \right)g~\]
done
clear
B)
\[\left( {{m}_{1}}-\text{ }{{m}_{2}} \right)\,\frac{g}{2}\]
done
clear
C)
\[\left( {{m}_{1}}+{{m}_{2}} \right)g\]
done
clear
D)
\[\left( {{m}_{1}}+{{m}_{2}} \right)\,\frac{g}{2}\]
done
clear
View Answer play_arrow
A marble block of mass 2 kg lying on ice when given a velocity of 6 m/s is stopped by friction in 10s. Then the coefficient of friction is-
A)
0.01
done
clear
B)
0.02
done
clear
C)
0.03
done
clear
D)
0.06
done
clear
View Answer play_arrow
A 500 g ball is released from a height of 4 m. Each time it makes contact with the ground it loses \[25\,%\] of its energy. Find the KE it possess after\[{{3}^{rd}}\]hit-
A)
15 J
done
clear
B)
11.25 J
done
clear
C)
8.44 J
done
clear
D)
none of these
done
clear
View Answer play_arrow
An inclined plane ends into a vertical loop of radius r as shown in figure. If a particle of mass m is released from topmost point. The point C on the track as shown in the figure and centre of circular tack are at same horizontal level. The normal force at point C is-
A)
8 mg
done
clear
B)
7 mg
done
clear
C)
1 mg
done
clear
D)
3 mg
done
clear
View Answer play_arrow
The energy required to accelerate a car from 10 m/s to 20 m/s is how many times the energy required to accelerate the car from rest to 10 m/s?
A)
equal
done
clear
B)
4 times
done
clear
C)
2 times
done
clear
D)
3 times
done
clear
View Answer play_arrow
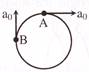
Find acceleration of CM and angular acceleration of cylinder while acceleration of points A & B are\[{{a}_{A}}\,\,=\,\,{{a}_{0}}\widehat{i}\,\,and\,\,\,{{a}_{B}}\,=\,{{a}_{0}}\,\widehat{j}\,\,\]
A)
\[0,\,\,\frac{{{a}_{0}}}{R}\]
done
clear
B)
\[{{a}_{0}},\,\,0\]
done
clear
C)
\[0,\,\,\frac{\sqrt{2}{{a}_{0}}}{R}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
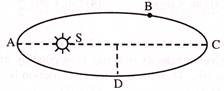
A planet revolves in elliptical orbit around the sun. The linear speed of the planet will be maximum at-
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
The period of simple harmonic oscillator is 24 sec. and the initial phase is zero. The time at which the oscillating point would have attained half the amplitude is:
A)
2 sec
done
clear
B)
2.4 sec
done
clear
C)
6 sec
done
clear
D)
12 sec
done
clear
View Answer play_arrow
Bulk modulus of water is\[2\,\,\times \,\,{{10}^{9}}\,Nm{{\,}^{-}}^{2}\]. The change in pressure required to increase the density of water by \[0.1\,\,%\] is
A)
\[2\,\,\times \,\,{{10}^{9}}\,N{{m}^{-}}^{2}\]
done
clear
B)
\[2\,\,\times \,\,{{10}^{8}}\,N{{m}^{-}}^{2}\]
done
clear
C)
\[2\,\,\times \,\,{{10}^{6}}\,N{{m}^{-}}^{2}\]
done
clear
D)
\[2\,\,\times \,\,{{10}^{4}}\,N{{m}^{-}}^{2}\]
done
clear
View Answer play_arrow
A 10 cm long wire is brought 20 cm up in a liquid of surface tension 40 dyne\[c{{m}^{-}}^{1}\]. Find the work done against surface tension-
A)
0.8 m J
done
clear
B)
1.6 m J
done
clear
C)
1.2 m J
done
clear
D)
3.2 m J
done
clear
View Answer play_arrow
In the figure shown the angle made by the light ray with the normal in the medium of refractive index \[\sqrt{2}\] is-
A)
\[30{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
none of these
done
clear
View Answer play_arrow
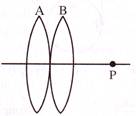
Two convex lenses placed in contact form the image of a distant object at P. If the lens B is moved to the right, the image will-
A)
move to the left
done
clear
B)
move to the right
done
clear
C)
remain at P
done
clear
D)
move either to the left or right, depending upon focal lengths of the lenses.
done
clear
View Answer play_arrow
A concave lens of glass, refractive index 1.5, has both surfaces of same radius of curvature R. On immersion in a medium of refractive index 1.75, it will behave as a-
A)
convergent lens of focal length 3.5 R
done
clear
B)
convergent lens of focal length 3.0 R
done
clear
C)
divergent lens of focal length 3.5 R
done
clear
D)
divergent lens of focal length 3.0 R
done
clear
View Answer play_arrow
A lens is placed between a source of light and a wall. It forms images of area \[{{A}_{1}}\] and \[{{A}_{2}}\] on the wall, for its two different positions. The area of the source of light is-
A)
\[\sqrt{{{A}_{1}}{{A}_{2}}}\]
done
clear
B)
\[\frac{{{A}_{1}}+{{A}_{2}}}{2}\]
done
clear
C)
\[{{\left( \frac{\sqrt{{{A}_{1}}}+\sqrt{{{A}_{2}}}}{2} \right)}^{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
For a prism, its refractive index is cot\[A/2\]. Then minimum angle of deviation is-
A)
\[180{}^\circ -A\]
done
clear
B)
\[180{}^\circ -2A\]
done
clear
C)
\[90{}^\circ -A\]
done
clear
D)
\[A/2\]
done
clear
View Answer play_arrow
The maximum intensity in Young?s double-slit experiment is Io. Distance between the slits is \[d\,\,=\,\,5\,\lambda \], where \[\lambda \] is the wavelength of monochromatic light used in the experiment. What will be the intensity of light in front of one of the slits on a screen at a distance\[D\,\,=\,\,10d\]?
A)
\[\frac{{{I}_{0}}}{2}\]
done
clear
B)
\[\frac{3}{4}{{I}_{0}}\]
done
clear
C)
\[{{I}_{0}}\]
done
clear
D)
\[\frac{{{I}_{0}}}{4}\]
done
clear
View Answer play_arrow
Two waves of equal amplitudes and wavelengths but differing in phase are superimposed. Amplitude of resultant wave is maximum when phase difference is-
A)
Zero
done
clear
B)
\[\pi /12\]
done
clear
C)
\[\pi \]
done
clear
D)
\[3\pi /2\]
done
clear
View Answer play_arrow
Two copper balls, each weighing 10 gm, are kept in air 10 cm apart. If one electron from every \[{{10}^{6}}\] atoms is transferred from one ball to the other, the coulomb force between them is - (Atomic weight of copper is 63.5)
A)
\[2.0\times {{10}^{4}}\,N\]
done
clear
B)
\[2.0\times {{10}^{6}}\]
done
clear
C)
\[2.0\times {{10}^{10}}N\]
done
clear
D)
\[2.0\times {{10}^{\text{8}}}\,N\]
done
clear
View Answer play_arrow
Two electric dipoles of moment p and 64p are placed in opposite direction on a line at a distance of 25 cm. The electric field will be zero at a point between the dipoles whose distance from the dipole of moment p is-
A)
5 cm
done
clear
B)
\[\frac{25}{9}\] cm
done
clear
C)
10 cm
done
clear
D)
\[\frac{4}{13}\] cm
done
clear
View Answer play_arrow
Two resistances are connected in two gaps of a meter bridge. The balance point is 20 cm from the zero end. A resistance of 15 ohm is connected in series with the smaller of the two. The null point shifts to 40 cm. The value of the smaller resistance in ohm is-
A)
3
done
clear
B)
6
done
clear
C)
9
done
clear
D)
12
done
clear
View Answer play_arrow
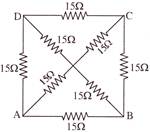
The equivalent resistance between the points A and B will be (each resistance is 15 \[\Omega \])-
A)
30\[\Omega \]
done
clear
B)
8\[\Omega \]
done
clear
C)
10\[\Omega \]
done
clear
D)
40\[\Omega \]
done
clear
View Answer play_arrow
Consider a capacitor charging circuit. Let \[{{Q}_{1}}\]) be the charge given to the capacitor in a time interval of 10 ms and \[{{Q}_{2}}\] be the charge given in the next time interval of 10 ms. Let 10 \[\mu C\] charge be deposited in time interval \[{{t}_{1}}\] and the next \[10\,\,\mu C\] charge deposited in next time interval \[{{t}_{2}}\], then-
A)
\[{{Q}_{1}}>{{Q}_{2}},\text{ }{{t}_{1}}>{{t}_{2}}\]
done
clear
B)
\[{{Q}_{1}}>{{Q}_{2}}.\text{ }{{t}_{1}}<\text{ }{{t}_{2}}\]
done
clear
C)
\[{{Q}_{1}}<{{Q}_{2}},{{t}_{1}}>{{t}_{2}}\]
done
clear
D)
\[{{Q}_{1}}<{{Q}_{2}},{{t}_{1}}<{{t}_{2}}\]
done
clear
View Answer play_arrow
A wire along x-axis carries a current 3.5 A. Find the force on a 1 cm section of the wire exerted by: \[B=0.74\text{ }T\text{ }\widehat{j}\,\,-\,\,0.3\text{ }T\text{ }\widehat{k}\]
A)
\[\left( 2.59\text{ }\widehat{k}+1.26\text{ }\widehat{j} \right)\times {{10}^{-}}^{2}\]
done
clear
B)
\[\left( 1.26\text{ }\widehat{k}-2.59\text{ }\widehat{j} \right)\times {{10}^{-}}^{2}\]
done
clear
C)
\[\left( -2.59\text{ }\widehat{k}-1.26\text{ }\widehat{j} \right)\times {{10}^{-}}^{2}\]
done
clear
D)
\[\left( -1.26\text{ }\widehat{k}+2.59\text{ }\widehat{j} \right)\times {{10}^{-}}^{2}\]
done
clear
View Answer play_arrow
Relative permittivity and permeability of a material \[{{\varepsilon }_{r}}\,\,and\,\,{{\mu }_{r}}\] respectively. Which of the following values of these quantities are allowed for a diamagnetic material?
A)
\[{{\varepsilon }_{r}}=0.5,\,\,\,{{\mu }_{r}}=1.5\]
done
clear
B)
\[{{\varepsilon }_{r}}=1.5,\,\,{{\mu }_{r}}=0.5\]
done
clear
C)
\[{{\varepsilon }_{r}}=0.5,\,\,{{\mu }_{r}}=0.5\]
done
clear
D)
\[{{\varepsilon }_{r}}=1.5,\,\,{{\mu }_{r}}=1.5\]
done
clear
View Answer play_arrow
Rate of change of torque (r) with deflection \[\theta \] is maximum for a magnet suspended in a uniform magnetic field B when-
A)
\[\theta =0{}^\circ \]
done
clear
B)
\[\theta =45{}^\circ \]
done
clear
C)
\[\theta =60{}^\circ \]
done
clear
D)
\[\theta =90{}^\circ \]
done
clear
View Answer play_arrow
A copper disc of radius 0.1 mis rotated about its centre with 20 revolution per second in a uniform magnetic field of 0.1 T with its plane perpendicular to the field. The emf induced across the radius of the disc is-
A)
\[\frac{\pi }{20}~volt\]
done
clear
B)
\[\frac{\pi }{10}~volt\]
done
clear
C)
\[20~\pi millivolt\]
done
clear
D)
\[100\,\,\pi \,\,millivolt\]
done
clear
View Answer play_arrow
For an alternating current-
A)
r.m.s. value may be equal to peak value
done
clear
B)
average value be equal to peak value
done
clear
C)
r.m.s. value be equal to average value
done
clear
D)
All of the above
done
clear
View Answer play_arrow
In an n-type semiconductor donor valence band is-
A)
above the conduction band of the host crystal
done
clear
B)
below the valence band of the host crystal
done
clear
C)
close to the conduction band of the host crystal
done
clear
D)
close to the valence band of the host crystal
done
clear
View Answer play_arrow
To measure light intensity we use-
A)
LED with forward bias
done
clear
B)
LED with reverse bias
done
clear
C)
photodiode with reverse bias
done
clear
D)
photodiode with forward bias
done
clear
View Answer play_arrow
Which of the following graph represents the variation of amount of chemisorptions of a gas by a solid with temperature under constant pressure?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In H-atom find out the ratio of time period when \[{{e}^{-}}\] revolves in \[n\,\,=\,\,2\] and \[n\,\,=\,\,3\] orbit:
A)
\[\frac{4}{27}\]
done
clear
B)
\[\frac{2}{27}\]
done
clear
C)
\[\frac{8}{27}\]
done
clear
D)
\[\frac{4}{9}\]
done
clear
View Answer play_arrow
1 mole of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] is completely react with x moles of FAS in acidic medium then find out the value of x:
A)
3
done
clear
B)
2
done
clear
C)
6
done
clear
D)
4
done
clear
View Answer play_arrow
The pH of \[{{10}^{-10}}\] \[M\text{ }{{H}_{2}}S{{O}_{4}}\] will be almost:
A)
\[> 7\]
done
clear
B)
\[7\]
done
clear
C)
\[< 7\]
done
clear
D)
\[10\]
done
clear
View Answer play_arrow
In which of the following \[{{K}_{p}}\] is less than\[{{K}_{c}}\]?
A)
\[{{N}_{2}}{{O}_{4}}\text{ }\rightleftharpoons 2N{{O}_{2}}\]
done
clear
B)
\[\,2HI\,\,\rightleftharpoons \,\,{{H}_{2}}\,+\,{{I}_{2}}\]
done
clear
C)
\[2S{{O}_{2}}+{{O}_{2}}\text{ }\rightleftharpoons \,\,2S{{O}_{3}}\]
done
clear
D)
\[\,{{N}_{2}}+{{O}_{2}}\,\,\rightleftharpoons \,\,2NO\]
done
clear
View Answer play_arrow
Consider a hypothetical reaction of the kind \[A{{B}_{2}}(g)+\frac{1}{2}\,\,{{B}_{2}}(g)\,\,\rightleftharpoons \,\,A{{B}_{3}}(g)\,\,;\,\,\Delta H\,\,=\,\,-\chi \,kJ\] More \[A{{B}_{3}}\] could be produced at equilibrium by
A)
using a catalyst
done
clear
B)
removing some of \[{{B}_{2}}\]
done
clear
C)
increasing the temperature
done
clear
D)
increasing the pressure
done
clear
View Answer play_arrow
Which among the following match is correct -
A)
\[{{d}^{6}}({{\Delta }_{0}}<P.E.)\,-\,{{t}_{2}}{{g}^{3}}e{{g}^{3}}\]
done
clear
B)
\[{{d}^{6}}(tetrahedral)\,-\,e{{g}^{3}}{{t}_{2}}{{g}^{3}}\]
done
clear
C)
\[{{d}^{6}}\left( {{\Delta }_{0}}>P.E. \right)-{{t}_{2}}{{g}^{4}}e{{g}^{2}}\]
done
clear
D)
None of them is correct
done
clear
View Answer play_arrow
Which represents incorrect order-
A)
\[Cl>F>Br>I\] (order of EA)
done
clear
B)
\[Li<Be<B<C<N\] (order of IE)
done
clear
C)
\[N>C>P>Si\] (order of EN)
done
clear
D)
\[MnO<M{{n}_{2}}{{O}_{3}}<Mn{{O}_{2}}<M{{n}_{2}}{{O}_{7}}\] (order of acidic strength)
done
clear
View Answer play_arrow
Enthalpy of neutralisation of HCl with NaOH is x. The heat evolved when 500 ml of 2N HCl are mixed with 250 ml of 4 N NaOH will be-
A)
500 x
done
clear
B)
100 x
done
clear
C)
x
done
clear
D)
10x
done
clear
View Answer play_arrow
In aqueous solution which has largest ionic radius-
A)
\[L{{i}^{+}}\]
done
clear
B)
\[C{{s}^{+}}\]
done
clear
C)
\[B{{e}^{2+}}\]
done
clear
D)
\[B{{a}^{2+}}\]
done
clear
View Answer play_arrow
\[M\,\,\left( OH \right)x\] has a \[{{K}_{sp}}\text{ }of\text{ }4\,\,\times \,\,{{10}^{-}}^{9}\] and its solubility is \[{{10}^{-3}}\] M. The value of x is:
A)
4
done
clear
B)
1
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
The correct order of the strength of H-bonds is
A)
\[H......F>H......O>H......N\]
done
clear
B)
\[H......N>H......O>H......F\]
done
clear
C)
\[H......O>H......N>H......F\]
done
clear
D)
\[H......F>H......N>H......O\]
done
clear
View Answer play_arrow
Which one of the following has largest number of isomers?
A)
\[{{\left[ Ir{{\left( P{{R}_{3}} \right)}_{2}}H\left( CO \right) \right]}^{2+}}\]
done
clear
B)
\[{{\left[ Co{{\left( N{{H}_{3}} \right)}_{5}}Cl \right]}^{2+}}\]
done
clear
C)
\[{{\left[ Ru{{\left( N{{H}_{3}} \right)}_{4}}C{{l}_{2}} \right]}^{+}}\]
done
clear
D)
\[\left[ Co{{\left( en \right)}_{2}}C{{l}_{2}}{{l}^{+}} \right]\]
done
clear
View Answer play_arrow
The correct order of bond angles (smallest first) in \[{{H}_{2}}S,\text{ }N{{H}_{3}},\text{ }B{{F}_{3}}\,\,and\text{ }Si{{H}_{4}}\] is-
A)
\[{{H}_{2}}S<N{{H}_{3}}<Si{{H}_{4}}<B{{F}_{3}}\]
done
clear
B)
\[N{{H}_{3}}<\text{ }{{H}_{2}}S\text{ }<\text{ }Si{{H}_{4}}<\text{ }B{{F}_{3}}\]
done
clear
C)
\[{{H}_{2}}S\text{ }<\text{ }Si{{H}_{4}}<\text{ }N{{H}_{3}}<\text{ }B{{F}_{3}}\]
done
clear
D)
\[{{H}_{2}}S\text{ }<\text{ }N{{H}_{3}}<\text{ }B{{F}_{3}}<\text{ }Si{{H}_{4}}\]
done
clear
View Answer play_arrow
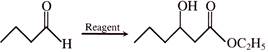
Diethyl ether combines with CO under specific conditions to form:
A)
ethyl propanoate
done
clear
B)
acetic acid
done
clear
C)
carbon dioxide
done
clear
D)
Diethyl ketone
done
clear
View Answer play_arrow
The major organic product in the reaction is- \[C{{H}_{3}}-O-CH{{\left( C{{H}_{3}} \right)}_{2}}+\text{ }HI\text{ }\to ?\]
A)
\[C{{H}_{3}}OH+{{\left( C{{H}_{3}} \right)}_{2}}CHI\]
done
clear
B)
\[C{{H}_{3}}I\,\,+\,\,{{\left( C{{H}_{3}} \right)}_{2}}CHOH\]
done
clear
C)
\[IC{{H}_{2}}OCH{{\left( C{{H}_{3}} \right)}_{2}}\]
done
clear
D)
\[\text{ }C{{H}_{3}}O\underset{\underset{C}{\mathop{|}}\,}{\mathop{C}}\,{{\left( C{{H}_{3}} \right)}_{2}}\]
done
clear
View Answer play_arrow
In a CCP lattice of X and Y, X atoms are present at the comers while Y atoms are at face centres. Then the formula of the compound would be if one of the X atoms from a comer is replaced by Z atoms (also monovalent)?
A)
\[{{X}_{7}}{{Y}_{24}}{{Z}_{2}}\]
done
clear
B)
\[{{X}_{7}}{{Y}_{24}}Z\]
done
clear
C)
\[{{X}_{24}}{{Y}_{7}}Z\]
done
clear
D)
\[Z{{Y}_{24}}Z\]
done
clear
View Answer play_arrow
\[F{{e}_{2}}{{\left( CO \right)}_{9}}\] is diamagnetic. Which of the following reasons is correct?
A)
Presence of one CO as bridge group
done
clear
B)
Presence of monodentate ligand
done
clear
C)
Metal-metal \[\left( Fe-Fe \right)\] bond in molecule
done
clear
D)
Resonance hybridization of CO
done
clear
View Answer play_arrow
The hybridization state of Fe in \[\left[ Fe{{\left( {{H}_{2}}O \right)}_{5}}NO \right]S{{O}_{4}}\text{ }is\text{ }\left( At.\text{ }No.\text{ }of\,\,Fe\,\,=\,\,26 \right)\]
A)
\[ds{{p}^{2}}\]
done
clear
B)
\[s{{p}^{3}}d\]
done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[\,{{d}^{2}}s{{p}^{3}}\]
done
clear
View Answer play_arrow
Which molecule is non-polar-
A)
\[PC{{l}_{3}}{{F}_{2}}\]
done
clear
B)
\[P-{{C}_{6}}{{H}_{4}}{{\left( SH \right)}_{2}}\]
done
clear
C)
\[C{{H}_{2}}C{{l}_{2}}\]
done
clear
D)
\[{{S}_{2}}C{{l}_{2}}\]
done
clear
View Answer play_arrow
The distance between an octahedral and tetrahedral void in fee lattice would be-
A)
\[\sqrt{3}\,a\]
done
clear
B)
\[\frac{\sqrt{3}a}{2}\]
done
clear
C)
\[\frac{\sqrt{3}a}{3}\]
done
clear
D)
\[\frac{\sqrt{3}a}{4}\]
done
clear
View Answer play_arrow
The molarity of a solution of ethanol in water in which the mole fraction of \[{{C}_{2}}{{H}_{5}}OH\] is 0.040 is- (assume density of solution to be 1 g/ml)
A)
2.09 M
done
clear
B)
2.31 M
done
clear
C)
20.9 M
done
clear
D)
23.1M
done
clear
View Answer play_arrow
Calculate the enthalpy of reaction at 298 K for the reaction- \[{{C}_{2}}{{H}_{4}}\left( g \right)\,\,+\,\,6{{F}_{2}}\left( g \right)\,\,\to \,\,2C{{F}_{4}}\left( g \right)+4HF\left( g \right)\] by using following equations
\[{{H}_{2}}\left( g \right)\,\,+\,\,{{F}_{2}}\left( g \right)\,\,\,\to \,\,2HF\left( g \right)\text{ }\Delta H\,\,=\,\,-537\text{ }kJ\] \[C\left( s \right)\,\,+\,\,2{{F}_{2}}\left( g \right)\,\,\to \,\,C{{F}_{4}}\left( g \right)\text{ }\Delta H\,\,=\,\,-680\text{ }kJ\] \[2C\left( s \right)\,\,+\,\,2{{H}_{2}}\left( g \right)\,\,\to \,\,{{C}_{2}}{{H}_{4}}\left( g \right)\text{ }\Delta H\,\,=\,\,52\text{ }kJ\]
A)
-1165 kJ
done
clear
B)
-2486 kJ
done
clear
C)
+1165 kJ
done
clear
D)
+2486 kJ
done
clear
View Answer play_arrow
Two mole of an ideal gas is expanded irreversibly and isothermally at \[37{}^\circ C\] until its volume is doubled and 3.41 kJ heat is absorbed from surrounding \[\Delta {{G}_{total}}\] is-
A)
-0.52 J/K
done
clear
B)
0.52 J/K
done
clear
C)
22.52 J/K
done
clear
D)
0
done
clear
View Answer play_arrow
The molar entropies of HI(g), H(g) and I(g) at 298 K are 206.5, 114.6 and 180.7 J \[mo{{l}^{-1}}{{K}^{-1}}\] respectively. Using the AG given below, calculate the bond energy of HI \[HI\left( g \right)\,\,\to \,\,H\left( g \right)+I\left( g \right)\text{ }\Delta G=271.8\text{ }kJ\]
A)
282.4 kJ
done
clear
B)
298.3 kJ
done
clear
C)
290.1 kJ
done
clear
D)
315.4 kJ
done
clear
View Answer play_arrow
\[{{H}_{2}}S\] gas when passed through a solution of cations containing HCl precipitates the cations of second group of qualitative analysis but not those belonging to the fourth group. It is because-
A)
Presence of HCl decreases the sulphide ion concentration
done
clear
B)
Sulphides of group IV cations are unstable in HCl
done
clear
C)
Solubility product of group II sulphides is more than that of group IV sulphides
done
clear
D)
Presence of HCl increases the sulphide ion concentration
done
clear
View Answer play_arrow
For the reaction: \[C{{H}_{4}}(g)+2{{O}_{2}}(g)\rightleftharpoons \,\,C{{O}_{2}}(g)+2{{H}_{2}}O(l)\] \[{{\Delta }_{r}}H\,\,\,=\,\,-170.8\,\,kJ\,\,mo{{l}^{-1}}\] Which of the following statements is not true-
A)
At equilibrium, the concentrations of \[C{{O}_{2}}\left( g \right)\] and \[{{H}_{2}}O\left( l \right)\] are not equal
done
clear
B)
The equilibrium constant for the reaction is given by \[{{K}_{p}}\,=\,\,\frac{\left[ C{{O}_{2}} \right]}{\left[ C{{H}_{4}} \right]\left[ {{O}_{2}} \right]}\]
done
clear
C)
Addition of \[C{{H}_{4}}\left( g \right)\] or \[{{O}_{2}}\left( g \right)\] at equilibrium will cause a shift to the right
done
clear
D)
The reaction is exothermic
done
clear
View Answer play_arrow
The unit of rate constant of elementary reaction depends upon the-
A)
Temperature of the reaction
done
clear
B)
Concentration of reactant
done
clear
C)
Activation energy of the reaction
done
clear
D)
Molecularity of the reaction
done
clear
View Answer play_arrow
Under which of the following conditions conductance, sp. Conductance and equivalent conductance are all equal-
A)
1000 cc of the solution contains 1 eq. the electrolyte
done
clear
B)
100 cc of the solution contains 1 eq. of the electrolyte
done
clear
C)
10 cc of the solution contain 1 eq. of me electrolyte
done
clear
D)
1 cc of the solution contain 1 eq. of the electrolyte
done
clear
View Answer play_arrow
When 20 g of naphthoic acid \[({{C}_{11}}{{H}_{8}}{{O}_{2}})\] is dissolved in 50 g of benzene\[({{K}_{f}}=\,\,1.72\text{ }K\text{ }kgmo{{l}^{-1}})\], a freezing point depression of 2K is observed. The Van?t Hoff factor (i) is-
A)
0.5
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
For the homogeneous reactions: \[xA+yB\,\,\to \,\,\ell Y+mZ\] \[\Delta H=-30\text{ }kJ\text{ }mo{{l}^{-1}},\text{ }\Delta S=-100\text{ }J{{k}^{-}}^{1}\,mo{{l}^{-1}}\] At what temperature the reaction is at equilibrium?
A)
\[50{}^\circ C\]
done
clear
B)
\[250{}^\circ C\]
done
clear
C)
\[100\text{ }K\]
done
clear
D)
\[27{}^\circ C\]
done
clear
View Answer play_arrow
Which is most stable species:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
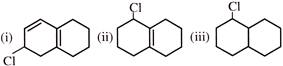
\[Phenol\,\,\xrightarrow{Zn-dust}\,A\,\xrightarrow[+{{H}_{2}}S{{O}_{4}}]{Conc.HN{{O}_{3}}}\,B\,\xrightarrow[NaOH]{Zn}C\] In the above reaction A, B and C are -
A)
benzene, nitrobenzene and aniline
done
clear
B)
benzene, dinitrobenzene and m-nitroaniline
done
clear
C)
Benzene, nitrobenzene and hydrazobenzene
done
clear
D)
toluene, m-nitrobenzene and m-toludine
done
clear
View Answer play_arrow
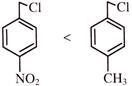
Identify correct reactivity order for \[{{S}_{N}}1\] reaction:
A)
done
clear
B)
done
clear
C)
done
clear
D)
All of these
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In the given reaction, [X] will be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
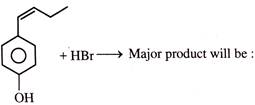
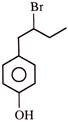
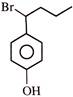
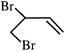
When
is treated with \[B{{r}_{2}}\] (1- equivalent), it would give as a major product:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
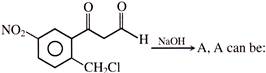
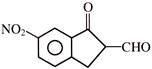
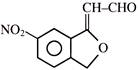
Which of the following reagent can be used for carrying out the reaction outlined below?
A)
\[BrMgC{{H}_{2}}CO{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[BrZnC{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
C)
\[LiC{{H}_{2}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
done
clear
D)
NOTA
done
clear
View Answer play_arrow
Which is true regarding the following structure?
A)
Optically active
done
clear
B)
Optically inactive
done
clear
C)
Both
done
clear
D)
None of these
done
clear
View Answer play_arrow
What could be the product for the following reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
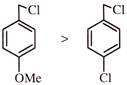
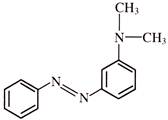
A compound
, shows a large dipole moment. Which of the following resonance structures can be used to adequately explain this observation?
A)
(ii) only
done
clear
B)
(iii) and (iv)
done
clear
C)
(ii) and (iii)
done
clear
D)
(iv) only
done
clear
View Answer play_arrow
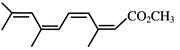
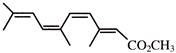
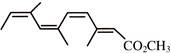
Which represents methyl-(2Z,4Z,6Z)-3,6,9- trimethy1-2,4,6,8-decatetraenoate:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
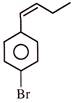
Identify correct reactivity order for \[{{E}^{2}}\] reaction with alcoholic KOH:
A)
\[\left( i \right)>\left( ii \right)>\left( iii \right)\]
done
clear
B)
\[\left( ii \right)>\left( iii \right)>\left( i \right)\]
done
clear
C)
\[\left( i \right)>\left( iii \right)>\left( ii \right)\]
done
clear
D)
\[\left( iii \right)>\left( i \right)>\left( ii \right)\]
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Hyaline cartilage, which is the most common form of cartilage:
A)
contains type I collagen
done
clear
B)
is a highly vascular tissue
done
clear
C)
contains matrix composed primarily of glycoproteins
done
clear
D)
is surrounded by a perichondrium except at articular surfaces
done
clear
View Answer play_arrow
Identify the animal tissues in given figure-
A)
A-Connective tissues, B-Muscular tissue
done
clear
B)
A-Epithelial tissues, B-Muscular tissue
done
clear
C)
A-Muscular tissues, B-Connective tissue
done
clear
D)
A-Epithelial tissues, B-Connective tissue
done
clear
View Answer play_arrow
Areolar connective tissue joins-
A)
Fat body with bone
done
clear
B)
Integument with muscles
done
clear
C)
Bones with muscles
done
clear
D)
Bones with bones
done
clear
View Answer play_arrow
A young infant may be feeding entirely on mother?s milk, which is white in colour but the stools, which the infant passes out is quite yellowish. What is this yellow colour due to?
A)
Intestinal juice
done
clear
B)
Bile pigments passed through bile juice
done
clear
C)
Undigested milk protein casein
done
clear
D)
Pancreatic juice poured into duodenum
done
clear
View Answer play_arrow
Vitamins sysnthesized by intestinal bacteria and absorbed in significant quantities include
A)
folic acid
done
clear
B)
riboflavin
done
clear
C)
thiamine
done
clear
D)
vitamin \[{{B}_{12}}\]
done
clear
View Answer play_arrow
The H-zone in the skeletal muscle fibre is due to
A)
The central gap between myosin filaments in the A-band
done
clear
B)
The central gap between actin filaments extending through myosin filaments in the A band.
done
clear
C)
Extension of myosin filaments in the central portion of the A-band
done
clear
D)
The absence of myofibriles in the central portion of A-band
done
clear
View Answer play_arrow
One of the examples of the action of the autonomous nervous system is-
A)
Knee-jerk response
done
clear
B)
Pupillary reflex
done
clear
C)
Swallowing of food
done
clear
D)
Peristalsis of the intestine
done
clear
View Answer play_arrow
An action potential in the nerve fibre is produced when positive and negative charges on the outside and the inside of the axon membrane are reversed, because-
A)
More potassium ions enter the axon as compared to sodium ions leaving it
done
clear
B)
More sodium ions enter the axon as compared to potassium ions leaving it
done
clear
C)
All potassium ions leave the axon
done
clear
D)
All sodium ions enter the axon
done
clear
View Answer play_arrow
The cornea is very important component of the human eye. The main function of the cornea is to-
A)
Bend the light before it reaches the lens
done
clear
B)
Provide structural support to the eye
done
clear
C)
Contain concentrated amount of cone cells in the correct orientation
done
clear
D)
Change the shape of the lens to enable the image to be focused on the retina
done
clear
View Answer play_arrow
Which part of the human ear plays no role in hearing as such, but is otherwise very much required?
A)
Eustachian tube
done
clear
B)
Organ of corti
done
clear
C)
Vestibular apparatus
done
clear
D)
Ear ossicles
done
clear
View Answer play_arrow
A person is having problem with calcium and phosphorus metabolism in his body. Which one of the following glands may not be functioning properly?
A)
Parotid
done
clear
B)
Pancreas
done
clear
C)
Adrenal cortex
done
clear
D)
Parathyroid
done
clear
View Answer play_arrow
Match the following columns
Column-I Column-II A Thyroid stimulating hormone I. Mammary glands B. Adenocorticotro-pic hormone II. Thyroid gland C. Prolactin III. Liver D. Growth hormone IV. Adrenal cortex E. Oestrogen V. Gonads F. FSH and LH VI. Anterior pituitary G. Androgens VII. Testis H. Gonadotropin Releasing hormone VIII. Ovary
A)
A. II B. IV C. I D. III E. VIII F. V G. VII H. VI
done
clear
B)
A. IV B. II C. I D. VIII E. III F. VII G. V H. VI
done
clear
C)
A. II B. IV C. I D. VIII E. III F. VI G. VII H. V
done
clear
D)
A. IV B. II C. I D. III E. VIII F. V G. VI H. VII
done
clear
View Answer play_arrow
Active transport-
I. Is a very specific process II. Is a passive process III Mainly it is due to lipid molecules IV It is a downhill transport V. It is a uphill transport
A)
All are correct
done
clear
B)
I, IV are correct
done
clear
C)
I and V are correct
done
clear
D)
All are correct except IV
done
clear
View Answer play_arrow
During the ascent of sap the water column does not break in xylem elements. It is due to?
A)
Transpiration pull
done
clear
B)
Cohesion and adhesion force
done
clear
C)
T.P gradient
done
clear
D)
Capillary force
done
clear
View Answer play_arrow
The following (P to U) are the main steps of chemiosmotic ATP synthesis in the light reaction. Which answer places them in correct order?
P. \[P.\text{ }{{H}^{+}}\] concentration gradient established Q. \[Q.\text{ }{{H}^{+}}\] diffuses through ATP synthetase R. Carriers use energy from electrons to move I-T across the membrane S. Electrons from PSII pass along electron transport chain T. Light excites electrons in PSII U. Energy of I-T flow is used by ATP synthetase to make ATP
A)
P \[\to \]Q\[\to \]T\[\to \]S\[\to \]R\[\to \]U
done
clear
B)
S\[\to \]T\[\to \]P\[\to \]Q\[\to \]R\[\to \]U
done
clear
C)
T\[\to \]S\[\to \]R\[\to \]P\[\to \]Q\[\to \]U
done
clear
D)
T\[\to \]S\[\to \]R\[\to \]U\[\to \]Q\[\to \]P
done
clear
View Answer play_arrow
A
Animal Species Members Bird I 1 Bird II 1 Bird III 4
B
Animal Species Members Bird I 2 Bird II 2 Mammal III 2
C
Animal Species Members Bird I 2 Mammal II 2 Insect III 2
Which of the following habitat (boxes) show maximum, medium and minimum biodiversity?
A)
A-Minimum diversity, B-Medium diversity, C-Maximum diversity
done
clear
B)
A-Maximum diversity, B- Medium diversity, C-Minimum diversity
done
clear
C)
A-Maximum diversity, B-Minimum diversity, C-Medium diversity
done
clear
D)
A-Minimum diversity, B-Maximum diversity C-Medium diversity
done
clear
View Answer play_arrow
Which is/are true regarding ecosystem?
A. It is a self-sustained unit B. Cyclic exchange of materials between living beings and environment C. Requirement of input of energy D. Characterized by a major vagetation type
A)
A, C
done
clear
B)
A, D
done
clear
C)
A, B, C
done
clear
D)
A, B, C, D
done
clear
View Answer play_arrow
Match list-I with list-II and select the correct answer-
List-I List-II [A] Auxin (i) Apical dominance [B] Giberelin (ii) Cell division [C] Cytokinin (iii) Fruit ripening [D] Ethylene (iv) Internodal elongation
A)
A-(i) B-(iv) C-(iii) D-(ii)
done
clear
B)
A-(iv) B-(i) C-(ii) D-(iii)
done
clear
C)
A-(iv) B-(i) C-(iii) D-(ii)
done
clear
D)
A-(i) B-(iv) C-(ii) D-(iii)
done
clear
View Answer play_arrow
During aerobic respiration, all the ATPs are synthesized as a result of-
A)
Oxidative phosphorylation
done
clear
B)
Oxidative and substrate level phosphorylation
done
clear
C)
Substrate level phosphorylation
done
clear
D)
Oxidative and photophosphorylation
done
clear
View Answer play_arrow
Which one of the following statement is best explanation of high photosynthetic efficiency of \[{{C}_{4}}\] plants-
A)
Presence of photosynthetic carbon oxidation cycle
done
clear
B)
Presence of bundle sheath cells
done
clear
C)
Absence of glycolate metabolism
done
clear
D)
High Km constant of enzyme Rubisco for carboxylation reaction
done
clear
View Answer play_arrow
Which of the following is incorrect about ecosystem?
A)
Continuous flow of energy in its biotic components
done
clear
B)
Cycling of nutrient among its biotic and abiotic components
done
clear
C)
Decomposition by decomposers regulates nutrients cycling
done
clear
D)
It can successfully thrive well prolonged in the absence of herbivores
done
clear
View Answer play_arrow
A plant species is extinct from the forest nearby to your village. Which is the probable cause of this biodiversity loss?
A)
Pollution
done
clear
B)
Alien species invasion
done
clear
C)
Habitat loss
done
clear
D)
Coextinction
done
clear
View Answer play_arrow
Choose the in situ conservation strategies-
[A] National Parks [B] Germplasm collection [C] Sacred Grooves [D] Arborata [E] Hot spots [F] Biosphere reserves
A)
A, B, C, E, F
done
clear
B)
A, B, D, E, F
done
clear
C)
A, C, E, F
done
clear
D)
A, B, E, F
done
clear
View Answer play_arrow
The ecologists discover that regardless of taxonomic group or region the value of ?Z? lies in the range of-
A)
0.6 to 1.2
done
clear
B)
1 to 1.15
done
clear
C)
0.1 to 0.2
done
clear
D)
0.5 to 0.8
done
clear
View Answer play_arrow
Secondary producers are-
A)
Herbivores
done
clear
B)
Carnivores
done
clear
C)
Producers
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Ecologically a ideal parasite is-
[A] Complete life cycle in host [B] Increase the fitness of host [C] Derive the nutrition from host but not harm the host [D] Exploit the host but not kill the host
A)
A, B, C, D
done
clear
B)
A, C, D
done
clear
C)
B, C
done
clear
D)
A, D
done
clear
View Answer play_arrow
The development of fruit without fertilization is called as-
A)
Parthenogenesis
done
clear
B)
Parthenocarpy
done
clear
C)
Vivipary
done
clear
D)
Apomixis
done
clear
View Answer play_arrow
Every living cell has a-
A)
Cell wall
done
clear
B)
Ribosome
done
clear
C)
Plasma membrane
done
clear
D)
[b] & [c] both
done
clear
View Answer play_arrow
Golgi body is the site for-
A)
Cholesterol synthesis
done
clear
B)
Glycolipid synthesis
done
clear
C)
Lipoprotein synthesis
done
clear
D)
All of the above
done
clear
View Answer play_arrow
The tonoplast facilitates the transport-of a number of ion and other molecule against concentration gradient into vacuole. This type of transport is known as-
A)
Simple diffusion
done
clear
B)
Facilitated diffusion
done
clear
C)
Active transport
done
clear
D)
Bulk transport
done
clear
View Answer play_arrow
In which of the following mitosis can be observed?
A)
Primary oocyte
done
clear
B)
Spore mother cell
done
clear
C)
Plant spore
done
clear
D)
Plant gamete
done
clear
View Answer play_arrow
How many bivalent will present in cell, if embryo contain 24 chromosome?
A)
36
done
clear
B)
24
done
clear
C)
12
done
clear
D)
Cannot be determined
done
clear
View Answer play_arrow
Which of the following is correctly matched-
A)
Toxin - Abrin
done
clear
B)
Lectins - Vinblastein
done
clear
C)
Alkaloid - Concanavalin -A
done
clear
D)
Pigments - Morphin
done
clear
View Answer play_arrow
Nucleic acids are polymer of -
A)
Nucleoside
done
clear
B)
Nucleotide
done
clear
C)
Nitrogen Base
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Reading of template during transcription.....A.....and synthesis of RNA is.....B..... What is A & B-
A)
\[A=5'\to 3,\,\,B\,\,3\to \,\,5\]
done
clear
B)
\[A=3\,\,\to \,\,5,\,\,B\,\,5\,\,\to \,\,3\]
done
clear
C)
1 & 2 Both
done
clear
D)
\[A=3'\to 5',\,\,B\,\,3'\to \,\,5'\]
done
clear
View Answer play_arrow
How many of the following have epipetalous stamen. Tulip, Petunia, Lily, Ashwagandha, Asparagus, Aloe, Indigofera, Sesbania
A)
Six
done
clear
B)
Three
done
clear
C)
Two
done
clear
D)
Four
done
clear
View Answer play_arrow
Supporting root coming out from lower node is present in-
A)
Sugarcan
done
clear
B)
Banyan
done
clear
C)
Gulmohar
done
clear
D)
All of the above
done
clear
View Answer play_arrow
A distinct monocot character shown by monocot families is
A)
Hypogynous flower
done
clear
B)
Actinomorphic flower
done
clear
C)
Trimerous flower
done
clear
D)
Bisexual flower
done
clear
View Answer play_arrow
An axillary bud is present at the base of -
A)
Simple leaf
done
clear
B)
Compound leaf
done
clear
C)
Leaflet
done
clear
D)
[a] & [b] both
done
clear
View Answer play_arrow
Identify the correct feature of the family to which given floral formula belongs.
A)
Presence of actinomorphic flowers and cruciform corolla
done
clear
B)
Androecium is commonly diadelphous or monadelphous
done
clear
C)
Presence of cymose inflorescence
done
clear
D)
Fruit is a berry or capsule
done
clear
View Answer play_arrow
Which of the following is correctly matched?
A. Collenchyma Mechanical support to young dicot root B. Parenchyma Storage and secretion of material C. Ray parenchyma Radical conduction of food & water D. Sclerenchyma Lignified and pitted cell wall
A)
A, B, C & D
done
clear
B)
B, C, D
done
clear
C)
B & D
done
clear
D)
A, B & D
done
clear
View Answer play_arrow
In papaya male and female flowers present on different plant. It will prevents-
A)
Autogamy
done
clear
B)
Xenogamy
done
clear
C)
Geitonogamy
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
Which of the following is not the feature of tapetum
A)
Dense cytoplasm
done
clear
B)
Multinucleate
done
clear
C)
Providing nutrition to megaspore
done
clear
D)
Inner most layer of anther
done
clear
View Answer play_arrow
Flower present in Commellina is -
A)
Cleistogamous
done
clear
B)
Chasmogamous
done
clear
C)
[a] & [b] both
done
clear
D)
Self incompatible
done
clear
View Answer play_arrow
A typical dicot embryo is characterized with
A)
Major part of root developed from hypophysis
done
clear
B)
Cotyledons formed from entire octant proembryo
done
clear
C)
Hypocotyl terminates as radicle
done
clear
D)
Embryonal axis on either side of cotyledons
done
clear
View Answer play_arrow
Floridean starch is structurally similar to-
A)
amylose
done
clear
B)
amylopectin
done
clear
C)
glycogen
done
clear
D)
Both [b] & [c]
done
clear
View Answer play_arrow
Select the correct options w.r.t bryophytes-
a. Usually occur in damp, humid and shaded localities. b. Less differentiated than algae c. Main plant body is diploid Gametophyte d. Have little economic importance e. Have great ecological importance
A)
a, d and e
done
clear
B)
a, c, d and e
done
clear
C)
a, c and d
done
clear
D)
a, b, d and e
done
clear
View Answer play_arrow
What is the origin of asexual spore and sexual spores produced in members of ascomycetes respectively?
A)
Exogenous, Endogenous
done
clear
B)
Endogenous, Exogenous
done
clear
C)
Exogenous, Exogenous
done
clear
D)
Endogenous, Endogenous
done
clear
View Answer play_arrow
Euglenoids are-
A)
Parasitic
done
clear
B)
Photosynthetic
done
clear
C)
Holozoic
done
clear
D)
Both [b] & [c]
done
clear
View Answer play_arrow
Which of the following group of diseases is caused by viruses?
A)
Mumps, smallpox, herpes, influenza
done
clear
B)
AIDS, diabetes, herpes, tuberculosis
done
clear
C)
Anthrax, cholera, tetanus, tuberculosis
done
clear
D)
Cholera, tetanus, smallpox, influenza
done
clear
View Answer play_arrow
The main plant body in pteridophyte is-
A)
Sporophyte (2n) which is differentiated into root, stem and leaf
done
clear
B)
Sporophyte having no root, stem and leaf
done
clear
C)
Gametophyte (n) which is differentiated into root, stem and leaf
done
clear
D)
Gametophyte having no root, stem and leaf
done
clear
View Answer play_arrow
In gymnosperm, the leaves are well-adapted to withstand extremes of temperature, humidity and wind. What are the xeric characters in conifers?
A)
Needle-like leaves
done
clear
B)
Thick cuticle
done
clear
C)
Sunken stomata
done
clear
D)
All of these
done
clear
View Answer play_arrow
Botanical garden mainly serve the purpose of providing
A)
beautiful area for recreation
done
clear
B)
reservoir for tropical plants
done
clear
C)
ex situ conservation of germplasm
done
clear
D)
natural habitat for wildlife
done
clear
View Answer play_arrow
Select the correct options w.r.t. bacteria-
[A] Most extensive metabolic diversity [B] Simple in behaviour [C] Complex in structure [D] Mostly heterotrophic
A)
a, b, c
done
clear
B)
a, d
done
clear
C)
b, c
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which of the most infectious disease is?
A)
Hepatitis-B
done
clear
B)
AIDS
done
clear
C)
Cough and cold
done
clear
D)
Malaria
done
clear
View Answer play_arrow
Interferons are synthesized in response to-
A)
Mycoplasma
done
clear
B)
Bacteria
done
clear
C)
Viruses
done
clear
D)
Fungi
done
clear
View Answer play_arrow
Read the given statements and select the correct option.
(i) Right end of a polysaccharide chain is called reducing end while left end is called non-reducing end. (ii) Starch can hold iodine molecules in its helical secondary structure but cellulose being non-helical, cannot hold iodine (iii) Starch and glycogen are branched molecules. (iv) Starch and glycogen are the reserve food materials of plants and animals, respectively.
A)
Statements (i) and (ii) are correct
done
clear
B)
Statements (ii) and (iii) are correct
done
clear
C)
Only statements (iv) is correct
done
clear
D)
All statements are correct
done
clear
View Answer play_arrow
Gynaecomastia state can be seen in-
A)
Down's syndrome
done
clear
B)
Klinefelter's syndrome
done
clear
C)
Turner syndrome
done
clear
D)
Edward's syndrome
done
clear
View Answer play_arrow
The allele for tallness is dominant over that of dwarfness. This is called-
A)
Law of independent assortment
done
clear
B)
Law of segregation
done
clear
C)
Law of unit character
done
clear
D)
Law of dominance
done
clear
View Answer play_arrow
What ratio is expected in offsprings if father is colour blind and mother's father was colour blind?
A)
50% daughter - colour blind
done
clear
B)
All the sons are colour blind
done
clear
C)
All the daughters colour blind
done
clear
D)
All the sons are normal
done
clear
View Answer play_arrow
If the sequence of bases in one strand of DNA is TGCATGCA, what would be the sequence of bases on complementary strand?
A)
ATGCATGCA
done
clear
B)
AUGCAUGCA
done
clear
C)
TACGTACGT
done
clear
D)
UACGUACGU
done
clear
View Answer play_arrow
Which of the following is true about SCP (Single Cell Proteins)?
A)
Substrate for SCP is saw dust, rice husk etc.
done
clear
B)
SCP are produced by yeast, algae, fungi
done
clear
C)
Not rich in fats and carbohydrates
done
clear
D)
None of these
done
clear
View Answer play_arrow
Golden rice is a transgenic crop of the future with the following improved trait-
A)
Insect resistance
done
clear
B)
High protein content
done
clear
C)
High vitamin-A content
done
clear
D)
High lysine (essential amino acid) content
done
clear
View Answer play_arrow
Archaeologists unearthed a human skull with a / small dried fragment of the scalp still attached. They extracted a tiny amount off DNA from the scalp tissue. How could they obtain sufficient DNA for an analysis of the ancient man?s genes?
A)
use a molecular probe
done
clear
B)
subject DNA to electrophoresis
done
clear
C)
use the polymerase chain reaction
done
clear
D)
subject DNA to restriction enzymes
done
clear
View Answer play_arrow
Similarity in DNA and RNA:
A)
Both are polymer of nucleotides
done
clear
B)
Both have similar pyrimidine
done
clear
C)
Both have similar sugar
done
clear
D)
Both are genetic material
done
clear
View Answer play_arrow
Length of one loop of B-DNA-
A)
3.4 nm.
done
clear
B)
0.34 nm.
done
clear
C)
20 nm.
done
clear
D)
10 nm.
done
clear
View Answer play_arrow
Method of DNA replication in which two strands of DNA separates and synthesize new strands -
A)
Dispersive
done
clear
B)
Conservative
done
clear
C)
Semiconservative
done
clear
D)
Non conservative
done
clear
View Answer play_arrow
Female genital pore in earthworm is located in-
A)
First clitellar segment
done
clear
B)
\[1{{8}^{th}}segment\]
done
clear
C)
\[1{{4}^{th}}clitellar segment\]
done
clear
D)
\[1{{3}^{th}}segment\]
done
clear
View Answer play_arrow
Which of the following structures is correctly matched with its description?
A)
Septal nephridia - Both are exonephric and pharyngeal nephridia
done
clear
B)
Typhlosole - Helps in grinding the soil particles and decaying leaves
done
clear
C)
Sensory system - Possesses light and touch receptors in earthworm
done
clear
D)
Gizzard - Internal median fold present in the dorsal wall of the intestine of earthworm
done
clear
View Answer play_arrow
Nephridia that opens on the body surface is-
A)
Integumentary
done
clear
B)
Septal
done
clear
C)
Pharyngeal
done
clear
D)
All
done
clear
View Answer play_arrow
In cockroach leathery & dark colour wings are present in-
A)
Prothorax
done
clear
B)
Mesothorax
done
clear
C)
Metathorax
done
clear
D)
Wings of cockroach are membranous not leathery
done
clear
View Answer play_arrow
Number of ganglia in thorax of cockroach is/are
A)
One
done
clear
B)
Four
done
clear
C)
Two
done
clear
D)
Three
done
clear
View Answer play_arrow
Silicous covering present in-
A)
Marine ciliates
done
clear
B)
Fresh water ciliates
done
clear
C)
Marine sarcodins
done
clear
D)
Fresh water sarcodins
done
clear
View Answer play_arrow
Coelentrates with skeleton composed of \[CaC{{O}_{3}}\] is-
A)
Polyp
done
clear
B)
Medusa
done
clear
C)
Corals
done
clear
D)
Anemones
done
clear
View Answer play_arrow
Development through many larval stages are present in-
A)
Nemathelminthese
done
clear
B)
Annelida
done
clear
C)
Mollusca
done
clear
D)
Platyhelminthese
done
clear
View Answer play_arrow
Basic characters of vertebrates are-
A)
Ventral heart
done
clear
B)
Kidney for excretion
done
clear
C)
Paired appendages
done
clear
D)
All of above
done
clear
View Answer play_arrow
Suctorial mouth is characteristic feature of-
A)
Cyclostomes
done
clear
B)
Chondrichthyse
done
clear
C)
Osteichthyse
done
clear
D)
Urochordates
done
clear
View Answer play_arrow
Number of gills cleft in osteichthyse are-
A)
6-15 pair
done
clear
B)
6-8 pair
done
clear
C)
4 pair
done
clear
D)
One pair
done
clear
View Answer play_arrow
Which of the following situations would result in the greatest degree of \[{{O}_{2}}\] saturation for haemoglobin, assuming \[P{{O}_{2}}\] remains constant-
A)
Increased \[C{{O}_{2}}\] levels, decreased temperature
done
clear
B)
Increased \[C{{O}_{2}}\] levels, increased temperature
done
clear
C)
Decreased \[C{{O}_{2}}\] levels, decreased temperature
done
clear
D)
Decreased \[C{{O}_{2}}\] levels, increased temperature
done
clear
View Answer play_arrow
What is the correct path of a reflex arc?
A)
Sensory stimulus \[\to \] Dendrite \[\to \] Axon
done
clear
B)
Motor nerves \[\to \] Acetylcholine \[\to \] Adjuster neuron
done
clear
C)
Efferent nerves \[\to \] Connector nerves \[\to \] Motor nerves
done
clear
D)
Afferent nerves \[\to \] Efferent nerves \[\to \] Connector nerves
done
clear
View Answer play_arrow
A human female is born with a million of eggs (primary oocyte) at the time of birth, only some 500 eggs get a chance of maturity. What is the density of rest of the eggs?
A)
Rest of the eggs differentiate back to thecal and granulosa cells.
done
clear
B)
Rest of the eggs nurture the dominant follicular cell
done
clear
C)
Rest of the eggs move out of the ovary and are destroyed by leucocytes.
done
clear
D)
Rest of the eggs break down and are absorbed i.e. degenerative follicular atresia
done
clear
View Answer play_arrow
If a woman consumes some drug which is rich in L.H., then which of the phenomenon occurs?
A)
Mensturation stops
done
clear
B)
Ovulation will occur
done
clear
C)
Activation of oestrogen occurs
done
clear
D)
Activation of FSH occurs
done
clear
View Answer play_arrow
Match the names of the parts listed under column-I with the names of the organs listed under column-II; Choose the answer which gives the correct combination of the alphabets.
Column-I Column-II (Parts) (Organs) [A] Bicuspid value [P] Brain [B] Nephron [Q] Liver [C] Alveoli [R] Heart [D] Cerebrum [S] Kidney
A)
A = R; B = T; C = S; D = P
done
clear
B)
A = R; B = S; C = T; D = P
done
clear
C)
A = S; B = Q; C = P; D = T
done
clear
D)
A = S; B = R; C = P; D = T
done
clear
View Answer play_arrow
Read the following statements carefully.
(i) In fishes the heart pumps out deoxygenated blood which is oxygenated by the gills and supplied to the body parts from where deoxygenated blood is returned to the heart. (ii) The openings of the right and the left ventricles into pulmonary artery and aorta respectively are provided with the mitral valves. The nodal musculature has the ability to generate action potentials without any external stimuli, i.e. it is autoexcitable (iv) The T-wave of ECG represents depolarisation of the ventricles. Which of the above two statements are incorrect?
A)
(i) and (iii)
done
clear
B)
(ii) and (iv)
done
clear
C)
(i) and (ii)
done
clear
D)
(iii) and (iv)
done
clear
View Answer play_arrow
Most terrestrial insects get rid of bulk of their nitrogenous wastes as-
A)
amino acids
done
clear
B)
urea
done
clear
C)
uric acid
done
clear
D)
ammonia
done
clear
View Answer play_arrow
The urination becomes voluntary when urine comes in-
A)
pelvis
done
clear
B)
ureter
done
clear
C)
urinary bladder
done
clear
D)
urethra
done
clear
View Answer play_arrow
Which one of the following phenomena supports Darwin's concept of natural selection in organic evolution?
A)
Development of transgenic animals
done
clear
B)
Production of 'Dolly', the sheep by cloning
done
clear
C)
Prevalence of pesticide resistant insects
done
clear
D)
Development of organs from 'stem cells' for organ transplantation
done
clear
View Answer play_arrow
Which one of the following sequences was proposed by Darwin and Wallace for organic evolution?
A)
Overproduction, variations, constancy of population size, natural selection
done
clear
B)
Variations, constancy of population size, overproduction, natural selection
done
clear
C)
Overproduction, constancy of population size, variations, natural selection
done
clear
D)
Variations, natural selection, overproduction, constancy of population size
done
clear
View Answer play_arrow
Read the following statements and select the option having both incorrect statements.
(i) Condoms decrease sperm motility (ii) Diaphragms, cervical caps and vaults are or both males and females (iii) IUDs are inserted by expert nurses (iv) Sterilisation is a terminal method to prevent further pregnancy
A)
(i) and (ii)
done
clear
B)
(i) and (ii)
done
clear
C)
(iii) and (iv)
done
clear
D)
(ii) and (iv)
done
clear
View Answer play_arrow
Match list-I with list-II and select the correct answer from the code given below this lists
List-I List-II A Neoteny I Axolotol larva B Parthenogenesis II Dragon fly C Retrogressive metamorphosis III Honey bee D Incomplete metamorphosis IV Ascidian larva
A)
A-I B-II C-III D-IV
done
clear
B)
A-IV B-III C-II D-I
done
clear
C)
A-IV B-III C-I D-II
done
clear
D)
A-I B-III C-IV D-II
done
clear
View Answer play_arrow
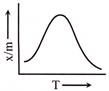
 From the above graph it can be concluded that-
From the above graph it can be concluded that- 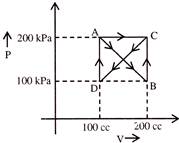


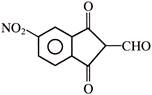
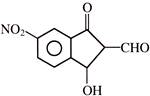

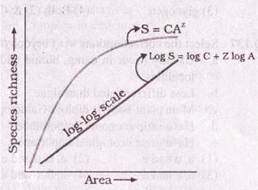 The ecologists discover that regardless of taxonomic group or region the value of ?Z? lies in the range of-
The ecologists discover that regardless of taxonomic group or region the value of ?Z? lies in the range of- 