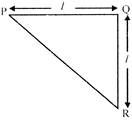
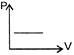
In the triangular sheet shown, \[PQ=QR=l,\]If m is the mass of the sheet, its moment of inertia about PR is
A)
\[\frac{m{{l}^{2}}}{24}\]
done
clear
B)
\[\frac{m{{l}^{2}}}{12}\]
done
clear
C)
\[\frac{m{{l}^{2}}}{6}\]
done
clear
D)
\[\frac{m{{l}^{2}}}{18}\]
done
clear
View Answer play_arrow
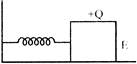
A spring-block system undergoes simple harmonic motion on a smooth horizontal surface. The block is now given some positive charge, and a uniform horizontal electric field to the right is switched on. As a result,
A)
The time period of oscillation will increase
done
clear
B)
The time period of oscillation will decrease
done
clear
C)
the time period of oscillation will remain unaffected
done
clear
D)
the mean position of simple harmonic motion will shift to the left
done
clear
View Answer play_arrow
On a smooth inclined plane a body of mass M is attached between two springs. The other ends of the springs are fixed to firm supports. If each spring has a force constant K, the period of oscillation of the body is (assuming the spring as massless).
A)
\[2\pi \sqrt{\frac{m}{2K}}\]
done
clear
B)
\[2\pi \sqrt{\frac{2m}{2K}}\]
done
clear
C)
\[2\pi \sqrt{\frac{m\,\sin \theta }{2K}}\]
done
clear
D)
\[2\pi \sqrt{\frac{2m\,\sin \theta }{K}}\]
done
clear
View Answer play_arrow
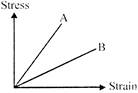
The stress strain graph for two materials is shown in the figure. If the stress Young's modulus for two materials are \[{{Y}_{A}}\] and \[{{Y}_{B}}\] then:
A)
\[{{Y}_{A}}={{Y}_{B}}\]
done
clear
B)
\[{{Y}_{A}}>{{Y}_{B}}\]
done
clear
C)
\[{{Y}_{A}}<{{Y}_{B}}\]
done
clear
D)
can't be predicted from the graph
done
clear
View Answer play_arrow
If n identical drops of mercury are combined to form a bigger drop, then the capacity of bigger drop, if capacity of each drop of mercury C=is
A)
\[{{n}^{1/3}}C\]
done
clear
B)
\[{{n}^{2/3}}C\]
done
clear
C)
\[{{n}^{1/4}}C\]
done
clear
D)
\[nC\]
done
clear
View Answer play_arrow
If R is the radius of earth, \[\omega \] is its angular velocity and \[{{g}_{p}}\] is the value of acceleration due to gravity at the poles, then effective value of acceleration due to gravity at the latitude, \[\lambda ={{60}^{o}}\] will be equal to:
A)
\[{{g}_{p}}-\frac{1}{4}R{{\omega }^{2}}\]
done
clear
B)
\[{{g}_{p}}-\frac{3}{4}\,\,{{\omega }^{2}}\]
done
clear
C)
\[{{g}_{p}}-R\,{{\omega }^{2}}\]
done
clear
D)
\[{{g}_{p}}+\frac{1}{2}R\,{{\omega }^{2}}\]
done
clear
View Answer play_arrow
A particle of mass is projected with velocity u at an angle \[\theta \] with horizontal. During the period when the particle descends from highest point to the position where its velocity vector makes an angle \[\frac{\theta }{2}\]with horizontal, the work done by the gravity force is:
A)
\[\frac{1}{2}m{{u}^{2}}{{\tan }^{2}}\frac{\theta }{2}\]
done
clear
B)
\[\frac{1}{2}m{{u}^{2}}{{\tan }^{2}}\theta \]
done
clear
C)
\[\frac{1}{2}m{{u}^{2}}co{{s}^{2}}\theta {{\tan }^{2}}\frac{\theta }{2}\]
done
clear
D)
\[\frac{\theta }{2}m{{u}^{2}}{{\cos }^{2}}\frac{\theta }{2}{{\sin }^{2}}\theta \]
done
clear
View Answer play_arrow
A particle is moving along a circular path of radius 5m and with uniform speed 5m/s. What will be the average acceleration, when the particles completes half revolution?
A)
zero
done
clear
B)
\[10m/{{s}^{2}}\]
done
clear
C)
\[10\pi m/{{s}^{2}}\]
done
clear
D)
\[\frac{10}{\pi }m/{{s}^{2}}\]
done
clear
View Answer play_arrow
We have error in the measurments of length, radius mass and current of a wire are 2%, 3% and 1% then error on its density will be:
A)
\[11%\]
done
clear
B)
\[8%\]
done
clear
C)
\[10%\]
done
clear
D)
\[7%\]
done
clear
View Answer play_arrow
The force F is given in terms of time t and dispa-cement x by the equation, \[f=A\,\cos Bx+C\,\sin \,Dt\]. The dimensions of \[\frac{D}{B}\] are:
A)
\[[{{M}^{o}}{{L}^{o}}{{T}^{o}}]\]
done
clear
B)
\[[{{M}^{o}}{{L}^{o}}{{T}^{-1}}]\]
done
clear
C)
\[[{{M}^{o}}{{L}^{-1}}{{T}^{o}}]\]
done
clear
D)
\[[{{M}^{o}}L{{T}^{-1}}]\]
done
clear
View Answer play_arrow
A cube of side b has a charge q at each of its virtues. The electric potential at the centre of the cube is:
A)
\[\frac{4q}{\sqrt{3\pi }{{\varepsilon }_{0}}b}\]
done
clear
B)
\[\frac{\sqrt{3}q}{\pi {{\varepsilon }_{0}}b}\]
done
clear
C)
\[\frac{2q}{\pi {{\varepsilon }_{0}}b}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
The potential energy U(x) of a particle executing SHM is given by:
A)
\[U(x)=\frac{K\,{{(x-a)}^{2}}}{2}\]
done
clear
B)
\[U(x)={{K}_{1}}x+{{K}_{2}}{{x}^{2}}+{{K}_{3}}{{x}^{3}}\]
done
clear
C)
\[U(x)=A{{e}^{-bx}}\]
done
clear
D)
U(x) = constant
done
clear
View Answer play_arrow
The energy required to remove a body of mass m from earth?s surface is equal to:
A)
\[2mg\,R\]
done
clear
B)
\[mg\text{ }R\]
done
clear
C)
\[-mg\text{ }R\]
done
clear
D)
zero
done
clear
View Answer play_arrow
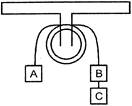
Three block A,B and C of equal to weights of mass 2 kg each are hanging on a string passing over a fixed pulley as shown in figure. What is the tension in the string connected between blocks B and C:
A)
zero
done
clear
B)
\[13\,N\]
done
clear
C)
\[3.3\,N\]
done
clear
D)
\[19.6\,N\]
done
clear
View Answer play_arrow
A uniform chain of length l and mass m overhangs a smooth table with its one third part lying on the table. The kinetic energy of the chain as it completely slips off the table is
A)
\[\frac{2}{9}mgl\]
done
clear
B)
\[\frac{4}{9}mgl\]
done
clear
C)
\[\frac{3}{4}mgl\]
done
clear
D)
\[\frac{5}{18}mgl\]
done
clear
View Answer play_arrow
Two vectors are .given as \[A=3i-5j+5k,\]\[B=5i-j+4k,\] choose the correct statements:
A)
Angle between A and B is \[0{}^\circ \]
done
clear
B)
Angle between A and B is \[90{}^\circ \]
done
clear
C)
Angle between A and B is greater than \[{{90}^{o}}\]
done
clear
D)
Angle between A and B is less than \[{{90}^{o}}\]
done
clear
View Answer play_arrow
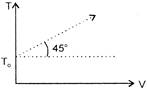
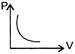
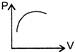
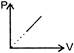
The given curve represents the variation of temp-erature as a function of volume for one mole of an ideal gas. Which of the following curves represents the variation of pressure as a function of volume?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The temperature of a substance increases by \[{{27}^{o}}C\]. On the Kelvin scale, this increase is equal to:
A)
\[300\,K\]
done
clear
B)
\[2.46\,K\]
done
clear
C)
27K
done
clear
D)
7K
done
clear
View Answer play_arrow
If \[{{\lambda }_{1}},{{\lambda }_{2}}\] and \[{{\lambda }_{3}}\] are the wavelengths of the waves giving responce with the fundamental, first and second overtones respectively of a closed organ pipe, then the ratio of wavelengths \[{{\lambda }_{1}}:{{\lambda }_{2}}:{{\lambda }_{3}}\] is:
A)
\[1:2:3\]
done
clear
B)
\[1:\frac{1}{3}:\frac{1}{5}\]
done
clear
C)
\[1:3:5\]
done
clear
D)
\[5:3:1\]
done
clear
View Answer play_arrow
The centre of a wheel rolling on a plane surface, moves with a speed \[{{V}_{0}}\]. A particle on their rim of the wheel at the same level as that centre will be moving at speed:
A)
zero
done
clear
B)
\[{{V}_{0}}\]
done
clear
C)
\[\sqrt{2{{V}_{0}}}\]
done
clear
D)
\[2{{V}_{0}}\]
done
clear
View Answer play_arrow
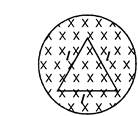
An equilateral triangular loop having a resistance R and length of each side I is placed in a magnetic field which is varying at \[\frac{d\,\,B}{dt}=\frac{l\,T}{S}\]. The induced current in the loop will be:
A)
\[\frac{\sqrt{3}}{4}\,\frac{{{l}^{2}}}{R}\]
done
clear
B)
\[\frac{4}{\sqrt{3}}\,\frac{{{l}^{2}}}{R}\]
done
clear
C)
\[\frac{\sqrt{3}}{4}\,\frac{R}{{{l}^{2}}}\]
done
clear
D)
\[\frac{4}{\sqrt{3}}\,\frac{R}{{{l}^{2}}}\]
done
clear
View Answer play_arrow
A conductor in the form of the right triangle ABC with \[AB=3cm\]and \[BC=4cm\]carries a current of 10A. There is a uniform magnetic field of 5T perpendicular to the plane of the conductor. The force on the conductor AC will be:
A)
\[1.5\,N\]
done
clear
B)
\[2.0\,N\]
done
clear
C)
\[2.5\,N\]
done
clear
D)
\[3.5\,N\]
done
clear
View Answer play_arrow
A particle of mass m and charge q moves with a constant velocity V along the positive x-direction. It enters a region containing a uniform magnetic field B directed along the negative z-direction, extending from X = a to X = b. The minimum value of V required, so that the particular can just enter the region \[X>b\] is:
A)
\[\frac{qbB}{m}\]
done
clear
B)
\[\frac{q(b-a)B}{m}\]
done
clear
C)
\[\frac{qa\,B}{m}\]
done
clear
D)
\[\frac{q\,(b+a)}{2m}\]
done
clear
View Answer play_arrow
The distance between two thin long straight parallel conducting wires is b. On passing the same current i with same same sense in them, the force per unit length between them will be:
A)
zero
done
clear
B)
\[\frac{{{\mu }_{0}}{{i}^{2}}}{2\pi b}\]
done
clear
C)
\[\frac{{{\mu }_{0}}i}{2\pi b}\]
done
clear
D)
\[\frac{{{\mu }_{0}}i}{2\pi }\]
done
clear
View Answer play_arrow
A moving coil voltmeter is generally used in laboratory to measure the potential difference across a conductor of resistance r, and carrying current I. The voltmeter has a resistance R and will measure the potential difference more accurately as:
A)
R approaches r
done
clear
B)
R becomes larger than r
done
clear
C)
R becomes smaller than r
done
clear
D)
R equal to zero
done
clear
View Answer play_arrow
The current at which a fuse wire melts docs not depend on:
A)
cross-sectional area
done
clear
B)
length
done
clear
C)
resistivity
done
clear
D)
density
done
clear
View Answer play_arrow
The current in a wire varies with time according to the equation \[\text{l}=4+2t,\]where l is in ampere and t is in second. The quantity of charge which has passed through a cross section of the wire during the time \[t=2S\]to \[t=6S\] will be:
A)
60 C
done
clear
B)
24 C
done
clear
C)
48 C
done
clear
D)
30 C
done
clear
View Answer play_arrow
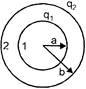
Two concentric conducting spheres having radii a and b are charged to \[{{q}_{1}}\] and \[{{q}_{2}}\] respectively. The potential difference between 1 and 2 will be:
A)
\[\frac{{{q}_{1}}}{4\pi {{\varepsilon }_{0}}a}-\frac{{{q}_{2}}}{4\pi {{\varepsilon }_{0}}b}\]
done
clear
B)
\[\frac{{{q}_{2}}}{4\pi {{\varepsilon }_{0}}}\left( \frac{1}{a}-\frac{1}{b} \right)\]
done
clear
C)
\[\frac{{{q}_{1}}}{4\pi {{\varepsilon }_{0}}}\left( \frac{1}{a}-\frac{1}{b} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Three charges Q, \[+q\] and \[+q\] are placed at the vertices of a right angle triangle (isosceles) as shown. The net electrostatic energy of the configuration is zero, if Q is equal to :
A)
\[\frac{-q}{1+\sqrt{2}}\]
done
clear
B)
\[\frac{2q}{2+\sqrt{2}}\]
done
clear
C)
\[-2q\]
done
clear
D)
\[+q\]
done
clear
View Answer play_arrow
A capacitors is connected to a battery. The force of attraction between the plates when the separation between them is halved:
A)
remains the same
done
clear
B)
becomes eight times
done
clear
C)
becomes four times
done
clear
D)
becomes two times
done
clear
View Answer play_arrow
Which of the following is known as universal logic gate?
A)
XOR
done
clear
B)
OR
done
clear
C)
NOR
done
clear
D)
None of these
done
clear
View Answer play_arrow
When \[_{3}^{7}Li\] nuclei are bombarded by protons, and the resultant nuclei are \[_{4}^{8}Be,\] the emitted particles will be:
A)
neutrons
done
clear
B)
alpha particle
done
clear
C)
beta particles
done
clear
D)
gamma rays
done
clear
View Answer play_arrow
A p - n junction diode cannot be used:
A)
as a rectifier
done
clear
B)
for getting light radiation
done
clear
C)
for converting light energy into electrical energy
done
clear
D)
as an amplifier
done
clear
View Answer play_arrow
Two photons of energy \[2.5\text{ }eV\] fall on a metal plate, which was work-function of\[4.0\text{ }eV\]. Number of electrons ejected from the metal surface is:
A)
one
done
clear
B)
two
done
clear
C)
zero
done
clear
D)
more than two
done
clear
View Answer play_arrow
If the de-Broglie wavelength of proton is \[1.0\times {{10}^{-13}}m,\]the electric potential through which it must have been accelerated is:
A)
\[4.07\times {{10}^{4}}V\]
done
clear
B)
\[8.2\times {{10}^{4}}V\]
done
clear
C)
\[8.2\times {{10}^{3}}V\]
done
clear
D)
\[4.07\times {{10}^{5}}V\]
done
clear
View Answer play_arrow
In an electromagnetic wave, the electric and magnetizing fields are \[100V{{m}^{-1}}\] and\[0.265\text{ }A{{m}^{-1}}\]. The maximum energy flow is:
A)
\[26.5\,W/{{m}^{2}}\]
done
clear
B)
\[26.4\,W/{{m}^{2}}\]
done
clear
C)
\[46.7\,W/{{m}^{2}}\]
done
clear
D)
\[765\,W/{{m}^{2}}\]
done
clear
View Answer play_arrow
In n-p-n transistor, the collector current is 10mA. If 90% of electrons emitted reach the collector, then:
A)
emitter current will be \[9\,mA\]
done
clear
B)
emitter current will be \[11.1\,mA\]
done
clear
C)
base current will be \[0.1\,mA\]
done
clear
D)
base current will be \[0.01\,mA\]
done
clear
View Answer play_arrow
A proton of mass m and charge \[+e\] is moving in a circular orbit in a magnetic field with energy \[1\,meV\]. What should be the energy of \[\alpha \] -particle (mass Am and charge \[=+2e\]), so that it can revolve in the path of same radius?
A)
\[1meV\]
done
clear
B)
\[4meV\]
done
clear
C)
\[4meV\]
done
clear
D)
\[0.5meV\]
done
clear
View Answer play_arrow
The critical angle of a medium is \[{{\sin }^{-1}}\left( \frac{3}{5} \right)\]. The polarizing angle of the medium will be:
A)
\[{{\sin }^{-1}}\left( \frac{4}{5} \right)\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{5}{3} \right)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \frac{3}{4} \right)\]
done
clear
D)
\[{{\tan }^{-1}}\left( \frac{4}{3} \right)\]
done
clear
View Answer play_arrow
In Young's double slit experiment the width of one slit is double that of the other, the ratio of intensity of a bright band to that of a dark band in the interference pattern will be:
A)
\[2:1\]
done
clear
B)
\[3:1\]
done
clear
C)
\[6:1\]
done
clear
D)
\[9:1\]
done
clear
View Answer play_arrow
Light travels through a glass plate of thickness t and having refractive index \[\mu \]. If c be the velocity of light in vacuum, the time taken by the light to travel this thickness of glass is:
A)
\[\frac{t}{\mu c}\]
done
clear
B)
\[t\,\mu c\]
done
clear
C)
\[\frac{\mu t}{c}\]
done
clear
D)
\[\frac{tc}{\mu }\]
done
clear
View Answer play_arrow
A ray of light falls normally on a refracting face of a prism of refractive index \[1.5\]. If the ray just fails to emerge from the prism. Then, the angle of prism is:
A)
\[{{\sin }^{-1}}\left( \frac{2}{3} \right)\]
done
clear
B)
\[{{\cos }^{-1}}\left( \frac{2}{3} \right)\]
done
clear
C)
\[si{{n}^{-1}}\left( \frac{1}{2} \right)\]
done
clear
D)
\[si{{n}^{-1}}\left( \frac{1}{3} \right)\]
done
clear
View Answer play_arrow
The alternating emf applied and the current flowing in an AC circuits are represented by \[\varepsilon ={{\varepsilon }_{0}}\,\sin \omega t\] and \[I={{I}_{0}}\,\sin \,\left( \omega t+\frac{\pi }{2} \right)\]respectively. The power loss in the circuit will be:
A)
\[\frac{{{\varepsilon }_{0}}{{I}_{0}}}{4}\]
done
clear
B)
\[\frac{{{\varepsilon }_{0}}{{I}_{0}}}{\sqrt{2}}\]
done
clear
C)
\[\frac{{{\varepsilon }_{0}}{{I}_{0}}}{2}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
An average emf of 20V is induced in an inductor when the current in it, changed from \[2.5\,A\]in one direction to the same value in opposite direction in \[0.1\text{ }S,\]the self-inductance of inductor is:
A)
\[0.4\,H\]
done
clear
B)
\[1\,H\]
done
clear
C)
\[2\,H\]
done
clear
D)
\[0.6\,H\]
done
clear
View Answer play_arrow
A flat coil carrying a current has a magnetic moment \[\mu \]. It is placed in a magnetic field B. The torque on the coil is \[\tau \]. then;
A)
\[\tau =\mu .B\]
done
clear
B)
\[\tau =B\times \mu \]
done
clear
C)
\[|\tau |=\mu .B\]
done
clear
D)
It is perpendicular to both \[\mu \] and B
done
clear
View Answer play_arrow
The product is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
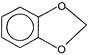
Which of the following is an aromatic compound?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following docs not give benzoic acid on hydrolysis?
A)
Phenyl cyanide
done
clear
B)
Bcnzoyl chloride
done
clear
C)
Benzyl chloride
done
clear
D)
Methyl benzoate
done
clear
View Answer play_arrow
The polymer which is used in non-sticky kit 'enwareis
A)
PVC
done
clear
B)
Teflon
done
clear
C)
rayon
done
clear
D)
isoprene
done
clear
View Answer play_arrow
Which one of the following statement is not correct?
A)
\[\alpha \]-carbon of \[\alpha \]-ammo acid is asymmetric
done
clear
B)
All proteins are found in Inform
done
clear
C)
I Turn an body can synthesise all proteins they need
done
clear
D)
At \[pH=7\]both ammo and carboxylic groups exist in ionised form.
done
clear
View Answer play_arrow
One 'S' orbital and one 'P? orbital hybridise then one gets:
A)
two orbitals at \[{{90}^{o}}\]
done
clear
B)
two hybrid orbitals which are mutually perpendicular
done
clear
C)
two hybrid orbitals at \[{{180}^{o}}\]
done
clear
D)
three orbitals in the same plane
done
clear
View Answer play_arrow
A bottle of dry ammonia and a bottle of dry hydrogen chloride connected through a long tube arc opened at the same time at both ends. The white ammonium chloride formed will be
A)
near the ammonia bottle
done
clear
B)
near the hydrogen chloride bottle
done
clear
C)
at the centre of the tube
done
clear
D)
through out the tube
done
clear
View Answer play_arrow
\[0.1M\] solution of an acid ionises to the extent of 10% at room temperature. The concentration of hydroxyl ions in g/ion/L would be:
A)
\[{{10}^{-2}}\]
done
clear
B)
\[{{10}^{-12}}\]
done
clear
C)
\[{{10}^{-11}}\]
done
clear
D)
\[{{10}^{-10}}\]
done
clear
View Answer play_arrow
Which one of the following statements regarding \[{{H}_{2}}{{O}_{2}}\]is false?
A)
It is a strong oxidising agent as well as reducing agent in acidic and also in basic medium.
done
clear
B)
It is decomposed in the presence of \[MnO\]
done
clear
C)
It is more stable in a basic solution
done
clear
D)
It behaves as a reducing agent towards acidified \[KMn{{O}_{4}}\],
done
clear
View Answer play_arrow
The two-third life of a first order reaction in which \[K=5.48\times {{10}^{-14}}\] per sec is:
A)
\[\frac{2.303}{5.48\times {{10}^{-14}}}\log 3\]
done
clear
B)
\[\frac{2.303}{5.48\times {{10}^{-14}}}\log 2\]
done
clear
C)
\[\frac{2.303}{5.48\times {{10}^{-14}}}\log \frac{1}{3}\]
done
clear
D)
\[\frac{2.303}{5.48\times {{10}^{-14}}}\log \frac{2}{3}\]
done
clear
View Answer play_arrow
For the reaction \[C{{u}_{(s)}}+2Ag_{(aq)}^{+}Cu_{(aq)}^{2+}+2A{{g}_{(g)}}\]. The equilibrium constant K is given by
A)
\[\frac{[C{{u}^{2+}}]\,{{[Ag]}^{2}}}{[Cu]\,[A{{g}^{2+}}]}\]
done
clear
B)
\[\frac{{{\left[ Ag \right]}^{2}}}{{{\left[ Ag_{2}^{+} \right]}^{2}}}\]
done
clear
C)
\[\frac{\left[ C{{u}^{2+}} \right]}{\left[ A{{g}^{+}} \right]}\]
done
clear
D)
\[\frac{\left[ C{{u}^{2+}} \right]}{\left[ Cu \right]}\]
done
clear
View Answer play_arrow
Find the pH value of the mixture containing \[50\text{ }c.c.\text{ }M-HCl\] and \[30\text{ }cc\text{ }M-NaOH\] solution assuming both to be completely ionised
A)
\[0.7051\]
done
clear
B)
\[0.6021\]
done
clear
C)
\[10.051\]
done
clear
D)
\[8.052\]
done
clear
View Answer play_arrow
For a d-electron the orbital angular momentum is
A)
\[\sqrt{6}\,\frac{h}{2\pi }\]
done
clear
B)
\[\sqrt{2}\,\frac{h}{2\pi }\]
done
clear
C)
\[\frac{\sqrt{h}}{2\pi }\]
done
clear
D)
\[\frac{2h}{2\pi }\]
done
clear
View Answer play_arrow
Molecular formula of glauber's salt is
A)
\[CuS{{O}_{4}}.5{{H}_{2}}O\]
done
clear
B)
\[ZnS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
C)
\[FeS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
D)
\[N{{a}_{2}}S{{O}_{4}}.10{{H}_{2}}O\]
done
clear
View Answer play_arrow
A solution contains \[F{{e}^{2+}}\], \[A{{g}^{+}},B{{i}^{3+}}\] and \[P{{b}^{2+}}\] ions in the same concentration. The value of reduction potential in volt are: \[Ag,\,A{{g}^{+}}=0.80\,V\] \[Fe,\,F{{e}^{3+}}=-0.44\,V\] \[Bi,\,B{{i}^{3+}}=0.21\,V\] \[Pb,\,P{{b}^{2+}}=-0.13\,V\] If sufficient voltage is applied to deposit any metal then the sequence of deposition will be
A)
\[Ag,Bi,Pb,Fe\]
done
clear
B)
\[Fe,Pb,Bi,Ag\]
done
clear
C)
\[Bi,Pb,Ag,Fe\]
done
clear
D)
\[Pb,Ag,Bi,Fe\]
done
clear
View Answer play_arrow
The colour of solution obtained by adding excess \[KI\] in the solution of \[HgC{{l}_{2}}\] is
A)
orange
done
clear
B)
brown
done
clear
C)
red
done
clear
D)
colourless
done
clear
View Answer play_arrow
Polyphosphates are used as water softening agents because they
A)
form soluble complexes with cationic species
done
clear
B)
precipitate cationic species
done
clear
C)
form soluble complexes with anionic species
done
clear
D)
precipitate anionic species
done
clear
View Answer play_arrow
Relative acidity of the following is in the order:
A)
\[RCOOH>{{H}_{2}}C{{O}_{3}}>{{C}_{6}}{{H}_{5}}OH>{{H}_{2}}O>ROH\]
done
clear
B)
\[RCOOH>ROH>{{H}_{2}}C{{O}_{3}}>{{C}_{6}}{{H}_{5}}OH>{{H}_{2}}O\]
done
clear
C)
\[ROH>RCOOH>{{H}_{2}}C{{O}_{3}}>{{C}_{6}}{{H}_{5}}OH>{{H}_{2}}O\]
done
clear
D)
\[RCOOH>{{C}_{6}}{{H}_{5}}OH>ROH>{{H}_{2}}C{{O}_{3}}>{{H}_{2}}O\]
done
clear
View Answer play_arrow
The bond energies of H-H and \[Cl-Cl\] are \[430\text{ }KJ\text{ }mo{{l}^{-1}}\] and \[242\text{ }KJ\text{ }mo{{l}^{-1}}1\] respectively. \[\Delta {{H}_{f}}\]. for \[HCl\]is\[91\text{ }KJ\text{ }mo{{l}^{-1}}\]. The bond energy of \[HCl\] will be:
A)
\[427\text{ }KJ\]
done
clear
B)
\[245\text{ }KJ\]
done
clear
C)
\[285\text{ }KJ\]
done
clear
D)
\[766\,KJ\]
done
clear
View Answer play_arrow
The reaction \[\underbrace{CO+{{H}_{2}}}_{water\,gas}+{{H}_{2}}\xrightarrow[673\,K,\,300\,atm]{Cr{{O}_{3}},\,ZnO}\] may be used for the manufacture of:
A)
\[HCHO\]
done
clear
B)
\[HCOOH\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow
Which is the least basic in nature?
A)
\[{{(C{{H}_{3}})}_{2}}NH\]
done
clear
B)
\[{{(C{{H}_{3}}C{{H}_{2}})}_{2}}NH\]
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Chlorophyll contains 2% by weight of magnesium. The number of magnesium atoms in 2g of chlorophyll is:
A)
\[{{10}^{21}}\]
done
clear
B)
\[{{10}^{23}}\]
done
clear
C)
\[{{10}^{24}}\]
done
clear
D)
\[6.023\times {{10}^{23}}\]
done
clear
View Answer play_arrow
The series of spectral lines which appear in the visible region of hydrogen spectrum is called:
A)
Balmer series
done
clear
B)
Brackett series
done
clear
C)
Lyman series
done
clear
D)
Pfund series
done
clear
View Answer play_arrow
Decomposition of \[PC{{l}_{5}}\] at 323 K is represented by \[PC{{l}_{5(g)}}PC{{l}_{3(g)}}+C{{l}_{2(g)}}\]. It the degree of \[PC{{l}_{5}}\] is \[\alpha \] and the total pressure developed is P, then the partial pressure of \[PC{{l}_{3}}\] at the given temperature
A)
\[\frac{\alpha }{1+\alpha }P\]
done
clear
B)
\[\frac{1-\alpha }{1+\alpha }P\]
done
clear
C)
\[P\alpha \]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Chlorination of benzene nucleus takes place in presence of lewis acid \[FeC{{l}_{3}}\]. The attacking species is
A)
\[C{{l}^{-}}\]
done
clear
B)
\[C{{l}^{+}}\]
done
clear
C)
\[Cl\]
done
clear
D)
\[FeCl_{4}^{-}\]
done
clear
View Answer play_arrow
Compound B is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Slaking is the process of adding water to
A)
\[CaO\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[CaS{{O}_{4}}\]
done
clear
D)
\[CaC{{l}_{2}}\]
done
clear
View Answer play_arrow
Which one of the following is incorrect?
A)
Boron halides are all monomeric while these of \[Al\] is dimeric
done
clear
B)
Boron halides and aluminium halides exist as monomeric.
done
clear
C)
Boron halides and aluminium halides are lewis acid
done
clear
D)
\[{{B}_{2}}{{O}_{3}}\] alone is acidic while \[A{{l}_{2}}{{O}_{3}}\] is amphoteric
done
clear
View Answer play_arrow
Which form of coal has the maximum percentage of carbon?
A)
Peat
done
clear
B)
Lignite
done
clear
C)
Anthracite
done
clear
D)
Bituminous
done
clear
View Answer play_arrow
Potassium chloride is used in the salt bridge because
A)
agar. agar forms a good jelly with it
done
clear
B)
it actively participates in the cell reaction
done
clear
C)
it is a strong electrolyte
done
clear
D)
\[{{K}^{+}}\] and \[C{{l}^{-}}\] ions have the same transference number.
done
clear
View Answer play_arrow
Which among the following will convert permanganate ion \[\left( MnO_{4}^{-} \right)\] to manganous \[\left( M{{n}^{2+}} \right)\] ion?
A)
\[PbC{{l}_{4}}\]
done
clear
B)
Cone. \[HCl\]
done
clear
C)
\[CrC{{l}_{3}}\]
done
clear
D)
Cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
Which of the following ligands is not a chelating agent?
A)
Pyridine
done
clear
B)
en
done
clear
C)
Oxalate
done
clear
D)
EDTA
done
clear
View Answer play_arrow
The metal that dissolves in liquid ammonia giving a dark blue coloured solution is
A)
tin
done
clear
B)
lead
done
clear
C)
sodium
done
clear
D)
silver
done
clear
View Answer play_arrow
The Lassaigne's extract is boiled with dil \[HN{{O}_{3}}\]before testing for halogens because
A)
Silver halides are soluble in \[HN{{O}_{3}}\]
done
clear
B)
\[N{{a}_{2}}S\] and \[NaCN\] are decomposed by \[HN{{O}_{3}}\]
done
clear
C)
\[A{{g}_{2}}CN\] is soluble in \[HN{{O}_{3}}\]
done
clear
D)
\[AgCN\] is soluble in \[HN{{O}_{3}}\]
done
clear
View Answer play_arrow
The ortho/para directing group among the following is
A)
\[COOH\]
done
clear
B)
\[COC{{H}_{3}}\]
done
clear
C)
\[CN\]
done
clear
D)
\[NHCOC{{H}_{3}}\]
done
clear
View Answer play_arrow
will give .......... by \[{{E}_{1}}\] reaction.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which one of the following reacts with \[HBr\] at the fastest rate?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The aldol condensation of \[C{{H}_{3}}-CHO\] results in the formation of
A)
\[C{{H}_{3}}\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{2}}-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,H\]
done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\underset{O}{\mathop{\underset{||}{\mathop{C}}\,}}\,H\]
done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}OH+C{{H}_{3}}OH\]
done
clear
View Answer play_arrow
Consider the following compounds
Which of the above compounds reacts with \[NaHC{{O}_{3}}\] giving \[C{{O}_{2}}\]?
A)
I, II and III
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I and II
done
clear
View Answer play_arrow
Which of the following compounds shows aromatic properties?
A)
Valine
done
clear
B)
Leucine
done
clear
C)
Serine
done
clear
D)
Tyrosine
done
clear
View Answer play_arrow
Which one of the following is mainly responsible for depletion of ozone layer?
A)
Methane
done
clear
B)
Carbon dioxide
done
clear
C)
Water
done
clear
D)
Chlorofluoro carbon
done
clear
View Answer play_arrow
Identify the least stable ion amongst the following.
A)
\[L{{i}^{-}}\]
done
clear
B)
\[B{{e}^{-}}\]
done
clear
C)
\[{{B}^{-}}\]
done
clear
D)
\[{{C}^{-}}\]
done
clear
View Answer play_arrow
Which of the following statement is false?
A)
Boric lotion is a solution of boric acid used as an antiseptic.
done
clear
B)
Drikold is solid sulphur dioxide.
done
clear
C)
Ammonia is used for removing grease.
done
clear
D)
None of these
done
clear
View Answer play_arrow
Amongst \[TiF_{6}^{2-},\] \[CoF_{6}^{3-},\] \[CuC{{l}_{2}}\] and \[NiCl_{4}^{2-},\] the colourless species are: (At. No. \[Ti=22,\text{ }Co=27,\text{ }Cu=29,\text{ }Ni=28\])
A)
\[CoF_{6}^{3-}\]and \[NiCl_{4}^{2-}\]
done
clear
B)
\[TiF_{6}^{2-}\]and \[CoF_{6}^{3-}\]
done
clear
C)
\[C{{u}_{2}}C{{l}_{2}}\]and \[NiCl_{4}^{2-}\]
done
clear
D)
\[TiF_{6}^{2-}\] and \[C{{u}_{2}}C{{l}_{2}}\]
done
clear
View Answer play_arrow
Horn silver ore dissolves in excess of sodium cyanide solution forming
A)
\[AgCN\]
done
clear
B)
\[Na[Ag\,{{(CN)}_{2}}]\]
done
clear
C)
\[N{{a}_{2}}[Ag\,{{(CN)}_{3}}]\]
done
clear
D)
\[N{{a}_{4}}[Ag\,{{(CN)}_{5}}]\]
done
clear
View Answer play_arrow
Formation of liquid endosperm in coconut takes place because:
A)
Karyokinesis is followed by cytokinesis
done
clear
B)
Formation of liquid endosperm is not dependent upon karyokinesis and cytokinesis
done
clear
C)
Karyokinesis is not followed by cytokinesis
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Select the incorrect statement regarding angiosperm:
A)
Megaspore is diploid
done
clear
B)
Megaspore is the first cell of female gametophyte
done
clear
C)
The pollen grain is the first cell of male gametophyte
done
clear
D)
All of the above
done
clear
View Answer play_arrow
In mammals, failure of testes to descend into the scrotum is known as:
A)
Castration
done
clear
B)
Paedogenesis
done
clear
C)
Impotancy
done
clear
D)
Cryptorchidism
done
clear
View Answer play_arrow
Location of ley dig cells and their secretions are:
A)
Ovary - Estrogen
done
clear
B)
Liver - Cholesterol
done
clear
C)
Pancreas - Glucagon
done
clear
D)
Testis - Testosterone
done
clear
View Answer play_arrow
In spermatogenesis, the phase of maturation involves:
A)
The growth of spermatogonia into primary spermatocyte
done
clear
B)
The formation of spermatogonia from gonocytes through mitosis
done
clear
C)
The formation of oogonia from the spermatocytes through meiosis
done
clear
D)
The formation of spermatids from primary spermatocytes through meiosis
done
clear
View Answer play_arrow
Match list I with list II and select the correct answer using the codes given below the lists:
List I List II A. Chorion 1. Nourishment B. Allantois 2. Protection C. Yolk sac 3. Fluid environment D. Amnion 4. Excretion
A)
A-4 B-2 C-1 D-3
done
clear
B)
A-2 B-4 C-1 D-3
done
clear
C)
A-2 B-4 C-3 D-1
done
clear
D)
A-1 B-3 C-2 D-4
done
clear
View Answer play_arrow
The organ which have duel origin from embryonic layers:
(A) Hypophysis (B) Sense organs (C) Adrenal gland (D) Pancreas
Select, codes that are correct
A)
B and C
done
clear
B)
B and D
done
clear
C)
A and C
done
clear
D)
A, B and D
done
clear
View Answer play_arrow
Assertion (A): Old age is not an illness. It is a continuation of the life with decreasing capacity for adaptation Reason (R): Cessation of mitosis is a normal genetically programmed event
A)
Both (A) and (R) are true and (R) is the correct explanation of the (A)
done
clear
B)
Both (A) and (R) are true but (R) is not the correct explanation of the (A)
done
clear
C)
(A) is true but (R) is false
done
clear
D)
Both (A) and (R) are false
done
clear
View Answer play_arrow
A pure tall and a pure dwarf plant were crossed to produce off springs. Of springs were self crossed, then find out the ratio between true breeding tall to true breeding dwarf?
A)
\[1:1\]
done
clear
B)
\[1:2:1\]
done
clear
C)
\[2:1\]
done
clear
D)
\[3:1\]
done
clear
View Answer play_arrow
Match the items given in Column I with those listed in Column II. Choose the answer with correct combination of alphabets of the two columns:
Column I Column II A. Monohybrid cross p. T and t B. Test cross q. TT C. Alleles r. \[TT\times tt\] D. Homozygous tall s. tt t. \[Tt\times tt\]
A)
A-r B-t C-s D-q
done
clear
B)
A-r B-t C-p D-q
done
clear
C)
A-t B-r C-q D-s
done
clear
D)
A-r B-p C-t D-q
done
clear
View Answer play_arrow
A genetically dwarf plant made tall by use of gibberellins was crossed with a plant purely tall. Then the progenies would be:
A)
All tall
done
clear
B)
All dwarf
done
clear
C)
50% tall and 50% dwarf
done
clear
D)
May be tall or dwarf
done
clear
View Answer play_arrow
When red and white flowered Mirabilis plants are crossed all pink flowers are seen in \[{{F}_{1}}\]generation. When \[{{F}_{1}}\] progeny is selfed, the expected phcnotypic and genotypic ratios are:
A)
\[3:1\] and \[2:1\]
done
clear
B)
\[1:1\] and \[1:1:1\]
done
clear
C)
\[1:2:1\] and \[3:1\]
done
clear
D)
\[1:2:1\] and \[1:2:1\]
done
clear
View Answer play_arrow
In which one of the following combinations (1-4) of the number of chromosomes is the present day hexaploid wheat correctly represented?
A)
Monosomic Haploid Nullisomic Trisomic 21 28 42 43
done
clear
B)
Monosomic Haploid Nullisomic Trisomic 7 28 40 42
done
clear
C)
Monosomic Haploid Nullisomic Trisomic 21 7 42 43
done
clear
D)
Monosomic Haploid Nullisomic Trisomic 41 21 40 43
done
clear
View Answer play_arrow
A segment of the DNA of following sequence AAG GAG GAG CAA CCA, which of the following sequences represents a frame shift mutation?
A)
AGG AGG ACC AAC CA
done
clear
B)
ACG GAG GAC GAG CCA
done
clear
C)
AAG GCG GAG CCA AC
done
clear
D)
AAG GAG GAC CAA CCA
done
clear
View Answer play_arrow
If the codones in DNA are ATG ATG ATG and a cytosine base is inserted at the beginning, select result from below
A)
A non sense mutation
done
clear
B)
C ATG ATG ATG
done
clear
C)
CATGATGATG
done
clear
D)
CAT GAT GAT G
done
clear
View Answer play_arrow
Diploid cells have
A)
Two pairs of homologus chromosomes
done
clear
B)
Two chromosomes
done
clear
C)
Two set of chromosomes
done
clear
D)
One set of chromosomes
done
clear
View Answer play_arrow
The number of linkage group in E.coli will be
A)
4000
done
clear
B)
1000
done
clear
C)
40
done
clear
D)
1
done
clear
View Answer play_arrow
A DNA has A = 30% then what would be percentage of G bore
A)
40%
done
clear
B)
30%
done
clear
C)
20%
done
clear
D)
10%
done
clear
View Answer play_arrow
During replication of DNA, Okajaki fragments are formed in the direction of
A)
\[3'\to 3'\]
done
clear
B)
\[5'\to 3'\]
done
clear
C)
\[5'\to 5'\]
done
clear
D)
\[3'\to 5'\]
done
clear
View Answer play_arrow
Out of 64 codons, 61 codons codes for 20 types of amino acids
A)
Overlapping of genes
done
clear
B)
Universality of codons
done
clear
C)
Degeneracy of codons
done
clear
D)
Wobbling of codons
done
clear
View Answer play_arrow
Match the following and select correct answer
Column I Column II A. UUU 1.Serine B. GGG 2.Methionine C. UCU 3. Phenylalanine D. CCC 4. Glycine E. AUG 5.Proline
A)
\[A=3,\text{ }B=4,\text{ }C=1\]
done
clear
B)
\[A=3,\text{ }B=1,\text{ }C=4\]
done
clear
C)
\[A=C,\text{ }B=4,C=3\]
done
clear
D)
\[A=2,\text{ }B=4,\text{ }C=1\]
done
clear
View Answer play_arrow
Consider the following lines
(A) r-RNA provides the template for synthesis of proteins (B) t-RNA brings amino acids and reads the genetic code (C) RNA polymerase binds to promoter and initiates transcription (D) A segment of DNA coding for polypeptide is called intron
A)
(B) and (C) are correct
done
clear
B)
(A) and (B) are correct
done
clear
C)
(A), (B) and (C) are correct
done
clear
D)
(A) (B) and (D) are correct
done
clear
View Answer play_arrow
In lac operon system lac i-gene codes for
A)
B galactosidase
done
clear
B)
Promoter
done
clear
C)
Represser
done
clear
D)
Inducer
done
clear
View Answer play_arrow
E-coli cells with a mutated Z gene of lac operon can not grow in medium containing only lactose as source of energy because
A)
They are unable to transport lactose from medium into cell
done
clear
B)
The lac operon is constitutively active in these cells
done
clear
C)
They can not synthesize functional B galactosidase
done
clear
D)
In the presence of glucose, E.Coli do not utilize lactose
done
clear
View Answer play_arrow
The genome of coenorhabditis eugans consist of
A)
Genes 13000, B. P. - 180 Million
done
clear
B)
Genes - 18000, B.P " 97 Million
done
clear
C)
Genes - 4000, B.P. 4.7 Million
done
clear
D)
Genes - 6000, B.P 10 Million
done
clear
View Answer play_arrow
Down syndrome is related to
A)
Decrease in 18th pair of autosomes
done
clear
B)
Increase in 18th pair of autosomes
done
clear
C)
Increase in 21st pair of autosomes
done
clear
D)
Increase in 13th pair of autosomes
done
clear
View Answer play_arrow
Select right combination, with respect to sex type in male and female
A. Man XO B. Drosophila XO C. Human XXY D. Drosophila XXY
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A man type A blood group marries a woman who has AB group. We do not know whether the man is homozygous or heterozygous for \[{{I}^{A}}\] allele. Which one of following types in the progeny of this couple would indicate that the man is heterozygous?
A)
Type B
done
clear
B)
Type A
done
clear
C)
Type O
done
clear
D)
Type AB
done
clear
View Answer play_arrow
Study the pedigree chart given blow:
What does it show?
A)
Inheritance of a recessive sex-linked disease like haemophilia
done
clear
B)
Inheritance of sex-linked inborn error of metabolism like phenylketonuria
done
clear
C)
The pedigree chart is wrong as this is not possible
done
clear
D)
Inheritance of a condition like phenylketonuria us an autosomal recessive trait
done
clear
View Answer play_arrow
Of the following which mixture, Miller used to solve the problem of origin of life?
A)
Methane, Ammonia, Oxygen, Water
done
clear
B)
Ethane, Ammonia, Hydrogen, Water
done
clear
C)
Ethane, Ammonia, Oxygen, Water
done
clear
D)
Methane, Ammonia, Hydrogen, Water
done
clear
View Answer play_arrow
Miller synthesized simple amino acids from one of the following mixtures in an experiment
A)
\[{{H}_{2}},{{O}_{2}},{{N}_{2}}\,(2:1:2)\]and water vapour
done
clear
B)
\[C{{H}_{4}},N{{H}_{3}},{{H}_{2}}\,(2:1:2)\] and water vapour
done
clear
C)
\[{{H}_{2}},{{O}_{2}},{{N}_{2}}\,(1:2:1)\] and water vapour
done
clear
D)
\[C{{H}_{4}},N{{H}_{3}},{{H}_{2}}\,(1:2:1)\] and water vapour
done
clear
View Answer play_arrow
Match the term listed under Column I with the description given under Column II; choose the answer which gives the correct combination of the alphabets of the two columns:
Column I (Term) Column II (Description) A. Fossil p. Accumulation of neutral mutations B. Stratification q. e.g., shark fins, penguin flippers C. Molecular clock r. stone-hard evidence of life in past D. Analogy s. layers of sedimentary rock
A)
A-s B-q C-r D-p
done
clear
B)
A-r B-s C-p D-q
done
clear
C)
A-s B-r C-q D-p
done
clear
D)
A-r B-q C-s D-p
done
clear
View Answer play_arrow
Age of fossils in the past was generally determined by radiocarbon method and other methods involving radioactive elements found in the rocks. More precise methods, which were used recently and led to the revision of the evolutionary periods for different groups of organisms includes:
A)
study of carbohydrates/proteins in rocks
done
clear
B)
study of the conditions offossilization
done
clear
C)
study of carbohydrates/proteins in fossils
done
clear
D)
electron-Spin Resonance (F3SR) and fossil DNA
done
clear
View Answer play_arrow
There are two opposing views about origin of modern man. According to one view Homo evectus in Asia were the ancestors suggested African origin of modern man. What kind of observation on DNA variation could suggest this?
A)
Greater variation in Africa than in Asia
done
clear
B)
Similar variation in Arica and Asia
done
clear
C)
Variation only in Asia and no variation in Africa
done
clear
D)
Greater variation in Asia than in Africa
done
clear
View Answer play_arrow
Match the following bacteria with the diseases and choose the correct option:
Column I Column II A.Treponema palladium 1. Plague B. Yersinia pestis 2. Anthrax C. Bacillus anthracis 3. Syphilis D. Vibrio 4. Choleva
A)
A-1 B-3 C-2 D-4
done
clear
B)
A-2 B-3 C-1 D-4
done
clear
C)
A-3 B-1 C-2 D-4
done
clear
D)
A-4 B-3 C-1 D-2
done
clear
E)
A-3 B-4 C-1 D-2
done
clear
View Answer play_arrow
Column I lists the components of body defence and column II lists the corresponding descriptions. Match the two columns. Choose the correct option from those given:
Column I Column II A. Active natural p. Injection of gamma immunity globulins B. First line of defence q. Complement proteins and interferons C. Passive natural r. Direct contact with the immunity pathogens that have entered inside D. Second line of defence s. Surface barriers t. Antibodies transferred through the placenta
A)
A-s B-r C-t D-q
done
clear
B)
A-r B-s C-q D-t
done
clear
C)
A-r B-s C-t D-q
done
clear
D)
A-t B-r C-q D-p
done
clear
View Answer play_arrow
Match the type of immunity listed in Column I with the examples listed in Column II. Choose the answer which gives the correct combination of alphabets of the columns:
Column I Type of Immunity Column II Example A. Natural Active p. Immunity developed by heredity B. Artificial Passive q. From mother to foetus through placenta C. Artificial active r. Injection of antiserum to travelers D. Natural passive s. Fighting infections naturally t. Induced by vaccination
A)
A-t B-s C-r D-p
done
clear
B)
A-s B-t C-q D-r
done
clear
C)
A-s B-r C-t D-q
done
clear
D)
A-p B-q C-r D-t
done
clear
View Answer play_arrow
It shows chronological order of the events occurring during callus culture:
A)
Callus \[\to \]Cell division \[\to \] explants \[\to \] addition of cytokinin \[\to \] cell acquired meristematic property
done
clear
B)
Explant \[\to \] callus \[\to \] cell division \[\to \] addition of cytokinin \[\to \] cell acquired meristematic property
done
clear
C)
Callus \[\to \] explants \[\to \] cell division \[\to \] addition of cytokinin \[\to \] cell acquired meristematic property
done
clear
D)
Explant \[\to \]cell division \[\to \] Callus \[\to \] addition of cytokinin \[\to \] cell acquired meristematic property
done
clear
View Answer play_arrow
In crop improvement programme, virus free clones can be obtained through:
A)
Embryo culture
done
clear
B)
Hybridization
done
clear
C)
Shoot apex culture
done
clear
D)
Grafting
done
clear
View Answer play_arrow
Single cell protein refers to
A)
A specific protein extracted from pure culture
done
clear
B)
Sources of mixed proteins extracted from pure or mixed culture of organisms or cells
done
clear
C)
A specific protein extracted from a single cell
done
clear
D)
Proteins extracted from a single cell
done
clear
View Answer play_arrow
Genetic engineering has been successfully used for producing
A)
Transgenic modals for studying new treatment for certain cardiac disease
done
clear
B)
Transgenic mice for testing safety of polio vaccine before use in humans
done
clear
C)
Transgenic Cow - Rosie which produces fat milk for making ghee
done
clear
D)
Animals like bulls for farm work as they have super power
done
clear
View Answer play_arrow
In genetic fingerprinting, the 'probe' refers to
A)
a radioactive, labelled, single stranded DNA
done
clear
B)
a radioactive, labelled, double stranded RNA molecule
done
clear
C)
a radioactive, labelled, single stranded RNA molecule
done
clear
D)
a radioactive, labelled double stranded DNA molecule
done
clear
View Answer play_arrow
Somaclones are obtained by:
A)
Genetic engineering
done
clear
B)
Plant breeding
done
clear
C)
Tissue culture
done
clear
D)
Irradiation
done
clear
View Answer play_arrow
Transgenic crops developed to tolerate herbicides are:
A)
Rice and Wheat
done
clear
B)
Tomato and Rice
done
clear
C)
Tomato and Tobacco
done
clear
D)
Maize and Sugarcane
done
clear
View Answer play_arrow
Growth rate for population in given time is
A)
\[\frac{dN}{dt}=rN\]
done
clear
B)
\[\frac{dN}{rN}=dt\]
done
clear
C)
\[\frac{dt}{dN}=rN\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
An interaction in which one organism is benefitted without affecting the other
A)
Symbiosis
done
clear
B)
Saprophytism
done
clear
C)
Commensalism
done
clear
D)
Parasitism
done
clear
View Answer play_arrow
Study of ecology of population is
A)
Ecotype
done
clear
B)
Autecology
done
clear
C)
Synecology
done
clear
D)
Demecology
done
clear
View Answer play_arrow
Pyramid of energy in pond ecosystem
A)
always up right
done
clear
B)
always inucrted
done
clear
C)
normally upright
done
clear
D)
spindle shaped
done
clear
View Answer play_arrow
Energy transfer from one trophic level to another is
A)
5%
done
clear
B)
10%
done
clear
C)
20%
done
clear
D)
40%
done
clear
View Answer play_arrow
Stable community in a ecological succession is
A)
Sere
done
clear
B)
Climax
done
clear
C)
Serai
done
clear
D)
Mesic
done
clear
View Answer play_arrow
Sucking roots are present in plant
A)
Solanum
done
clear
B)
Mangifera
done
clear
C)
Trapa
done
clear
D)
Cuscnta
done
clear
View Answer play_arrow
Number of plant species reported from India are
A)
50000
done
clear
B)
40000
done
clear
C)
45000
done
clear
D)
1 lac
done
clear
View Answer play_arrow
Which of these is not a green house gas
A)
CFC
done
clear
B)
\[S{{O}_{2}}\]
done
clear
C)
\[{{O}_{2}}\]
done
clear
D)
\[{{N}_{2}}O\]
done
clear
View Answer play_arrow
BOD of an eutrophic lake will be
A)
Moderate
done
clear
B)
Higher
done
clear
C)
Lower
done
clear
D)
Can not say
done
clear
View Answer play_arrow
Chipko movement was launched for the protection of
A)
Ponds
done
clear
B)
Rivers
done
clear
C)
Forests
done
clear
D)
Tribals
done
clear
View Answer play_arrow
Select correct for viruses
A)
They have either DNA or RNA
done
clear
B)
They multiply inside the living cell only
done
clear
C)
They arc in between living and non living
done
clear
D)
All of these
done
clear
View Answer play_arrow
Process which are basic to classification
A)
Characterization, identification, nomenclature, classification
done
clear
B)
Characterization, identification, nomenclature
done
clear
C)
Only nomenclature and naming
done
clear
D)
Only identification
done
clear
View Answer play_arrow
Select odd with respect to Binomial epithet
A)
Genus
done
clear
B)
Species
done
clear
C)
Scientist name
done
clear
D)
Family
done
clear
View Answer play_arrow
Select wrong match with respect shape of bacteria
A)
Coma like - Vibrio
done
clear
B)
Rod like -Bacillus
done
clear
C)
Round-Spirillum
done
clear
D)
Pleomorphic - No fixed shape
done
clear
View Answer play_arrow
Animals radially symmetrical in adults and late rally symmetrical in larval are
A)
Hemichordates
done
clear
B)
Echinodermates
done
clear
C)
Polychaete
done
clear
D)
Coelentrates
done
clear
View Answer play_arrow
Notochord occurs in adult animal of
A)
Amphibia
done
clear
B)
Chondrichthyes
done
clear
C)
Protochordata
done
clear
D)
Osteichthyes
done
clear
View Answer play_arrow
Salvinia is a
A)
Liverwort
done
clear
B)
Moss
done
clear
C)
Tree fern
done
clear
D)
Water fern
done
clear
View Answer play_arrow
Phyco crythrin is found in
A)
Polysiptonia
done
clear
B)
Chlorella
done
clear
C)
Fucus
done
clear
D)
Dictyota
done
clear
View Answer play_arrow
Walking fern is
A)
Dryopteris
done
clear
B)
Pteris
done
clear
C)
Adiantum
done
clear
D)
Ophioglossum
done
clear
View Answer play_arrow
Alternaria, Trichoderma, collctotrichum belong to how many class of fungi
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
Cones are visible in
A)
Pincers
done
clear
B)
Cedrus
done
clear
C)
Horsetail
done
clear
D)
All of these
done
clear
View Answer play_arrow
Gemma helps in___ and found in____ respectively
A)
Vegetative reproduction, marchantia
done
clear
B)
A sex. Reproduction, Moss
done
clear
C)
Sex. Reproduction, Raccia
done
clear
D)
Vegetative reproduction, Anthoceros
done
clear
View Answer play_arrow
Translation takes place on the surface of
A)
Mitochondria
done
clear
B)
RER
done
clear
C)
Ribosomes
done
clear
D)
SER
done
clear
View Answer play_arrow
Semiautonomous all organelle is/are
A)
Chloroplast
done
clear
B)
Mitochondria
done
clear
C)
Nucleus
done
clear
D)
More than one
done
clear
View Answer play_arrow
When potato are exposed to air
A)
Lencoplast are converted to chromoplast
done
clear
B)
Chromoplast are converted to lencoplast
done
clear
C)
Lencoplast are converted to chloroplast
done
clear
D)
Chloroplast give rise to chromoplast
done
clear
View Answer play_arrow
A monomer unit of fungal cellulose is
A)
NAM
done
clear
B)
NAG
done
clear
C)
AMA
done
clear
D)
All of these
done
clear
View Answer play_arrow
Select odd one with respect to mucopolysaccharide
A)
Alginic acid
done
clear
B)
Resin
done
clear
C)
Agar
done
clear
D)
Carrageenan
done
clear
View Answer play_arrow
Asymmatric carbon atom is found in
A)
Histidine
done
clear
B)
Isolencine
done
clear
C)
Valine
done
clear
D)
Serine
done
clear
View Answer play_arrow
A protein having both structural and enzymatic traits is
A)
Actin
done
clear
B)
Collagen
done
clear
C)
Trypsin
done
clear
D)
Myosin
done
clear
View Answer play_arrow
Synapsis is seen in
A)
Leptonema
done
clear
B)
Zygonema
done
clear
C)
Pachytcne
done
clear
D)
Diakinesis
done
clear
View Answer play_arrow
Two diploid sets of daughter chromosomes gathered at opposite poles during
A)
Metaphase
done
clear
B)
Prophase
done
clear
C)
Anaphase
done
clear
D)
Telophase
done
clear
View Answer play_arrow
Select the wrong match
A)
Tuberous root - sweet potato
done
clear
B)
Prop roots - Banyan
done
clear
C)
Reproductive roots ? sweet potato
done
clear
D)
Pillar roots - Maize
done
clear
View Answer play_arrow
Select correct match with respect to stem
A)
Celm - Palms
done
clear
B)
Caudex - Bamboo
done
clear
C)
Excurrent - Pinus
done
clear
D)
Deliquescent - Banyan
done
clear
View Answer play_arrow
Ziminkand and colocasia have
A)
Rhizome
done
clear
B)
Corm
done
clear
C)
Bulb
done
clear
D)
Runner
done
clear
View Answer play_arrow
Select the odd with respect to tendril
A)
Grapevine
done
clear
B)
Watermelon
done
clear
C)
Citrus
done
clear
D)
Pumpkin
done
clear
View Answer play_arrow
Leaf bladder is modification of
A)
Lamina
done
clear
B)
Petiole
done
clear
C)
Leaf apex
done
clear
D)
Stipule
done
clear
View Answer play_arrow
The filaments are free but anthers are fused as in
A)
Compositac
done
clear
B)
Malvaceae
done
clear
C)
Fabaccae
done
clear
D)
All of these
done
clear
View Answer play_arrow
Asteraccae has _______ placentation
A)
Basal
done
clear
B)
Axile
done
clear
C)
Parietal
done
clear
D)
Superficial
done
clear
View Answer play_arrow
\[{{P}_{3+3}}\] condition is related to
A)
Lily family
done
clear
B)
Mustard family
done
clear
C)
Grass family
done
clear
D)
Sunflower family
done
clear
View Answer play_arrow
Phlocmparenchyma is absent in
A)
Monocot
done
clear
B)
Dicat
done
clear
C)
Monocot root only
done
clear
D)
Leaf only
done
clear
View Answer play_arrow
Plants with distinct early wood and late wood grow in
A)
Temperate regions
done
clear
B)
Alpine regions
done
clear
C)
Tropical regions
done
clear
D)
Subtropics
done
clear
View Answer play_arrow
Nucleus is absent in
A)
Sieve tube
done
clear
B)
Trachieds
done
clear
C)
Vessels
done
clear
D)
All of these
done
clear
View Answer play_arrow
Earthworm is
A)
Unisexual
done
clear
B)
Hermaphrodite
done
clear
C)
Bisexual
done
clear
D)
More than one are correct
done
clear
View Answer play_arrow
Web spining gland in spider is
A)
Nervous gland
done
clear
B)
Abdominal gland
done
clear
C)
Anal gland
done
clear
D)
Sting gland
done
clear
View Answer play_arrow
Stomach in mammals is lined by
A)
Stratified epithelium
done
clear
B)
Columnar epithelium
done
clear
C)
Squamous epithelium
done
clear
D)
Cuboidal epithelium
done
clear
View Answer play_arrow


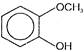
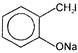
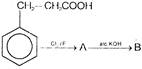 Compound B is
Compound B is 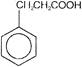
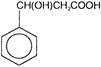
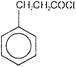
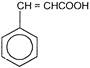
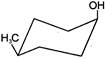
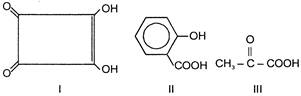 Which of the above compounds reacts with \[NaHC{{O}_{3}}\] giving \[C{{O}_{2}}\]?
Which of the above compounds reacts with \[NaHC{{O}_{3}}\] giving \[C{{O}_{2}}\]?  What does it show?
What does it show? 