A body is projected horizontally from the top of a tower with an initial velocity of 18 m/s. It hits the ground at an angle of\[45{}^\circ \]. What is the vertical component of velocity when the body strikes the ground?
A)
\[9\,m/s\]
done
clear
B)
\[9\sqrt{2}\,m\]
done
clear
C)
\[18\,m/s\]
done
clear
D)
\[18\sqrt{2}\,m/s\]
done
clear
View Answer play_arrow
A car accelerates from rest at a constant rate a for some time, after which it decelerates at a constant rate \[\beta \] and comes to rest. to the total time elapsed is t, then the maximum velocity acquired by the car is-
A)
\[\left( \frac{{{\alpha }^{2}}+{{\beta }^{2}}}{\alpha \beta } \right)t\]
done
clear
B)
\[\left( \frac{{{\alpha }^{2}}-{{\beta }^{2}}}{\alpha \beta } \right)t\]
done
clear
C)
\[\frac{(\alpha -\beta )t}{\alpha \beta }\]
done
clear
D)
\[\frac{\alpha \beta t}{\alpha +\beta }\]
done
clear
View Answer play_arrow
Given: \[\overrightarrow{C}=\overrightarrow{A}+\overrightarrow{B}\]. Also, the magnitudes of \[\overrightarrow{A},\,\,\overrightarrow{B}\] and \[\overrightarrow{C}\]are 12, 5 and 13 units respectively. The angle between \[\overrightarrow{A}\text{ }and\text{ }\overrightarrow{B}\] is-
A)
\[0{}^\circ \]
done
clear
B)
\[\frac{\pi }{4}\]
done
clear
C)
\[\frac{\pi }{2}\]
done
clear
D)
\[\pi \]
done
clear
View Answer play_arrow
Two objects A and B each of mass m are connected by a light inextensible string. They are restricted to move on a frictionless ring of radius R in a vertical plane (as shown in figure). The objects are released from rest at the position shown. Then, the tension in the cord just after release is-
A)
zero
done
clear
B)
mg
done
clear
C)
\[\sqrt{2}\,mg\]
done
clear
D)
\[mg/\sqrt{2}\]
done
clear
View Answer play_arrow
A car of mass 1500 kg is moving with a speed of \[12.5\text{ }m{{s}^{-}}^{1}\] on a circular path of radius 20 m on a level road. What should be the value of coefficient of friction to attain this centripetal force?
A)
0.2
done
clear
B)
0.4
done
clear
C)
0.6
done
clear
D)
0.8
done
clear
View Answer play_arrow
A body of mass 2 kg is moved from a point A to a point B by an external agent in a conservative force field. If the velocity of the body at the points A and B are 5 m/s and 3 m/s respectively and the work done by the external agents is -10 J, then the change in potential energy between points A and B is-
A)
6 J
done
clear
B)
36 J
done
clear
C)
16 J
done
clear
D)
None of these
done
clear
View Answer play_arrow
A ball of mass 10 g is moving with a velocity of 10 m/s. If strikes another ball of mass 5 kg which is moving in the same direction with a velocity of 4 m/s. If the collision is elastic their velocities after the collision will be respectively-
A)
6 m/s, 12 m/s
done
clear
B)
12 m/s, 6 m/s
done
clear
C)
12 m/s, 10 m/s
done
clear
D)
12 m/s, 25 m/s
done
clear
View Answer play_arrow
Two spheres A and B have densities in the ratio 2:1, diameters in the ratio 1 : 2 and specific heats in the ratio 1:3, find ratio of their thermal capacities-
A)
\[1\text{ }:\text{ }6\]
done
clear
B)
\[1\text{ }:\text{ }12\]
done
clear
C)
\[1\text{ }:\text{ }3\]
done
clear
D)
\[1\text{ }:\text{ }4\]
done
clear
View Answer play_arrow
A vessel is filled with a gas at a pressure of 76 cm of mercury at a certain temperature. The mass of the gas is increased by \[50\,%\] by introducing more gas in the vessel at the same temperature. The resultant pressure, in cm of Hg, is-
A)
76
done
clear
B)
152
done
clear
C)
114
done
clear
D)
1117
done
clear
View Answer play_arrow
Temperature of 1000 grams of nitrogen is raised from \[-20{}^\circ \,C\text{ }to\text{ }100{}^\circ \,C\] at constant pressure. Given: specific heat at constant volume \[{{C}_{v}}\text{ }=5\text{ }cal/mole\text{ }{}^\circ C\text{ }and\text{ }R=2\,Cal/mole\,{}^\circ C\]
[a] Heat required in the process is 30 K Cal [b] Increase in the internal energy of gas is 21 Kcal [c] If process is at constant volume then heat required is 21 Kcal [d] external work done is 10 KCal
A)
a, d
done
clear
B)
a, b, c
done
clear
C)
c, d
done
clear
D)
b, d
done
clear
View Answer play_arrow
A cylinder of radius R made of material of thermal conductivity \[{{K}_{1}}\] is surrounded by a cylindrical shell of inner radius R and outer radius 3R made of a material of thermal conductivity\[{{K}_{2}}\]. The two ends of the combined system are maintained at two different temperature. What is the effective thermal conductivity of the system?
A)
\[{{K}_{1}}+{{K}_{2}}\]
done
clear
B)
\[\frac{{{K}_{1}}+8{{K}_{2}}}{9}\]
done
clear
C)
\[\frac{{{K}_{1}}{{K}_{2}}}{{{K}_{1}}+{{K}_{2}}}\]
done
clear
D)
\[\frac{8{{K}_{1}}+{{K}_{2}}}{9}\]
done
clear
View Answer play_arrow
Density of substance at \[0{}^\circ C\] is \[10\text{ }gm/cc\] and at\[100{}^\circ \,C\], its density is \[9.7\text{ }gm/cc\]. The coefficient of linear expansion of the substance will be-
A)
\[{{10}^{2}}\]
done
clear
B)
\[{{10}^{-}}^{2}\]
done
clear
C)
\[{{10}^{-3}}~\]
done
clear
D)
\[{{10}^{-}}^{4}\]
done
clear
View Answer play_arrow
If a string of length 133 cm is divided in 3 segments having fundamental frequency in the ratio of \[1:3:4\] then lengths of these segments are-
A)
84 cm, 28cm, 21 cm
done
clear
B)
90 cm, 22 cm, 21 cm
done
clear
C)
78 cm, 34 cm, 21 cm
done
clear
D)
50 cm, 72 cm, 11 cm
done
clear
View Answer play_arrow
A disc of radius R is rotating uniformly with angular frequency co. A source of sound is fixed to the rim of the disc. The ratio of maximum and minimum frequencies heard by stationary observer, far away from the disc and in the plane of disc is \[-\left( v=-speed\text{ }of\text{ }sound \right)\]
A)
\[\left( \frac{v}{v-R\omega } \right)\]
done
clear
B)
\[\left( \frac{v}{v+R\omega } \right)\]
done
clear
C)
\[\left( \frac{v-R\omega }{v+R\omega } \right)\]
done
clear
D)
\[\left( \frac{v+R\omega }{v-R\omega } \right)\]
done
clear
View Answer play_arrow
Potential barrier developed across a pn-junction
A)
Hinders the movement of both majority as well as minority charge-carrier
done
clear
B)
Supports the movement of both majority as well as minority charge-carriers
done
clear
C)
Hinders the movement of majority charge- carriers but supports the movement of minority charge-carrier
done
clear
D)
Supports the movement of majority charge- carriers, but hinders the movement of minority charge-carriers
done
clear
View Answer play_arrow
A wooden cube just floats inside water when a 200 g mass is placed on it. When the mass is removed, the cube is 2 cm above the water level. What is the size of each sides of the cube?
A)
6 cm
done
clear
B)
8 cm
done
clear
C)
10 cm
done
clear
D)
12 cm
done
clear
View Answer play_arrow
If h is the height of capillary rise and r be the radius of capillary tube, then which of the following relation will be correct?
A)
\[hr=constant\]
done
clear
B)
\[\frac{h}{{{r}^{2}}}=constant\]
done
clear
C)
\[h{{r}^{2}}=constant\]
done
clear
D)
\[\frac{h}{r}=constant\]
done
clear
View Answer play_arrow
A system is shown in the figure. The time period for small oscillations of the two blocks will be-
A)
\[2\pi \sqrt{\frac{3m}{k}}\]
done
clear
B)
\[2\pi \sqrt{\frac{3m}{4k}}\]
done
clear
C)
\[2\pi \sqrt{\frac{3m}{8k}}\]
done
clear
D)
\[2\pi \sqrt{\frac{3m}{2k}}\]
done
clear
View Answer play_arrow
If 'g' is the acceleration due to gravity on the earth's surface, the gain in the potential energy of an object of mass 'm' raised from the surface of the earth to a height equal to the radius 'R' of the earth is-
A)
\[2\text{ }mgR\]
done
clear
B)
\[1/2mgR\]
done
clear
C)
\[1/4\text{ }mgR\]
done
clear
D)
\[mgR\]
done
clear
View Answer play_arrow
Four particles each of mass m are placed at the comers of a square of side length\[\ell \]. The radius of gyration of the system about an axis perpendicular to the square and passing through its centre is-
A)
\[\frac{\ell }{\sqrt{2}}\]
done
clear
B)
\[\ell \]
done
clear
C)
\[\frac{\ell }{2}\]
done
clear
D)
\[(\sqrt{2})\,\ell \]
done
clear
View Answer play_arrow
A simple pendulum of length t and mass m is initially at its lowest position. It is given the minimum horizontal speed necessary to move in a circular path about the point of suspension. The tension in the string at the lowest position of the bob is-
A)
3mg
done
clear
B)
4mg
done
clear
C)
5 mg
done
clear
D)
6 mg
done
clear
View Answer play_arrow
Potential difference between cathode and anode is 200 kV and the distance between them is 25 cm. The electric field and the energy imparted to the electrons as they move from cathode to anode is-
A)
\[8\times {{10}^{5}}\,V/m;\text{ }0.8\times {{10}^{-}}^{14}J\]
done
clear
B)
\[4\times {{10}^{5}}\,V/m;\,\,1.6\times {{10}^{-}}^{14}J\]
done
clear
C)
\[8\times {{10}^{5}}\,V/m;\,\,3.2\times {{10}^{-}}^{14}J\]
done
clear
D)
\[4\times {{10}^{5}}\,V/m;\,\,0.8\times {{10}^{-}}^{14}J\]
done
clear
View Answer play_arrow
A particle of mass 'm' is projected from ground with velocity 'u' making angle '\[\theta \]' with the vertical. The de Broglie wavelength of the particle at the highest point is-
A)
\[\infty \]
done
clear
B)
\[h/mu\text{ }sin\text{ }\theta \]
done
clear
C)
\[h/mu\text{ }cos\text{ }\theta \]
done
clear
D)
\[h/mu\]
done
clear
View Answer play_arrow
In YDSE, having slits of equal widths, let \[\theta \] be the fringe width and \[{{I}_{0}}\] be the maximum intensity. At a distance x from the central bright fringe, the intensity will be:
A)
\[{{I}_{0}}\,\cos \,\left( \frac{x}{\beta } \right)\]
done
clear
B)
\[{{I}_{0}}\,{{\cos }^{2}}\,\left( \frac{2\pi x}{\beta } \right)\]
done
clear
C)
\[{{I}_{0}}\,{{\cos }^{2}}\,\left( \frac{\pi x}{\beta } \right)\]
done
clear
D)
\[\frac{{{I}_{0}}}{4}\,{{\cos }^{2}}\,\frac{\pi x}{\beta }\]
done
clear
View Answer play_arrow
An astronomical telescope has an angular magnification of magnitude 5 for distant objects. The separation between the objective and the eye-piece is 36 cm and the final image is formed at infinity. The focal length \[{{f}_{0}}\] of the objective and \[{{f}_{e}}\] of the eye-piece are-
A)
\[{{f}_{0}}=45\text{ }cm\text{ }and\text{ }{{f}_{e}}=-9\text{ }cm\]
done
clear
B)
\[{{f}_{0}}=50\text{ }cm\text{ }and\text{ }{{f}_{e}}=10\text{ }cm\]
done
clear
C)
\[{{f}_{0}}=7.2\text{ }cm\text{ }and\,\,{{f}_{e}}=5\,\,cm\]
done
clear
D)
\[{{f}_{0}}=30\text{ }cm\text{ }and\text{ }{{f}_{e}}=6\text{ }cm\]
done
clear
View Answer play_arrow
The angle of incidence at which reflected light is totally polarized for reflection from air to glass (refractive index n) is:
A)
\[{{\sin }^{-1}}\,(n)\]
done
clear
B)
\[{{\sin }^{-1}}\,(1/n)\]
done
clear
C)
\[{{\tan }^{-1}}\,(1/n)\]
done
clear
D)
\[{{\tan }^{-1}}\,(n)\]
done
clear
View Answer play_arrow
A point source S is placed at a distance of 21 cm from a convex mirror of focal length 12 cm. What is the distance (in cm) of a converging lens of focal length 10 cm from mirror, placed between the source and the mirror to get the final image on the source itself-
A)
12 cm
done
clear
B)
10 cm
done
clear
C)
6 cm
done
clear
D)
5 cm
done
clear
View Answer play_arrow
One of the refracting surfaces of a prism of angle of \[30{}^\circ \] is silvered. A ray of light incident at an angle of \[60{}^\circ \] retraces its path. The refractive index of the material of the prism is-
A)
\[\sqrt{2}\]
done
clear
B)
\[\sqrt{3}\]
done
clear
C)
\[3/2\]
done
clear
D)
\[2\]
done
clear
View Answer play_arrow
If time in object clock is 3 : 20, then time in image clock is-
A)
\[8\text{ }:\text{ }40\]
done
clear
B)
\[9\text{ }:\text{ }40\]
done
clear
C)
\[9\text{ }:\text{ }20\]
done
clear
D)
\[8\,\,:\,\,20\]
done
clear
View Answer play_arrow
In the disintegration series \[_{92}^{238}\,U\xrightarrow{\alpha }\,X\,\xrightarrow{{{\beta }^{-}}}\,_{Z}^{A}\,Y\] The values of Z and A respectively will be-
A)
92,236
done
clear
B)
88,230
done
clear
C)
90,234
done
clear
D)
91,234
done
clear
View Answer play_arrow
In coolidge tube experiment, if applied voltage is increased to three times, the short wavelength limit of continuous X-ray spectrum shifts by 20 pm. What is the initial voltage applied to the tube?
A)
20 kV
done
clear
B)
41 Kv
done
clear
C)
52 kV
done
clear
D)
102 kV
done
clear
View Answer play_arrow
The relation gives the value of x as, \[x=\frac{{{a}^{3}}{{b}^{3}}}{c\sqrt{d}}\]. Find the maximum percentage error in x, if the percentage error in a, b, c, d are \[2%,\text{ }1%,\text{ }2%\text{ }\And \text{ }4%\] respectively
A)
\[13%\]
done
clear
B)
\[5%\]
done
clear
C)
\[9%\]
done
clear
D)
\[8\,%\]
done
clear
View Answer play_arrow
When the speed of electron increase, then the value of its specific charge-
A)
Increases
done
clear
B)
Decreases
done
clear
C)
Remains unchanged
done
clear
D)
Increases upto some velocity and then begins to decrease
done
clear
View Answer play_arrow
An electric field is expressed as\[E=2\widehat{i}+3\widehat{j}\]. Find the potential difference\[({{V}_{A}}-{{V}_{B}})\] between two points A and B whose position vectors are given by \[{{r}_{A}}=\widehat{i}+2\widehat{j}\text{ }and\text{ }{{r}_{B}}=2\widehat{i}+\widehat{j}+3\widehat{k}.\]
A)
-1 V
done
clear
B)
1 V
done
clear
C)
2 V
done
clear
D)
3 V
done
clear
View Answer play_arrow
Two parallel metal plates carry charges +Q and -Q respectively. A test charge \[{{q}_{0}}\] placed between them experiences a force F. Now the separation between the plates is doubled, than the force on the test charge will be-
A)
F
done
clear
B)
2F
done
clear
C)
F/2
done
clear
D)
F/4
done
clear
View Answer play_arrow
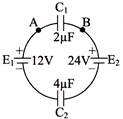
Two capacitors \[{{C}_{1}}\text{ }and\text{ }{{C}_{2}}\] are connected in a circuit as shown in figure. The potential difference \[{{V}_{A}}-{{V}_{B}}\] is-
A)
8 V
done
clear
B)
-8 V
done
clear
C)
12 V
done
clear
D)
-12 V
done
clear
View Answer play_arrow
A battery has an emf of 15 V and internal resistance of\[1\,\Omega \]. Is the terminal to terminal potential difference less than equal to or greater than 15V if the current in the battery is-
(i) from negative to positive terminal, (ii) from positive to negative terminal, (iii) zero current?
A)
Less, Greater, Equal
done
clear
B)
Less, Less, Equal
done
clear
C)
Greater, Greater, Equal
done
clear
D)
Greater, Less, Equal
done
clear
View Answer play_arrow
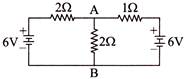
The potential difference between the points A and B in the following circuit will be-
A)
zero
done
clear
B)
2 V
done
clear
C)
3.5 V
done
clear
D)
4.5 V
done
clear
View Answer play_arrow
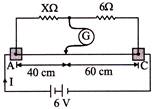
In the given circuit, a meter bridge is shown in balanced state. The resistance of bridge wire is\[1\,\Omega /cm\]. The current I drawn from the battery of negligible internal resistance is-
A)
0.22A
done
clear
B)
0.33A
done
clear
C)
0.44A
done
clear
D)
0.66A
done
clear
View Answer play_arrow
In a cyclotron, if a deuteron can gain an energy of 40 MeV, then a proton can gain an energy of
A)
40 MeV
done
clear
B)
80 MeV
done
clear
C)
20 MeV
done
clear
D)
60 MeV
done
clear
View Answer play_arrow
A proton, a deuteron and an \[\alpha -particle\] having the same kinetic energy are moving in circular trajectories in a constant magnetic field. If \[{{r}_{p}},\,\,{{r}_{d}}\,\,and\,\,{{r}_{\alpha }}\] denote respectively the radii of the trajectories of these particles, then-
A)
\[{{r}_{\alpha }}=\,\,{{r}_{p}}\,\,<\,\,{{r}_{d}}\]
done
clear
B)
\[{{r}_{\alpha }}>{{r}_{d}}\,>\,\,{{r}_{p}}\]
done
clear
C)
\[{{r}_{\alpha }}=\,\,{{r}_{d}}\,\,>\,\,{{r}_{p}}\]
done
clear
D)
\[{{r}_{p}}=\,\,{{r}_{d}}\,\,>\,\,{{r}_{\alpha }}\]
done
clear
View Answer play_arrow
A magnetic needle is kept in a non-uniform magnetic field. It may experiences-
A)
A force and torque
done
clear
B)
A force but not a torque
done
clear
C)
A torque but not a force
done
clear
D)
Neither a torque nor a force
done
clear
View Answer play_arrow
A metallic circular loop of radius r is placed in uniform magnetic field B acting perpendicular to the plane of the loop. A naughty boy pulls diametrically opposite corner so that after sometime the loop changes into an ellipse of major and minor radius a and b. If total resistance of loop is R and it remains constant during the pulling, the average charge flowing through loop during pulling is-
A)
\[\frac{B(\pi ab)}{R}\]
done
clear
B)
\[\frac{B(\pi ab-\pi {{r}^{2}})}{R}\]
done
clear
C)
\[\frac{B\pi {{r}^{2}}}{R}\]
done
clear
D)
\[\frac{B\pi br}{R}\]
done
clear
View Answer play_arrow
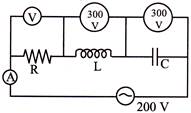
In the series circuit shown in the figure the voltmeter reading will be-
A)
300V
done
clear
B)
900V
done
clear
C)
200V
done
clear
D)
100V
done
clear
View Answer play_arrow
Electromagnetic wave consists of periodically oscillating electric and magnetic vectors-
A)
in mutually perpendicular planes but vibrating with a phase difference of \[\pi \]
done
clear
B)
in mutually perpendicular planes but vibrating with a phase difference of \[\frac{\pi }{2}\]
done
clear
C)
in randomly oriented planes but vibrating in phase
done
clear
D)
in mutually perpendicular planes but vibrating in phase
done
clear
View Answer play_arrow
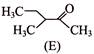
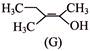
Which is correct statement concerning the structures E, F and G is-
A)
E, F and E, G are tautomers
done
clear
B)
F and G are geometrical isomers
done
clear
C)
F and G are diastereomers
done
clear
D)
All of the above
done
clear
View Answer play_arrow
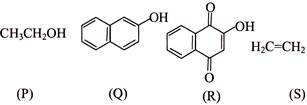
Arrange the following compounds in decreasing order of acidity-
A)
\[R>Q>P>S\]
done
clear
B)
\[Q>R>P>S\]
done
clear
C)
\[S>R>Q>P\]
done
clear
D)
\[Q>P>R>S\]
done
clear
View Answer play_arrow
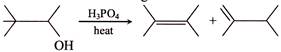
Consider the following reaction:
Which response contains all the correct statement about this process? [a] Dehydration [b] \[{{E}_{2}}\] mechanism [c] Rearrangement of carbocation [d] Most stable alkene will form [e] Single-step reaction
A)
a, c
done
clear
B)
a, b, c
done
clear
C)
a, b, e
done
clear
D)
a, c, d
done
clear
View Answer play_arrow
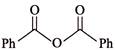
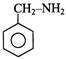
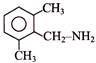
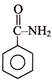
Consider the following two reaction sequences.
The two products (P) and (Q) are, respectively -
A)
\[PhCOC{{H}_{3}}\text{ }and\text{ }PhC{{H}_{2}}CHO\]
done
clear
B)
\[PhC{{H}_{2}}CHO\text{ }and\text{ }PhC{{H}_{2}}CHO\]
done
clear
C)
\[PhC{{H}_{2}}CHO\text{ }and\text{ }PhCOC{{H}_{3}}\]
done
clear
D)
\[PhCOC{{H}_{3}}\text{ }and\text{ }PhCOC{{H}_{3}}\]
done
clear
View Answer play_arrow
The compound (X) is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Consider the following reaction
Br
Which of the following base will give the best yield of the alkene II as the major product-
A)
\[C{{H}_{3}}{{O}^{-}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}O{{~}^{-}}\]
done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{3}}C{{O}^{-}}\]
done
clear
D)
\[{{\left( {{C}_{2}}{{H}_{5}} \right)}_{3}}C{{O}^{-}}\]
done
clear
View Answer play_arrow
Consider the following compounds,
What is the correct order of basicity of the above compounds?
A)
\[I>II>III~\]
done
clear
B)
\[III>I>II\]
done
clear
C)
\[\text{II}>I>III\]
done
clear
D)
\[\text{I}>III>II\]
done
clear
View Answer play_arrow
Trichloroacetaldehyde was subjected to cannizzaro's reaction by using NaOH. The mixture of the products contains sodium trichloroacetate ion and another compound. The other compound is:
A)
2, 2, 2-trichloroethanol
done
clear
B)
trichloromethanol
done
clear
C)
2, 2, 2-trichloropropanol
done
clear
D)
chloroform
done
clear
View Answer play_arrow
Which of the following statements are not correct?
A)
When phenol vapour is passed over Zn dust, benzene is produced
done
clear
B)
The phenolic -OH group is ortho-and para- directing
done
clear
C)
o-Nitrophenol is more acidic than p- nitrophenol
done
clear
D)
Phenol is more acidic than o-cresol
done
clear
View Answer play_arrow
The structure of compound Y:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following compounds can form dyes on reaction with \[HN{{O}_{2}}\,\,\And \,\,\beta -Naphthol\,\,at\,\,0-5{}^\circ C\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following give positive isocyanide test?
A)
done
clear
B)
\[C{{H}_{3}}-N{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}-NH-C{{H}_{3}}\]
done
clear
D)
done
clear
View Answer play_arrow
Which of the following drugs combination is not correct?
A)
Ranitidine - Tranquilizer
done
clear
B)
Chloremphenicol - Antibiotics
done
clear
C)
Equanil - Antidepressant
done
clear
D)
Aspirin - Antipyretic
done
clear
View Answer play_arrow
Nylon-6,6 is so named because it is a polyamide, formed from a
A)
six-carbon dibase and a six-carbon diamine
done
clear
B)
six-carbon diacid and a six-carbon diamine
done
clear
C)
six-carbon diacid and a six-carbon diamide
done
clear
D)
six-carbon e-caprolactum and a six-carbon diamine
done
clear
View Answer play_arrow
How many lines in the spectrum will be observed when electrons return from 7th shell to 2nd shell?
A)
13
done
clear
B)
14
done
clear
C)
15
done
clear
D)
16
done
clear
View Answer play_arrow
Which of the following expression is not applicable on the equilibrium for hydrolysis of
\[C{{N}^{-}}\] \[C{{N}^{-}}~+{{H}_{2}}O\rightleftharpoons HCN+O{{H}^{-}}\]
A)
\[{{K}_{h}}=\frac{{{K}_{w}}}{{{K}_{a}}}\]
done
clear
B)
\[h=\sqrt{\frac{{{K}_{H}}}{C}}~\]
done
clear
C)
\[pH=\frac{1}{2}p{{K}_{a}}\]
done
clear
D)
\[[{{H}^{+}}]=\frac{{{K}_{w}}{{K}_{a}}}{C}\]
done
clear
View Answer play_arrow
\[{{F}_{2}}\left( g \right)+2HCl\left( g \right)\to 2HF\left( g \right)+C{{l}_{2}}\left( g \right)\Delta H{}^\circ =-352\]\[\Delta {{H}_{f{}^\circ HF}}=-\,268.3\text{ }kJ\] The heat of formation of HCl will be-
A)
\[-22\text{ }kJ\text{ }mo{{l}^{-}}^{1}\]
done
clear
B)
\[88\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[-92.2\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
D)
\[-183.8\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
What would be the amount of heat released when an aqueous solution containing 0.5 mole of HNOs is mixed with 0.3 mole of OHT (enthalpy of neutralisation is -57.1 kJ)?
A)
28.5 kJ
done
clear
B)
17.1 kJ
done
clear
C)
45.7 kJ
done
clear
D)
1.7 kJ
done
clear
View Answer play_arrow
The decomposition of certain mass of \[CaC{{O}_{3}}\] gave \[11.2\text{ }d{{m}^{3}}\] of \[C{{O}_{2}}\] gas at STP. The mass of KOH required to completely neutralize the gas is
A)
56 g
done
clear
B)
28 g
done
clear
C)
42 g
done
clear
D)
20 g
done
clear
View Answer play_arrow
The metal which displaces hydrogen from a caustic soda solution is-
A)
Na
done
clear
B)
Zn
done
clear
C)
Mg
done
clear
D)
Fe
done
clear
View Answer play_arrow
Which quantity of nickel has the largest mass?
A)
one mole
done
clear
B)
\[6.02\times {{10}^{23}}atoms\]
done
clear
C)
58.7gm
done
clear
D)
22.4 moles
done
clear
View Answer play_arrow
\[N{{a}_{2}}S\]give purple colour with sodium nitroprusside. Purple colour has formulae:
A)
\[N{{a}_{2}}\left[ Fe{{\left( CN \right)}_{5}}S \right]\]
done
clear
B)
\[N{{a}_{2}}\left[ Fe{{\left( CN \right)}_{5}}NOS \right]\]
done
clear
C)
\[N{{a}_{4}}\left[ Fe{{\left( CN \right)}_{5}}NOS \right]\]
done
clear
D)
\[KFe\left[ Fe{{\left( CN \right)}_{5}}NO \right]\]
done
clear
View Answer play_arrow
(1) Interstitial compounds have high melting points, higher than those of pure metals. (2) Permangnate titrations in presence of hydrochloric acid are unsatisfactory (3) \[KMn{{O}_{4}}\] does not act as an oxidizing agent in strong alkaline medium (4) \[M{{n}^{2+}}\] compounds are more stable than \[F{{e}^{2+}}\] towards +2 states
A)
TTFT
done
clear
B)
TFFT
done
clear
C)
TFTT
done
clear
D)
FFTF
done
clear
View Answer play_arrow
\[C{{r}_{2}}O_{7}^{-2}\,+{{H}^{+}}+\,\,{{H}_{2}}{{O}_{2}}\to \,\,\,\,\,\underset{\,(deep\,\,blue\,\,colour)}{\mathop{'{A}'}}\,\,\,+\,\,\,{{H}_{2}}O\,\,\,\,\]
\[Mn{{O}_{2}}+KOH+{{O}_{2}}\to \text{ }\,\,\,\underset{(green)}{\mathop{'B'}}\,\,\,+\,\,{{H}_{2}}O~~~~~~~~~~~\left( green \right)\] \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\,\,\xrightarrow{\Delta }\,\,{{K}_{2}}Cr{{O}_{4}}+\,\,C{{r}_{2}}{{O}_{3}}\,+\,\,\,\,\,\underset{(Colourless\,\,gas)}{\mathop{'C'}}\,\]\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\,\,+\,\,{{H}_{2}}S{{O}_{4}}+NaCl\text{ }\to \] \[\begin{array}{*{35}{l}} {} \\ KHS{{O}_{4}}\,\,+\,\,NaHS{{O}_{4}}+{{H}_{2}}O \\ {} \\ \end{array}+\,\,\underset{\left( Reddish\text{ }brown\,\,vapour, \right)}{\mathop{'D'}}\,\]
A, B, C and D are
A)
\[C{{r}^{3+}},\,\,{{K}_{2}}Mn{{O}_{4}},\,\,{{O}_{2}},\,\,C{{l}_{2}}\]
done
clear
B)
\[C{{r}_{2}}{{O}_{3}},\,\,{{K}_{2}}Mn{{O}_{4}},\,\,{{O}_{2}},\,\,Cr{{O}_{2}}C{{l}_{2}}\]
done
clear
C)
\[CrO{{\left( {{O}_{2}} \right)}_{2}},\text{ }KMn{{O}_{4}},\text{ }{{O}_{2}},\text{ }Cr{{O}_{2}}C{{l}_{2}}\]
done
clear
D)
\[Cr{{O}_{5}},\text{ }{{K}_{2}}Mn{{O}_{4}},{{O}_{2}},Cr{{O}_{2}}C{{l}_{2}}\]
done
clear
View Answer play_arrow
Which reaction take place in Bessemer converter
A)
\[2C{{U}_{2}}O+C{{u}_{2}}S\,\,\to \,\,6Cu+S{{O}_{2}}\]
done
clear
B)
\[2C{{U}_{2}}S+3{{O}_{2}}\to 2C{{u}_{2}}O+2S{{O}_{2}}\]
done
clear
C)
\[CuO+C\to Cu+CO\]
done
clear
D)
\[CuC{{O}_{3}}\,\,\to \,\,CuO+C{{O}_{2}}\]
done
clear
View Answer play_arrow
The compound \[{{\left( Si{{H}_{3}} \right)}_{3}}N\] is expected to be:
A)
pyramidal and more basic than \[{{(C{{H}_{3}})}_{3}}N\]
done
clear
B)
planar and less basic than \[{{(C{{H}_{3}})}_{3}}N\]
done
clear
C)
pyramidal and less basic than \[{{(C{{H}_{3}})}_{3}}N\]
done
clear
D)
planer and more basic than \[{{(C{{H}_{3}})}_{3}}N\]
done
clear
View Answer play_arrow
Which represents correct order?
A)
\[\,N{{F}_{3}}>NC{{l}_{3}}>NB{{r}_{3}}>N{{I}_{3}}\] (order of lewis basic strength)
done
clear
B)
\[{{F}_{2}}<C{{l}_{2}}<B{{r}_{2}}<{{I}_{2}}\] (order of solubility in water)
done
clear
C)
\[N{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}>Sb{{H}_{3}}>Bi{{H}_{3}}\] (order of thermal stability)
done
clear
D)
\[{{F}^{-}}>C{{l}^{-}}>B{{r}^{-}}>{{I}^{-}}\] (order of reducing power)
done
clear
View Answer play_arrow
In a hydrogen-oxygen cell, 672 litre of \[{{H}_{2}}\] at NTP is used in 15 minutes. What is the average current produced?
A)
549.4 amp
done
clear
B)
643.33 amp
done
clear
C)
965 amp
done
clear
D)
129.8 amp
done
clear
View Answer play_arrow
An element X occurs in II short period having configuration \[n{{s}^{2}}\] \[n{{p}^{1}}\]. The formula and nature of its oxide is:
A)
\[X{{O}_{3}}\], basic
done
clear
B)
\[X{{O}_{3}}\], acidic
done
clear
C)
\[{{X}_{2}}{{O}_{3}}\], amphoteric
done
clear
D)
\[{{X}_{2}}{{O}_{3}}\], basic
done
clear
View Answer play_arrow
Find the species/molecule is having maximum number of lone pair on the central atom.
A)
\[ClOF_{4}^{-}\]
done
clear
B)
\[ClOF_{2}^{+}\]
done
clear
C)
\[BH_{4}^{-}\]
done
clear
D)
\[XeO{{F}_{2}}\]
done
clear
View Answer play_arrow
Which one of the following complete its octet.
A)
\[BeC{{l}_{2}}\left( g \right)\]
done
clear
B)
\[BeC{{l}_{2}}\left( s \right)\]
done
clear
C)
\[BC{{l}_{3}}\]
done
clear
D)
\[AlC{{l}_{3}}\]
done
clear
View Answer play_arrow
In which of the following pairs, both the species have the same hybridisation-
(I) \[S{{F}_{4}},\,\,Xe{{F}_{4}}\] (II) \[I_{3}^{-},\,\,Xe{{F}_{2}}\] (III) \[ICl_{_{4}}^{+},\,\,\,SiC{{l}_{4}}\] (IV) \[Cl{{O}^{-}}_{3},PO_{4}^{3-}\,\]
A)
I, II
done
clear
B)
II, III
done
clear
C)
II, IV
done
clear
D)
I, II, III
done
clear
View Answer play_arrow
The standard reduction potentials for \[Z{{n}^{2+}}/Zn,\,\,N{{i}^{2+}}/Ni,\,\,and\,\,F{{e}^{2+}}/Fe\] are -0.76, -0.23 and -0.44 V respectively. The reaction \[X+{{Y}^{2+}}\to {{X}^{2+}}+Y\] will be spontaneous when-
A)
\[X=Ni,Y=Zn\]
done
clear
B)
\[X=Fe,Y=Zn\]
done
clear
C)
\[X=Zn,Y=Ni\]
done
clear
D)
\[X=Ni,Y=Fe\]
done
clear
View Answer play_arrow
\[Zn|Z{{n}^{2+}}\,({{c}_{1}})\parallel Z{{n}^{2+}}\,({{c}_{2}})|Z\] for this cell \[\Delta G\] is negative if-
A)
\[{{C}_{1}}={{C}_{2}}\]
done
clear
B)
\[{{C}_{1}}>{{C}_{2}}\]
done
clear
C)
\[{{C}_{2}}>{{C}_{1}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
In a first order reaction the reactant substance has half-life period of ten minutes. What fraction of the substance will be left after an hour the reaction has occurred?
A)
1/6 of initial concentration
done
clear
B)
1/64 of initial concentration
done
clear
C)
1/12 of initial concentration
done
clear
D)
1/32 of initial of concentration
done
clear
View Answer play_arrow
The reaction of hydrogen, and iodine monochloride is represented by the equation:
\[{{H}_{2}}\left( g \right)+2ICl\left( g \right)\to 2HCl\left( g \right)+{{I}_{2}}(g)\] This reaction is first order in \[{{H}_{2}}\] (g) and also first order in ICl(g). Which of these proposed mechanism can be consistent with the given information about this reaction?
Mechanism I: \[{{H}_{2}}\left( g \right)+2ICl\left( g \right)\to 2HCl\left( g \right)+{{I}_{2}}\left( g \right)\]
Mechanism II: \[{{H}_{2}}(g)+ICl(g)\xrightarrow{Slow}\,\,HCl(g)+HI(g)\] \[HI(g)+ICl(g)\,\,\xrightarrow{fast}\,\,HCl(g)+{{I}_{2}}(g)\]
A)
I only
done
clear
B)
II only
done
clear
C)
both I and II
done
clear
D)
neither I nor II
done
clear
View Answer play_arrow
\[PtC{{l}_{4}}.\,6{{H}_{2}}O\] can exist as a hydrated complex 1 molal aqueous solution has depression in freezing point of \[3.72{}^\circ \]. Assume \[100%\] ionisation and \[{{K}_{f}}\left( {{H}_{2}}O \right)=1.86{}^\circ \text{ }mo{{l}^{-1}}kg\] kg, then complex is-
A)
\[\left[ Pt{{\left( {{H}_{2}}O \right)}_{6}} \right]C{{l}_{4}}\]
done
clear
B)
\[\left[ Pt{{\left( {{H}_{2}}O \right)}_{4}}C{{l}_{2}} \right]C{{l}_{2}}2{{H}_{2}}O\]
done
clear
C)
\[\left[ Pt{{\left( {{H}_{2}}O \right)}_{3}}C{{l}_{3}} \right]C{{l}_{3}}{{H}_{2}}O\]
done
clear
D)
\[\left[ Pt{{\left( {{H}_{2}}O \right)}_{4}}C{{l}_{4}} \right]4{{H}_{2}}O\]
done
clear
View Answer play_arrow
Shortest distance between octahedral & tetrahedral voids in C.C.P. will be
A)
\[\frac{a}{\sqrt{3}}\]
done
clear
B)
\[\frac{a\sqrt{3}}{4}\]
done
clear
C)
\[\frac{a\sqrt{3}}{2}\]
done
clear
D)
\[\frac{a}{\sqrt{2}}\]
done
clear
View Answer play_arrow
Which of the following statement is not correct in respect of hydrophilic sol.-
A)
The particles are hydrated
done
clear
B)
They are quite stable and are not easily coagulated
done
clear
C)
They are irreversible
done
clear
D)
There are considerable interactions between the dispersed phase and dispersion medium
done
clear
View Answer play_arrow
If the pressure of a given mass of gas is reduced to half and temperature is doubled simultaneously, the volume will be-
A)
Same as before
done
clear
B)
Twice as before
done
clear
C)
Four time as before
done
clear
D)
One fourth as before
done
clear
View Answer play_arrow
0.116 g of \[{{C}_{4}}{{H}_{4}}{{O}_{4}}(A)\] is neutralised by 0.074 g of\[Ca{{\left( OH \right)}_{2}}\]. Hence protonic hydrogen \[({{H}^{+}})\] in will be-
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
In the nuclear reaction\[_{92}^{238}\,U\,\,\,\to \,\,_{82}^{206}\,\,Pb\]. The number of a and P particles emitted are-
A)
\[7\alpha ,\,\,5\beta \]
done
clear
B)
\[6\alpha ,\,\,4\beta \]
done
clear
C)
\[4\alpha ,\,\,3\beta \]
done
clear
D)
\[8\alpha ,\,\,6\beta \]
done
clear
View Answer play_arrow
\[{{N}_{2}}+3{{H}_{2}}\rightleftharpoons 2N{{H}_{3}}\] Starting with one mole of nitrogen and 3 moles of hydrogen, at equilibrium \[50%\] of each had reached. If the equilibrium pressure is P, the pressure of hydrogen at equilibrium would be-
A)
P/2
done
clear
B)
P/3
done
clear
C)
P/4
done
clear
D)
P/6
done
clear
View Answer play_arrow
Consider the heterogeneous equilibrium in a closed container
\[N{{H}_{4}}HS\,(s)\,\rightleftharpoons \,N{{H}_{3}}(g)\,\,+\,\,{{H}_{2}}S(g)\] If more \[N{{H}_{4}}HS\] is added to the equilibrium
A)
Partial pressure of \[N{{H}_{3}}\] increases
done
clear
B)
Partial pressure of \[{{H}_{2}}S\] increases
done
clear
C)
Total pressure in the container increases
done
clear
D)
No effect on partial pressure of \[N{{H}_{3}}\] and \[{{H}_{2}}S\]
done
clear
View Answer play_arrow
On adding few drops of dil HCl to freshly precipitated ferric hydroxide, a red coloured colloidal solution is obtained. This phenomenon is known is-
A)
Peptisation
done
clear
B)
Dialysis
done
clear
C)
Protective action
done
clear
D)
Dissolution
done
clear
View Answer play_arrow
Process of photosynthesis in plant involved two major steps light reaction and dark reaction. The dark reaction of photosynthesis occur during:
A)
Daytime only
done
clear
B)
Night time only
done
clear
C)
Both day and night time
done
clear
D)
Morning and evening hrs. only
done
clear
View Answer play_arrow
If the source is opposite to the direction of sun, then honey bee will convey the direction by-
A)
Clockwise round dance
done
clear
B)
Upright down tail wagging dance
done
clear
C)
Anticlockwise round dance
done
clear
D)
Opposite to [b]
done
clear
View Answer play_arrow
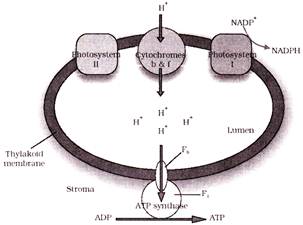
Select the number of correct statements regarding the given diagram-
A. The NADP reductase enzyme is located on the stroma side of the membrane. B. The primary electron acceptor of photosystem-II is located towards the outer side of membrane. C. Energy is used to pump protons across the membrane to create a high concentration of protons within the thylakoid lumen. D. The pH of stroma is acidic while of lumen of thylakoid is alkaline E. The chemiosmosis of protons from stroma to lumen leads to synthesis of ATP
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
2
done
clear
View Answer play_arrow
The end product of glycolysis is
A)
\[4\,\,ATP+2\,\,NADPH\,\,+\,\,2PA\]
done
clear
B)
\[2\,\,ATP+2\,\,NA{{D}^{+}}+2\,\,PA\]
done
clear
C)
\[ATP\,\,+\,\,4\,\,NA{{D}^{+}}+\,\,\,4\,\,PA\]
done
clear
D)
\[2\,ATP+2\,\,NAD{{H}_{2}}\,+\,\,2\,\,PA\]
done
clear
View Answer play_arrow
During the photophosphorylation the concentration of protons in stroma of chloroplast is low as protons move to lumen of thylakoid from stroma through-
A)
\[C{{f}_{0}}-C{{f}_{1}}\text{ }particle\]
done
clear
B)
Plastocyanin
done
clear
C)
Plastoquinone
done
clear
D)
Pheophytin
done
clear
View Answer play_arrow
From unripe opium fruit, the opium is extracted. From this which one of following groups of narcotic drugs are also prepared-
A)
Hashish, morphine, heroin
done
clear
B)
Hashish, codeine, pethidine and methadone
done
clear
C)
Codeine, pethidine, methadone, morphine and heroin
done
clear
D)
Codeine, pethidine, methadone and cocaine
done
clear
View Answer play_arrow
Match List-I with List - II and select correct answer
List-I (Variety) List-II (Crop, resistance to disease) A Pusa shubhra I Brassica, white rust B Pusa komal II Cauliflower, Black rot & curl blight C Pusa sadabahar III Cowpea, Bacterial blight D Pusa swarnim IV Chilli and Tobacco mosaic virus
A)
A-II B-IV C-III D-I
done
clear
B)
A-II B-III C-I D-IV
done
clear
C)
A-II B-I C-III D-IV
done
clear
D)
A-II B-III C-IV D-I
done
clear
View Answer play_arrow
A biennial plant can be changed into annual plant by which of the following treatment
A)
Low temperature treatment in second season
done
clear
B)
Cytokinin treatment in first season
done
clear
C)
Low temperature treatment in first season
done
clear
D)
Removal of axillary bud
done
clear
View Answer play_arrow
Which one of the following will reduce the rate of transpiration
A)
Increase in wind velocity
done
clear
B)
Rise in temperature
done
clear
C)
Increase in water uptake by plants
done
clear
D)
Increase in relative humidity
done
clear
View Answer play_arrow
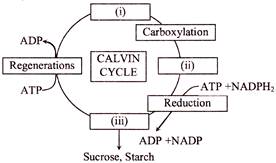
Choose the correct combinations of labeling the carbohydrate molecule involved in the Calvin cycle
A)
RuBP (ii) Triose phosphate (iii) PGA
done
clear
B)
PGA (ii) RuBP (iii) Triose phosphate
done
clear
C)
RuBP (ii) PGA (iii) Triose phosphate
done
clear
D)
Triose phosphate (ii) PGA (iii) RuBP
done
clear
View Answer play_arrow
I. No endosperm II. Endosperm present III. Cotyledons-thin IV. Cotylendons thick V. Food is stored in cotyledons VI. Food is stored in endosperms
Sort out correct points (from I to VI) for Albuminous and Exibuminous seeds (B).
A)
A- II, VI, VI B- I, III, IV
done
clear
B)
A-I, IV, V B- II, III, VI
done
clear
C)
A-I, III, V B- II, IV, VI
done
clear
D)
A-II, III, VI B- I, IV, V
done
clear
View Answer play_arrow
The separation of Archaebacteria into a separate domain indicates-
A)
That these organisms are very different from eubacteria and from eukaryotes
done
clear
B)
That early taxonomists rushed to classify them without closely examining them
done
clear
C)
That organisms should never be classified according to how they appear
done
clear
D)
None of these
done
clear
View Answer play_arrow
Out of the total cost of various ecosystem services established by R.constanza and collegue's highest cost is of which of the following service-
A)
Habitat for wild life
done
clear
B)
Pollination
done
clear
C)
Soil formation
done
clear
D)
Nutrient cycling
done
clear
View Answer play_arrow
Choose the feature not associated with organisms found in tropical deserts-
A)
Small body size
done
clear
B)
Large surface area relative to their volume
done
clear
C)
Large body extremities
done
clear
D)
Small surface area relative to their volume
done
clear
View Answer play_arrow
Sun light is the only source of energy for all ecosystems on earth except deep sea hydrothermal ecosystem. What is the source of energy in such ecosystems?
A)
Organic compound
done
clear
B)
Inorganic compound
done
clear
C)
Water currents
done
clear
D)
Warm vents
done
clear
View Answer play_arrow
Which of the following part of Funaria, sporophyte is involved in the dispersal of spores?
A)
Calyptra
done
clear
B)
Operculum
done
clear
C)
Peristoma
done
clear
D)
Annulus
done
clear
View Answer play_arrow
Cells are the basic unit of life of all living organism is evidenced by-
A. Most of cells are totipotent B. Omnis cellula e cellula C. Subcellular component can regenerate whole cell D. Anything less than a complete cell ensure independent existence
A)
A & B
done
clear
B)
A, B & D
done
clear
C)
A, B, C & D
done
clear
D)
A, B & C
done
clear
View Answer play_arrow
Which of the following are correct statement?
A. Cell cycle in organism is under genetic control B. Diplontic life cycle is present in angiosperm C. Xylan, mannan and galactan are present in cell wall of algae D. DNA polymerase synthesis, DNA replication and centriole duplication occurs in S-phase
A)
A, B, C & D
done
clear
B)
A, B & C
done
clear
C)
A & D
done
clear
D)
A, B & D
done
clear
View Answer play_arrow
How many of the following are present in eukaryotes and prokaryotes both Flagella, centriole, cell membrane, cell wall, 70s ribosome, 80s ribosome, DS DNA, sap vacuole
A)
Four
done
clear
B)
Five
done
clear
C)
Six
done
clear
D)
Three
done
clear
View Answer play_arrow
Fluidity of membrane can be measured by-
A)
Formation of inter cellular junction
done
clear
B)
Cell growth
done
clear
C)
Movement of molecule across membrane
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which of the following is involved in inheritance of character in animal?
A)
Nucleus
done
clear
B)
Chloroplast and nucleus
done
clear
C)
Nucleus and mitochondria
done
clear
D)
Nucleus, mitochondria and chloroplast
done
clear
View Answer play_arrow
Which of the following is a character of prophase-
A. Initiation of condensation of chromosome B. Centriole duplication and movement towards poles C. Dissolution of cytoskeleton D. Formation of spindle fiber with the help of microfilament
A)
A and C
done
clear
B)
B & C
done
clear
C)
A & B
done
clear
D)
All
done
clear
View Answer play_arrow
Segregation of homologous chromosome occur in
A)
Anaphase
done
clear
B)
Anaphase I
done
clear
C)
Anaphase II
done
clear
D)
[b] & [c] both
done
clear
View Answer play_arrow
The tendency of the recombined homologous chromosome of the bivalent to separate from each other during-
A)
Pachytene
done
clear
B)
Diplotene
done
clear
C)
Dikinesis
done
clear
D)
Zygotene
done
clear
View Answer play_arrow
According to binomial nomenclature, two words used for naming a plant or animal are
A)
Species and genus
done
clear
B)
Genus and species
done
clear
C)
Species and family
done
clear
D)
Genus and family
done
clear
View Answer play_arrow
Silk is the product of-
A)
Salivary gland of the larva
done
clear
B)
Cuticle of the adult
done
clear
C)
Cuticle of the larva
done
clear
D)
Salivary gland of the adult
done
clear
View Answer play_arrow
In Spirogyra, ladder like structure is formed in
A)
Lateral conjugation
done
clear
B)
Fragmentation
done
clear
C)
Palmella stage
done
clear
D)
Scalariform conjugation
done
clear
View Answer play_arrow
Unique features of living organisms are
A)
Ability to self-replicate
done
clear
B)
Self-organise
done
clear
C)
Interaction
done
clear
D)
All of these
done
clear
View Answer play_arrow
A. Eukaryotes B. Polysaccharide cell wall present C. Sexual reproduction present D. Multicellular E. Phototrophs F. Heterotrophs
How many of them are commonly present in plantae and fungi both-
A)
Three
done
clear
B)
Four
done
clear
C)
Two
done
clear
D)
Five
done
clear
View Answer play_arrow
Bryophytes are dependent on water because-
A)
Archegonium has to remain filled with water for fertilization
done
clear
B)
Water is essential for fertilization for their homosporous nature
done
clear
C)
Water is essential for their vegetative propagation
done
clear
D)
The sperms can easily reach upto egg in the archegonium
done
clear
View Answer play_arrow
Common feature between albugo and tobacco mosaic virus-
A)
Both are obligate parasite of plant
done
clear
B)
DNA is genetic material in both
done
clear
C)
Both are living
done
clear
D)
[a] & [b] both
done
clear
View Answer play_arrow
Which of the following is true during ageing
A)
Decreased blood Urea and GFR.
done
clear
B)
Increased calcium content of arteries and cartilage
done
clear
C)
Decreased cholesterol content of cornea and lens
done
clear
D)
Decreased calcium content of arteries and cartilage
done
clear
View Answer play_arrow
Read the following statements
a. Second stage of gametophyte development is leafy stage in mosses. b. Mosses have elaborate mechanism of spore dispersal.
A)
Both a & b are correct
done
clear
B)
Only a is correct
done
clear
C)
Both a & b are incorrect
done
clear
D)
Only b is correct
done
clear
View Answer play_arrow
In Spirogyra, ladder like structure is formed in
A)
Lateral conjugation
done
clear
B)
Fragmentation
done
clear
C)
Palmella stage
done
clear
D)
Scalariform conjugation
done
clear
View Answer play_arrow
When a genetically dwarf pea plant is treated with giberellin, it becomes as tall as the genetically tall plant. Suppose this gibberellin treated tall looking pea plant is crossed with a genetic dwarf pea plant, the progeny will consist of-
A)
3 tall : 1 dwarf
done
clear
B)
2 tall : 2 dwarf
done
clear
C)
All tall only
done
clear
D)
All dwarf only
done
clear
View Answer play_arrow
Which of the following is correctly matched?
A)
Chlorophyceae - Chl.a + Chl.b + Pectocellulose cell wall
done
clear
B)
Pheophyceae - Chl. A + Chl. C + cellulose + Carregenin cell wall
done
clear
C)
Rhodophycae - Chl. a + Chl. b +Pectocellulose cellwall
done
clear
D)
[a] & [b] both
done
clear
View Answer play_arrow
Complex tissue are
A)
Present only in sporophytes of tracheophyta
done
clear
B)
Heterogenous in structure and function
done
clear
C)
Made up of living and dead cell both
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Dicot stem vascular bundle form secondary tissue thus they are referred as-
A)
Open
done
clear
B)
Closed
done
clear
C)
Open or closed
done
clear
D)
Partially open
done
clear
View Answer play_arrow
Thorn in bougainvillea is for
A)
Reduction in transpiration
done
clear
B)
Protection against browsing
done
clear
C)
[a] & [b] Both
done
clear
D)
Vegetative reproduction
done
clear
View Answer play_arrow
If a annual plant is kept on compensation point for 20 days then-
A)
Survive successfully
done
clear
B)
Survive successfully, only weight loss
done
clear
C)
Survive successfully without exchange of gaseous from environment
done
clear
D)
Died due to lack of protoplasmic constituents
done
clear
View Answer play_arrow
A type of mental illness in which patients lose touch with reality is known as-
A)
Epilepsy
done
clear
B)
Neurosis
done
clear
C)
Mental deficiency
done
clear
D)
Pscychosis
done
clear
View Answer play_arrow
A. Flower is modified shoot wherein floral apical meristem changes to shoot apical meristem B. The apex produces different kind of floral appendages terminally at successive nodes
A)
A is true B is false
done
clear
B)
B is true A is false
done
clear
C)
A and B both are true
done
clear
D)
Both statements are false
done
clear
View Answer play_arrow
What type of phyllotaxy is shown in figure given-
A)
Opposite
done
clear
B)
Whorled
done
clear
C)
Alternate
done
clear
D)
Spiral
done
clear
View Answer play_arrow
Which of the following is correct floral formula of family fabaceae?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Correct ascending order of different region of root is
A)
Root cap, zone of meristematic activity, elongation zone, zone of differentiation
done
clear
B)
Root cap, zone of meristematic activitiy, zone of differentiation, elongation zone
done
clear
C)
Maturation zone, elongation zone, meristematic zone, root cap
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The female genital pore of Pheritima psothuma located upon the segment?
A)
\[{{14}^{th}}\]
done
clear
B)
\[{{16}^{th}}\]
done
clear
C)
\[{{18}^{th}}\]
done
clear
D)
\[{{15}^{th}}\]
done
clear
View Answer play_arrow
Which of the following would not be a response stimulated by a large fall in blood pressure?
A)
Constriction of afferent renal arteriole
done
clear
B)
Increased release of renin
done
clear
C)
Increased release of antidiuretic hormone
done
clear
D)
Increased thirst
done
clear
View Answer play_arrow
Which of these will be completely reabsorbed form glomerular filtrate under normal conditions in the nephrons?
A)
Urea
done
clear
B)
Salts
done
clear
C)
Glucose
done
clear
D)
Uric acid
done
clear
View Answer play_arrow
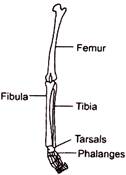
Given below is a diagram of the bones of the left human hindlimb as seen from front. It has certain mistakes in labelling. Two of the wrongly labelled bones are:
A)
Tibia & tarsals
done
clear
B)
Femur & fibula
done
clear
C)
Fibula & phalanges
done
clear
D)
Tarsals & femur
done
clear
View Answer play_arrow
Which of the following cardiac effects can be observed if the potassium concentration is increased two to three times the normal value-
A)
Weakness of heart
done
clear
B)
Abnormal rhythm
done
clear
C)
Death
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which of the following is an Non-proteinaceous enzyme-
A)
Ribo nuclease-P
done
clear
B)
Ribozyme
done
clear
C)
Lysozyme
done
clear
D)
above [a] & [b]
done
clear
View Answer play_arrow
Function of Ascending arm of Rente's loop is that-
A)
It loses urea to the renal medulla, helping this tissue to maintain it?s cone. gradient of solutes.
done
clear
B)
It provides water for reabsorption by the interstitial fluid and Capillaries
done
clear
C)
It helps to maintain the concentration gradient of NaCl in the interrstitial fluid. Thus increasing the water reabsorption
done
clear
D)
It absorps some drugs and poisons from the surrounding capillaries.
done
clear
View Answer play_arrow
Inhibition of secretion of which of the following hormone is necessary for disintegration of corpus luteum-
A)
LH
done
clear
B)
Progesterone
done
clear
C)
LTH
done
clear
D)
FSH
done
clear
View Answer play_arrow
Malarial parasites with 48 h cycle are-
A)
Plasmodium malaria, P.falciparum and-P.ovale
done
clear
B)
Plasmodium malariae, P.falciparwn and P.vivax
done
clear
C)
Plasmodium malariae, P.ovate and P.vivax
done
clear
D)
Plasmodium falciparum, P.ovale and P.vivax
done
clear
View Answer play_arrow
Which of the following will increase alveolar\[{{P}_{{{O}_{2}}}}\]?
A)
Increase in metabolism and no change in alveolar ventilation
done
clear
B)
Breathing air with 15 per cent oxygen at sea level
done
clear
C)
Increase in alveolar ventilation matched by an increase in metabolism
done
clear
D)
Increased alveolar ventilation with no change in metabolism
done
clear
View Answer play_arrow
ECG is least effective in detecting abnormalities in
A)
cardiac rhythm
done
clear
B)
coronary blood flow
done
clear
C)
cardiac contractility
done
clear
D)
atrioventricular conduction
done
clear
View Answer play_arrow
During fertilization, the enzyme which facilitates penetration of the egg by the spermatozoan is:
A)
hyaluronidase
done
clear
B)
acid phosphatase
done
clear
C)
acetylcholinesterase
done
clear
D)
alkaline phosphatase
done
clear
View Answer play_arrow
Which of the following variations are temporary and have nothing to do with the last or next generation-
A)
Hereditary variations
done
clear
B)
Discontinuous variations
done
clear
C)
Environmental variations
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which one of the following statements is incorrect about menstruation?
A)
During normal menstruation about 80 ml blood is lost
done
clear
B)
The menstrual fluid can easily clot
done
clear
C)
At menopause in the female, there is especially abrupt increase in gonadotropic hormones
done
clear
D)
The beginning of the cycle of menstruation is called menarche
done
clear
View Answer play_arrow
Vision in Cockroach is
A)
In bright light vision is mosaic while in olim light it is super position.
done
clear
B)
During day time it is mosaic, while in night it is super position.
done
clear
C)
Super position
done
clear
D)
Mosaic
done
clear
View Answer play_arrow
If cell A with \[OP=5\] and \[TP=4\] is surrounded by cell with \[OP-3\] and \[TP=1\], what will be the direction of water movement-
A)
From cell A to other cells
done
clear
B)
From other cells to cell A
done
clear
C)
Water will not move
done
clear
D)
Water will move up
done
clear
View Answer play_arrow
The characteristic larva of phylum ?cnidaria? is-
A)
planula
done
clear
B)
cysticercus
done
clear
C)
rhabditiform
done
clear
D)
wriggler
done
clear
View Answer play_arrow
Blood is a kind of-
A)
Liquid matrix
done
clear
B)
Fluid connective tissue
done
clear
C)
Cell mass
done
clear
D)
An organ
done
clear
View Answer play_arrow
Ligaments join-
A)
Bones to bones
done
clear
B)
Muscles to muscles
done
clear
C)
Bones to muscles
done
clear
D)
Muscles to cartilages
done
clear
View Answer play_arrow
Sarcolemma is a membrane found around-
A)
Nerve fibre
done
clear
B)
Cardiac muscle cell
done
clear
C)
Muscle fibre
done
clear
D)
Heart
done
clear
View Answer play_arrow
AZT is the treatment of:
A)
TB
done
clear
B)
AIDS
done
clear
C)
Malaria
done
clear
D)
Kala-azar
done
clear
View Answer play_arrow
Given below is a table comparing the effects of sympathetic and parasympathetic nervous system for four features\[\left( 1-4 \right)\]. Which one feature is correctly described-
Feature Sympathetic nervous system Parasympathetic nervous system 1 Salivary glands Stimulus secretion Inhibits secretion 2 Anal sphincter Dilate Constricts 3 Gastric gland Decreases gastric juice Increases gastric juice 4 Intestinal peristalsis Stimulus Inhibits
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following controls and coordinates voluntary muscles?
A)
Medulla oblongata
done
clear
B)
Sympathetic N.S.
done
clear
C)
Parasympathetic N.S.
done
clear
D)
Central nervous system
done
clear
View Answer play_arrow
An action potential in the nerve fibre is produced when positive and negative charges on the outside and the inside of the axon membrane are reversed because-
A)
More potassium ions enter the axon than sodium ions leaving it
done
clear
B)
More sodium ions enter the axon
done
clear
C)
All potassium ions leave the axon
done
clear
D)
All sodium ions exit the axon
done
clear
View Answer play_arrow
Cerebrospinal fluid is secreted by-
A)
Cerebrum
done
clear
B)
Cerebellum
done
clear
C)
Olfactory lobe
done
clear
D)
Choroid plexus
done
clear
View Answer play_arrow
Net Primary productivity (NPP) is equal to:
A)
GPP + Loss in respiration
done
clear
B)
GPP - Loss in respiration
done
clear
C)
Secondary Productivity - GPP
done
clear
D)
Secondary Productivity + GPP
done
clear
View Answer play_arrow
Choose the correct statement regarding the ZIFT procedure
A)
Ova collected from the female donor are transferred to the fallopian tube to facilitate zygote formation
done
clear
B)
Zygote is collected from a test tube and transferred to the fallopian tube
done
clear
C)
Zygote is collected from a female donor and transferred to the uterus
done
clear
D)
Ova collected from a female donor and transferred to the uterus
done
clear
View Answer play_arrow
After tubectomy what will not occur in a female-
A)
Ovulation
done
clear
B)
Menstrual cycle
done
clear
C)
Bleeding at end of menstrual cycle
done
clear
D)
Fertilization
done
clear
View Answer play_arrow
Nerve cells do not occur in
A)
Nematodes
done
clear
B)
Mosquitoes
done
clear
C)
Sponges
done
clear
D)
Coelenterates
done
clear
View Answer play_arrow
?Venus flower basket? is the name of the dried skeleton of
A)
Euspongia
done
clear
B)
Euplectella
done
clear
C)
Spongilla
done
clear
D)
Leucosolenia
done
clear
View Answer play_arrow
Overall ......... nerves slow down the body and divert energy to digestion and other basic house -keeping Tasks; and........ nerves slow down house-keeping tasks and increase overall activity in times of heightened awareness or excitement.
A)
Peripheral; central
done
clear
B)
Parasympathetic; sympathetic
done
clear
C)
Sympathetic; Parasympathetic
done
clear
D)
Autonomic; somatic
done
clear
View Answer play_arrow
Match the following and choose the correct combination from the options given-
Column-I
Column-II
A.
Escherichia coli
1.
'Nif' gene
B.
Rhizobium meliloti
2.
Digestive hydrocarbons of crude oil
C.
Bacillus thurigiensis
3.
Human insulin production
D.
Pseudomonas putida
4.
Biocontrol of fungal disease
5.
Biodegradable insecticide
A)
A=3, B=1, C=5, D=4
done
clear
B)
A=1, B=2, C=3, D=4
done
clear
C)
A=2, B=1, C=3, D=4
done
clear
D)
A=3, B=1, C=5, D=2
done
clear
View Answer play_arrow
Comparing the somatic nervous system with the autonomic nervous system, which of the following statements is true-
A)
Autonomic nervous system Produces involuntary movement in smooth muscle, viscera and glands.
done
clear
B)
Autonomic nervous system produces voluntary movement in smooth muscle and glands.
done
clear
C)
Somatic nervous system produces voluntary movement in glands and smooth muscles.
done
clear
D)
Autonomic nervous system produces involuntary movement in skeletal muscle.
done
clear
View Answer play_arrow
Match the following in column-I with column-II and choose the correct combination:
Column-I Column-II A. Termination 1. Aminoacyi t-RNA synthetase B. Translation 2. Okazaki fragments C. Transcription 3. UAA D. DNA replication 4. RNA polymerase
A)
A =2, B=3, C=1, D=4
done
clear
B)
A=1, B=4, C=4, D=3
done
clear
C)
A=3, B=1, C=4, D=2
done
clear
D)
A =4, B=2, C=1, D=3
done
clear
View Answer play_arrow
P.C.R modification is
A)
A.F.L.P.
done
clear
B)
R.A.P.D.
done
clear
C)
Both of above
done
clear
D)
R.F.L.P.
done
clear
View Answer play_arrow
Cleavage is a unique form of mitotic cell- division in that-
A)
Spindle formation do not take place
done
clear
B)
There is no growth of cells
done
clear
C)
The nucleus does not participate
done
clear
D)
Plasma membrane of daughter cells do not separate
done
clear
View Answer play_arrow
In operon concept, the operator gene combines with-
A)
Regulator gene to switch off structural gene transcription
done
clear
B)
Regulator protein to switch on structural gene transcription
done
clear
C)
Regulator protein to switch off structural gene transcription
done
clear
D)
Inducer to switch off structural gene transcription
done
clear
View Answer play_arrow
Match the list-I and List-11 and select the answer using the code given below:
List-I (Scientist) List-II (Discovery) A Fredrick Griffith 1 Phenomenon of transformation B Gamow 2 A sequence of 3 nucleotide codes for a single amino acid C F. Miescher 3 Isolated DNA for the first time
Code:
A)
A-1 B-2 C-3
done
clear
B)
(b) A-2 B-3 C-1
done
clear
C)
A-3 B-2 C-1
done
clear
D)
A-2 B-1 C-3
done
clear
View Answer play_arrow
The idea of Natural Selection as the fundamental process of evolutionary changes was reached-
A)
By Alfred Russel Wallace in 1901
done
clear
B)
Independently by Charles Darwin and Alfred Russel Wallace in 1859
done
clear
C)
Independently by Charles Darwin and Alfred Russel Wallace in 1900
done
clear
D)
By Charles Darwin in 1866
done
clear
View Answer play_arrow
\[N{{a}^{+}}-~{{K}^{\text{+}}}\] pump is found in membranes of many cells, like nerve cells. It works against electro chemical gradient and involve an integral protein ATPase. For each molecule of ATP used-
A)
3 ions of \[N{{a}^{+}}\] are pumped out and two \[{{K}^{+}}\] are taken in
done
clear
B)
3 ions of \[N{{a}^{+}}\] are taken in and \[2{{K}^{+}}\] are pumped out
done
clear
C)
2 ions of \[N{{a}^{+}}\] are thrown out \[3{{K}^{+}}\] are absorbed
done
clear
D)
3 ions of \[{{K}^{+}}\] are absorbed and \[3N{{a}^{+}}\] are pumped out
done
clear
View Answer play_arrow
Match List-I and List-II and select the correct answer using the codes given below the lists :
List-I (Peculiarity of male-determining sperm) List-II (Organism in which it is seen) A No Y-chromosome is needed at all 1. Grasshopper B Necessarily with a Y-chromosome 2. Male honeybee C With haploid set of autosomes 3. Birds D. With W-chromosome 4. Drosophila 5. Human
A)
A=2, B=1, C=3, D=4
done
clear
B)
A=5, B=2, C=4, D=3
done
clear
C)
A=3, B=5, C=1, D=4
done
clear
D)
A=1, B=5, C=2, D=3
done
clear
View Answer play_arrow
Which one of the following pairs is not correctly matched-
A)
Plasmid Small piece of extra chromosomal DNA in bacteria
done
clear
B)
Interferon An enzyme that interferes with DNA replication
done
clear
C)
Cosmid A vector for carrying large DNA fragments into host cells
done
clear
D)
Myeloma Anitbody-producing tumour cells
done
clear
View Answer play_arrow
What is the purpose of tubectomy-
A)
To prevent embryonic development
done
clear
B)
To prevent sexual intercourse
done
clear
C)
To prevent formation of eggs
done
clear
D)
To prevent fertilization
done
clear
View Answer play_arrow
Which of the following type of gynoecium is associated by wind pollination
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Intra-ovarian fertilization means fertilization
A)
Outside ovule
done
clear
B)
Outside embryo sac
done
clear
C)
By putting pollens directly into ovary wall
done
clear
D)
Between male gamete and synergids
done
clear
View Answer play_arrow
 The two products (P) and (Q) are, respectively -
The two products (P) and (Q) are, respectively - 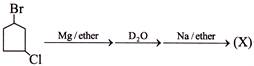 The compound (X) is:
The compound (X) is: 
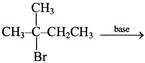 Br
Br 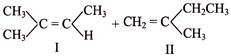 Which of the following base will give the best yield of the alkene II as the major product-
Which of the following base will give the best yield of the alkene II as the major product- 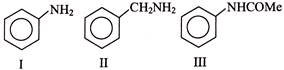 What is the correct order of basicity of the above compounds?
What is the correct order of basicity of the above compounds? 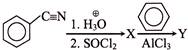 The structure of compound Y:
The structure of compound Y: