If F, V and T were chosen as fundamental units of force, velocity and time respectively, the dimensions of mass would be represented as:
A)
\[\text{FVT }\!\!~\!\!\text{ }\]
done
clear
B)
\[FV{{T}^{-1}}\]
done
clear
C)
\[F{{V}^{-1}}T\]
done
clear
D)
\[F{{V}^{-1}}{{T}^{-1}}\]
done
clear
View Answer play_arrow
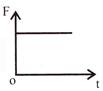
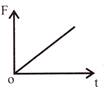
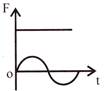
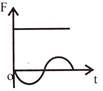
The displacement time graph of a particle executing S.H.M is as shown in the figure. The corresponding force-time graph of the particle is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A projectile is thrown with an initial velocity of\[\left( a\hat{i}+b\hat{j} \right)m{{s}^{-1}}\]. If the range of the projectile is twice the maximum height reached by it, then:
A)
\[b=\frac{a}{2}\]
done
clear
B)
\[\operatorname{b} = a\]
done
clear
C)
\[\operatorname{b} = 2a\]
done
clear
D)
\[\operatorname{b} = 4a\]
done
clear
View Answer play_arrow
A wire of length L and cross sectional area A is made of a material of young's modulus Y. If the wire is stretched by an amount x, the work done is:
A)
\[\frac{YA{{x}^{2}}}{2L}\]
done
clear
B)
\[\frac{YAx}{2{{L}^{2}}}\]
done
clear
C)
\[\frac{YAx}{2L}\]
done
clear
D)
\[\frac{YA{{x}^{2}}}{L}\]
done
clear
View Answer play_arrow
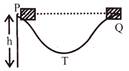
A block is placed at point P of a track shown in the figure. It is slightly pushed by a force toward right. The speed of block just reaching point Q is:
A)
Zero
done
clear
B)
\[\sqrt{2gh}\]
done
clear
C)
\[\sqrt{gh}\]
done
clear
D)
\[\frac{\sqrt{3gh}}{2}\]
done
clear
View Answer play_arrow
A ball is thrown vertically upwards with a velocity of 25m/s from the top of a tower of height 30m. How long will it travel before it hits ground:
A)
1 sec
done
clear
B)
6 sec
done
clear
C)
2 sec
done
clear
D)
5 sec
done
clear
View Answer play_arrow
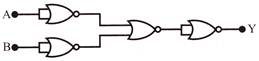
The circuit as shown in the figure is equivalent to:
A)
AND
done
clear
B)
NOR
done
clear
C)
OR
done
clear
D)
NAND
done
clear
View Answer play_arrow
Two non-mixing liquids of densities \[\rho and \]\[n\rho \left( n > 1 \right)\]are put in a container. The height of each liquid is h. A solid cylinder of length L and density d is put in this container. The cylinder floats with its axis vertical and length pL(p < 1) in the denser liquid. The density d is equal to:
A)
\[\left\{ 1+\left( n+1 \right)p \right\}\rho \]
done
clear
B)
\[\left\{ 2+\left( n+1 \right)p \right\}\rho \]
done
clear
C)
\[\left\{ 2+\left( n-1 \right)p \right\}\]
done
clear
D)
\[\left\{ 1+\left( n-1 \right)p \right\}\rho \]
done
clear
View Answer play_arrow
If the kinetic energy of the particle is increased to 16 times its previous value, wavelength of the particle is:
A)
25
done
clear
B)
75
done
clear
C)
60
done
clear
D)
50
done
clear
View Answer play_arrow
A magnetic needle suspended parallel to a magnetic field require \[\sqrt{3}J\] of work to turn it through\[60{}^\circ \]. The torque needed to maintain the needle in this position will be:
A)
\[2\sqrt{3}J\]
done
clear
B)
\[3J\]
done
clear
C)
\[\sqrt{3}J\]
done
clear
D)
\[\frac{3J}{2}\]
done
clear
View Answer play_arrow
Hydrogen atom in ground state is excited by a monochromatic radiation of\[~\lambda = 975 \overset{\operatorname{o}}{\mathop{A}}\,\]. Number of spectral lines in the resulting spectrum emitted will be:
A)
3
done
clear
B)
2
done
clear
C)
6
done
clear
D)
10
done
clear
View Answer play_arrow
A closed loop PQRS carrying a current is placed in a uniform magnetic field. If the magnetic forces on segments PS, SR and RQ are\[{{\operatorname{F}}_{1}} = 2N\], \[{{\operatorname{F}}_{2}} = 6N and {{F}_{3}}= 3N\]and are in the plane of the paper and along the direction shows, the force on the segment QP is:
A)
3N
done
clear
B)
5N
done
clear
C)
4N
done
clear
D)
IN
done
clear
View Answer play_arrow
The vessels A and B of equal volume and weight are immersed in water to depth h. The vessel A has an opening at the bottom through which water can enter. If the work done in immersing A and B are W and Wg respectively then:
A)
\[{{\operatorname{W}}_{A}} = {{W}_{B}}\]
done
clear
B)
\[{{\operatorname{W}}_{A}}< {{W}_{B}}\]
done
clear
C)
\[{{\operatorname{W}}_{A}}> {{W}_{B}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
de-broglie wavelength of an electron and the wavelength of a photon are the same. The ratio between the energy of that photon and the momentum of that electron is:
A)
h
done
clear
B)
c
done
clear
C)
\[\frac{1}{h}\]
done
clear
D)
\[\frac{1}{c}\]
done
clear
View Answer play_arrow
When cooking oil is heated in a frying pan, the oil moves around in the pan more easily when it is hot. The main reason for this is that with rise in temperature, there is decrease in:
A)
Surface tension
done
clear
B)
Viscosity
done
clear
C)
Angle of contact
done
clear
D)
Density
done
clear
View Answer play_arrow
A tank full of water has a small hole in its bottom. If one-fourth of the tank is emptied in \[{{t}_{1}}\]seconds and the remaining three fourths of the tank is emptied in \[{{t}_{2}}\] seconds, then ratio \[\frac{{{t}_{1}}}{{{t}_{2}}}\]is:
A)
\[\sqrt{3}\]
done
clear
B)
\[\sqrt{2}\]
done
clear
C)
\[\frac{2-\sqrt{2}}{\sqrt{2}}\]
done
clear
D)
\[\frac{2-\sqrt{3}}{\sqrt{3}}\]
done
clear
View Answer play_arrow
The acceleration due to gravity on the planet A is 9 times the acceleration due to gravity on planet B. A man jumps from a height of 2m on the surface of. What is the height jump by the same person on planet B?
A)
\[\text{6m}\]
done
clear
B)
\[\frac{2}{3}m\]
done
clear
C)
\[\frac{2}{9}m\]
done
clear
D)
\[18m\]
done
clear
View Answer play_arrow
The primary and secondary coils of a transformer have 50 and 1500 turns, respectively. If the magnetic flux \[\phi \] linked \[\phi ={{\phi }_{\circ }}+4t,\]with the primary coil is given by where is in Wb, t is time in second and \[{{\phi }_{\circ }}\]is a constant, then the output voltage across the secondary coil is:
A)
90V
done
clear
B)
120V
done
clear
C)
220V
done
clear
D)
30V
done
clear
View Answer play_arrow
If the phase difference between two sound waves of wavelength is \[60{}^\circ \], the corresponding path difference is:
A)
\[\frac{\lambda }{2}\]
done
clear
B)
\[\frac{\lambda }{6}\]
done
clear
C)
\[2\lambda \]
done
clear
D)
\[\frac{\lambda }{4}\]
done
clear
View Answer play_arrow
A potentiometer wire has length 4m and resistance\[8\Omega \]. The resistance that must be connected in series with the wire and an accumulator of emf 2V, so as to get a potential gradient 1mV per cm on the wire is:
A)
\[48\Omega \]
done
clear
B)
\[32\Omega \]
done
clear
C)
\[40\Omega \]
done
clear
D)
\[44\Omega \]
done
clear
View Answer play_arrow
A mixture consists of two radioactive materials \[{{A}_{1}}\] and \[{{A}_{2}}\] with half-lives of 20 sec and 10 sec, respectively. Initially, the mixture has 40g of \[{{A}_{1}}\]and 160g of\[{{A}_{2}}\]. The amount of the two in the mixture will become equal after:
A)
60 sec
done
clear
B)
80 sec
done
clear
C)
20 sec
done
clear
D)
40 sec
done
clear
View Answer play_arrow
A gas is heated at constant pressure. The fraction of heat supplied used for external work is:
A)
\[\frac{1}{\gamma }\]
done
clear
B)
\[1-\frac{1}{\gamma }\]
done
clear
C)
\[\gamma -1\]
done
clear
D)
\[1-\frac{1}{{{\gamma }^{2}}}\]
done
clear
View Answer play_arrow
A particle moves along the X-axis. The position x of a particle w.r.t. time from origin given by\[x={{b}_{\circ }}+{{b}_{1}}t+{{b}_{2}}{{t}^{2}}\]. The acceleration of the particle is:
A)
\[{{b}_{{}^\circ }}\]
done
clear
B)
\[{{b}_{1}}\]
done
clear
C)
\[{{b}_{2}}\]
done
clear
D)
\[2{{b}_{2}}\]
done
clear
View Answer play_arrow
The energy required to move a satellite of mass m from orbit of radius 2R to 3R around earth of mass M is:
A)
\[\frac{GMm}{12R}\]
done
clear
B)
\[\frac{GMm}{R}\]
done
clear
C)
\[\frac{GMm}{8R}\]
done
clear
D)
\[\frac{GMm}{2R}\]
done
clear
View Answer play_arrow
A ball falls from a height of 20m on the floor and rebounds to a height of 5m. Time of contact is 0.02 seFind the acceleration during impact?
A)
\[12m/{{s}^{2}}\]
done
clear
B)
\[1000m/{{s}^{2}}\]
done
clear
C)
\[2000m/{{s}^{2}}\]
done
clear
D)
\[1500m/{{s}^{2}}\]
done
clear
View Answer play_arrow
If \[{{n}_{1}},{{n}_{2}},{{n}_{3}}\]are the fundamental frequencies of three segments into which a string is divided, then the original fundamental frequency n of the string is given by:
A)
\[\frac{1}{n}=\frac{1}{{{n}_{1}}}+\frac{1}{{{n}_{2}}}+\frac{1}{{{n}_{3}}}\]
done
clear
B)
\[\frac{1}{\sqrt{n}}=\frac{1}{{{\sqrt{n}}_{1}}}+\frac{1}{\sqrt{{{n}_{2}}}}+\frac{1}{\sqrt{{{n}_{3}}}}\]
done
clear
C)
\[\sqrt{n}=\sqrt{{{n}_{1}}}+\sqrt{{{n}_{2}}}+\sqrt{{{n}_{3}}}\]
done
clear
D)
\[n={{n}_{1}}+{{n}_{2}}+{{n}_{3}}\]
done
clear
View Answer play_arrow
If a battery of 6V is connected to the terminals of 3m long wire of uniform thickness and resistance of the order of \[100\Omega \] The difference of potential difference between the two points separated by 50 cm in the wire is:
A)
1V
done
clear
B)
1.5V
done
clear
C)
2V
done
clear
D)
3V
done
clear
View Answer play_arrow
A body moves with uniform acceleration, then which of the following graphs is correct:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The temperature inside a refrigerator is \[{{t}_{2}}^{\circ }C\] and the room temperature is \[{{t}_{1}}^{\circ }C\] The amount of heat delivered to the room for each joule of electrical energy consumed ideally will be:
A)
\[\frac{{{t}_{1}}+{{t}_{2}}}{{{t}_{1}}+273}\]
done
clear
B)
\[\frac{{{t}_{1}}}{{{t}_{1}}+{{t}_{2}}}\]
done
clear
C)
\[\frac{{{t}_{1}}+273}{{{t}_{1}}+{{t}_{2}}}\]
done
clear
D)
\[\frac{{{t}_{2}}+273}{{{t}_{1}}+{{t}_{2}}}\]
done
clear
View Answer play_arrow
If Young's double slit experiment is performed in water instead of air, then:
A)
No fringe would be seen
done
clear
B)
Fringe width would decrease
done
clear
C)
Fringe width would increase
done
clear
D)
Fringe width would remain unchanged
done
clear
View Answer play_arrow
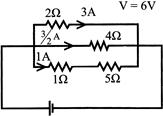
A current of 3A flows through the 20, resistor shown in the circuit. The power dissipated in the 50 resistor is:
A)
4W
done
clear
B)
2W
done
clear
C)
1W
done
clear
D)
5W
done
clear
View Answer play_arrow
The apparent depth of water in cylindrical water tank of diameter 2R cm is reducing at the rate of x cm/min when water is being drained out at a constant rate. The amount of water drained in cc per minute is [\[{{n}_{1}}\]= refractive index of air, \[{{n}_{2}}\]= refractive index of water]
A)
\[n\frac{x\pi {{R}^{2}}{{n}_{1}}}{{{n}_{2}}}\]
done
clear
B)
\[n\frac{x\pi {{R}^{2}}{{n}_{2}}}{{{n}_{2}}}\]
done
clear
C)
\[n\frac{2\pi R{{n}_{1}}}{{{n}_{2}}}\]
done
clear
D)
\[\pi {{R}^{2}}x\]
done
clear
View Answer play_arrow
Two block of masses 7kg and 5kg are placed in contact with each other on a smooth surface. If a force of 6N is applied on the heavier mass, the force on the lighter mass is:
A)
3.5N
done
clear
B)
2.5N
done
clear
C)
7N
done
clear
D)
5N
done
clear
View Answer play_arrow
A solid sphere of volume V and density p floats at the interface of two immiscible liquids of densities \[{{p}_{1}}\]and \[{{p}_{2}}\]respectively. If \[{{p}_{1}}<p<{{r}_{2}}\]then the ratio of volume of the parts of the sphere in upper and lower liquids is:
A)
\[\frac{p-{{p}_{1}}}{{{p}_{2}}-p}\]
done
clear
B)
\[\frac{{{p}_{2}}-p}{p-{{p}_{1}}}\]
done
clear
C)
\[\frac{p+p}{p+{{p}_{2}}}\]
done
clear
D)
\[\sqrt{\frac{{{p}_{1}}{{p}_{2}}}{p}}\]
done
clear
View Answer play_arrow
Eight oil drops of same size are charged to a potential of 50V each. These oil drops are merged into one single large drop. What will be the potential of the large drop?
A)
50V
done
clear
B)
100V
done
clear
C)
200V
done
clear
D)
400V
done
clear
View Answer play_arrow
If resistance\[R=10\Omega \], inductance \[L=2mH\]and capacitance \[C=5\mu F\]are connected in series to an AC source of frequency 50Hz, then impedance at resonance will be:
A)
\[Zero\]
done
clear
B)
\[10\Omega \]
done
clear
C)
\[1k\Omega \]
done
clear
D)
\[110k\Omega \]
done
clear
View Answer play_arrow
If the angular momentum of a rotating body about a fixed axis is increased by 10%. Its kinetic energy will be increased by:
A)
10%
done
clear
B)
20%
done
clear
C)
21%
done
clear
D)
5%
done
clear
View Answer play_arrow
The electric field due to an electric dipole at a distance r from its centre in axial position is E. If the dipole is rotated through an angle of \[90{}^\circ \]about its perpendicular axis, the magnitude of electric field at the same point is:
A)
\[E\]
done
clear
B)
\[\frac{E}{4}\]
done
clear
C)
\[\frac{E}{2}\]
done
clear
D)
\[2E\]
done
clear
View Answer play_arrow
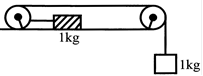
Consider the system shown in figure. The pulley and the string and all the surfaces are frictionless. The tension in the string is (in Newton)
A)
0
done
clear
B)
1
done
clear
C)
2
done
clear
D)
5
done
clear
View Answer play_arrow
The primary winding of transformer has 500 turns whereas its secondary has 5000 turns. The primary is connected to an AC supply of 20V, 50Hz. The secondary will have an output of:
A)
2V, 5Hz
done
clear
B)
200V, 500Hz
done
clear
C)
2V, 50Hz
done
clear
D)
200V, 50Hz
done
clear
View Answer play_arrow
A cyclic process is shown in the figure. Work done during the cyclic process ABCDA is:
A)
160J
done
clear
B)
150J
done
clear
C)
600J
done
clear
D)
900J
done
clear
View Answer play_arrow
In the system shown in figure, tension \[{{T}_{3}}\] is:
A)
5g
done
clear
B)
3g
done
clear
C)
g
done
clear
D)
6g
done
clear
View Answer play_arrow
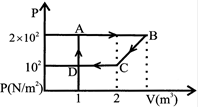
The ad joining figure shows graph of pressure and volume of a gas at two temperature \[{{T}_{1}}\]and \[{{T}_{2}}\]which inference is true:
A)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}<{{T}_{2}}\]
done
clear
D)
\[None\]
done
clear
View Answer play_arrow
A body of mass 2kg is floating in water with half its volume submerge What would be the force required to wholly submerge it into the water:
A)
2N
done
clear
B)
9.8N
done
clear
C)
19.6N
done
clear
D)
4.9N
done
clear
View Answer play_arrow
A block of weight 4kg is resting on a smooth horizontal plane. If it is struck by a jet of water at the rate of 2kg/s and at the speed of 10 m/s, then the initial acceleration of the block is:
A)
\[15m/{{s}^{2}}~\]
done
clear
B)
\[10m/{{s}^{2}}\]
done
clear
C)
\[2.5m/{{s}^{2~}}\]
done
clear
D)
\[5m/{{s}^{2}}\]
done
clear
View Answer play_arrow
The volume of water to be added \[100\,c{{m}^{3}}\]of \[0.5\,N{{H}_{2}}S{{O}_{4}}\] to get deci normal concentration is:
A)
\[100\,c{{m}^{3}}\]
done
clear
B)
\[450\,c{{m}^{3}}\]
done
clear
C)
\[500\,c{{m}^{3}}\]
done
clear
D)
\[400\text{ }c{{m}^{3}}\]
done
clear
View Answer play_arrow
If the electron falls from n = 3 to n = 2 in H-atom, then emitted energy is:
A)
10.2ev
done
clear
B)
12.09ev
done
clear
C)
1.9ev
done
clear
D)
0.65ev
done
clear
View Answer play_arrow
A body of mass 10 mg is moving with a velocity of\[100\,m{{s}^{-1}}\]. The wavelength of the de-Broglie wave associated with it:
A)
\[6.63\times {{10}^{-7}}\]
done
clear
B)
\[6.63\times {{10}^{-31}}m\]
done
clear
C)
\[6.63\times {{10}^{-4}}m\]
done
clear
D)
\[6.63\times {{10}^{-35}}m\]
done
clear
View Answer play_arrow
The increasing order of the first ionization enthalpy of the elements B, P, S and F (lowest first) is:
A)
B<P<S<F
done
clear
B)
B<S<P<F
done
clear
C)
F<S<P<B
done
clear
D)
P<S<B<F
done
clear
View Answer play_arrow
Which of the following contains maximum number of lone pairs on the central atom?
A)
\[CIO_{^{3}}^{-}\]
done
clear
B)
\[Xe{{F}_{4}}\]
done
clear
C)
\[S{{F}_{4}}\]
done
clear
D)
\[I_{3}^{-}\]
done
clear
View Answer play_arrow
If \[{{V}_{rms}}\]is\[30{{R}^{1/2}}\]at \[{{27}^{\circ }}C\], than the molar mass of the gas in kilogram is:
A)
1
done
clear
B)
2
done
clear
C)
4
done
clear
D)
0.001
done
clear
View Answer play_arrow
Among the following, the molecule with highest dipole moment is:
A)
\[C{{H}_{3}}CI\]
done
clear
B)
\[C{{H}_{2}}C{{I}_{2}}\]
done
clear
C)
\[CHC{{I}_{3}}\]
done
clear
D)
\[CC{{I}_{4}}\]
done
clear
View Answer play_arrow
Which of the following is not a state function?
A)
\[\Delta G\]
done
clear
B)
\[\Delta E\]
done
clear
C)
\[W\]
done
clear
D)
\[\Delta H\]
done
clear
View Answer play_arrow
Atomic orbitals of carbon in \[C{{O}_{2}}\]are:
A)
\[sphybridised\]
done
clear
B)
\[s{{p}^{3}}dhybridised\]
done
clear
C)
\[s{{p}^{2}}hybridised\]
done
clear
D)
\[s{{p}^{3}}-ybridised\]
done
clear
View Answer play_arrow
Among \[{{H}_{2}}O,\text{ }{{H}_{2}}S,\text{ }{{H}_{2}}Se\]and \[{{H}_{2}}Te,\]the one with highest boiling point is:
A)
\[{{H}_{2}}O\]because of hydrogen bonding
done
clear
B)
\[{{H}_{2}}Te\]because of higher molecular weight
done
clear
C)
\[{{H}_{2}}S\]because of hydrogen boning
done
clear
D)
\[{{H}_{2}}Se\]because of lower molecular weight
done
clear
View Answer play_arrow
The following equilibrium constants are given:
\[{{N}_{2}}+3{{H}_{3}}\overset{{}}{leftrightarrows}2{{N}_{3}};{{K}_{1}}\] \[{{N}_{2}}+{{O}_{2}}\overset{{}}{leftrightarrows}2NO;{{K}_{2}}\] \[{{H}_{2}}+\frac{1}{2}{{O}_{2}}\overset{{}}{leftrightarrows}{{H}_{2}}O;{{K}_{3}}\] The equilibrium constant for the oxidation of \[N{{H}_{3}}\]by oxygen to give NO is:
A)
\[{{K}_{1}}\times {{K}_{2}}/{{K}_{3}}\]
done
clear
B)
\[{{K}_{2}}\times \,K_{3}^{3}/{{K}_{1}}\]
done
clear
C)
\[{{K}_{2}}\times \,K_{3}^{2}/{{K}_{1}}\]
done
clear
D)
\[K_{2}^{2}\,{{K}_{3}}/{{K}_{1}}\]
done
clear
View Answer play_arrow
In which of the following acid - base titration, pH is greater than 8 at the equivalence point?
A)
Acetic acid versus ammonia
done
clear
B)
Acetic acid versus sodium hydroxide
done
clear
C)
Hydrochloric acid versus ammonia
done
clear
D)
Hydrochloric acid versus sodium hydroxide
done
clear
View Answer play_arrow
The values of x and y in the following redox reaction, \[xC{{l}_{2}}+6O{{H}^{-}}\to CIO_{3}^{-}+yC{{I}^{-}}+3{{H}_{2}}O\]are:
A)
x = 2, y = 4
done
clear
B)
x = 5, y = 3
done
clear
C)
x = 3, y = 5
done
clear
D)
x = 4, y = 2
done
clear
View Answer play_arrow
The correct increasing order of the acidity of\[C{{O}_{2}}\],\[{{H}_{2}}O\]and \[{{H}_{2}}{{O}_{2}}\]is:
A)
\[C{{O}_{2}}<{{H}_{2}}{{O}_{2}}<{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}O<{{H}_{2}}{{O}_{2}}<C{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}O<{{H}_{2}}{{O}_{2}}>C{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}{{O}_{2}}>C{{O}_{2}}>{{H}_{2}}O\]
done
clear
View Answer play_arrow
On dissolving moderate amount of sodium metal in liquid \[N{{H}_{3}}\] at low temperature, which of the following does not occur?
A)
Blue colored solution is formed
done
clear
B)
\[N{{a}^{+}}\]ions are formed in the solution
done
clear
C)
Liquid ammonia becomes good conductor of electricity
done
clear
D)
Liquid ammonia remains diamagnetic
done
clear
View Answer play_arrow
Borax is used as a cleansing agent because on dissolving in water, it gives:
A)
Alkaline solution
done
clear
B)
Acidic solution
done
clear
C)
Bleaching solution
done
clear
D)
Amphoteric solution
done
clear
View Answer play_arrow
The IUPAC for the hydrocarbon represented by the swastika design:
A)
Neononane
done
clear
B)
Tetraethyl carbon
done
clear
C)
2-ethylpentane
done
clear
D)
3, 3-diethylpentane
done
clear
View Answer play_arrow
Naphthalene can be easily purified by:
A)
Sublimation
done
clear
B)
Crystallisation
done
clear
C)
Distillation
done
clear
D)
Extraction with a solvent
done
clear
View Answer play_arrow
Consider the following carbocations.
[a] \[CI_{3}^{+}C\] [b] \[C{{I}_{2}}\overset{+}{\mathop{C}}\,H\] [c] \[CICH_{2}^{+}\] [d] \[\overset{+}{\mathop{C}}\,{{H}_{3}}\] The stability sequence follows:
A)
iv < i < ii < iii
done
clear
B)
i < ii < iii < iv
done
clear
C)
iv < i < iii < ii
done
clear
D)
iv < ii < i < iii
done
clear
View Answer play_arrow
1-Butyne reacts with cold alkaline \[KMn{{O}_{4}}\] to produce:
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COOH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COOH+C{{O}_{2}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH+HCOOH\]
done
clear
View Answer play_arrow
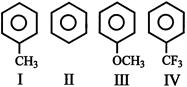
Among the following compounds, the decreasing order of reactivity towards electrophilic substitution is:
A)
Ill > I > II > IV
done
clear
B)
IV > I > II > III
done
clear
C)
I > II > III > IV
done
clear
D)
II > I > III > IV
done
clear
View Answer play_arrow
'White lung cancer' is caused by:
A)
Asbestos
done
clear
B)
Silica
done
clear
C)
Textiles
done
clear
D)
Paper
done
clear
View Answer play_arrow
Which has no rotation of symmetry?
A)
Hexagonal
done
clear
B)
Orthorhombic
done
clear
C)
Cubic
done
clear
D)
Triclinic
done
clear
View Answer play_arrow
The \[C{{a}^{2+}}\] and \[{{F}^{-}}\] ion are located in \[Ca{{F}_{2}}\]crystal, respectively at fee lattice points and in:
A)
Tetrahedral voids
done
clear
B)
Half of tetrahedral voids
done
clear
C)
Octahedral voids
done
clear
D)
Half of octahedral voids
done
clear
View Answer play_arrow
A 5% solution of cane sugar in water has freezing point of 271K and freezing point of pure water is 273.15K. The freezing point of a 50% solution (by mass) of glucose in water is:
A)
271K
done
clear
B)
273.15K
done
clear
C)
269.07K
done
clear
D)
277.23K
done
clear
View Answer play_arrow
Which will form maximum boiling azeotrope?
A)
\[HN{{0}_{3}}+{{H}_{2}}\text{O }Solution\text{ }\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH+{{H}_{2}}O\,solution\]
done
clear
C)
\[{{C}_{6}}{{H}_{6}}+{{C}_{6}}{{H}_{5}}C{{H}_{3}}\,solution\]
done
clear
D)
\[None\]
done
clear
View Answer play_arrow
Time required to deposit one mill mole of aluminium metal by the passage of 9.65 ampere through aqueous solution of aluminium ion is:
A)
30s
done
clear
B)
10s
done
clear
C)
30, 000s
done
clear
D)
10,000s
done
clear
View Answer play_arrow
Decomposition of \[N{{H}_{3}}\]on the surface of tungsten is a reaction of:
A)
0 order
done
clear
B)
1 order
done
clear
C)
2 order
done
clear
D)
Fractional order
done
clear
View Answer play_arrow
Unit of rate constant for zero order reaction is:
A)
\[Mole\,{{L}^{-1}}{{s}^{-1}}\,\]
done
clear
B)
\[Mol{{e}^{-1}}L{{s}^{-1}}\]
done
clear
C)
\[{{s}^{-1}}~\]
done
clear
D)
\[Mol{{e}^{-2}}{{L}^{2}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
Gold number of protective colloids A, B, C and D are 0.50, 0.01, 0.10 and 0.005 respectively. The correct order of their protective powers is:
A)
A<C<B<D
done
clear
B)
B<D<A<C
done
clear
C)
D<A<C<B
done
clear
D)
C<B<D<A
done
clear
View Answer play_arrow
Cassiterite is an ore of:
A)
Pb
done
clear
B)
Zn
done
clear
C)
Sn
done
clear
D)
Mn
done
clear
View Answer play_arrow
Which two of the following salts are used for preparing iodized salt?
1. \[KI{{O}_{3}}\] 2. \[KI\] 3. \[{{I}_{2}}\] 3. \[HI\]
A)
i and ii
done
clear
B)
i and iii
done
clear
C)
ii and iv
done
clear
D)
iii and iv
done
clear
View Answer play_arrow
Which inert gas has abnormal behaviour on liquefication?
A)
Xe
done
clear
B)
He
done
clear
C)
Ar
done
clear
D)
Kr
done
clear
View Answer play_arrow
Among the following pairs of ions, the lower oxidation state in aqueous solution is more stable than the other, in:
A)
\[T{{i}^{+}},T{{i}^{3+}}\]
done
clear
B)
\[C{{u}^{2+}},C{{u}^{+}}\]
done
clear
C)
\[C{{r}^{2+}},C{{r}^{3+}}\]
done
clear
D)
\[{{V}^{2+}},V{{O}^{2+}}\]
done
clear
View Answer play_arrow
The transition metals are mostly:
A)
Diamagnetic
done
clear
B)
Paramagnetic
done
clear
C)
Neither diamagnetic nor paramagnetic
done
clear
D)
Both diamagnetic and paramagnetic
done
clear
View Answer play_arrow
Which of the following shall form an octahedral complex?
A)
\[{{d}^{4}}\,(low\,spin)\]
done
clear
B)
\[{{d}^{8}}\,(high\,spin)\]
done
clear
C)
\[{{d}^{6}}\,(low\,spin)\]
done
clear
D)
\[~All\text{ }of\text{ }these\]
done
clear
View Answer play_arrow
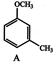
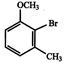
Themajor -productobtainedon monobromination with \[B{{r}_{2}}/FeB{{r}_{3}}\] of the following compound A is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
On mercuration-demercuration produces:
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of these
done
clear
View Answer play_arrow
Aldol condensation will not take place in:
A)
\[HCHO\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
View Answer play_arrow
The most suitable reagent for the conversion of \[RC{{H}_{2}}OH\to RCHO\]is:
A)
\[KMn{{O}_{4}}\]
done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
C)
\[Cr\,{{O}_{3}}\]
done
clear
D)
\[PCC\text{ (}pyridinium\text{ }chlorochromate\text{)}\]
done
clear
View Answer play_arrow
In the following sequence of reactions, what is D?
A)
\[{{1}^{{}^\circ }}\]amine
done
clear
B)
An amide
done
clear
C)
Phenyl isocyanate
done
clear
D)
Chain lengthened hydrocarbon
done
clear
View Answer play_arrow
Which of the following on reduction with lithium aluminium hydride yields a secondary amine?
A)
Methyl cyanide
done
clear
B)
Nitro ethane
done
clear
C)
Methyl isocyanides
done
clear
D)
Acetamide
done
clear
View Answer play_arrow
Lysine is least soluble in water in the pH range:
A)
3 to 4
done
clear
B)
5 to 6
done
clear
C)
6 to 7
done
clear
D)
8 to 9
done
clear
View Answer play_arrow
Natural rubber is a polymer so:
A)
Butadiene
done
clear
B)
Ethyene
done
clear
C)
Styrene
done
clear
D)
Isoprene
done
clear
View Answer play_arrow
The compound
is used as
A)
Antiseptic
done
clear
B)
Antibiotic
done
clear
C)
Analgesic
done
clear
D)
Pesticide
done
clear
View Answer play_arrow
Where maturity does is observed in the sporophyte stage of the plants?
A)
In gemmae
done
clear
B)
In primary structures
done
clear
C)
In sporophylls
done
clear
D)
In eggs
done
clear
View Answer play_arrow
Which structure pushes the developing embryo towards endosperm to get nutrition:
A)
Hypophyis
done
clear
B)
Terminoil ocant
done
clear
C)
Proembryo
done
clear
D)
Suspensor
done
clear
View Answer play_arrow
What is meant by term Darwin fitness:
A)
Ability to survive and reproduce
done
clear
B)
High aggressiveness
done
clear
C)
Healthy appearance
done
clear
D)
Physical strength
done
clear
View Answer play_arrow
A polygenic trait is controlled by 3 genes A, B & In a cross AaBbCc X AaBb Cc, the phenol typic ratio of the offspring was observed as 1 : 6 x : 20 : x : 6 :1. What is the possible value of x?
A)
3
done
clear
B)
9
done
clear
C)
15
done
clear
D)
25
done
clear
View Answer play_arrow
Endemic species are:
A)
Rare species
done
clear
B)
Species localised in a specific region
done
clear
C)
Cosmopolitan in distribution
done
clear
D)
Critically endangered species
done
clear
View Answer play_arrow
The sequence of structural gene is lac operon concept is:
A)
lac Y, lac Z, lac A
done
clear
B)
lac Z, lac Y, lac A
done
clear
C)
lac A, lac Y, lac Z
done
clear
D)
lac A/lac Z, lay Y
done
clear
View Answer play_arrow
What is true for cleavage?
A)
Size of embryo increases
done
clear
B)
Size of cells increases
done
clear
C)
Size of cells decreases
done
clear
D)
Size of embryo decreases
done
clear
View Answer play_arrow
Thermo stable DNA polymerase:
A)
Taq polymerase
done
clear
B)
Vent polymerase
done
clear
C)
Pfu polymerase
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following cell are found in porifera only:
A)
Choanocytes
done
clear
B)
Cnidoblasts
done
clear
C)
Sensory cells
done
clear
D)
Gland cells
done
clear
View Answer play_arrow
The function of ligament is to join:
A)
Muscle to muscle
done
clear
B)
Muscle to bone
done
clear
C)
Bone to bone
done
clear
D)
Bone to skin
done
clear
View Answer play_arrow
The unit membrane concept was given by:
A)
Danielli davson
done
clear
B)
Robertson
done
clear
C)
Singer
done
clear
D)
Nicolson
done
clear
View Answer play_arrow
The first product of \[{{C}_{4}}\] pathway is:
A)
Oxaloacetic acid
done
clear
B)
RuBP
done
clear
C)
PGA
done
clear
D)
PGAL
done
clear
View Answer play_arrow
The alimentary canal of hematodes has:
A)
Only mouth
done
clear
B)
Only anus
done
clear
C)
Both mouth & anus
done
clear
D)
No intestine
done
clear
View Answer play_arrow
A plant cell attain turgidity due to:
A)
Electrolysis
done
clear
B)
Exosmosis
done
clear
C)
Plasmolysis
done
clear
D)
Hydrolysis
done
clear
View Answer play_arrow
The calvin cycle takes place:
A)
Only in light
done
clear
B)
Only in darkness
done
clear
C)
Light dependent
done
clear
D)
Light independent
done
clear
View Answer play_arrow
All hoofed mammals:
A)
Have horns
done
clear
B)
Are cud - chewing
done
clear
C)
Are herbivores
done
clear
D)
Have four chambered stomach
done
clear
View Answer play_arrow
The readily available water for the plants is:
A)
Gravitational water
done
clear
B)
Hygroscopic water
done
clear
C)
Capillary water
done
clear
D)
Water in water table
done
clear
View Answer play_arrow
Which of the following cell do not have a nucleus:
A)
WBCs
done
clear
B)
RBCs
done
clear
C)
Neurons
done
clear
D)
Bone cell
done
clear
View Answer play_arrow
The drinking cell is also termed as:
A)
Exocytosis
done
clear
B)
Pinocytosis
done
clear
C)
Phagocytosis
done
clear
D)
Endocytosis
done
clear
View Answer play_arrow
The first acceptor of \[C{{O}_{2}}\]in the calvin cycle is:
A)
PGA
done
clear
B)
RuBP
done
clear
C)
PGAL
done
clear
D)
RUP
done
clear
View Answer play_arrow
Motile zygote of Plasmodium is:
A)
Liver
done
clear
B)
Human blood
done
clear
C)
Gut of female anopheles
done
clear
D)
Salivary glands of anopheles
done
clear
View Answer play_arrow
In plants the gene for cytoplasmic male sterility are generally located in:
A)
Nuclease - genome
done
clear
B)
Ribosomas genome
done
clear
C)
Mitochondrial genome
done
clear
D)
Cytosol
done
clear
View Answer play_arrow
Who is the father of tissue culture?
A)
Bonner
done
clear
B)
Haberlandt
done
clear
C)
Laibach
done
clear
D)
Gautheret
done
clear
View Answer play_arrow
Production of which enzyme was blocked in flower saver tomoto:
A)
Ligase
done
clear
B)
Invertase
done
clear
C)
Peptidase
done
clear
D)
Poly galactouranase
done
clear
View Answer play_arrow
Who showed that saccharomyces cerevisiae caused fomentation forming products such as beer and butter milk?
A)
Louis Pasteur
done
clear
B)
Schatz
done
clear
C)
Alexander fleming
done
clear
D)
Selman Waksman\
done
clear
View Answer play_arrow
A genetically engineered micro organismused successfully in biosemediation of oil spills is a species as:
A)
Trichoderma
done
clear
B)
Bacillus
done
clear
C)
Xanthomonas
done
clear
D)
Pseudomonas
done
clear
View Answer play_arrow
Tapewarm infection usually comes under:
A)
Droplet infection
done
clear
B)
Vector
done
clear
C)
Measly pork
done
clear
D)
Contaminated water
done
clear
View Answer play_arrow
First life of on earth was:
A)
Cyanobactes
done
clear
B)
Chemo heterotrophs
done
clear
C)
Autographs
done
clear
D)
Photo autogtrophs
done
clear
View Answer play_arrow
Trysin acts on:
A)
Protein in acidic medium
done
clear
B)
Protein in alkaline medium
done
clear
C)
Dipeptide in acidic medium
done
clear
D)
Dipeptide in alkaline medium
done
clear
View Answer play_arrow
HC1 acts on the following to convert it into active form:
A)
Pepsin
done
clear
B)
Trypsin
done
clear
C)
Chymotrypsinogen
done
clear
D)
Pepsinogen
done
clear
View Answer play_arrow
The site for gaseous exchange in lungs is:
A)
Bronchi
done
clear
B)
Bronchioles
done
clear
C)
Alveoli
done
clear
D)
Trachea
done
clear
View Answer play_arrow
The second heart sound was called?
A)
hub
done
clear
B)
lub
done
clear
C)
dub
done
clear
D)
kub
done
clear
View Answer play_arrow
Which of the following hastens the ripening of fruits?
A)
Auxins
done
clear
B)
Gibberlins
done
clear
C)
Cyto kinins
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Tracheal system is found in:
A)
Flatworms
done
clear
B)
Insects
done
clear
C)
Prawns
done
clear
D)
Frogs
done
clear
View Answer play_arrow
The function of vagus nerve innervating the heart is to:
A)
Initiate the heartbeat
done
clear
B)
Reduce the heartbeat
done
clear
C)
Accelerate the heartbeat
done
clear
D)
Keep the heartbeat constant
done
clear
View Answer play_arrow
Full form of JGA:
A)
Juxta golumerular apparatus
done
clear
B)
Juxtagelemorular apparatus
done
clear
C)
Juxtaglomerular apparatus
done
clear
D)
Juxtagallomerula apparatus
done
clear
View Answer play_arrow
A steriosclerosis refer to:
A)
Hypertension
done
clear
B)
Plaque
done
clear
C)
Hardening of arteries
done
clear
D)
PDGF
done
clear
View Answer play_arrow
Protonephridia are excretory organs of:
A)
Coelenterates
done
clear
B)
Platyhelminthes
done
clear
C)
Annelids
done
clear
D)
Crustaceans
done
clear
View Answer play_arrow
What is critical for flowering of short day plant?
A)
Short days
done
clear
B)
Long nights without interruptions
done
clear
C)
Lung night
done
clear
D)
Long days with interruption
done
clear
View Answer play_arrow
The total of all harmful and lethal genes which are present in individuals are called:
A)
Genome
done
clear
B)
Gene port
done
clear
C)
Genetic drift
done
clear
D)
Genetic load
done
clear
View Answer play_arrow
Starfish eggs are:
A)
Microlecithol
done
clear
B)
Isolecithel
done
clear
C)
Alecithal
done
clear
D)
Mesolectithel
done
clear
View Answer play_arrow
Somaclonal variations are the ones:
A)
Induced during sexual embryo eras
done
clear
B)
Caused by UV radiations
done
clear
C)
Produce during tissue culture
done
clear
D)
Caused by mutogens
done
clear
View Answer play_arrow
The rotenone is:
A)
A natural herbicide
done
clear
B)
An insect hormone
done
clear
C)
A bionerbicide
done
clear
D)
A natural insecticide
done
clear
View Answer play_arrow
Contraceptive pills does not:
A)
Engulfs the sperm
done
clear
B)
After the quality of urnical mucus
done
clear
C)
Prevents the entry of sperm
done
clear
D)
Inhibits implantation
done
clear
View Answer play_arrow
Hemolytic anemia disease caused by:
A)
AIDS
done
clear
B)
Leprosy
done
clear
C)
ABO incompatibility
done
clear
D)
Liver cirrhosis
done
clear
View Answer play_arrow
Vir gene expression enhances by:
A)
Cyanidin
done
clear
B)
Acetosyringone
done
clear
C)
Dextran
done
clear
D)
Glutennin
done
clear
View Answer play_arrow
Enzyme used in reverse transcription is:
A)
RNA dependent DNA polymerase
done
clear
B)
DNA dependent RNA polymerase
done
clear
C)
Reverse transcriptase
done
clear
D)
Primase
done
clear
View Answer play_arrow
Vocal cords are present in:
A)
Glottis
done
clear
B)
Epiglottis
done
clear
C)
Larynx
done
clear
D)
Trachea
done
clear
View Answer play_arrow
An electrical device which is place in the chest of the patient.
A)
Sinoatrial node
done
clear
B)
Artificial pacemaker
done
clear
C)
Pacemaker
done
clear
D)
Depolariser
done
clear
View Answer play_arrow
Arteriosclerosis refer to:
A)
Hypertension
done
clear
B)
Plaque
done
clear
C)
Hardening of arteries
done
clear
D)
PDGF
done
clear
View Answer play_arrow
Heart of cockroach is present in __ sinus:
A)
Meta cardial
done
clear
B)
Cetacardial
done
clear
C)
Pericardial
done
clear
D)
Total cardial
done
clear
View Answer play_arrow
__ mL of oxygen is transported per decilitre of blood:
A)
46 mL
done
clear
B)
0.46 Ml
done
clear
C)
4.6mL
done
clear
D)
4.06mL
done
clear
View Answer play_arrow
The heart sound dub is produced by closing of:
A)
Semilunar value
done
clear
B)
Bicuspid value
done
clear
C)
Tricuspid value
done
clear
D)
Aorta
done
clear
View Answer play_arrow
The lactic acid formation leads to net yield of:
A)
\[ZATP\]
done
clear
B)
\[1FAD{{H}_{2}}\]
done
clear
C)
\[2GTP\]
done
clear
D)
\[1NADP{{H}_{2}}\]
done
clear
View Answer play_arrow
Bolting in rosette plants can be induced by:
A)
Auxins
done
clear
B)
Gibberellins
done
clear
C)
Cytokinins
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Pyloric sphincter is present b/w:
A)
Stomach & duodenum
done
clear
B)
Duodenum & jejunum
done
clear
C)
Jejumum & ileum
done
clear
D)
Ileum & caecum
done
clear
View Answer play_arrow
Seedless fruits can be obtained by spraying:
A)
Auxins
done
clear
B)
Bibberllins
done
clear
C)
Cytokinin
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
During krebs cycle, succinate is converted to:
A)
Succinyl COA
done
clear
B)
Fumarate
done
clear
C)
Malate
done
clear
D)
Water
done
clear
View Answer play_arrow
Fats are absorbed by:
A)
Arteries
done
clear
B)
Veins
done
clear
C)
Capillaries
done
clear
D)
Lacteals
done
clear
View Answer play_arrow
Deep black soil is productive due to high proportion by:
A)
Sand and zinc
done
clear
B)
Gravel and calcium
done
clear
C)
Clay and humus
done
clear
D)
Silt and earthworm
done
clear
View Answer play_arrow
Correct order is:
A)
Poleozoic > Mesozoic > Coenozoic
done
clear
B)
Archaeozoic > Coenozoic > Poleozoic
done
clear
C)
Coenozoic > Paleozoic > Azchaeozoic
done
clear
D)
Mesozoic > Archaeozic > Coenozoic
done
clear
View Answer play_arrow
Which is wrong?
A)
Alkaptonuria is due to a recessive gene
done
clear
B)
Gene controls in the metabolism
done
clear
C)
In eukaryates DNA synthesis is faster than prokaryotes
done
clear
D)
Homo gentisic acid is excreted in the wine in alkaptonuria patients
done
clear
View Answer play_arrow
Psychomotor is found in:
A)
Cystic fibrosis
done
clear
B)
Phenylketonuria
done
clear
C)
Down syndrome
done
clear
D)
Truner's syndrome
done
clear
View Answer play_arrow
The first transgenic plant was:
A)
Flax
done
clear
B)
Tobacco
done
clear
C)
Pea
done
clear
D)
Cotton
done
clear
View Answer play_arrow
The gradual physiological adjustment to slowly changing new environmental conditions is known as:
A)
Selection
done
clear
B)
Introduction
done
clear
C)
Acclimatization
done
clear
D)
Quarantine
done
clear
View Answer play_arrow
Pyramid of numbers in a pond ecosystem is:
A)
Irregular
done
clear
B)
Inverted
done
clear
C)
Upright
done
clear
D)
Spindle shaped
done
clear
View Answer play_arrow
The first CFC was synthesised in:
A)
1892
done
clear
B)
1920
done
clear
C)
1980
done
clear
D)
1800
done
clear
View Answer play_arrow
Which of the following bases in plasmids discovered so for:
A)
5000 kilo base
done
clear
B)
500 kilo base
done
clear
C)
50 base
done
clear
D)
5000 base
done
clear
View Answer play_arrow
Biogas can be good substitute for:
A)
Fuel wood
done
clear
B)
Coal
done
clear
C)
Charcol
done
clear
D)
Oiland petrol
done
clear
View Answer play_arrow
Prostate gland produces a secretion of:
A)
Attracting sperms
done
clear
B)
Stimulating sperm activity
done
clear
C)
Inhibiting sperm activity
done
clear
D)
None of the above
done
clear
View Answer play_arrow
What is true of ecosystem?
A)
Primary consumers are least dependent upon producers
done
clear
B)
Primary consumers out - number produces
done
clear
C)
Produces are more than primary consumes
done
clear
D)
Suoudary consumers are the largest and most powerful
done
clear
View Answer play_arrow
Sounds above what level are considered hazardous noise poll".
A)
Above 80 dB
done
clear
B)
Above 150 dB
done
clear
C)
Above 30 dB
done
clear
D)
Above 120dB
done
clear
View Answer play_arrow
For providing the semi conservative mode of replication of DNA E. colt, were first grown in the medium containing.
A)
\[^{14}N{{H}_{3}}\]
done
clear
B)
\[_{^{14}}N{{H}_{4}}CI\]
done
clear
C)
\[^{15}N{{H}_{4}}CI\]
done
clear
D)
\[{{(N{{H}_{4}})}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
All of this obeys Mendel?s laws excepts:
A)
Linkage
done
clear
B)
Independence assortment
done
clear
C)
Dominance
done
clear
D)
Purity of gametes
done
clear
View Answer play_arrow
Ecological niche of an organism represents:
A)
The resources it utilizes
done
clear
B)
Functional role in the ecological system
done
clear
C)
The range of conditions that it can tolerate
done
clear
D)
All of these
done
clear
View Answer play_arrow
Sweet serve to eliminate mainly ______ and
A)
NaCI & Water
done
clear
B)
NaCI & Uric acid
done
clear
C)
NaCI & HCI
done
clear
D)
Water & HCI
done
clear
View Answer play_arrow
Kelp is a kind of:
A)
An aquatic plant
done
clear
B)
A moss
done
clear
C)
An algae
done
clear
D)
A fungus
done
clear
View Answer play_arrow
Whale belongs to group:
A)
Edentata
done
clear
B)
Cetaceans
done
clear
C)
Carnivorous
done
clear
D)
Rodenticide
done
clear
View Answer play_arrow
Sea urchin & sea cucumber belong to phylum:
A)
Hemichordata
done
clear
B)
Chordates
done
clear
C)
Echinodermata
done
clear
D)
Urochordata
done
clear
View Answer play_arrow
Which of the following is under control of ADH:
A)
PCT
done
clear
B)
Descending limb
done
clear
C)
Ascending limb
done
clear
D)
DCT
done
clear
View Answer play_arrow
Agar - agar is produced from:
A)
Blue green algae
done
clear
B)
Red algae
done
clear
C)
Brown algae
done
clear
D)
Green algae
done
clear
View Answer play_arrow
____ act as lubricant in a synovial joints:
A)
Myosin
done
clear
B)
Synovial fluid
done
clear
C)
Actin
done
clear
D)
Myoglobin
done
clear
View Answer play_arrow
Another name of collar bone:
A)
Scapula
done
clear
B)
Clavicle
done
clear
C)
Ischium
done
clear
D)
Ileum
done
clear
View Answer play_arrow
_____ is like hammer shape in the middle ear:
A)
Stapes
done
clear
B)
Callous
done
clear
C)
Malleus
done
clear
D)
Dilate
done
clear
View Answer play_arrow
A fertile agricultural soil appears deep coloured at the surface as deep compared to soil one metre down. The reason for colour of top soil is:
A)
More moisture
done
clear
B)
Rich in organic matter
done
clear
C)
Recent formation
done
clear
D)
Rich in iron, calcium & magnesium
done
clear
View Answer play_arrow
Which of the following statements about ozone is true?
A)
Ozone is a mazor constituent of photo chemical smog
done
clear
B)
Ozone is highly reactive
done
clear
C)
Ozone protects us from the harmful UV radiation of sun
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which one of the following is not observed in biodiversity hotspots?
A)
Sunder bans
done
clear
B)
Western ghats
done
clear
C)
Eastern ghats
done
clear
D)
Gangetic plain
done
clear
View Answer play_arrow
How many times more acidic a rain having pH = 5.6 will be in comparison to a neutral rain having pH 7?
A)
\[72/5.62=1.56\]
done
clear
B)
\[7/5.6=1.25\]
done
clear
C)
\[107/105.6=20~~\]
done
clear
D)
\[5.6/7=0.8\]
done
clear
View Answer play_arrow
Global warming can be controlled by:
A)
Reducing deforestation, cutting down use of fossil fuel
done
clear
B)
Reducing reforestation, increasing the use of fossil fuel
done
clear
C)
Increasing deforestation, slowing down the growth of human population
done
clear
D)
Increasing deforestation, reducing efficiency of energy usage
done
clear
View Answer play_arrow
The two sub-units of ribosome remain united at a critical ion level of:
A)
Copper
done
clear
B)
Molybedanum
done
clear
C)
Boron
done
clear
D)
Magnesium
done
clear
View Answer play_arrow



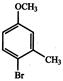

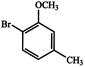

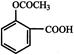 is used as
is used as 