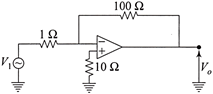
The voltage gain of the following amplifier is
A)
10
done
clear
B)
100
done
clear
C)
1000
done
clear
D)
9.9
done
clear
View Answer play_arrow
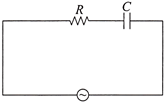
A 50 Hz AC source of 20 V is connected across R and C as shown in the figure. The voltage across R is 12 V. The voltage across C is
A)
8 V
done
clear
B)
16 V
done
clear
C)
10 V
done
clear
D)
not possible to determine unless values of R and C are given
done
clear
View Answer play_arrow
In the figure given, the position-time graph of a particle of mass 0.1 kg is shown. The impulse at \[\operatorname{t}= 2 s\] is
A)
\[0.2\,\,kg\,\,m{{s}^{-}}^{1}\]
done
clear
B)
\[-0.2\,\,kg\,\,m{{s}^{-}}^{1}\]
done
clear
C)
\[0.1\,\,kg\,\,m{{s}^{-}}^{1}\]
done
clear
D)
\[-\,0.4\,\,kg\,\,m{{s}^{-}}^{1}\]
done
clear
View Answer play_arrow
A block of mass 10 kg is moving in x-direction with a constant speed of 10 m/s. It is subjected p to a retarding force \[\operatorname{F}= - 0.1\,x\,\,J/m\] during its travel from\[\operatorname{x}= 20 m to\,\,x =30 m\]. Its final kinetic energy will be
A)
475 J
done
clear
B)
450 J
done
clear
C)
275 J
done
clear
D)
250 J
done
clear
View Answer play_arrow
For a constant hydraulic stress on an object, the fractional change in the object?s volume \[(\Delta V/V)\] and its bulk modulus [B] are related as
A)
\[\frac{\Delta V}{V}\propto B\]
done
clear
B)
\[\frac{\Delta V}{V}\propto \frac{1}{B}\]
done
clear
C)
\[\frac{\Delta V}{V}\propto {{B}^{2}}\]
done
clear
D)
\[\frac{\Delta V}{V}\propto {{B}^{-2}}\]
done
clear
View Answer play_arrow
Which of the following functions represents a simple harmonic oscillation?
A)
\[\sin \,\,\omega t - cos \omega t\]
done
clear
B)
\[si{{n}^{2}}\,\omega t\]
done
clear
C)
\[\sin \,\,\omega \,t +sin 2\,\,\omega t\]
done
clear
D)
\[\sin \,\,\omega \,t -sin 2\,\,\omega t\]
done
clear
View Answer play_arrow
In case of linearly polarised light, the magnitude of the electric field vector
A)
does not change with time
done
clear
B)
varies periodically with time
done
clear
C)
increases and decrease linearly with time
done
clear
D)
is parallel to the direction of propagation
done
clear
View Answer play_arrow
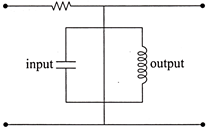
The circuit shown below acts as
A)
tuned filter
done
clear
B)
low pass filter
done
clear
C)
high pass filter
done
clear
D)
rectifier
done
clear
View Answer play_arrow
Which of the following logic gates is a universal gate?
A)
OR
done
clear
B)
NOT
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
What should be the maximum acceptance angle at the air-core interface of an optical fibre if and are the refractive indices of the core and the cladding, respectively?
A)
\[{{\sin }^{-1}}\,\left( {{n}_{2}}/{{n}_{1}} \right)\]
done
clear
B)
\[{{\sin }^{-1}}\,\sqrt{n_{1}^{2}-n_{2}^{2}}\]
done
clear
C)
\[\left[ {{\tan }^{-1}}\frac{{{n}_{2}}}{{{n}_{1}}} \right]\]
done
clear
D)
\[\left[ {{\tan }^{-1}}\frac{{{n}_{1}}}{{{n}_{2}}} \right]\]
done
clear
View Answer play_arrow
A conducting ring of radius 1 m is placed in an uniform magnetic field B of 0.01 T oscillating with frequency 100 Hz with its plane at right angle to B. What will be the induced electric field?
A)
\[\pi \,V/m\]
done
clear
B)
\[2\,V/m\]
done
clear
C)
\[10\,V/m\]
done
clear
D)
\[62\,V/m\]
done
clear
View Answer play_arrow
Consider an n-p-n transistor amplifier in common-emitter configuration. The current gain of the transistor is 100. If the collector current changes by 1 mA, what will be the change in emitter current?
A)
1.1 mA
done
clear
B)
1.01 mA
done
clear
C)
0.01 mA
done
clear
D)
10 mA
done
clear
View Answer play_arrow
A telescope has an objective lens of focal length 200 cm and an eye piece with focal length 2 cm. If this telescope is used to see a 50 m tall building at a distance of 2 km, what is the height of the image of the building formed by the objective lens?
A)
5 cm
done
clear
B)
10 cm
done
clear
C)
1 cm
done
clear
D)
2 cm
done
clear
View Answer play_arrow
Solid targets of different elements are bombarded by highly energetic electron beams. The frequency (f) of the characteristic X-rays emitted from different targets varies with atomic number Z as
A)
\[f\propto \sqrt{Z}\]
done
clear
B)
\[f\propto {{Z}^{2}}\]
done
clear
C)
\[f\propto Z\]
done
clear
D)
\[f\propto {{Z}^{3/2}}\]
done
clear
View Answer play_arrow
The magnetic moment \[\left( \mu \right)\] of a revolving electron around the nucleus varies with principal quantum number n as
A)
\[\mu \propto n\]
done
clear
B)
\[\mu \propto 1/n\]
done
clear
C)
\[\mu \propto {{n}^{2}}\]
done
clear
D)
\[\mu \propto 1/{{n}^{2}}\]
done
clear
View Answer play_arrow
A radioactive material has half-life of 10 days. What fraction of the material would remain after 30 days?
A)
0.5
done
clear
B)
0.25
done
clear
C)
0.125
done
clear
D)
0.33
done
clear
View Answer play_arrow
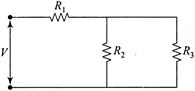
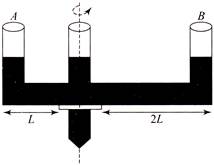
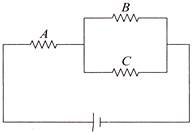
For ensuring dissipation of the same energy in all three resistors \[\left( {{R}_{1}},\,\,{{R}_{2}},\,\,{{R}_{3}} \right)\] connected as show in the figure, their values must be related as
A)
\[{{R}_{1}}=\,\,{{R}_{2}}\,\,=\,\,{{R}_{3}}\]
done
clear
B)
\[{{R}_{2}}={{R}_{3}}\,\,and\,\,{{R}_{1}}=4{{R}_{2}}\]
done
clear
C)
\[{{R}_{2}}={{R}_{3}}\,\,and\,\,{{R}_{1}}=\left( 1/4 \right){{R}_{2}}\]
done
clear
D)
\[{{R}_{1}}={{R}_{2}}+{{R}_{3}}\]
done
clear
View Answer play_arrow
The apparent depth of water in cylindrical water tank of diameter 2 R cm is reducing at the rate of x cm/min when water is being drained out at a constant rate. The amount of water drained in cc per minute is (\[{{n}_{1}}\] = refractive index of air, \[{{n}_{2}}\] = refractive index of water)
A)
\[\frac{x\,\pi \,{{R}^{2}}\,{{n}_{1}}}{{{n}_{2}}}\]
done
clear
B)
\[\frac{x\,\pi \,{{R}^{2}}\,{{n}_{2}}}{{{n}_{1}}}\]
done
clear
C)
\[\frac{2\pi \,\,R\,{{n}_{1}}}{{{n}_{2}}}\]
done
clear
D)
\[\pi {{R}^{2}}x\]
done
clear
View Answer play_arrow
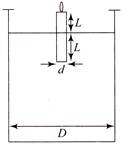
A candle of diameter d is floating on a liquid in a cylindrical container of diameter \[D\left( D>>d \right)\] as shown in the figure. If it is burning at the rate of 2 cm/h then the top of the candle will
A)
remain at the same height
done
clear
B)
fall at the rate of 1 cm/h
done
clear
C)
fall at the rate of 2 cm/h
done
clear
D)
go up at the rate of 1 cm/h
done
clear
View Answer play_arrow
A given shaped glass tube having uniform cross-section is filled with water and is mounted on a rotatable shaft as shown in the figure. If the tube is rotated with a constant angular velocity \[\omega \], then
A)
water levels in both sections A and B go up
done
clear
B)
water level in section A goes up and that in B comes down
done
clear
C)
water level in section A comes down and that in B it goes up
done
clear
D)
water levels remain the same in both sections
done
clear
View Answer play_arrow
When a ball is thrown up vertically with velocity\[{{\nu }_{0}}\]. It reaches a maximum height of h. If one wishes to triple the maximum height, then the ball should be thrown with velocity
A)
\[\sqrt{3}\,{{\nu }_{0}}\]
done
clear
B)
\[3\,{{\nu }_{0}}\]
done
clear
C)
\[9\,{{\nu }_{0}}\]
done
clear
D)
\[3/2\,{{\nu }_{0}}\]
done
clear
View Answer play_arrow
A solid sphere is rolling on a frictionless surface, shown in the figure with a translational velocity v m/s. If it is to climb the inclined surface, then v should be
A)
\[\ge \,\sqrt{\frac{10}{7}\,gh}\]
done
clear
B)
\[\ge \,\sqrt{2\,\,gh}\]
done
clear
C)
2 gh
done
clear
D)
\[\frac{10}{7}\,\,gh\]
done
clear
View Answer play_arrow
A horizontal platform is rotating with uniform angular velocity around the vertical axis passing through its centre. At some instant of time a viscous fluid of mass m is dropped at the centre and is allowed to spread out and finally fall. The angular velocity during this period
A)
decreases continuously
done
clear
B)
decreases initially and increases again\
done
clear
C)
remains unaltered
done
clear
D)
increases continuously
done
clear
View Answer play_arrow
A particle having charge q and mass m is projected with velocity \[\vec{\nu }=2\hat{i}-3\hat{j}\] in uniform electric field \[\vec{E}={{E}_{0}}\,.\,\hat{j}\]. Change in momentum \[\left| \Delta \vec{p} \right|\] during any time interval t is given by
A)
\[\sqrt{13}\,\,M\]
done
clear
B)
\[q{{E}_{0}}t\]
done
clear
C)
\[\frac{q{{E}_{0}}t}{m}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
There is a current of 1.344 amp in a copper wire whose area of cross-section normal to the length of the wire is 1 mm. If the number of free electrons per \[c{{m}^{3}}\] is \[8.4 \times 1{{0}^{22}}\], then the drift velocity would be
A)
1.0 mm/sec
done
clear
B)
1.0 m/sec
done
clear
C)
0.1 mm/sec
done
clear
D)
0.01 mm/sec
done
clear
View Answer play_arrow
A particle of mass m moving with horizontal speed 6 m/sec as shown in the figure. If \[m<M\], then for one dimensional elastic collision, the speed of lighter particle after collision will be
A)
2 m/sec in original direction
done
clear
B)
2 m/sec opposite to the original direction
done
clear
C)
4 m/sec opposite to the original direction
done
clear
D)
4 m/sec in original direction
done
clear
View Answer play_arrow
The focal length of an objective of a telescope is metre diameter 15 cm. Assuming for a normal eye, the diameter of the pupil is 3 mm for its complete use, the focal length of eye piece must be
A)
6 cm
done
clear
B)
6.3 cm
done
clear
C)
20 cm
done
clear
D)
60 cm
done
clear
View Answer play_arrow
If two bulbs of wattage 25 and 100 respectively each rated at 220 volt are connected in series with the supply of 440 volt, then which bulbs will fuse
A)
100 watt bulb
done
clear
B)
25 watt bulb
done
clear
C)
None of them
done
clear
D)
Both of them
done
clear
View Answer play_arrow
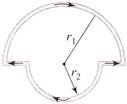
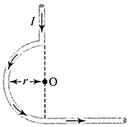
In the figure shown there are two semicircles of radii\[{{r}_{1}}\,\,and\,\,{{r}_{2}}\], in which a current i is flowing. The magnetic induction at the centre O will be
A)
\[\frac{{{\mu }_{0}}i}{r}\left( {{r}_{1}}+{{r}_{2}} \right)\]
done
clear
B)
\[\frac{{{\mu }_{0}}i}{4}\left( {{r}_{1}}-{{r}_{2}} \right)\]
done
clear
C)
\[\frac{{{\mu }_{0}}i}{4}\left( \frac{{{r}_{1}}+{{r}_{2}}}{{{r}_{1}}{{r}_{2}}} \right)\]
done
clear
D)
\[\frac{{{\mu }_{0}}i}{4}\left( \frac{{{r}_{2}}+{{r}_{1}}}{{{r}_{1}}{{r}_{2}}} \right)\]
done
clear
View Answer play_arrow
A straight wire of length \[\left( {{\pi }^{2}} \right)\] metre is carrying a current of 2 A and the magnetic field due to it is measured at a point distant 1 cm from it. If the wire is to be bent into a circle and is to carry the same current as before, the ratio of the magnetic field at its centre to that obtained in the first case would be
A)
\[50 : 1\]
done
clear
B)
\[1 : 50\]
done
clear
C)
\[100 : 1\]
done
clear
D)
\[1 : 100\]
done
clear
View Answer play_arrow
Two like magnetic poles of strength 10 and 40 SI units are separated by a distance 30 cm. The intensity of magnetic field is zero on the line joining them
A)
At a point 10 cm from the stronger pole
done
clear
B)
At a point 20 cm from the stronger pole
done
clear
C)
At the mid-point
done
clear
D)
At infinity
done
clear
View Answer play_arrow
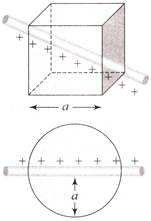
A linear charge having linear charge density \[\lambda \] penetrates a cube diagonally and then it penetrates a sphere diametrically as shown. What will be the ratio of flux coming cut of cube and sphere?
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{2}{\sqrt{3}}\]
done
clear
C)
\[\frac{\sqrt{3}}{2}\]
done
clear
D)
\[\frac{1}{1}\]
done
clear
View Answer play_arrow
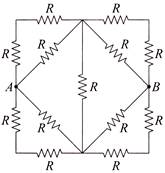
Thirteen resistances each of resistance R ohm are connected in the circuit as shown in the figure. The effective resistance between A and B is
A)
\[2\,\,R\Omega \]
done
clear
B)
\[\frac{4\,R}{3}\,\Omega \]
done
clear
C)
\[\frac{2\,R}{3}\,\Omega \]
done
clear
D)
\[R\,\Omega \]
done
clear
View Answer play_arrow
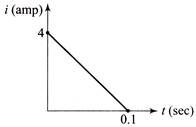
Some magnetic flux is changed from a coil of resistance 10 ohm. As a result an induced current is developed in it, which varies with time as shown in the figure. The magnitude of change in flux through the coil in webers is
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
None of these
done
clear
View Answer play_arrow
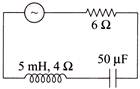
In the circuit shown below, the ac source has voltage \[V=20\text{ }cos\left( \omega t \right)\] volts with \[\omega =2000 rad/sec.\] The amplitude of the current will be nearest to
A)
\[2\,A\]
done
clear
B)
3.3. A
done
clear
C)
\[\frac{2}{\sqrt{5}A}\]
done
clear
D)
\[\sqrt{5}\,A\]
done
clear
View Answer play_arrow
In an LR-circuit, the inductive reactance is equal to the resistance R of the circuit. An e.m.f. \[E={{E}_{0}}\cos \,(\omega t)\] applied to the circuit. The power consumed in the circuit is
A)
\[\frac{E_{0}^{2}}{R}\]
done
clear
B)
\[\frac{E_{0}^{2}}{2\,R}\]
done
clear
C)
\[\frac{E_{0}^{2}}{4\,R}\]
done
clear
D)
\[\frac{E_{0}^{2}}{8\,R}\]
done
clear
View Answer play_arrow
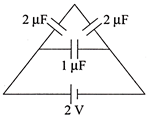
The charge on any one of the \[2\,\mu F\] capacitors and \[1\,\mu F\] capacitor will be given respectively (in \[\left( in\,\,\mu C \right)\] as
A)
1, 2
done
clear
B)
2, 1
done
clear
C)
1, 1
done
clear
D)
2, 2
done
clear
View Answer play_arrow
Light of wavelength \[2475\text{ }\overset{o}{\mathop{A}}\,\] is incident on barium. Photoelectrons emitted describe a circle of radius 100 cm by a magnetic field of flux density \[\frac{1}{\sqrt{17}}\,\,=\,\,\times {{10}^{-}}^{5}\] Tesla. Work function of the barium is \[\left( Given \frac{e}{m} = 1.7 \times 1{{0}^{11}} \right)\]
A)
1.8 eV
done
clear
B)
2.1 eV
done
clear
C)
4.5 eV
done
clear
D)
3.3 eV
done
clear
View Answer play_arrow
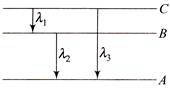
Energy levels A, B, C of a certain atom corresponding to increasing values of energy, i.e., \[{{E}_{A}}<{{E}_{B}}<{{E}_{C}}\]. If \[{{\lambda }_{1}},\,\,{{\lambda }_{2}}\,and\,{{\lambda }_{3}}\] are the wave- lengths of radiations corresponding to the transitions C to B, B to A and C to A respectively, which of the following statements is correct?
A)
\[{{\lambda }_{3}}=\,\,{{\lambda }_{1}}\,+\,{{\lambda }_{2}}\]
done
clear
B)
\[{{\lambda }_{3}}=\,\,\frac{{{\lambda }_{1}}\,{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\]
done
clear
C)
\[{{\lambda }_{1}}+{{\lambda }_{2}}+{{\lambda }_{3}}=0\]
done
clear
D)
\[{{\lambda }_{3}}^{2}+{{\lambda }^{2}}_{1}\,+\lambda _{2}^{2}\]
done
clear
View Answer play_arrow
The percentage errors in the measurement of mass and speed are \[2\,% and 3\,%\] respectively. How much will be the maximum error in the estimation of the kinetic energy obtained by measuring mass and speed?
A)
\[11%\]
done
clear
B)
\[8\,%\]
done
clear
C)
\[5\,%\]
done
clear
D)
\[1%\]
done
clear
View Answer play_arrow
A rubber ball is dropped from a height of 5 m on a planet where the acceleration due to gravity is not known. On bouncing, it rises to 1.8 m. The ball loses its velocity on bouncing by a factor of
A)
16/25
done
clear
B)
2/5
done
clear
C)
3/5
done
clear
D)
9/25
done
clear
View Answer play_arrow
If a 30 V, 90 W bulb is to be worked on a 120 line, a resistance of how many ohms should be connected in series with the bulb?
A)
10 ohm
done
clear
B)
20 ohm
done
clear
C)
30 ohm
done
clear
D)
40 ohm
done
clear
View Answer play_arrow
Three identical resistances A, B and C are connected as shown in the given figure. The heat produced will be maximum
A)
In B
done
clear
B)
In B and C
done
clear
C)
In A
done
clear
D)
Same for A, B and C
done
clear
View Answer play_arrow
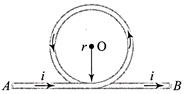
A part of a long wire carrying a current i is bent into a circle of radius r as shown in the figure. The net magnetic field at the centre O of the circular loop is
A)
\[\frac{{{\mu }_{0}}i}{4r}\]
done
clear
B)
\[\frac{{{\mu }_{0}}i}{2r}\]
done
clear
C)
\[\frac{{{\mu }_{0}}i}{2\pi r}(\pi +1)\]
done
clear
D)
\[\frac{{{\mu }_{0}}i}{2\pi r}(\pi -1)\]
done
clear
View Answer play_arrow
In the figure, what is the magnetic field at the point O
A)
\[\frac{{{\mu }_{0}}I}{4\pi r}\]
done
clear
B)
\[\frac{{{\mu }_{0}}I}{4\pi r}+\frac{{{\mu }_{0}}I}{2\pi r}\]
done
clear
C)
\[\frac{{{\mu }_{0}}I}{4r}+\frac{{{\mu }_{0}}I}{4\pi r}\]
done
clear
D)
\[\frac{{{\mu }_{0}}I}{4r}-\frac{{{\mu }_{0}}I}{4\pi r}\]
done
clear
View Answer play_arrow
0.6 g of a solute is dissolved in 0.1 L of a solvent which develops an osmotic pressure of 1.23 atm at\[27{}^\circ C\]. The molecular mass of the substance is
A)
\[\,149.5 \,g\, mole{{\,}^{-}}^{1}\]
done
clear
B)
\[120 \,g\, mole{{\,}^{-}}^{1}\]
done
clear
C)
\[430 \,g\, mole{{\,}^{-}}^{1}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
In fluorite structure \[(Ca{{F}_{2}})\]
A)
\[C{{a}^{++}}\] ions are ccp and \[{{F}^{-}}\] ions are present in all the tetrahedral voids
done
clear
B)
\[C{{a}^{++}}\] ions are ccp and \[{{F}^{-}}\] ions are present in all the octahedral voids
done
clear
C)
\[C{{a}^{++}}\] ions are ccp and \[{{F}^{-}}\] ions are present in all the octahedral voids and half of ions are in the tetrahedral voids
done
clear
D)
None
done
clear
View Answer play_arrow
Boiling point of chloroform was raised by 0.323 K, when 0.5143 g of anthracene was dissolved in 35 g of chloroform. Molecular mass of anthracene is \[({{K}_{b}}\,\,for\,\,CHC{{l}_{3}}\,\,=\,\,3.9\,\,kg\,\,mo{{l}^{-1}})\]
A)
79.42 g/mol
done
clear
B)
132.32 g/mol
done
clear
C)
177.42 g/mol
done
clear
D)
242.32 g/mol
done
clear
View Answer play_arrow
The boiling point of a solution of 0.1050 g of a substance in 15.84 g of ether was found to be \[100{}^\circ C\] higher than that of pure ether. What is the molecular weight of the substance [Molceular elevation constant of ether per\[100\,g=21.6\]]?
A)
144.50
done
clear
B)
143.18
done
clear
C)
140.28
done
clear
D)
146.66
done
clear
View Answer play_arrow
A solution is obtained by dissolving 12 g of urea (mol. wt. 60) in a litre of water. Another solution is obtained by dissolving 68.4 g of cane sugar (mol. wt. 342) in a litre of water at the same temperature. The lowering of vapour pressure in the first solution is
A)
Same as that of 2nd solution
done
clear
B)
Nearly one-fifth of the 2nd solution
done
clear
C)
Double that of 2nd solution
done
clear
D)
Nearly five times that of 2nd solution
done
clear
View Answer play_arrow
Then during electrolysis of a solution of \[AgN{{O}_{3}}\], 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposited in the cathode will be
A)
1.08 g
done
clear
B)
10.8 g
done
clear
C)
21.6 g
done
clear
D)
108 g
done
clear
View Answer play_arrow
Which of the following liberate hydrogen on reaction with dilute\[{{H}_{2}}S{{O}_{4}}\]?
A)
Fe
done
clear
B)
Cu
done
clear
C)
Al
done
clear
D)
Hg
done
clear
View Answer play_arrow
The arrangement of \[C{{l}^{-}}\] ions in \[CsCl\] structure is
A)
hcp
done
clear
B)
fcc
done
clear
C)
bcc
done
clear
D)
Simple cubic
done
clear
View Answer play_arrow
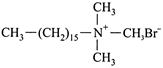
Which one of the following is not a surfactant?
A)
done
clear
B)
\[C{{H}_{3}}-{{(C{{H}_{2}})}_{14}}-C{{H}_{2}}-N{{H}_{2}}\]
done
clear
C)
\[C{{H}_{3}}-{{(C{{H}_{2}})}_{16}}-C{{H}_{2}}OS{{O}^{-}}_{2}N{{a}^{+}}\]
done
clear
D)
\[OHC-{{(C{{H}_{2}})}_{14}}-C{{H}_{2}}-CO{{O}^{-}}\,N{{a}^{+}}\]
done
clear
View Answer play_arrow
The Arrhenius equation for trans isomerization of 2-butene and 2-butene nitrile are given as follows:
[A] For 2-butene \[{{\operatorname{CH}}_{3}}CH=CHC{{H}_{3}}\,K({{S}^{-1}})\] \[K({{S}^{-1}})={{10}^{13.8}}\,\exp {{\,}^{-263.5\,\,}}{{^{KJ\,}}^{mol{{\,}^{-1}}/RT}}\] [B] For 2-butene nitrile \[C{{H}_{3}}CH=CH\cdot CN\] \[K({{S}^{-1}})={{10}^{11}}\,\,\exp {{\,}^{-214.5\,\,}}{{^{KJ\,}}^{mol{{\,}^{-1}}/RT}}\] The temperature at which K = K?
A)
913.87 K
done
clear
B)
533.43 K
done
clear
C)
1000.02 K
done
clear
D)
407.05 K
done
clear
View Answer play_arrow
Size of colloidal particles is
A)
\[0.1\text{ }\mu m\text{ }to\text{ }0.001\text{ }\mu m\]
done
clear
B)
\[10\,\mu \,\,to\,\,20\,\,\mu \]
done
clear
C)
\[0.5 \mu \,m\,\,to\,\,0.1 \mu \,m\]
done
clear
D)
\[25 \mu \,\,to\,\,30 \mu \]
done
clear
View Answer play_arrow
Thermal decomposition of a compound is of first order. If \[50\,%\] of a sample of the compound is decomposed in 120 minutes, how long will it take for \[90\,%\] of the compound to decompose?
A)
399 min
done
clear
B)
410 min
done
clear
C)
250 min
done
clear
D)
120 min
done
clear
View Answer play_arrow
In which of the following process, roasting and self-reduction is required?
A)
\[C{{u}_{2}}S\to C{{u}_{2}}O\]
done
clear
B)
\[\operatorname{CaC}{{O}_{3}}\to CaO\]
done
clear
C)
\[ZnS\,\,\to \,\,Zn\]
done
clear
D)
\[PbS\to Pb\]
done
clear
View Answer play_arrow
For respiration, deep sea divers used a mixture of
A)
Oxygen and argon
done
clear
B)
Oxygen and helium
done
clear
C)
Oxygen and nitrogen
done
clear
D)
Oxygen and hydrogen
done
clear
View Answer play_arrow
The 3d metal ions form coloured compounds because the energy corresponding to the following lies in the visible range of electromagnetic spectrum
A)
Free energy change of complex formation by 3d metal ions
done
clear
B)
d - d transitions of 3d electrons
done
clear
C)
Heat of hydration of 3d metal ions
done
clear
D)
Ionisation energy of 3d metal ions
done
clear
View Answer play_arrow
If the impurity in a metal has a greater affinity for oxygen and is more easily oxidised than the metal, then the purification of metal may be carried out by:
A)
cupellation
done
clear
B)
electrolytic refining
done
clear
C)
zone refining
done
clear
D)
poling
done
clear
View Answer play_arrow
Which of the following is not obtained by direct reaction of constituent elements?
A)
\[Xe{{F}_{2}}\]
done
clear
B)
\[Xe{{F}_{4}}\]
done
clear
C)
\[Xe{{O}_{3}}\]
done
clear
D)
\[Xe{{F}_{6}}\]
done
clear
View Answer play_arrow
Identity the transition element.
A)
\[1{{s}^{2}},2{{s}^{2}},\,\,2{{p}^{6}},\,\,3{{s}^{2}},\,\,\,3{{p}^{6}},\,\,4{{s}^{2}}\]
done
clear
B)
\[1{{s}^{2}},2{{s}^{2}},\,\,2{{p}^{6}},\,\,3{{s}^{2}},\,\,\,3{{p}^{6}},\,\,3{{d}^{2}},\,4{{s}^{2}}\]
done
clear
C)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}},\,\,\,3{{p}^{6}}3{{d}^{10}},\,4{{s}^{2}}4{{p}^{2}}\]
done
clear
D)
\[1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},\,\,3{{s}^{2}},\,\,\,3{{p}^{6}}3{{d}^{10}},\,4{{s}^{2}}4{{p}^{1}}\]
done
clear
View Answer play_arrow
Replacement of Cl of chlorobenzene to give phenol requires drastic conditions but chlorine of 2, 4-dinitrochlorobenzene is readily replaced because
A)
\[N{{O}_{2}}\] makes ring electron rich at ortho and para
done
clear
B)
\[N{{O}_{2}}\] withdraws \[{{e}^{-}}\] from meta position
done
clear
C)
\[N{{O}_{2}}\] denotes \[{{e}^{-}}\] at meta position
done
clear
D)
\[N{{O}_{2}}\] withdraws \[{{e}^{-}}\] from ortho/para positions
done
clear
View Answer play_arrow
The boiling points to isomeric alcohols follow the order
A)
\[primary > secondary >tertiary\]
done
clear
B)
\[tertiary > secondary > primary\]
done
clear
C)
\[secondary > tertiary > primary\]
done
clear
D)
does not follow any order
done
clear
View Answer play_arrow
On hydrolysis \[{{(Me)}_{2}}SiC{{l}_{\text{2}}}\] will produce
A)
\[\left( M{{e}_{2}} \right)Si{{\left( OH \right)}_{2}}\]
done
clear
B)
\[{{\left( Me \right)}_{2}}Si=O\]
done
clear
C)
\[-{{[-O-{{(Me)}_{2}}Si-O-]}_{n}}\]
done
clear
D)
\[{{\operatorname{Me}}_{2}}SiCl\left( OH \right)\]
done
clear
View Answer play_arrow
The alcohol that produces turbidity immediately with \[ZnC{{l}_{2}}+conc.\] HCl at room temperature
A)
1 -hydroxybutane
done
clear
B)
2-hydroxybutane
done
clear
C)
2-hydroxy-2-methylpropane
done
clear
D)
1-hydroxy-2-methylpropane
done
clear
View Answer play_arrow
Among the following one with the highest percentage of chlorine is
A)
Chloral
done
clear
B)
Pyrene
done
clear
C)
PVC
done
clear
D)
Gammexene
done
clear
View Answer play_arrow
Give the decreasing order of the following towards NA reaction.
A)
\[\left( I \right)>\left( II \right)>\left( in \right)>\left( IV \right)\]
done
clear
B)
\[\left( IV \right) > \left( III \right) >\left( II \right)> \left( I \right)\]
done
clear
C)
\[\left( II \right) > \left( III \right) > \left( IV \right) > \left( I \right)\]
done
clear
D)
\[\left( I \right) > \left( IV \right) > \left( III \right) > \left( II \right)\]
done
clear
View Answer play_arrow
The more the -I effect, the more reactive is the aldehyde towards NA reaction\[[(II)>(III)>(IV)>(I)]\]. In order to distinguish between \[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] and, which of the following reagents is useful?
A)
Hinsberg reagent
done
clear
B)
\[p-Naphthol\]
done
clear
C)
Benzene diazonium chloride
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Kwashiorkor is caused by the deficiency of
A)
vitamins
done
clear
B)
hormones
done
clear
C)
amino acids
done
clear
D)
essential amino acids
done
clear
View Answer play_arrow
Types of isomerism shown by \[\left[ Cr{{(N{{H}_{3}})}_{5}}N{{O}_{2}} \right]Cl\] is
A)
Optical
done
clear
B)
Ionisation
done
clear
C)
Geometrical
done
clear
D)
Linkage
done
clear
View Answer play_arrow
Which of the following is not an ambident nucleophile?
A)
\[NO_{2}^{\Theta }\]
done
clear
B)
\[\overset{\Theta }{\mathop{O}}\,H\]
done
clear
C)
\[CS{{N}^{\Theta }}\]
done
clear
D)
\[C{{N}^{\Theta }}\]
done
clear
View Answer play_arrow
Which of the following is the best method for the synthesis of ester (I) \[(M{{e}_{3}}C-COOMe)\]?
A)
\[M{{e}_{3}}C-COCl+MeOH\,\,\xrightarrow{{}}\]
done
clear
B)
\[M{{e}_{3}}C-COOH+MeOH\,\,\xrightarrow{{}}\]
done
clear
C)
\[{{\operatorname{Me}}_{3}}C- COOH + C{{H}_{2}}N{{H}_{2}}\,\xrightarrow{{}}\]
done
clear
D)
\[{{\operatorname{Me}}_{3}}C-COCl+MeONa\,\,\xrightarrow{{}}\]
done
clear
View Answer play_arrow
Which of the following compound is used as a body deodorant?
A)
Aspirin
done
clear
B)
Omeprazole
done
clear
C)
\[Indigosol-O\]
done
clear
D)
\[p-Chlorometaxylenol\]
done
clear
View Answer play_arrow
Arrange the following in the order of decreasing mass
(i) IF atom (ii) IN atom (iii) 1 0 atoms (iv) 1 H atom
A)
\[\left( i \right)>\left( iii \right)>\left( iv \right)>\left( ii \right)\]
done
clear
B)
\[\left( iv \right)<\left( ii \right)<\left( iii \right)<\left( i \right)\]
done
clear
C)
\[\left( i \right) > \left( iii \right) > \left( ii \right) > \left( iv \right)\]
done
clear
D)
\[\left( iii \right)>\left( i \right)>\left( ii \right)>\left( iv \right)\]
done
clear
View Answer play_arrow
Perlon is
A)
Rubber
done
clear
B)
Nylon-6
done
clear
C)
Terylene
done
clear
D)
Oxlon
done
clear
View Answer play_arrow
The number of millimoles contained in 0.160 g of NaOH is
A)
0.04
done
clear
B)
0.4
done
clear
C)
4
done
clear
D)
40
done
clear
View Answer play_arrow
The frequency of one of the lines in Paschen series of hydrogen atom is \[2.340 \times 1{{0}^{11}}\,Hz\]. The quantum number \[{{n}_{2}}\] which produces this transition is
A)
6
done
clear
B)
5
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
What will be the order of 1st ionisation energy?
A)
\[Li>Na>K\]
done
clear
B)
\[K>Li>Na\]
done
clear
C)
\[Na>Li>K\]
done
clear
D)
\[Li>K>Na\]
done
clear
View Answer play_arrow
The maximum number of hydrogen bonds formed by a water molecule in ice is
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
The number of nodal planes in \[a\,{{p}_{x}}\] is
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Zero
done
clear
View Answer play_arrow
Which of the following configurations represents atoms of the elements having the highest second ionization energy?
A)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{4}}\,\,\]
done
clear
B)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\,\,\]
done
clear
C)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{1}}\]
done
clear
D)
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}\]
done
clear
View Answer play_arrow
The correct order of increasing \[\operatorname{C} - O\] bond, length of \[\operatorname{CO}, CO_{3}^{2-},\,\,C{{O}_{2}}\] is
A)
\[CO_{3}^{2-}<C{{O}_{2}}<CO\]
done
clear
B)
\[CO_{2}^{{}}<CO_{3}^{2-}<CO\]
done
clear
C)
\[CO<CO_{3}^{2-}<C{{O}_{2}}\]
done
clear
D)
\[CO<CO_{2}^{{}}<CO_{3}^{2-}\]
done
clear
View Answer play_arrow
Two gaseous equilibria \[S{{O}_{2}}(g)+\frac{1}{2}\,{{O}_{2}}(g)\rightleftharpoons S{{O}_{3}}(g)\] and \[2S{{O}_{3}}(g)\rightleftharpoons 2S{{O}_{2}}(g)+{{O}_{2}}(g)\] have equilibrium constants \[{{K}_{1}}\,and\,\,{{K}_{\text{2}}}\] respectively at 298 K. Which of the following relationships between \[{{K}_{1}}\,and\,\,{{K}_{\text{2}}}\] is correct?
A)
\[{{K}_{1}}\,=\,\,{{K}_{\text{2}}}\]
done
clear
B)
\[{{K}_{2}}\,=\,\,K_{1}^{2}\]
done
clear
C)
\[{{K}_{2}}\,=\,\,\frac{1}{K_{1}^{2}}\]
done
clear
D)
\[{{K}_{2}}\,=\,\,\frac{1}{K_{1}^{{}}}\]
done
clear
View Answer play_arrow
In which of the following equilibria, the value of \[{{K}_{p}}\] is less than \[{{K}_{c}}\].
A)
\[{{H}_{2}}+{{I}_{2}}\rightleftharpoons 2HI\]
done
clear
B)
\[{{N}_{2}}+3{{H}_{2}}\rightleftharpoons 2N{{H}_{3}}\]
done
clear
C)
\[{{N}_{2}}+{{O}_{2}}\rightleftharpoons 2NO\]
done
clear
D)
\[CO+{{H}_{2}}O\rightleftharpoons C{{O}_{2}}+{{H}_{2}}\]
done
clear
View Answer play_arrow
For two gases of molecular weights\[{{M}_{A}}\,\,and\,\,{{M}_{B}}\], at temperature\[{{T}_{A}}\,\,and\,\,{{T}_{B}}\],\[{{T}_{A}}{{M}_{B}}={{T}_{B}}{{M}_{A}}\], then which property has the same magnitude for both the gases.
A)
density
done
clear
B)
pressure
done
clear
C)
KE per mol
done
clear
D)
\[{{V}_{rms}}\]
done
clear
View Answer play_arrow
Entropy change involved in conversion of one mole of liquid water at 373 K to vapour at the same temperature (latent heat of vaporisation of\[\operatorname{water} = 2.257 kJ\,{{g}^{-}}^{1}\]).
A)
\[30.7 J{{K}^{-1}} mol{{e}^{\,-1}}\]
done
clear
B)
\[60.3 J{{K}^{-1}} mol{{e}^{\,-1}}\]
done
clear
C)
\[90.8 J{{K}^{-1}} mol{{e}^{\,-1}}\]
done
clear
D)
\[108.9 J{{K}^{-1}} mol{{e}^{\,-1}}\]
done
clear
View Answer play_arrow
The K.E. of N molecules of \[{{O}_{2}}\] is x joules a\[-123{}^\circ C\]. Another sample of \[{{O}_{2}}\text{ }at\text{ }27{}^\circ C\] has a KE of 2x joules. The latter sample contains
A)
\[N\,\,molecules\,\,of\,\,{{O}_{2}}\]
done
clear
B)
\[2/N\,\,molecules\,of\,\,{{O}_{2}}\]
done
clear
C)
\[N/2\,\,molecules\,of\,\,{{O}_{2}}\]
done
clear
D)
\[N/4\,\,molecules\,of\,\,{{O}_{2}}\]
done
clear
View Answer play_arrow
5 mole of an ideal gas expand reversibly from a volume of \[8 d{{m}^{3}}\,to 80 d{{m}^{3}}\] at a temperature of\[27{}^\circ C\]. Calculate the change in entropy.
A)
\[70.26 J{{K}^{-}}^{1}\]
done
clear
B)
\[82.55 J{{K}^{-}}^{1}\]
done
clear
C)
\[95.73 J{{K}^{-}}^{1}\]
done
clear
D)
\[107.11 J{{K}^{-}}^{1}\]
done
clear
View Answer play_arrow
Number of base pairs in human chromosomes
A)
\[3\,\,\times \,\,1{{0}^{9}}\]
done
clear
B)
\[3\,\,\times \,\,1{{0}^{7}}\]
done
clear
C)
\[6\,\,\times \,\,1{{0}^{8}}\]
done
clear
D)
\[6\,\,\times \,\,1{{0}^{7}}\]
done
clear
View Answer play_arrow
Total amount of \[C{{O}_{2}}\] fixed annually by plants
A)
\[7\times 1{{0}^{23}}ton\]
done
clear
B)
\[7\times 1{{0}^{13}}ton\]
done
clear
C)
\[7\times 1{{0}^{10}}ton\]
done
clear
D)
\[7\times 1{{0}^{11}}ton\]
done
clear
View Answer play_arrow
Most stable pesticides are
A)
Organophosphates
done
clear
B)
Organochlorines
done
clear
C)
Bordeaux mixture
done
clear
D)
Azadirachtin
done
clear
View Answer play_arrow
Best economic method to harvest the solar energy
A)
Solar cell
done
clear
B)
Energy plantation
done
clear
C)
Cultivation of sugar cane then energy obtain by burning it
done
clear
D)
Solar cooker
done
clear
View Answer play_arrow
Main reason of disturbance of biological diversity
A)
Greenhouse effect
done
clear
B)
Hunting
done
clear
C)
Soil erosion
done
clear
D)
Destruction of natural habitats
done
clear
View Answer play_arrow
Best method to preserve the wild relatives of plants
A)
By growing them in natural habitats
done
clear
B)
Gene library
done
clear
C)
By storing seeds
done
clear
D)
Cryopreservation
done
clear
View Answer play_arrow
Practical purpose of taxonomy or classification
A)
Facilitate the identification of unknown species
done
clear
B)
Explain the origin of organisms
done
clear
C)
To know the evolutionary history
done
clear
D)
Identification of medicinal plants
done
clear
View Answer play_arrow
Koch's postulates are not applicable to
A)
Mycobacterium leprae
done
clear
B)
Tuberculosis
done
clear
C)
Pneumonia
done
clear
D)
Cholera
done
clear
View Answer play_arrow
Amount of cellular DNA increases during
A)
Cytokinesis
done
clear
B)
Fertilisation
done
clear
C)
Mutation
done
clear
D)
Respiration
done
clear
View Answer play_arrow
Initiation codon in eukaryotes
A)
UGA
done
clear
B)
CCA
done
clear
C)
AGA
done
clear
D)
AUG
done
clear
View Answer play_arrow
Transition of exarch bundles of root to endarch bundles of stem occurs in
A)
Epicotyl
done
clear
B)
Hypocotyl
done
clear
C)
Apical bud
done
clear
D)
Coleoptile
done
clear
View Answer play_arrow
Which induces the development of corpus luteum?
A)
LH
done
clear
B)
Oestrogen
done
clear
C)
FSH
done
clear
D)
LTH
done
clear
View Answer play_arrow
Plant pathogenic bacteria are mostly
A)
Gram + Non-spore forming
done
clear
B)
Gram - Non-spore forming
done
clear
C)
Gram + spore forming
done
clear
D)
Gram (-) spore forming
done
clear
View Answer play_arrow
First transgenic plant is
A)
Potato
done
clear
B)
Tomato
done
clear
C)
Tobacco
done
clear
D)
Maize
done
clear
View Answer play_arrow
Dolly sheep was obtained by
A)
Cloning the udder cell (somatic cell) fused with unnucleated oocyte
done
clear
B)
Cloning of gametes
done
clear
C)
Tissue culture
done
clear
D)
None
done
clear
View Answer play_arrow
CCK and secretin secreted by
A)
Stomach
done
clear
B)
Ileum
done
clear
C)
Duodenum
done
clear
D)
Colon
done
clear
View Answer play_arrow
Suspensory ligaments are found in
A)
Brain
done
clear
B)
Eyes
done
clear
C)
Liver
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
Life span of worker honey bee is:
A)
30 days
done
clear
B)
15 days
done
clear
C)
90 days
done
clear
D)
10 days
done
clear
View Answer play_arrow
Parathormone deficiency leads to
A)
Decrease of \[C{{a}^{+2}}\] level in blood
done
clear
B)
Increase of \[C{{a}^{+2}}\] level in blood
done
clear
C)
Osteoporosis
done
clear
D)
Hypercalemia
done
clear
View Answer play_arrow
Gene is composed of
A)
Amino acids
done
clear
B)
Polynucleotide
done
clear
C)
Fatty acid
done
clear
D)
Nitrogen bases
done
clear
View Answer play_arrow
Omithophily takes place in
A)
Yellow flower having nectarines
done
clear
B)
Scented flower
done
clear
C)
Flower with charming colour
done
clear
D)
Modified corolla tube
done
clear
View Answer play_arrow
Bhopal gas tragedy is related with
A)
Methane
done
clear
B)
Carbon monooxide
done
clear
C)
Methyl isocyanate (MIC)
done
clear
D)
\[S{{O}^{2}}\]
done
clear
View Answer play_arrow
Concentration of DDT is highest in
A)
Primary consumer
done
clear
B)
Producers
done
clear
C)
Top consumer
done
clear
D)
Decomposers
done
clear
View Answer play_arrow
Percentage energy transferred to higher tropic level in food chain is
A)
\[1\,%\]
done
clear
B)
\[10\,\,%\]
done
clear
C)
\[90\,%\]
done
clear
D)
\[100\,%\]
done
clear
View Answer play_arrow
What change occurs by changing one base in DNA?
A)
Always a change of one amino acid in protein
done
clear
B)
Change in complex sequence of amino acid
done
clear
C)
Always a change in property of protein
done
clear
D)
Does not necessarily change the pheno-type
done
clear
View Answer play_arrow
HIV infects
A)
RBC
done
clear
B)
T-helper cells
done
clear
C)
B-cells
done
clear
D)
Basophils
done
clear
View Answer play_arrow
Which of the following statement is true for bryophyta?
A)
Along with water absorption roots also provide anchoring to plants
done
clear
B)
Sporophyte is dominant
done
clear
C)
Gametophyte is dominant and sporophyte is mostly parasitic
done
clear
D)
Gametophyte is parasitic
done
clear
View Answer play_arrow
Lichens can be used as
A)
Bioindicator for water and air pollution
done
clear
B)
Initial vegetation for waste lands
done
clear
C)
Source of wood
done
clear
D)
To check the air pollution
done
clear
View Answer play_arrow
Biotic and abiotic components form
A)
Community
done
clear
B)
Society
done
clear
C)
Population
done
clear
D)
Species
done
clear
View Answer play_arrow
Endosperm in gymnosperm is
A)
Polyploid
done
clear
B)
Diploid
done
clear
C)
Triploid
done
clear
D)
Haploid
done
clear
View Answer play_arrow
The plant having the largest flower is
A)
Total stem parasite
done
clear
B)
Epiphyte
done
clear
C)
Total root parasite
done
clear
D)
Partial stem parasite
done
clear
View Answer play_arrow
Anabaena is associated with Azolla's
A)
Stem
done
clear
B)
Leaves
done
clear
C)
Roots
done
clear
D)
Flowers
done
clear
View Answer play_arrow
The allele for tallness is dominant over that of dwarfness. This is called
A)
Law of independent assortment
done
clear
B)
Law of segregation
done
clear
C)
Law of unit character
done
clear
D)
Law of dominance
done
clear
View Answer play_arrow
Oxytocin mainly helps in
A)
Milk production
done
clear
B)
Child birth
done
clear
C)
Diuresis
done
clear
D)
Gametogenesis
done
clear
View Answer play_arrow
What ratio is expected in offspring if father is colour blind and mother's father was colour blind?
A)
\[50%\] daughters are colour blind
done
clear
B)
All the sons are colour blind
done
clear
C)
All the daughters are colour blind
done
clear
D)
All the sons are normal
done
clear
View Answer play_arrow
When AABBcc is crossed with AaBbCc, then the ratio of hybrid for all the three genes is
A)
1/8
done
clear
B)
1/4
done
clear
C)
1/16
done
clear
D)
1/32
done
clear
View Answer play_arrow
Which hormone is concerned with the concentration of urine?
A)
Oxytocin
done
clear
B)
Vasopressin
done
clear
C)
Prolactin
done
clear
D)
Cortisol
done
clear
View Answer play_arrow
Ventricular contraction is in command of
A)
SA Node
done
clear
B)
AV Node
done
clear
C)
Purkinje fibers
done
clear
D)
Papillary muscles
done
clear
View Answer play_arrow
Which of the following does not contain metal?
A)
Glycoproteins
done
clear
B)
Ferritin
done
clear
C)
Cytochromes
done
clear
D)
Chromoproteins
done
clear
View Answer play_arrow
Double unit membrane is absent in
A)
Ribosomes
done
clear
B)
Nucleus
done
clear
C)
Plastids
done
clear
D)
ER
done
clear
View Answer play_arrow
Function of nucleases is
A)
Break the polynucleotide chain by breaking each terminal nucleotide
done
clear
B)
Breaks phosphodiester bond
done
clear
C)
Breaks peptide bonds
done
clear
D)
Breaks ester bonds
done
clear
View Answer play_arrow
What is phytotron?
A)
A device to grow the plants in controlled environment
done
clear
B)
Growing plants in greenhouse
done
clear
C)
Radiation chamber to induce the mutations
done
clear
D)
Apparatus to study the effect of light on plants
done
clear
View Answer play_arrow
Species diversity is maximum in
A)
Tropical rain forest
done
clear
B)
Temperate forest
done
clear
C)
Deserts
done
clear
D)
Hill slops
done
clear
View Answer play_arrow
Exponential growth is shown by
A)
Unicellular forms
done
clear
B)
A cell in tissue culture
done
clear
C)
Embryo
done
clear
D)
Multicellular plants
done
clear
View Answer play_arrow
Which of the following is secondary pollutant?
A)
PAN
done
clear
B)
CO
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
According to Forestry Commission Report 1997 the total forest cover of India:
A)
\[11\,%\]
done
clear
B)
\[19.5\,%\]
done
clear
C)
\[17\,%\]
done
clear
D)
\[18.7\,%\]
done
clear
View Answer play_arrow
During injury mast cells secrete
A)
Histamine
done
clear
B)
Heparin
done
clear
C)
Prothrombin
done
clear
D)
Antibodies
done
clear
View Answer play_arrow
Nitrogen fixing bacteria converts
A)
\[{{\operatorname{N}}_{2}}\to N{{H}_{3}}\]
done
clear
B)
\[{{\operatorname{N}}_{4}}^{+}\to \,\,Nitrates\]
done
clear
C)
\[N{{O}_{2}}\,\,\to \,\,N{{O}_{3}}\]
done
clear
D)
\[\operatorname{N}{{O}_{3}}\to {{N}_{2}}\]
done
clear
View Answer play_arrow
Insulin differs from growth hormone in
A)
Increases activity of m-RNA and Ribosomes
done
clear
B)
Increase the permeability of cell membrane
done
clear
C)
Affects metabolism of fats by inducing lipogenesis
done
clear
D)
Increasing protein synthesis
done
clear
View Answer play_arrow
Homologous organs are
A)
Wings of cockroach and wings of bats
done
clear
B)
Wings of insects and wings of birds
done
clear
C)
Air bladder of fishes and lungs of frog
done
clear
D)
Pectoral fins of fishes and forelimbs of horse
done
clear
View Answer play_arrow
Which arrangement is in correct ascending order?
A)
\[Species < genus < order < family\]
done
clear
B)
\[Genus < species < family < order\]
done
clear
C)
\[Order < family < genus < species\]
done
clear
D)
\[Species < genus < family < order\]
done
clear
View Answer play_arrow
In stomach after physical and chemical digestion food is called
A)
Chyme
done
clear
B)
Chyle
done
clear
C)
Amino acid
done
clear
D)
Bolus
done
clear
View Answer play_arrow
Exchange of bicarbonates and chloride ions between RBC and plasma is called
A)
Chloride shift
done
clear
B)
Bohr?s effect
done
clear
C)
Haldane?s effect
done
clear
D)
Intracellular respiration
done
clear
View Answer play_arrow
Which gland decreases in size with increasing age?
A)
Thyroid
done
clear
B)
Adrenal
done
clear
C)
Thymus
done
clear
D)
Pituitary
done
clear
View Answer play_arrow
Which of following occurs in maximum concentration in blood plasma (ECF)?
A)
\[{{K}^{+}}\]
done
clear
B)
\[M{{g}^{+2}}\]
done
clear
C)
\[C{{a}^{+2}}\]
done
clear
D)
\[N{{a}^{+}}\]
done
clear
View Answer play_arrow
Large-scale death of fishes occur in
A)
Saline lake
done
clear
B)
Oligotrophic lake
done
clear
C)
Eutrophic lake
done
clear
D)
Shallow lake
done
clear
View Answer play_arrow
A normal human being requires how much calories per day:
A)
2500 kcal
done
clear
B)
4000 kcal
done
clear
C)
5000 kcal
done
clear
D)
686 kcal
done
clear
View Answer play_arrow
Which of the following yield maximum energy?
A)
By glycolysis in a sprinter
done
clear
B)
Aerobic respiration in germinating seeds
done
clear
C)
Fermentation by yeast
done
clear
D)
Anaerobic respiration
done
clear
View Answer play_arrow
Main reason of water bloom in rivers, lakes, sea, etc., is
A)
Brown algae and green algae
done
clear
B)
Cyanobacteria and dinoflagellates
done
clear
C)
Eicchornia
done
clear
D)
Fishes
done
clear
View Answer play_arrow
Insectivorous plants grow in the soil which is deficient in
A)
Mg
done
clear
B)
Ca
done
clear
C)
P
done
clear
D)
N
done
clear
View Answer play_arrow
Which pair is of insectivorous plants?
A)
Drosera and Vallisneria
done
clear
B)
Utricularia and Hydrilla
done
clear
C)
Allobandra and Utricularia
done
clear
D)
Rafflesia and Dionaea
done
clear
View Answer play_arrow
What shall be the water potential of a root hair cell absorbing water from the soil?
A)
Zero
done
clear
B)
Less than zero
done
clear
C)
More than zero
done
clear
D)
Infinite
done
clear
View Answer play_arrow
Deficiency of oxygen affects most the
A)
Brain
done
clear
B)
Skin
done
clear
C)
Kidney
done
clear
D)
Intestine
done
clear
View Answer play_arrow
Maximum DDT in birds feeding on
A)
Fishes
done
clear
B)
Meat
done
clear
C)
Insects
done
clear
D)
Seeds
done
clear
View Answer play_arrow
Fully digested food reaches to liver by
A)
Hepatic portal vein
done
clear
B)
Hepatic artery
done
clear
C)
Hepatic vein
done
clear
D)
All the above
done
clear
View Answer play_arrow
One of fraternal twin is haemophilic while baby's brother is normal, then which statement is true
A)
Baby is male
done
clear
B)
Baby is female
done
clear
C)
Mother is heterozygous
done
clear
D)
Mother is homozygous
done
clear
View Answer play_arrow
Which one is associated with occupational hazard?
A)
Fluorosis
done
clear
B)
Pneumoconiosis
done
clear
C)
Silicosis
done
clear
D)
Asthma
done
clear
View Answer play_arrow
Azolla is used in the cultivation of
A)
Maize
done
clear
B)
Sorghum
done
clear
C)
Wheat
done
clear
D)
Rice
done
clear
View Answer play_arrow
Which one produce gas by decomposing the gobar (Dung) in gobar gas?
A)
Fungus
done
clear
B)
Virus
done
clear
C)
Methanogenic bacteria
done
clear
D)
Algae
done
clear
View Answer play_arrow
Pantothenic acid and biotin are associated with
A)
Vitamin D
done
clear
B)
Vitamin B complex
done
clear
C)
Vitamin K
done
clear
D)
Vitamin E
done
clear
View Answer play_arrow
Which one is wrong pair?
A)
Scurvy - Vitamin C
done
clear
B)
Rickets - Vitamin D
done
clear
C)
Night blindness (Xerophthalmia) - Vitamin A
done
clear
D)
Beriberi - Vitamin K
done
clear
View Answer play_arrow
Maximum photosynthesis takes place by
A)
Phytoplankton
done
clear
B)
Zooplankton
done
clear
C)
Marsh plants
done
clear
D)
Woody plants
done
clear
View Answer play_arrow
Reptiles like mammals are originated in
A)
Jurassic
done
clear
B)
Triassic
done
clear
C)
Cretaceous
done
clear
D)
Permian
done
clear
View Answer play_arrow
Dental formula of adolescent human being before seventeen year is
A)
21222122
done
clear
B)
21232123
done
clear
C)
21022102
done
clear
D)
10232023
done
clear
View Answer play_arrow
Molecular weight of DNA in yeast is
A)
\[2.56 \times 1{{0}^{9}}\]
done
clear
B)
\[0.5 \times 1{{0}^{9}}\]
done
clear
C)
\[7 \times 1{{0}^{7}}\]
done
clear
D)
\[6 \times 1{{0}^{6}}\]
done
clear
View Answer play_arrow
Minute quantity of hormones and steroid is detected by
A)
Electrophoresis
done
clear
B)
Radio immunoassay
done
clear
C)
Electro encephalogram
done
clear
D)
Fractional analysis
done
clear
View Answer play_arrow
Hybridoma is
A)
Collection of DNA from DNA
done
clear
B)
Collection of RNA from DNA
done
clear
C)
A fusion of tumour sex cell with non-tumour sex cell
done
clear
D)
A fusion of tumour somatic cell with non tumour somatic cell
done
clear
View Answer play_arrow
Which substance can be used as male contraceptive in future?
A)
FSH
done
clear
B)
LH
done
clear
C)
Testosterone
done
clear
D)
Progesterone
done
clear
View Answer play_arrow
Genetic material of prokaryotic cell is
A)
Non-histonic double stranded DNA
done
clear
B)
Histonic double stranded DNA
done
clear
C)
Histone and DNA both are absent
done
clear
D)
Histone without DNA
done
clear
View Answer play_arrow
Ligament consists of
A)
\[Yellow fibres + Elastic fibres\]
done
clear
B)
\[Yellow fibres + Collagen \left( white \right) fibres\]
done
clear
C)
\[Yellow fibres + Muscle fibres\]
done
clear
D)
\[White fibres + Muscle fibres\]
done
clear
View Answer play_arrow
Tendon consists of
A)
Non-elastic connective tissue
done
clear
B)
White elastic tissue
done
clear
C)
Collagen (white) fibres + Muscle fibres
done
clear
D)
Only collagen fibres
done
clear
View Answer play_arrow
Industrial melanism is example of
A)
Natural selection
done
clear
B)
Mutation
done
clear
C)
Racial difference
done
clear
D)
Predation
done
clear
View Answer play_arrow
Casparian bands are found in
A)
Endodermis
done
clear
B)
Pericycle
done
clear
C)
Periderm
done
clear
D)
Cortex
done
clear
View Answer play_arrow
Funaria's male gametes are
A)
Polyflagellate
done
clear
B)
Monoflagellate
done
clear
C)
Biflagellate
done
clear
D)
Tetraflagellate
done
clear
View Answer play_arrow
E. coli are used in production of
A)
Rifampicin
done
clear
B)
LH
done
clear
C)
Ecdysone
done
clear
D)
Interferon
done
clear
View Answer play_arrow
Which one is obtained by S. Miller in his experiments on origin of life before 1953?
A)
Simple sugars
done
clear
B)
Amino acids
done
clear
C)
Nucleotide
done
clear
D)
Peptides
done
clear
View Answer play_arrow
Which protein is found in maximum amount?
A)
Catalase
done
clear
B)
Zinc carbonic anhydrase
done
clear
C)
Transferase
done
clear
D)
RUBISCO
done
clear
View Answer play_arrow
After ovulation follicles are converted into
A)
Corpus luteum
done
clear
B)
Corpus albicans
done
clear
C)
Corpus cavemosa
done
clear
D)
Corpus callosum
done
clear
View Answer play_arrow
Minor change in gene's structure is called
A)
Reversible mutation
done
clear
B)
Point mutation
done
clear
C)
Forward mutation
done
clear
D)
Backward mutation
done
clear
View Answer play_arrow
Greenhouse effect is
A)
Gardening outside the house
done
clear
B)
Global cooling
done
clear
C)
Global warming
done
clear
D)
Green colour house
done
clear
View Answer play_arrow