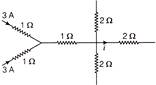
Figure shows current in a part of electrical circuit, then-value of current is
A)
6 A
done
clear
B)
3 A
done
clear
C)
2 A
done
clear
D)
Insufficient information
done
clear
View Answer play_arrow
A particle is revolving in a circular path completes first half of circumference in 4s and next half in 2s, then the average angular velocity of particle is
A)
\[\frac{2\pi }{3}rad/s\]
done
clear
B)
\[\frac{\pi }{3}rad/s\]
done
clear
C)
\[\pi \,\,rad/s\]
done
clear
D)
\[2\pi \,\,rad/s\]
done
clear
View Answer play_arrow
An electric kettle takes 4A current at 220 V. How much time will it take to boil 1 kg of water from temperature\[20{}^\circ C\]? \[\left( Boiling point of water = 100{}^\circ C \right)\]
A)
8.4 min
done
clear
B)
6.3 min
done
clear
C)
4.2 min
done
clear
D)
12.6 min
done
clear
View Answer play_arrow
A physical quantity x is calculated from relation\[\lambda ={{(ab)}^{2}}\,\sqrt{c}\]. If a, b and c are measured respectively with error 1%, 3% and 2%, then the percentage error in calculation of x is
A)
\[4\,%\] %
done
clear
B)
\[9\,%\]%
done
clear
C)
\[12\,%\] %
done
clear
D)
None of these
done
clear
View Answer play_arrow
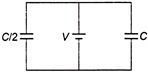
Two capacitors having capacity C and \[\frac{C}{2}\] are connected to a V volt battery, as shown in figure. Then work done charging both the capacitors fully is
A)
\[\frac{3}{4}C{{V}^{2}}\]
done
clear
B)
\[\frac{1}{4}C{{V}^{2}}\]
done
clear
C)
\[2\,C{{V}^{2}}\]
done
clear
D)
\[\frac{1}{2}\,C{{V}^{2}}\]
done
clear
View Answer play_arrow
The electric potential at a point in free space due to charge Q Coulomb is Q X K^V. The electric field at that poi is
A)
\[4\pi {{\varepsilon }_{0}} Q \times 1{{0}^{22}}\,V/m\]
done
clear
B)
\[4\pi {{\varepsilon }_{0}} Q \times 1{{0}^{20}}\,V/m\]
done
clear
C)
\[12\pi {{\varepsilon }_{0}} Q \times 1{{0}^{22}}\,V/m\]
done
clear
D)
\[12\pi {{\varepsilon }_{0}} Q \times 1{{0}^{20}}\,V/m\]
done
clear
View Answer play_arrow
A car accelerates from rest at a constant rate \[\alpha \] for some time after which it deaccelerate at constant rate \[\beta \] and finally comes to rest. If total time elapsed is T, then maximum velocity attained by car is
A)
\[\frac{\alpha +\beta }{\alpha \beta }T\]
done
clear
B)
\[\frac{\alpha \beta }{\alpha +\beta }T\]
done
clear
C)
\[\frac{\alpha }{\alpha +\beta }T\]
done
clear
D)
\[\frac{\beta }{\alpha +\beta }T\]
done
clear
View Answer play_arrow
If a proton having kinetic energy 0.5 MeV approaches a gold nucleus. Then, the distance of closest approach w be \[\left( Z=79 \right)\]
A)
\[2.3\,\,\times \,\,1{{0}^{-10}}m\]
done
clear
B)
\[2.3\,\,\times \,\,1{{0}^{-14}}m\]
done
clear
C)
\[4.8\,\,\times \,\,1{{0}^{-10}}m\]
done
clear
D)
\[4.8\,\,\times \,\,1{{0}^{-14}}m\]
done
clear
View Answer play_arrow
The position of a particle moving along x-axis depend on time in accordance with following equation\[\operatorname{x}=a{{t}^{2}}+b{{t}^{3}}\]. Then, dimensions of a and b are respectively
A)
\[\left[ L{{T}^{-2}} \right];\,\, \left[ L{{T}^{-3}} \right]\]
done
clear
B)
\[\left[ L{{T}^{-3}} \right];\,\, \left[ L{{T}^{-2}} \right]\]
done
clear
C)
\[\left[ L{{T}^{-2}} \right];\,\, \left[ L{{T}^{-2}} \right]\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A ball falling from rest was observed to fall through 78.4 m in 2 s. The time for which it had been falling before this observation is
A)
5 s
done
clear
B)
4 s
done
clear
C)
2 s
done
clear
D)
3 s
done
clear
View Answer play_arrow
If an X-rays tube operates at 18 kV, then the maximum speed of electron striking the anti-cathode is
A)
\[2\times 1{{0}^{7}}m/s\]
done
clear
B)
\[3\times 1{{0}^{8}}m/s\]
done
clear
C)
\[8\times 1{{0}^{7}}m/s\]
done
clear
D)
\[8\times 1{{0}^{10}}m/s\]
done
clear
View Answer play_arrow
Lucknow is at a distance of 200 km from Delhi. If A sets out from Delhi to Lucknow at a speed of 30 km/h and B sets out at the same time from Lucknow to Delhi at a speed of 20 km/h. The time after which they will meet is
A)
2 h
done
clear
B)
3 h
done
clear
C)
4 h
done
clear
D)
None of these
done
clear
View Answer play_arrow
When a charged particle moving with velocity v is subjected to a magnetic field of induction B, the force on it is non-zero. This implies that
A)
angle between them is either zero or \[180{}^\circ \]
done
clear
B)
angle between them can have any value other than zero or \[180{}^\circ \]
done
clear
C)
angle between them is necessary \[90{}^\circ \]
done
clear
D)
angle between them can have any value other than \[90{}^\circ \]
done
clear
View Answer play_arrow
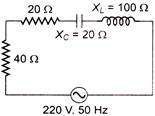
The power factor of the circuit shown in figure is
A)
0.2
done
clear
B)
0.8
done
clear
C)
0.4
done
clear
D)
0.6
done
clear
View Answer play_arrow
What is the ratio of circumference of the first Bohr orbit for the electron in the hydrogen atom to the de-Broglie wavelength of electrons having the same velocity at the electron in the first Bohr orbit of the hydrogen atom?
A)
\[2:1\]
done
clear
B)
\[1:1\]
done
clear
C)
\[1:4\]
done
clear
D)
\[1:2\]
done
clear
View Answer play_arrow
Zener diode is used for
A)
producing oscillation in an oscillator
done
clear
B)
amplification
done
clear
C)
stabilisation
done
clear
D)
rectification
done
clear
View Answer play_arrow
When boron is added as sea impurity to silicon the resulting material is
A)
n-type conductor
done
clear
B)
p -type conductor
done
clear
C)
n-type semiconductor
done
clear
D)
p type semiconductor
done
clear
View Answer play_arrow
The logic operation performed by the circuit given here is
A)
NAND
done
clear
B)
NOR
done
clear
C)
OR
done
clear
D)
NOT
done
clear
View Answer play_arrow
If a force \[\operatorname{F}=6{{t}^{2}} \hat{i}+4t\,\hat{j}\] is acting on a particle of mass 3kg, then velocity of particle at \[\operatorname{t}= 3s\] is (at \[\operatorname{t} =0\], particle is at rest)
A)
\[2\hat{i} + 3\hat{j}\]
done
clear
B)
\[4\hat{i} + 6\hat{j}\]
done
clear
C)
\[18\hat{i} + 6\hat{j}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two bodies are thrown with same initial velocity at angles \[\theta \] and \[\left( 90{}^\circ - \theta \right)\] with horizontal. The ratio of maximum height attained by them is
A)
\[{{\sin }^{2}}\theta \]
done
clear
B)
\[{{\cos }^{2}}\theta \]
done
clear
C)
\[{{\tan }^{2}}\theta \]
done
clear
D)
1
done
clear
View Answer play_arrow
The temperature of a wire is doubled. The Young's modulus of elasticity of the given wire
A)
will also double
done
clear
B)
will become four times
done
clear
C)
will remain same
done
clear
D)
will decrease
done
clear
View Answer play_arrow
If an \[\alpha \] particle is moving in a magnetic field of \[\left( 3\hat{i}+2\hat{j} \right)T\] with a velocity of \[5\times {{10}^{5}}\text{ }\hat{i}.m/s\]. The magnetic force acting on the particle will be
A)
\[3.2\,\,\times \,\,{{10}^{-}}^{13}\,N\text{ }in\text{ }z-direction~\]
done
clear
B)
\[3.2\,\,\times \,\,{{10}^{-}}^{12}\,dyne\text{ }in\text{ }z-direction~\]
done
clear
C)
\[3.2\,\,\times \,\,{{10}^{-}}^{13}\,N\text{ }in\text{ negative}\,\,z-direction~\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If \[{{f}_{v}}\,\,and\,\,{{f}_{R}}\] are the focal length of a convex lens for violet and red light respectively and \[{{F}_{v}}\,\,and\,\,{{F}_{R}}\] are the focal length of a concave lens for violet and red light respectively, then we must have
A)
\[{{\operatorname{f}}_{v}} < {{f}_{R}}\,\,and\,\,\,{{F}_{v}} > {{F}_{R}}\]
done
clear
B)
\[{{\operatorname{f}}_{v}} > {{f}_{R}}\,\,and\,\,\,{{F}_{v}} < {{F}_{R}}\]
done
clear
C)
\[{{\operatorname{f}}_{v}} > {{f}_{R}}\,\,and\,\,\,{{F}_{v}} > {{F}_{R}}\]
done
clear
D)
\[{{\operatorname{f}}_{v}} < {{f}_{R}}\,\,and\,\,\,{{F}_{v}} < {{F}_{R}}\]
done
clear
View Answer play_arrow
Two sound waves with wavelength 5 m and 5.5 m respectively are propagating in a gas with velocity 330 m/s. We expect the following number of beats per second.
A)
1
done
clear
B)
0
done
clear
C)
6
done
clear
D)
12
done
clear
View Answer play_arrow
A string is cut into three parts having fundamental frequency \[{{n}_{1}},\,\,{{n}_{2}}\,\,and\,\,{{n}_{3}}\] respectively. Then, original fundamental frequency n related by the expression as
A)
\[n={{n}_{1}}\times \,\,{{n}_{2}}\,\times \,\,{{n}_{3}}\]
done
clear
B)
\[\frac{1}{n}=\frac{1}{{{n}_{1}}}+\frac{1}{{{n}_{2}}}+\frac{1}{{{n}_{3}}}\]
done
clear
C)
\[n={{n}_{1}}+\,\,{{n}_{2}}\,+\,\,{{n}_{3}}\]
done
clear
D)
\[n=\frac{{{n}_{1}}+{{n}_{2}}+{{n}_{3}}}{3}\]
done
clear
View Answer play_arrow
If the magnetic dipole moment of an atom of diamagnetic material, paramagnetic material and ferromagnetic material or denoted by \[{{M}_{d}},\,\,{{M}_{p}},\,\,{{M}_{f}}\] respectively, then
A)
\[{{M}_{d}}=\,0,\,\,{{M}_{p}}\ne \,\,0\]
done
clear
B)
\[{{M}_{p}}=\,0,\,\,{{M}_{f}}\ne \,\,0\]
done
clear
C)
\[{{M}_{d}}\ne \,\,0,\,\,{{M}_{p}}=\,\,0\]
done
clear
D)
\[{{M}_{d}}=\,\,0,\,\,{{M}_{f}}\ne \,\,0\]
done
clear
View Answer play_arrow
For protecting a sensitive equipment from the external magnetic field, it should be
A)
placed inside an iron can
done
clear
B)
surrounded with fine copper sheet
done
clear
C)
wrapped with insulation around it when passing current through it
done
clear
D)
placed inside an aluminium can
done
clear
View Answer play_arrow
In a pure silicon \[({{n}_{i}}=1{{0}^{16}}/{{m}^{3}})\] crystals at 300 K, \[{{10}^{21}}\] atoms of phosphorous are added per cubic metre. The new hole concentration will be
A)
\[{{10}^{5}}per\,{{m}^{3}}\]
done
clear
B)
\[{{10}^{11}}\,per\,{{m}^{3}}\]
done
clear
C)
\[{{10}^{19}}\,per\,{{m}^{3}}\]
done
clear
D)
\[{{10}^{21}}\,per\,{{m}^{3}}\]
done
clear
View Answer play_arrow
The torque of a force \[\operatorname{F} =- 3\hat{i} + \hat{j} + 5\hat{k}\] acting at the point \[\operatorname{r}=7\,\widehat{i}+3\,\widehat{j}+\widehat{k}\] is
A)
\[4\,\widehat{i}+4\,\widehat{j}+6\,\hat{k}\]
done
clear
B)
\[-21\,\,\widehat{i}+4\,\widehat{j}+4\,\hat{k}\]
done
clear
C)
\[-14\,\,\widehat{i}+34\,\widehat{j}-16\,\hat{k}\]
done
clear
D)
\[14\,\,\widehat{i}-38\,\widehat{j}+16\,\hat{k}\]
done
clear
View Answer play_arrow
The mass number of He is 4 and that of sulphur is 32. The radius of sulphur nucleus is larger than that of helium by the factor of
A)
2
done
clear
B)
4
done
clear
C)
\[\sqrt{8}\]
done
clear
D)
8
done
clear
View Answer play_arrow
A charge q is enclosed by a Gaussian spherical surface of radius R. If the radius is doubled then outward electric flux will
A)
be reduced to half
done
clear
B)
increase four times
done
clear
C)
remain same
done
clear
D)
be doubled
done
clear
View Answer play_arrow
The inward and outward electric flux for a closed surface in units of \[N-{{m}^{2}}/C\] are respectively \[8\times 1{{0}^{3}}\,N-{{m}^{2}}/C\] and\[4 \times 1{{0}^{3}}\,N-{{m}^{2}}/C\]. Then, total charge inside the surface is
A)
\[4\,\,\times \,\,1{{0}^{3}}\,\,C\]
done
clear
B)
\[-\,4\,\,\times \,\,1{{0}^{3}}\,\,C\]
done
clear
C)
\[-(\,4\,\,\times \,\,1{{0}^{3}})\,{{\varepsilon }_{0}}C\]
done
clear
D)
\[-\,\frac{4\,\,\times \,\,1{{0}^{3}}}{{{\varepsilon}_{0}}}\,C\];
done
clear
View Answer play_arrow
In Davisson and Germer experiment, the velocity of electrons emitted from the electron gun can be increased by
A)
increasing the filament current
done
clear
B)
decreasing the filament current
done
clear
C)
decreasing the potential difference between the anode and filament
done
clear
D)
increasing the potential different between the anode and filament
done
clear
View Answer play_arrow
If in a common emitter configuration, transistor has \[\beta =50\] and input resistance\[1\,k\,\Omega \]. If peak value of input AC is 0.01, then the peak value of collector current is
A)
\[500\,\mu A\]
done
clear
B)
\[250\,\,\mu A\]
done
clear
C)
\[0.01\,\,\mu A\]
done
clear
D)
\[0.25\,\,\mu A\]
done
clear
View Answer play_arrow
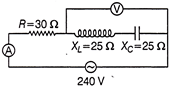
In the given circuit (as shown in figure) neglecting source resistance, then voltmeter and ammeter reading will be
A)
120 V, 3 A
done
clear
B)
120 V, 6 A
done
clear
C)
0 V, 8 A
done
clear
D)
0 V, 3 A
done
clear
View Answer play_arrow
At what height from above the surface of earth, then value of acceleration due to gravity reduces to half of its present value (radius of earth =6400 km)
A)
2660.0 km
done
clear
B)
2689.0 km
done
clear
C)
2559.6 km
done
clear
D)
2649.6 km
done
clear
View Answer play_arrow
The activity of a radioactive substance decreases to one-third of the original activity number \[{{N}_{0}}\] in a period of 9 yr. After a further lapse of 9 yr, its activity will be
A)
\[\frac{3}{4}{{N}_{0}}\]
done
clear
B)
\[\frac{{{N}_{0}}}{9}\]
done
clear
C)
\[{{N}_{0}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A cylinder containing 1 mole of gas was compressed adiabatically until its temperature rises from 300 K to 370 K. Then, heat produced in the gas is \[\left( given r=1.5 \right)\]
A)
276.7 cal
done
clear
B)
376.7 cal
done
clear
C)
298.5 cal
done
clear
D)
398.5 cal
done
clear
View Answer play_arrow
Assuming Bohr model of hydrogen atom. If an electron jumps from first excited state to the ground state of hydrogen atom. What will be the percentage change in the speed of electron?
A)
\[25\]%
done
clear
B)
\[50\]%
done
clear
C)
\[100\,\]%
done
clear
D)
\[200\,\]%
done
clear
View Answer play_arrow
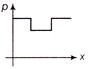
Water is flowing through a frictionless tube having varying cross-sectional are (as shown in fig.) variation of pressure p at points along x-axis is best represented by graph
A)
done
clear
B)
done
clear
C)
done
clear
D)
,
done
clear
View Answer play_arrow
A wire x has a resistance of\[20\,\Omega \]. Another wire y of same material but length twice that of x resistance of \[8\,\Omega \]. If r is the radius of cross-section of x, then radius of cross-section of y is
A)
r
done
clear
B)
\[\frac{r}{\sqrt{2}}\]
done
clear
C)
\[\sqrt{5}\,r\]
done
clear
D)
2r
done
clear
View Answer play_arrow
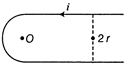
In the given figure, then magnetic field induction at point O will be
A)
\[\frac{{{\mu }_{0}}i}{2\pi r}\]
done
clear
B)
\[\left( \frac{{{\mu }_{0}}i}{4\pi r} \right)\,(\pi +2)\]
done
clear
C)
\[\left( \frac{{{\mu }_{0}}i}{4\pi r} \right)\,(\pi +1)\]
done
clear
D)
\[\left( \frac{{{\mu }_{0}}i}{4\pi r} \right)\,(\pi -2)\]
done
clear
View Answer play_arrow
The frequency of fundamental note in a wire stretched under tension T is f. If the tension is increased to 25 T, then the frequency of the fundamental note will be
A)
5 f
done
clear
B)
f
done
clear
C)
25 f
done
clear
D)
\[\frac{f}{5}\]
done
clear
View Answer play_arrow
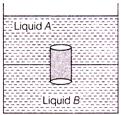
If a glass jar is filled with two non-mixing liquids A and B having densities \[{{D}_{A}}\,\,and\,\,{{D}_{B}}\] respectively. A solid cylinder made of a material of density \[{{D}_{C}}\] is dropped in the jar, it comes to equilibrium position as shown f figure. Which of the following relation is true for densities?
A)
\[\] \[{{\operatorname{D}}_{A}}>{{D}_{C}}>{{D}_{B}}\]
done
clear
B)
\[{{\operatorname{D}}_{A}}={{D}_{B}}>{{D}_{C}}\]
done
clear
C)
\[{{\operatorname{D}}_{A}}<{{D}_{C}}>{{D}_{B}}\]
done
clear
D)
\[{{\operatorname{D}}_{A}}>{{D}_{C}}>{{D}_{B}}\]
done
clear
View Answer play_arrow
If a transformer is used to light a 100 W and 110 V lamp from a 220 V mains. If the main current is 0.5 A, t efficiency of the transformer is approximately.
A)
\[90\,\]%
done
clear
B)
\[10\,%\]%
done
clear
C)
\[30\,%\]%
done
clear
D)
\[50\,%\]%
done
clear
View Answer play_arrow
Determine the time required to \[75\,%\] complete the first order reaction. If the reaction \[20\,%\] complete in 10 min. Choose the correct option.
A)
0.233 min
done
clear
B)
62.18 min
done
clear
C)
112.12 min
done
clear
D)
36.18 min
done
clear
View Answer play_arrow
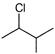
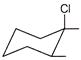
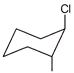
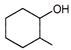
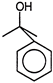
Which of the following will undergo fastest elimination to give desired product?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If the bond forming entities are dissimilar with different electronegativity. The bond formed has partial ionic character as the electron pair is attracted by more electronegativity. Such a bond is called polar covalent bond. Which of the following have lowest degree of covalency?
A)
NaCl
done
clear
B)
\[MgC{{l}_{2}}\]
done
clear
C)
AgCl
done
clear
D)
CsCl
done
clear
View Answer play_arrow
The another name of Nessler?s reagent is ammonia kit is obtained by the reaction of KI with mercuric chloride and NaOH solution. Which of the following is known as Nessler?s reagent?
A)
\[{{K}_{2}}Hg{{l}_{4}}\]
done
clear
B)
\[{{K}_{2}}Hg{{l}_{4}}+KOH\]
done
clear
C)
\[{{K}_{2}}Hg{{l}_{4}}+Hg\]
done
clear
D)
\[{{K}_{2}}Hg{{l}_{4}}+{{H}_{2}}\]
done
clear
View Answer play_arrow
Number of unpaired electrons of Ni in \[{{\left[ Ni{{(CN)}_{4}} \right]}^{2-}}\] is
A)
0
done
clear
B)
1
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following is incorrect regarding detergent?
A)
Detergent is more-better cleansing agent than soap
done
clear
B)
Detergent can be used in hard water also
done
clear
C)
Detergent is soluble in water due to present of long hydrocarbon chain
done
clear
D)
Detergent is insoluble in water due to presence of long hydrocarbon chain.
done
clear
View Answer play_arrow
Aspartame is one of the good artificial sweeteners whose use is limited to cold foods and soft drinks because
A)
It has very low boiling point
done
clear
B)
it gets dissociated at cooking temperature
done
clear
C)
It is sweetener at low temperature only
done
clear
D)
It is insoluble at high temperatures
done
clear
View Answer play_arrow
Bakelite with low degree of polymerisation is used in making glues, in varnishes and lacquers and bakelite with high degree of polymerisation is widely used for making electrical goods, fountain pen barrels, combs, et [c] It is an example of
A)
condensation polymerisation
done
clear
B)
addition polymerisation
done
clear
C)
free radical polymerisation
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following reagent is used to convert glucose to gluconic acid?
A)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
\[B{{r}_{2}}/{{H}_{2}}O\]
done
clear
C)
\[HN{{O}_{3}}\]
done
clear
D)
dil. HCL
done
clear
View Answer play_arrow
Caprolactum may be obtained from
A)
\[fl. hexane + cyclohexane\]
done
clear
B)
\[adipic acid + terphthalic acid\]
done
clear
C)
adipic acid and hexamethylene diammine
done
clear
D)
\[\operatorname{cyclohexanone} + hexamethylene diammine\]
done
clear
View Answer play_arrow
What will be the product B in the following reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
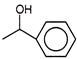
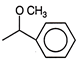
What will be the product formed when acetone is treated with phenyl magnesium bromide followed by ad hydrolysis?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Phenol is heated with phthalic anhydride in presence of\[\operatorname{conc}.{{H}_{2}}S{{O}_{4}}\]. The product gives pink colour with alkali. The product is
A)
phenol phthalein
done
clear
B)
bake lite
done
clear
C)
salicylic acid
done
clear
D)
fluorescein
done
clear
View Answer play_arrow
Product of the following reaction is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
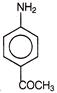
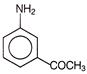
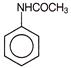
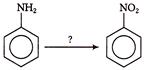
Which reagent will be used to complete below conversion?
A)
\[KMn{{O}_{4}}\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[{{P}_{2}}{{O}_{5}}\]
done
clear
D)
\[{{\operatorname{CF}}_{3}}COOOH\]
done
clear
View Answer play_arrow
What is the relation between following these two isomers?
A)
Enantiomers
done
clear
B)
Diastereomers
done
clear
C)
Mesomers
done
clear
D)
Position isomers
done
clear
View Answer play_arrow
When sodium nitroprusside is added to sodium extract of organic compound it turns pink colouration. This test confirm presence of
A)
sulphur
done
clear
B)
chlorine
done
clear
C)
nitrogen
done
clear
D)
None of these
done
clear
View Answer play_arrow
Conjugated dienes are more stable than non-conjugated dienes (both isolated and cumulated) due to factors such as delocalization of charge through resonance and hybridisation energy. Which of the following compound is most stable?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
What will be the product obtained when A reacts with heavy water,
Choose the correct option regarding product
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The \[As{{F}_{5}}\] molecule is trigonal bipyramidal. The hybrid orbitals used by the As atoms for bonding are
A)
\[{{d}_{{{x}^{2}}-{{y}^{2}}}},\,\,d{{z}^{2}},\,\,s,\,{{p}_{x}},\,\,{{p}_{y}}\]
done
clear
B)
\[{{d}_{xy}},\,\,s,\,{{p}_{x}},\,\,{{p}_{y}},\,\,{{p}_{z}}\]
done
clear
C)
\[s,\,\,{{p}_{x}},\,\,{{p}_{y}},\,\,pz,\,\,d{{z}^{2}}\]
done
clear
D)
\[{{d}_{{{x}^{2}}-{{y}^{2}}}},\,s,\,{{p}_{x}},\,\,{{p}_{y}}\]
done
clear
View Answer play_arrow
Which of the following pair is iso electronics?
A)
\[{{\operatorname{CO}}_{2}} and N{{O}_{2}}\]
done
clear
B)
\[{{\operatorname{BF}}_{3}}\,\,and N{{N}_{3}}\]
done
clear
C)
\[NO_{3}^{-}\,\,and CC{{l}_{4}}\]
done
clear
D)
\[NO_{{}}^{+}\,\,and Sb{{H}_{5}}\]
done
clear
View Answer play_arrow
Choose the correct order of radii of these isoelectronic species.
A)
\[O_{2}^{-}<{{F}^{-}}<N{{a}^{+}}<M{{g}^{2+}}<A{{l}^{3+}}\]
done
clear
B)
\[O_{2}^{-}>{{F}^{-}}>N{{a}^{+}}>M{{g}^{2+}}>A{{l}^{3+}}\]
done
clear
C)
\[O_{2}^{-}>{{F}^{-}}<N{{a}^{+}}<M{{g}^{2+}}<A{{l}^{3+}}\]
done
clear
D)
\[{{F}^{-}}>O_{2}^{-}>N{{a}^{+}}>A{{l}^{3+}}>M{{g}^{2+}}\]
done
clear
View Answer play_arrow
Gold is a chemical element with the symbol Au and atomic number 79. It is used to make ornamental objects a jewellry four thousands of years 18 carat gold contain
A)
\[75\,\]% gold
done
clear
B)
\[20\,\]% gold
done
clear
C)
\[80\,\] % gold
done
clear
D)
\[38.07\,\]% gold
done
clear
View Answer play_arrow
Which of the following order of element is correct regarding 1st ionisation enthalpy?
A)
\[B<C<N<O<F\]
done
clear
B)
\[B<C<N>O<F\]
done
clear
C)
\[B > C > N > O > F\]
done
clear
D)
\[B >C < N > O > F\]
done
clear
View Answer play_arrow
When \[(n-1)d\] orbitals are used in bond formation, the complex formed is called inner orbital complex. Sometimes, in place of \[(n-1)d\] orbitals, outturned orbitals are used for hybridization. The complex thus formed is called out orbital complexes. Hybridisation involved in \[{{\left[ Fe{{(CN)}_{5}}({{H}_{2}}O) \right]}^{3+}}\] is
A)
\[s{{p}^{3}}{{d}^{2}}\]
done
clear
B)
\[{{d}^{2}}s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}{{d}^{3}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[\left[ Co{{(N{{H}_{3}})}_{2}}C{{l}_{2}} \right]\] shows
A)
functional isomerism
done
clear
B)
geometrical isomerism
done
clear
C)
optical isomerism
done
clear
D)
structural isomerism
done
clear
View Answer play_arrow
Orbital angular momentum \[(L)=-h\,\sqrt{l(l+1),}\,\,l=\]orbital angular momentum quantum number. For d-electron, the angular momentum is
A)
\[\sqrt{6}\frac{h}{2\,\pi }\]
done
clear
B)
\[\sqrt{2}\frac{h}{2\,\pi }\]
done
clear
C)
\[\frac{h}{2\,\pi }\]
done
clear
D)
\[2\frac{h}{2\,\pi }\]
done
clear
View Answer play_arrow
Consider the reaction equilibrium, \[2S{{O}_{2}},\,\,(g)\,\,{{O}_{2}},\,\,(\,g)\rightleftharpoons 2\,S{{O}_{3}}(g),\,\,\Delta H{}^\circ =-198\,\,KJ\]on the basis of Le-chatelier's principle, the condition favourable for the forward reaction is
A)
lowering of temperature as well as pressure
done
clear
B)
lowering the temperature and increasing the pressure
done
clear
C)
increasing the temperature and pressure
done
clear
D)
any value of temperature and pressure
done
clear
View Answer play_arrow
Distillation is a method of separating mixture based on differences in volatility of components in a boiling liquid mixture. Which of the following is purified by distillation method?
A)
Zn
done
clear
B)
Al
done
clear
C)
Fe
done
clear
D)
None of these
done
clear
View Answer play_arrow
The compound A on heating gives a colourless gas and a residue that is dissolved in water to obtain Excess \[C{{O}_{2}}\] is bubbled through aqueous solution of B, C is formed which recovered in the solid form. Solid C on gen heating gives back A The compound is
A)
\[\operatorname{CaS}{{O}_{4}}-2{{H}_{2}}O\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[{{K}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
Among the following statement [s] which is/are correct I. Alkali metal hydrides are less reactive than alkaline earth metal hydrides II. Alkali metals have more reducing power than alkaline earth metal III. Alkali metals are more basic than alkaline earth metals IV. Alkali metal have lower ionisation energy than alkaline earth metal. Choose the correct choice among following four choices
A)
I and I
done
clear
B)
only I
done
clear
C)
I, II and III
done
clear
D)
II, III and IV
done
clear
View Answer play_arrow
Which of the following statement is incorrect regarding boric acid?
A)
Boric acid has layered structure
done
clear
B)
It is prepared by \[BC{{l}_{3}}\,and\,{{H}_{2}}O\]
done
clear
C)
It is used as mild anticeptic in washing eye
done
clear
D)
It has square planar structure
done
clear
View Answer play_arrow
The repeating unit present in asbestos is
A)
\[{{\left[ S{{i}_{4}}{{O}_{11}} \right]}^{6\,-}}\]
done
clear
B)
\[{{\left[ Si{{O}_{4}} \right]}^{4\,-}}\]
done
clear
C)
\[{{\left[ S{{i}_{2}}{{O}_{7}} \right]}^{2\,-}}\]
done
clear
D)
\[{{\left[ S{{i}_{3}}{{O}_{9}} \right]}^{3\,-}}\]
done
clear
View Answer play_arrow
Given, \[E_{C{{r}^{3\,+}}/Cr}^{{}^\circ }=-\,0.72\,\,V\] \[E_{F{{e}^{2\,+}}/Fe}^{{}^\circ }=-\,0.42\,\,V\] the potential for the cell \[\operatorname{Cr}|C{{r}^{3+}}\,(0.01\,\,M)\,\,\parallel F{{e}^{2+}}\,\,(0.01\,\,M)Fe\] is
A)
- 0.26 V
done
clear
B)
0.26 V
done
clear
C)
0.339 V
done
clear
D)
- 0.339 V
done
clear
View Answer play_arrow
The solubility product of a salt having general formula\[{{\operatorname{MX}}_{2}} is 4 \times \,\,{{10}^{-}}^{12}\]. Their concentration of \[{{M}^{2+}}\] ion in the aqueous solution of the salt is
A)
\[2\times {{10}^{-\,6}}\,M\]
done
clear
B)
\[1\times {{10}^{-\,4}}\,M\]
done
clear
C)
\[1.6\times {{10}^{-\,4}}\,M\]
done
clear
D)
\[4\times {{10}^{-\,6}}\,M\]
done
clear
View Answer play_arrow
According to Hund's rule of maximum multiplicity, "The pairing of electrons in the orbitals of a particular sub-shell (p, d or f) does not take place until all the orbitals of a sub-shell are singly occupied." The ground state electronic configuration of nitrogen \[(N=1{{s}^{2}}, 2{{s}^{2}},\,\,2{{p}^{3}})\] can be represented as
A)
done
clear
B)
done
clear
C)
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
Hydrocarbon on reaction with steam produces hydrogen gas. Chemical reaction occuring in this process is given by \[{{\operatorname{H}}_{2}}O\,(g)+C{{H}_{4}}(g) \,\rightleftharpoons \,\,CO\,(g)\,+\,3{{H}_{2}}\,(g)\] If \[{{\operatorname{K}}_{C}}=3.0\times 1{{0}^{-}}^{4}\] at 1000 K, then the \[{{K}_{p}}\] would be
A)
202
done
clear
B)
2.02
done
clear
C)
20.2
done
clear
D)
0.6740
done
clear
View Answer play_arrow
In AgBr crystal, the ion size lies in the order\[A{{g}^{+}}<<B{{r}^{-}}\]. The AgBr crystal should have the following characteristics.
A)
Perfect crystal
done
clear
B)
Schottky defect
done
clear
C)
Frenkel defect
done
clear
D)
Both Schottky and Fernkel defect
done
clear
View Answer play_arrow
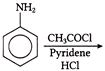
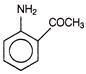
The number of coulombs required to reduce 12.3 g of nitrobenzene to aniline, is
A)
96500 C
done
clear
B)
5790 C
done
clear
C)
95700 C
done
clear
D)
57900 C
done
clear
View Answer play_arrow
For ZnS, the coordination number of cation and anion are respectively
A)
4, 4
done
clear
B)
4, 6
done
clear
C)
6, 6
done
clear
D)
4, 8
done
clear
View Answer play_arrow
The law of definite proportions states, "A sample of a pure compound always consists of the same elements combined in same proportions by mass, whatever be its source. The law of definite proportions is not applicable to nitrogen oxide because
A)
nitrogen atomic weight is not constant
done
clear
B)
nitrogen molecular weight is variable
done
clear
C)
nitrogen equivalent weight is variable
done
clear
D)
oxygen atomic weight is variable
done
clear
View Answer play_arrow
If 10 mol of an ideal gas expands reversibly and isothermally from 10 L to 100 L at 300 K, then entropy change will be
A)
\[-191 -47J {{K}^{-}}^{1}\,mo{{l}^{-}}^{1}\]
done
clear
B)
\[191.24 J{{K}^{-}}^{1}\]
done
clear
C)
\[83.03 J{{K}^{-}}^{1}\]
done
clear
D)
\[83.03 J{{K}^{-}}^{1}\,mo{{l}^{-}}^{1}\]
done
clear
View Answer play_arrow
Which of the following is the property of an isothermal expansion process?
A)
\[\Delta U=0\]
done
clear
B)
\[\operatorname{W}=Q\]
done
clear
C)
\[W=-RT\,\,In\,\,\frac{{{V}_{2}}}{{{V}_{1}}}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following statement is correct regarding catalyst?
A)
Catalyst decreases rate of reaction
done
clear
B)
Catalyst is consumed during course of reaction
done
clear
C)
Catalyst decreases activation energy of reaction
done
clear
D)
Catalyst increases activation energy of reaction
done
clear
View Answer play_arrow
For which of the following order of reaction, the unit of rate constant and rate of reaction is
A)
Zero
done
clear
B)
First
done
clear
C)
Second
done
clear
D)
Third
done
clear
View Answer play_arrow
The insect resistance property of Bacillus thuringiensis, was discovered by whom and when?
A)
Shigetane Ishiwatori; 1901
done
clear
B)
AM Chakarabarty; 1905
done
clear
C)
Zakhayas; 1910
done
clear
D)
Liewellyn; 1920
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Rann of Kutch 1. Tiger B. Corbett park 2. Elephant C. Kaziranga 3. Rhinoceros D. Periyar 4. Wild Ass
Codes
A)
A-4, B-1, C-3, D-2
done
clear
B)
A-2, B-1, C-3, D-4
done
clear
C)
A-1, B-4, C-2, D-3
done
clear
D)
A-3, B-1, C-4, D-2
done
clear
View Answer play_arrow
In DNA model of Watson and Crick, the major grooves represent one of the following sites.
A)
binding of histone proteins
done
clear
B)
binding of acidic proteins
done
clear
C)
binding of RNA molecules
done
clear
D)
binding of glycoproteins
done
clear
View Answer play_arrow
Which of the following can be used to test the DNA?
A)
Iodine
done
clear
B)
Feulgen reaction
done
clear
C)
Polish test
done
clear
D)
Miclion?s reagent
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Prolactin 1. Proteinaceous hormone B. Thryoxine 2. Peptide hormone C. Insulin 3. Amino acid derivative
Codes
A)
A-2, B-1, C-3
done
clear
B)
A-3, B-2, C-1
done
clear
C)
A-3, B-1, C-2
done
clear
D)
A-2, B-3, C-1
done
clear
View Answer play_arrow
There is some permissible latitude in the pairing of the third base in the codon. This is known as one of the following by points.
A)
Wobble hypothesis
done
clear
B)
Lyon's hypothesis
done
clear
C)
Khorana's hypothesis
done
clear
D)
Watson's hypothesis
done
clear
View Answer play_arrow
Which of the following regions of our country are hot spots?
A)
Western ghats and Deccan plateau
done
clear
B)
Western ghats and Indo Burma Himalayan
done
clear
C)
Eastern Himalayas and Gangetic plain
done
clear
D)
Trans Himalayas and Deccan Peninsula
done
clear
View Answer play_arrow
The given figure shows a part of nephron. Identify and choose it from the given options
A)
Thin descending limb
done
clear
B)
Thin ascending limb
done
clear
C)
Thick ascending limb
done
clear
D)
Proximal convulated tubules
done
clear
View Answer play_arrow
Which of the following is correct? Critically endangered - Sus salvinus Endangered - Lion tailed Macaque Rare species - Great Indian Bustard
A)
I and II
done
clear
B)
III and II
done
clear
C)
All of these
done
clear
D)
I and III
done
clear
View Answer play_arrow
Consider the following statements. I. In cyclic photophosphorylation, oxygen is not released and NADPH is also not produced II. Photosynthesis in \[{{C}_{4}}\]-plants is relatively less limited by atmospheric carbon dioxide levels because the primary fixation of carbon dioxide is mediated via PEP carboxylase. III. The portion of the spectrum between 100-200 nm is also referred to as Photosynthetically Active Radiation (PAR).
A)
I and II is true, III is false
done
clear
B)
I and III is true, II is false
done
clear
C)
II is true, I and III is false
done
clear
D)
Ill is true, I and II is false
done
clear
View Answer play_arrow
You got an opportunity to visit a crime scene. The dead body lying there wax in Pugilistic position (boxers position). Your junior asked the reason for this typical position. You explained that this is due to
A)
Chill of death
done
clear
B)
Rigor Mortis
done
clear
C)
Algor Mortis
done
clear
D)
Liror Mortis
done
clear
View Answer play_arrow
Huntington's disease is caused by which of the following reason?
A)
Autosomal gene on chromosome 7
done
clear
B)
Sex-linked gene on X-chromosome
done
clear
C)
Autosomal gene on chromosome 4
done
clear
D)
Sex chromosomal mono so my
done
clear
View Answer play_arrow
A man with blood group AB, marries a woman with 'O' blood group. Which of the following blood group [s] a be found in their progeny
A)
III and IV
done
clear
B)
II and IV
done
clear
C)
I and II
done
clear
D)
All can be found
done
clear
View Answer play_arrow
Which one of the following is the most important for transfection in animal?
A)
Papillora virus
done
clear
B)
Adeno virus
done
clear
C)
Agrobacterium
done
clear
D)
Retro virus
done
clear
View Answer play_arrow
Complete the reaction with correct option. \[\operatorname{Citrulline} + Asparatic acid \to \,\,......\]
A)
Arginine
done
clear
B)
Omithine
done
clear
C)
Urea
done
clear
D)
Arginosucdnic acidc
done
clear
View Answer play_arrow
Inactive enzyme precursors, such as pepsinogen for pepsin are called
A)
activase
done
clear
B)
polyglycoids
done
clear
C)
zymogens
done
clear
D)
cholenzymes
done
clear
View Answer play_arrow
Which one of the statement given below is not correct?
A)
As a result of light reaction, oxygen, NADPH and ATP are formed Oxygen is released into the atmosphere, while NADPH and ATP are utilised for reduction of carbon dioxide to carbohydrate in dark reaction.
done
clear
B)
In \[{{\operatorname{C}}_{3}}-plants\], carbon dioxide, combines with RuBP to form phosphoglyceric acid, on the other hand in\[{{C}_{4}}-plants\], carta dioxide is first picked up by phosphoenol pyruvate to form oxaloacetic acid.
done
clear
C)
Cytochrome plays very important role in ETS of photosynthesis as well as respiration.
done
clear
D)
Energy is released during photorespiration and ammonia is not released from photorespiration.
done
clear
View Answer play_arrow
Micropyle helps in
A)
growth of pollen tube
done
clear
B)
allowing entry of pollen tube
done
clear
C)
coming out of pollen tube from pollen grains
done
clear
D)
germination of pollen grains
done
clear
View Answer play_arrow
Fossils of ancestor of man found in Shivalik hills are
A)
Sinathropus
done
clear
B)
Rama pithecus
done
clear
C)
Pithecanthropus
done
clear
D)
Australopithecus
done
clear
View Answer play_arrow
Assertion [A] Nerves fibres in the CNS can regenerate after injury. Reason [R] Neurilemma around them brings about regeneration.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true, but R is false
done
clear
D)
A is false, but R is true
done
clear
View Answer play_arrow
Sour taste of vinegar is due to which one of the following acid?
A)
lactic acid
done
clear
B)
acetic acid
done
clear
C)
butyric acid
done
clear
D)
fumaricacid
done
clear
View Answer play_arrow
When gram seeds and wheat grains are soaked in water, pea seeds showed more swelling than the wheat. The reason is
A)
Imbibition capacity of proteins is more than that of starch
done
clear
B)
Presence of less hydrophilic colloids in the wheat grains
done
clear
C)
Cell membrane of pea seeds is less permeable and cell wall of wheat grains are more permeable
done
clear
D)
Cell membrane of pea seeds is more permeable and cell wall of wheat grains are less permeable
done
clear
View Answer play_arrow
Which one of the following is refer to the term Atavism in man
A)
appearance of hidden characters
done
clear
B)
evolution of existing characters
done
clear
C)
reappearance of ancestral characters
done
clear
D)
loss of pre-existing characters
done
clear
View Answer play_arrow
Which of the following is a similarity between apes and man?
A)
Presence of tail
done
clear
B)
Tendency to live in pairs as couples, menstruation in females
done
clear
C)
Smaller head and neck
done
clear
D)
Small brain and cranial cavity
done
clear
View Answer play_arrow
Statement [A] Bilirubin is transported in blood by being attached to plasma albumin. Statement [B] Bilirubin is insoluble in water.
A)
A and B both are correct, and B is the correct explanation of [A]
done
clear
B)
A and B both are correct, but B is not the correct explanation of [A]
done
clear
C)
A is true, but B is false.
done
clear
D)
Both are false
done
clear
View Answer play_arrow
Match the following evidences from embryology.
Column I Column II A. Similarity in early development 1. Tail in tadpole B. Progressive metamorphosis 2. Morula C. Retrogressive metamorphosis 3. Branchiostoma D. Temporary embryonic structures 4. Sacculina
Codes
A)
A-2, B-3, C-4, D-1
done
clear
B)
A-1, B-4, C-3, D-2
done
clear
C)
A-4, B-3, C-2, D-1
done
clear
D)
A-2, B-1, C-4, D-3
done
clear
View Answer play_arrow
Consider the following statements.
I. Passive absorption of mineral salts is dependent on diffusion. II. Movement in cells against concentration gradient is called active transport. III. Water potential of impure water is higher than that of pure water. Which of the statements given above are correct.
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Darwin's finches were an excellent evidence of evolution from which of the following the field
A)
Anatomy
done
clear
B)
Biogeography
done
clear
C)
Embryology
done
clear
D)
Paleontology
done
clear
View Answer play_arrow
In the above cycle identify the marking A, B, C with their number of carbons. If (1) ribulose 5-phosphate (2) glyceraldehyde 3-phosphate (3) 1, 3-diphosphoglycerate. Where figures given in the bracket shows the number of carbon in that compound.
A)
A-2(5C) B-1(5C) C-3(5C)
done
clear
B)
A-3(5C) B-1(3C) C-2(3C)
done
clear
C)
A-2(3C) B-1(5C) C-3(3C)
done
clear
D)
A-1(5C) B-3(3C) C-2(3C)
done
clear
View Answer play_arrow
Whenever, we exercise, our \[{{O}_{2}}\] demand of the body increases. To compensate this increase demand, our respiratory rates increase. Suppose, after running 200 meter race, respiratory rate of Ramu become 40 times pa- minute. What would be the minute respiratory volume of Ramu after race?
A)
20 thousand liters
done
clear
B)
10 thousand liters
done
clear
C)
40 thousand liters
done
clear
D)
25 thousand liters
done
clear
View Answer play_arrow
Which of the following forms the most stable product with haemoglobin?
A)
\[{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
CO
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following species is/are critically endangered species in India (as per Red Data Book of IUCN)?
I. Great Indian Bustard (Ardeotis nigriceps) II. Pondicherry Shark (Carcharhinus hemiodon) III. Gharial (Gavialis gangeticus) IV. Himalayan Wolf (Canis himalayensis)
A)
I and II
done
clear
B)
All of these
done
clear
C)
II and IV
done
clear
D)
III and IV
done
clear
View Answer play_arrow
The force of tension cohesion exceeds root pressure on a
A)
foggy morning
done
clear
B)
humid day
done
clear
C)
at night
done
clear
D)
sunny day
done
clear
View Answer play_arrow
Which of the following is related to tetradynamous condition
A)
Androecium
done
clear
B)
Gynoecium
done
clear
C)
Pappus
done
clear
D)
Perianth,
done
clear
View Answer play_arrow
A 36 year old woman have active bilateral synouitis in her hand, wrist, and ankles and nodules on her left elbow. On blood test her RF is found to be 344 (normal 0-29 14/mL.) Most probably she is suffering from
A)
hypertension
done
clear
B)
Acute bronchial asthma
done
clear
C)
Arthritis
done
clear
D)
Leukaemia
done
clear
View Answer play_arrow
Identify the correct chronological sequence periods of Palaeozoic and Mesozoic era
I. Cretaceous II. Permian III. Jurassic IV. Carboniferous V. Triassic
A)
\[I\to II\to III\to IV\to V\]
done
clear
B)
\[I\to IV\to II\to V\to III\]
done
clear
C)
\[IV\to II\to V\to III\to I\]
done
clear
D)
\[IV\to III\to II\to V\to I\]
done
clear
View Answer play_arrow
One of the following is true for Mendible
A)
skull bone
done
clear
B)
lower jaw bone
done
clear
C)
nasal bone
done
clear
D)
neck bone
done
clear
View Answer play_arrow
Cross of \[{{\operatorname{F}}_{1}}-progeny\] back to one of recessive parent is
A)
test cross
done
clear
B)
back cross
done
clear
C)
dihybrid cross
done
clear
D)
trihydrid cross
done
clear
View Answer play_arrow
Choose the incorrect option.
A)
Amorphophallus is largest inflorescence.
done
clear
B)
Lodicules are two scale like strictures that lie at the base of the ovary of a grass flower
done
clear
C)
The spadix contains a large green or coloured bracts called spathe
done
clear
D)
Fruit in members of Solana ceae is pepo.
done
clear
View Answer play_arrow
DNA forms the chemical or molecular basis of heredity. The above statement was given in accordance with the result of experiment conducted by
A)
Astburg
done
clear
B)
Wilkins and Franklin
done
clear
C)
Leuene and Miescher
done
clear
D)
Avery, McCarthy and MacLeod
done
clear
View Answer play_arrow
In which one pair both the plants can be vegetatively propagated by stem cutting?
A)
Croton and Asparagus
done
clear
B)
Bougainvillea and Duranta
done
clear
C)
Bryophyllum and rose
done
clear
D)
Rose and sweet potato
done
clear
View Answer play_arrow
Surface architecture is studied by the help of one of the following method
A)
Polarisation microscopy
done
clear
B)
Phase contract microscopy
done
clear
C)
Scanning electron microscopy
done
clear
D)
Transmission electron microscopy
done
clear
View Answer play_arrow
Match the following epistatic interactions to their ratios.
Column I Column II A. Dominant epistasis 1. \[9:3:4\] B. Recessive epistasis 2. \[15:1\] C. Duplicate recessive genes 3. \[12:3:1\] D. Duplicate dominant genes 4. \[13:3\] 5. \[12:3\]
Codes
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-5, B-2, C-1, D-3
done
clear
C)
A-3, B-1, C-2, D-4
done
clear
D)
A-3, B-1, C-5, D-2
done
clear
View Answer play_arrow
Your grandmother snores so loudly is the right that you cannot have your sleep properly. She also has irregular breathing and elevated blood pressure 152/92 mmHg. She might be suffering from
A)
Pneumonia
done
clear
B)
Bronchial asthma
done
clear
C)
Malignant hypertension
done
clear
D)
Obstructive sleep apnea
done
clear
View Answer play_arrow
Which of the following forms stable colloids?
A)
Carbohydrates
done
clear
B)
Proteins
done
clear
C)
Fats
done
clear
D)
Amino acids
done
clear
View Answer play_arrow
The given pie chart shows the relative contribution of various greenhouse gases to total global warming. Identify the markings correctly if
I. Methane II. \[CF{{C}_{5}}\] III. \[C{{O}_{2}}\] IV. \[{{N}_{2}}O\]
A)
A-IV, B-I, C-III, D-II
done
clear
B)
A-II, B-I, C-IV, D-III
done
clear
C)
A-III, B-II, C-IV, D-I
done
clear
D)
A-I, B-IV, C-III, D-II
done
clear
View Answer play_arrow
Which of the following is/are not characteristic features of family-Cucurbitaceous?
I. Cypseta type of fruit II. Flower are unisexual II. Flower are unisexual III. Ovary superior IV. Placentation parietal
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Anemophily 1. By bats B. Entomophily 2. By ants C. Chiropterophily 3. By insect D. Myrmecophily 4. By wind
Codes
A)
A-3, B-1, C-2, D-4
done
clear
B)
A-4, B-3, C-1, D-2
done
clear
C)
A-3, B-2, C-4, D-1
done
clear
D)
A-4, B-1, C-3, D-2
done
clear
View Answer play_arrow
Which of the following is true for deuteronopia
A)
blindness to all colours
done
clear
B)
red blindness
done
clear
C)
green blindness
done
clear
D)
inability of see in dark
done
clear
View Answer play_arrow
Which of the following is a congenital disease?
A)
Marasmus
done
clear
B)
Colour blindness
done
clear
C)
Pericarditis
done
clear
D)
Rabies
done
clear
View Answer play_arrow
Boron is present in the soil in very small amounts. Boron in green plants assists in
A)
Activation of hormones
done
clear
B)
Activation of enzymes
done
clear
C)
Sugar transport
done
clear
D)
Water transport
done
clear
View Answer play_arrow
Which one of the following disease is caused by endotoxins?
A)
Botulism
done
clear
B)
Cholera
done
clear
C)
Diphtheria
done
clear
D)
Scarlet fever
done
clear
View Answer play_arrow
Without exception, which one of the following is true for all birds?
A)
Omnivorous
done
clear
B)
Have feathers and fly
done
clear
C)
Have calcareous shelled egg
done
clear
D)
Form nest and care them
done
clear
View Answer play_arrow
Genetically engineered maturation of proinsulin into insulin takes place after one of the following step.
A)
After joining of C-peptide
done
clear
B)
removal of disulphide bridge
done
clear
C)
after removal of C-peptide
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which one of the following is caused by Lyssa virus?
A)
Mumps
done
clear
B)
Measles
done
clear
C)
Trachoma
done
clear
D)
Rabies
done
clear
View Answer play_arrow
Taylor experiment on Viciafaba (broad bean) root tips using audio radiography demonstrates that
A)
DNA replication is continuous
done
clear
B)
DNA replicates in discontinuous manner
done
clear
C)
Chromosomes show semiconservative replication
done
clear
D)
DNA replicates in conservative manner
done
clear
View Answer play_arrow
A person is gaming weight day-by-day and he always has the feeling of letharginess. One of the most probable cause is
A)
myopathy
done
clear
B)
neuropathy
done
clear
C)
thyroid related problem
done
clear
D)
Polyploidy
done
clear
View Answer play_arrow
Which among the following is natural auxin?
A)
IPA
done
clear
B)
IBA
done
clear
C)
IAA
done
clear
D)
NAA
done
clear
View Answer play_arrow
In sewage/effluent treatment plant, biological treatment occurs in
A)
primary treatment
done
clear
B)
secondary treatment
done
clear
C)
tertiary treatment
done
clear
D)
None of these
done
clear
View Answer play_arrow
In which of the following organ of human body Muller?s fibres are present?
A)
Heart
done
clear
B)
Kidney
done
clear
C)
Pancreas
done
clear
D)
Retina
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Minamata disease 1. Cadmium in water B. Itai-Itai disease 2. Excess of nitrate in drinking water C. Knock-knees disease 3. Mercury in water D. Methaemoglobinemia 4. Excess fluoride
Codes
A)
A-1, B-4, C-2, D-3
done
clear
B)
A-3, B-1, C-4, D-2
done
clear
C)
A-2, B-3, C-4, D-1
done
clear
D)
A-3, B-4, C-1, D-2
done
clear
View Answer play_arrow
If a person is feeding only on meat, egg and milk, then he will suffer from
A)
night blindness
done
clear
B)
scurvy
done
clear
C)
rickets
done
clear
D)
beri-beri
done
clear
View Answer play_arrow
Which of the following is not a control of air pollution?
A)
Combustion technique
done
clear
B)
Scrubbers
done
clear
C)
Effluent treatment plant
done
clear
D)
Gravity setting chamber
done
clear
View Answer play_arrow
Respiration can be called as a
A)
chemical charge
done
clear
B)
physical process
done
clear
C)
vital process
done
clear
D)
mechanical process
done
clear
View Answer play_arrow
The below figure shows the conversion of a substate to product by an enzyme. Identify the components labelled as A, B, C and D
I. Potential energy II. Transition energy III. Activation energy with enzyme IV. Activation energy without enzyme Starting from A, the components are
A)
\[I\to II\to IV\to III\]
done
clear
B)
\[II\to I\to IV\to III\]
done
clear
C)
\[I\to II\to III\to IV\]
done
clear
D)
\[III\to II\to IV\to I\]
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Sago 1. Adiantum capillus veneris B. Chilgoza 2. Sphagnum C. Maidan hair fern 3. Pinus gerardiana D. Bogmoss 4. Cycas circinalis
Codes
A)
A-2, B-4, C-3, D-1
done
clear
B)
A-4, B-3, C-1, D-2
done
clear
C)
A-3, B-1, C-2, D-4
done
clear
D)
A-1, B-2, C-4, D-3
done
clear
View Answer play_arrow
Type of immunoglobulin present in colostrum is
A)
IgG
done
clear
B)
IgD
done
clear
C)
IgA
done
clear
D)
IgE
done
clear
View Answer play_arrow
Which of the following statement is not true with reference to neurotrans mitters?
A)
Norepinephrine is inactivated by an enzyme monoamine oxidase
done
clear
B)
Cholinergic fibres release acetylcholine
done
clear
C)
Glycine is a neurotransmitter
done
clear
D)
GABA promotes postsynaptic regeneration of action potential
done
clear
View Answer play_arrow
Consider the following statements.
I. In Rhizobium cobalt play an important role in nitrogen fixation and is an essential constituents of vitamin-B. II. Cobalt found in ferns, takes part in growth. III. The elements taken in the form of gas by IV. Prokaryotes only in hydrogen. Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Nucleolar organiser 1. Histones \[{{H}_{2}}A\], \[{{H}_{2}}B\], \[{{H}_{3}}\] and \[{{H}_{4}}\] B. Facultative heterochromatin 2. Repetitive sequence of DNA C. Constitutive heterochromatin 3. Secondary constrictions D. 4. Condensed chromosomes-X
Codes
A)
A-3, B-4, C-2
done
clear
B)
A-1, B-4, C-3
done
clear
C)
A-3, B-4, C-1
done
clear
D)
A-1, B-2, C-3
done
clear
View Answer play_arrow
What is common among amylase, rennin and trypsin?
A)
These are produced by stomach
done
clear
B)
These acts at a pH lower than 7
done
clear
C)
These all are proteins
done
clear
D)
These all are proteolytic enzymes
done
clear
View Answer play_arrow
One of the following is true for taste blindness for phenylthiocarbamide (PTC)
A)
genetic dominant trait
done
clear
B)
genetic recessive trait
done
clear
C)
caused by pseudo alleles
done
clear
D)
caused by inhibitor genes
done
clear
View Answer play_arrow
How many percentage of blood is made up of water?
A)
60
done
clear
B)
45
done
clear
C)
80
done
clear
D)
90
done
clear
View Answer play_arrow
During dark reaction for fixation of carbon, the three carbon atoms of each molecule of PGA are derived from.
A)
\[\operatorname{RuBP}+C{{O}_{2}}\]
done
clear
B)
\[\operatorname{RuBP}+{{H}_{2}}O\]
done
clear
C)
only RuBP
done
clear
D)
\[only\text{ }C{{O}_{2}}\]
done
clear
View Answer play_arrow
ADA is an enzyme, which is deficient in a genetic disorder SCID. ADA stands for adenosine deaminase is also known as
A)
adenosine transferase
done
clear
B)
adenosine transaminase
done
clear
C)
adenosine am in hydrolase
done
clear
D)
adenosine deoxyaminase
done
clear
View Answer play_arrow
Which one of the following is correctly matched?
A)
Vitamin-E-to copherol
done
clear
B)
Vitamin-D-riboflavin
done
clear
C)
Vitamin-B-calciferol
done
clear
D)
Vitamin-A-thiamine
done
clear
View Answer play_arrow
Hair cells in lateral line sense organs in fish possesses one of the following receptors.
A)
Cal receptors
done
clear
B)
Frigid receptors
done
clear
C)
Stat receptors
done
clear
D)
Chemoreceptors
done
clear
View Answer play_arrow
How many amino acids are present in calcitonin?
A)
10
done
clear
B)
12
done
clear
C)
20
done
clear
D)
32
done
clear
View Answer play_arrow
Which of the following statement is/are correct? I. Animals of class-Mammalia have seven cervical vertebrae. II. Homodont type of dentition is a mammalian character. III. All mammals are characterised by viviparity and biconcave red blood cells. IV. Whale is an egg laying mammal.
A)
All of these
done
clear
B)
Only I
done
clear
C)
II, III and IV
done
clear
D)
None of these
done
clear
View Answer play_arrow
Match the following column.
Column I (Diseases) Column II (Element) A. Whiptail disease in cauliflower 1. Copper B. White bud disease in maize 2. Boron C. Internal cork of apple 3. Zinc D. Die back disease in citrus 4. Molybdenum
Codes
A)
A-3, B-4, C-1, D-2
done
clear
B)
A-4, B-3, C-2, D-1
done
clear
C)
A-3, B-2, C-1, D-4
done
clear
D)
A-4, B-2, C-3, D-1
done
clear
View Answer play_arrow
Identify the given figure.
A)
In dole 3 acetic acid
done
clear
B)
In dole 3 propionic acid
done
clear
C)
\[\alpha \] naphthoxy acetic acid
done
clear
D)
2, 4-dichlorophenoxy acetic acid
done
clear
View Answer play_arrow
Respiration that arose first, had one of the following characteristics. It, was
A)
aerobic as it is more complex
done
clear
B)
aerobic as it releases more energy
done
clear
C)
anaerobic as it releases more energy
done
clear
D)
anaerobic as early atmosphere contained little or no oxygen
done
clear
View Answer play_arrow
Who provided experimental evidence for 'selection' in bacteria using replica planting technique?
A)
Charles Darwin
done
clear
B)
Louis Pasteur
done
clear
C)
RA Fischer
done
clear
D)
Lederberg
done
clear
View Answer play_arrow
Complete the sequence of developmental process in plant cell.
A)
Differentiation Maturation Senescence Expansion
done
clear
B)
Expansion Senescence Differentiation Maturation
done
clear
C)
Differentiation Expansion Maturation Senescence
done
clear
D)
Senescence Expansion Differentiation Maturation
done
clear
View Answer play_arrow
Which one of the following taxonomic aid can give a comprehensive account of the complete information of an genus or family at a particular time?
A)
Herbarium
done
clear
B)
Taxonomic key
done
clear
C)
Museum
done
clear
D)
Monograph
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Carboniferous period 1. Age of fishes B. Devonian period 2. Age of reptiles C. Jurassic period 3. Age of mammals D. Triassic period 4. Age of amphibians
Codes
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-4, B-1, C-2, D-3
done
clear
C)
A-4, B-3, C-2, D-1
done
clear
D)
A-2, B-4, C-1, D-3
done
clear
View Answer play_arrow
In Plasmodium, ookinete is formed by
A)
zygote
done
clear
B)
mast cells
done
clear
C)
sporozoite
done
clear
D)
mezozoite
done
clear
View Answer play_arrow
In the given diagram identify.
I. Couchette II. Perineum III. Opening of Bartholdi gland IV. Clitoris
A)
A-V, B-II, C-III, D-IV
done
clear
B)
A-IV, B-III, C-II, D-I
done
clear
C)
A-VI, B-V, C-I, D-III
done
clear
D)
A-V, B-VI, C-IV, D-I
done
clear
View Answer play_arrow
Match the following autoimmune diseases with location self-antigens.
Column I
Column II
A.
Grave's disease
1.
\[\beta -cells\](pancreas)
B.
Rheumatoid arthritis
2.
IgG
C.
Type I diabetes mellitus
3.
Thyroid cells
D.
Addison?s disease
4.
Adrenal cortex
Codes
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-3, B-2, C-1, D-4
done
clear
C)
A-4, B-3, C-2, D-1
done
clear
D)
A-2, B-1, C-4, D-3
done
clear
View Answer play_arrow
Recombinant DNA technology is used firstly to produce one of the following vaccine
A)
AIDS vaccine
done
clear
B)
Polio vaccine
done
clear
C)
MMR vaccine
done
clear
D)
Hepatitis B vaccine
done
clear
View Answer play_arrow

 ,
, 


 In the above cycle identify the marking A, B, C with their number of carbons. If (1) ribulose 5-phosphate (2) glyceraldehyde 3-phosphate (3) 1, 3-diphosphoglycerate. Where figures given in the bracket shows the number of carbon in that compound.
In the above cycle identify the marking A, B, C with their number of carbons. If (1) ribulose 5-phosphate (2) glyceraldehyde 3-phosphate (3) 1, 3-diphosphoglycerate. Where figures given in the bracket shows the number of carbon in that compound. 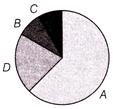
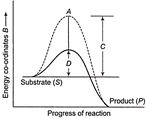 I. Potential energy II. Transition energy III. Activation energy with enzyme IV. Activation energy without enzyme Starting from A, the components are
I. Potential energy II. Transition energy III. Activation energy with enzyme IV. Activation energy without enzyme Starting from A, the components are