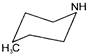
An isolated particle of mass m is moving in horizontal plane (X-Y), along the X-axis, at a certain height above ground. It suddenly explodes into two fragments of masses m/4 and 3m/4). An instant later, the smaller fragment is at \[Y=+15\,cm.\]. The larger fragment at this instant is at:
A)
\[Y=-5cm\]
done
clear
B)
\[Y=+20cm\]
done
clear
C)
\[Y=+5cm\]
done
clear
D)
\[Y=-20cm\]
done
clear
View Answer play_arrow
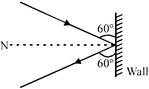
A 3kg ball strikes a heavy rigid wall with a speed of \[10m/s\] at an angle of \[{{60}^{o}}\] with the wall. It gets reflected with the same speed at \[{{60}^{o}}\] with ball. If the ball is in contact with the wall or \[0.2s,\] the average force exerted on the ball by the wall is:
A)
300N
done
clear
B)
zero
done
clear
C)
\[150\sqrt{3N}\]
done
clear
D)
150 N
done
clear
View Answer play_arrow
Two simple harmonic motions are represted by the equations \[{{Y}_{1}}=a\sin (nt+\pi /3)\] and \[{{Y}_{2}}=a\cos \pi t.\]The phase difference of velocity of particle 1 with respect to velocity of particle 2 is:
A)
\[-\frac{\pi }{3}\]
done
clear
B)
\[\frac{\pi }{6}\]
done
clear
C)
\[-\frac{\pi }{6}\]
done
clear
D)
\[\frac{\pi }{3}\]
done
clear
View Answer play_arrow
A metal wire of length L and radius r is clamped at one end to a rigid fixed support. A force F is applied at another and so that its length increase by L. The increases in length of another metal wire of length 2L and radius 2r, when stretched by a force 2F, will be:
A)
\[2L\]
done
clear
B)
\[L\]
done
clear
C)
\[L/2\]
done
clear
D)
\[L/4\]
done
clear
View Answer play_arrow
Two water of pipes of diameters \[2cm\] and \[4cm\] are connected with the main supply line. The velocity of flow of water in the pipe of \[2cm\] diameter is:
A)
4 times that in the other pipe
done
clear
B)
\[{\scriptstyle{}^{1}/{}_{4}}\] times that in the other pipes
done
clear
C)
2 times that in the other pipe
done
clear
D)
\[{\scriptstyle{}^{1}/{}_{2}}\] times that in the other pipes
done
clear
View Answer play_arrow
The height of the point vertically above the earth's surface at which the acceleration due to gravity becomes 1% of its value at the surface (R is the radius of the earth)
A)
\[8\,R\]
done
clear
B)
\[9\,R\]
done
clear
C)
\[10\,R\]
done
clear
D)
\[20\,R\]
done
clear
View Answer play_arrow
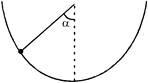
An insect crawls up a hemispherical surface very slowly. The coefficient of friction between the insect and the surface is 1/3. If the line joining the centre of the hemispherical surface to the insect, makes an angle a with the verticle, the maximum value of a is given by:
A)
\[\cot \alpha =3\]
done
clear
B)
\[\sec \alpha =3\]
done
clear
C)
\[cosec\alpha =3\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A particle is projected from a point A with a velocity V at an angle \[\theta \] (upward) with the horizontal. At a certain point B, it moves at right angle to its initial direction. It follows that:
A)
velocity of the particle at B is V
done
clear
B)
velocity of the particle at B is V \[\cos \theta \]
done
clear
C)
velocity of the particle at B is \[V\tan \theta \]
done
clear
D)
the time of flight from A to B is \[\frac{V}{g\sin \theta }\]
done
clear
View Answer play_arrow
If there is a positive error of 50% in the measurement of velocity of a body, then the error in the measurement of kinetic energy is:
A)
\[15%\]
done
clear
B)
50%
done
clear
C)
\[100%\]
done
clear
D)
125%
done
clear
View Answer play_arrow
What are the dimensions of electrical permittivity:
A)
\[[M{{L}^{-2}}{{T}^{-2}}{{Q}^{-2}}]\]
done
clear
B)
\[[{{M}^{-1}}{{L}^{-2}}{{T}^{-3}}{{Q}^{-1}}]\]
done
clear
C)
\[[{{M}^{-1}}{{L}^{-3}}{{T}^{2}}{{Q}^{2}}]\]
done
clear
D)
\[[{{M}^{-1}}{{L}^{-3}}{{T}^{-2}}{{Q}^{-2}}]\]
done
clear
View Answer play_arrow
The electric potential at a point (x, y) is given by \[V=Kxy\]. The electric field intensity at a distance r from the origin varies as:
A)
\[{{r}^{2}}\]
done
clear
B)
\[r\]
done
clear
C)
2r
done
clear
D)
\[2{{r}^{2}}\]
done
clear
View Answer play_arrow
Two small spheres each having the charge \[+Q\] are suspended by insulting threads of length L from a hook. This arrangement is taken is space where there is no gravitational effect, then the angle between the two threads and the tension in each will be:
A)
\[{{180}^{o}}\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{{{Q}^{2}}}{{{(2L)}^{2}}}\]
done
clear
B)
\[{{90}^{o}},\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{{{Q}^{2}}}{{{L}^{2}}}\]
done
clear
C)
\[{{180}^{o}},\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{{{Q}^{2}}}{2{{L}^{2}}}\]
done
clear
D)
\[{{180}^{o}},\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{{{Q}^{2}}}{{{L}^{2}}}\]
done
clear
View Answer play_arrow
A cricket ball is hit for a six leaving the bat at an angle of \[{{45}^{o}}\] to the horizontal with kinetic energy K. At the top position, the kinetic energy of the ball is:
A)
zero
done
clear
B)
K
done
clear
C)
\[K/2\]
done
clear
D)
\[K/\sqrt{2}\]
done
clear
View Answer play_arrow
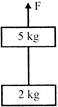
Two blocks of masses 5kg and 2kg are connected by a massles a string as shown in the figure. A verticle force F is applied on the 5kg block. Find tghe value of F, its tension in the string is \[40N\] \[(g=10m/{{s}^{2}})\].
A)
\[140\,N\]
done
clear
B)
\[70\,N\]
done
clear
C)
\[40\,N\]
done
clear
D)
\[100\,N\]
done
clear
View Answer play_arrow
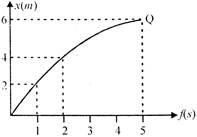
What is the average velocity during the interval \[t=2s\] tot \[=5s\], In the following position-time curve?
A)
\[2m/s\]
done
clear
B)
\[2/3m/s\]
done
clear
C)
\[1.2m/s\]
done
clear
D)
\[0.4m/s\]
done
clear
View Answer play_arrow
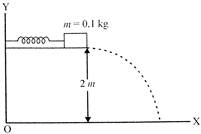
A small block of mass \[0.1\text{ }kg\] is pressed against a horizontal spring fixed at one end to compress the spring through \[5.0cm\]as shown. The spring constant is \[100N/m.\]. When released the WE block moves horizonally till it leaves the spring. It will hit the ground 2m below the spring.
A)
at a horizontal distance of \[1m\] from free end of the spring
done
clear
B)
at a horizontal distance of \[2m\] from free and of the springs
done
clear
C)
vertically below the edge on which the mass is resting
done
clear
D)
at a horizontal distance of \[\sqrt{2}m\] from free end of the spring
done
clear
View Answer play_arrow
The root mean square velocity of the gas molecules is \[300m/s\]. What will be root mean square speed of the molecules if the atomic weight is double and absolute temperature havled?
A)
\[300m/s\]
done
clear
B)
\[150m/s\]
done
clear
C)
\[600m/s\]
done
clear
D)
\[75m/s\]
done
clear
View Answer play_arrow
If Y be the ratio of specific heats of a perfect gas, the number of degree of freedom of a molecule of the gas is:
A)
\[(Y-1)\]
done
clear
B)
\[\frac{3Y-1}{2Y-1}\]
done
clear
C)
\[\frac{2}{Y-1}\]
done
clear
D)
\[\frac{9}{2}\,(Y-1)\]
done
clear
View Answer play_arrow
The driver of a car travelling with speed \[30m/s\]towards a hill, sounds a horns of frequency \[600Hz\]. If the velocity of sound in air is \[330Ws,\]the frequency of reflected sound as heard by the driver is:
A)
\[720\,Hz\]
done
clear
B)
\[555.5Hz\]
done
clear
C)
\[550Hz\]
done
clear
D)
\[500Hz\]
done
clear
View Answer play_arrow
A thin circular ring of mass m and radius R is rotating about its axis with a constant angular \[\omega \]. Two objects each of mas m are attached gently to the opposite ends of a diameter of the ring. The ring now rotates with an angular velocity \[\omega =\]
A)
\[\frac{\omega m}{(m+2m)}\]
done
clear
B)
\[\frac{\omega (m+2m)}{m}\]
done
clear
C)
\[\frac{\omega (m-2m)}{(m+2m)}\]
done
clear
D)
\[\frac{\omega m}{(m+m)}\]
done
clear
View Answer play_arrow
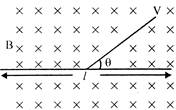
A particle of charge per unit mass, a is released from the origin width a velocity \[V={{V}_{0}},\] in a uniform magnetic field \[B=-{{B}_{0}}K\]. If the particle posses through \[(0,y,0)\], then Y is equal to:
A)
\[\frac{2{{V}_{0}}}{{{B}_{0}}a}\]
done
clear
B)
\[\frac{{{V}_{0}}}{{{B}_{0}}a}\]
done
clear
C)
\[\frac{2{{V}_{0}}}{{{B}_{0}}a}\]
done
clear
D)
\[\frac{{{V}_{0}}}{{{B}_{0}}a}\]
done
clear
View Answer play_arrow
A circular current carrying loop radius R, carries a current \[i,\] the magnetic field at a point on the axis of coil is \[\frac{1}{\sqrt{8}}\] times the value of magnetic field of the centre. Distance of point from centre is:
A)
\[\frac{R}{\sqrt{2}}\]
done
clear
B)
\[\frac{R}{\sqrt{3}}\]
done
clear
C)
\[R\sqrt{2}\]
done
clear
D)
\[R\]
done
clear
View Answer play_arrow
If a copper wire is stretched to make it \[0.1%\]longer. The percentage change in its resistance is:
A)
\[0.2%\]increases
done
clear
B)
\[0.2%\]decrease
done
clear
C)
\[0.1%\]increase
done
clear
D)
\[0.1%\] decrease
done
clear
View Answer play_arrow
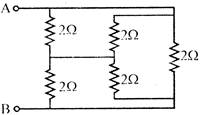
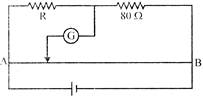
Find the equivalent resistance across AB
A)
\[1\,\Omega \]
done
clear
B)
\[2\,\Omega \]
done
clear
C)
\[3\,\Omega \]
done
clear
D)
\[4\,\Omega \]
done
clear
View Answer play_arrow
A, B and C are resistors of resistances R, \[1.5R\]and 3R respectively. When some potential difference is applied between X and Y, the voltmeter reading are \[{{V}_{A}},{{V}_{B}}\] and \[{{V}_{C}}\] respectively:
A)
\[{{V}_{A}}={{V}_{B}}={{V}_{C}}\]
done
clear
B)
\[{{V}_{A}}\ne {{V}_{B}}={{V}_{C}}\]
done
clear
C)
\[{{V}_{A}}={{V}_{B}}\ne {{V}_{C}}\]
done
clear
D)
\[{{V}_{B}}\ne {{V}_{A}}={{V}_{C}}\]
done
clear
View Answer play_arrow
AB is wire of uniform resistance. The galvanometer G shows zero current when the length \[AC=20cm\]and \[CB=80cm\], The resistance R is equal to:
A)
\[2\,\Omega \]
done
clear
B)
\[8\Omega \]
done
clear
C)
\[20\Omega \]
done
clear
D)
\[40\Omega \]
done
clear
View Answer play_arrow
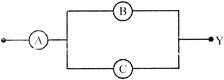
The total current supplied to the circuit by the battery is:
A)
1A
done
clear
B)
2A
done
clear
C)
4A
done
clear
D)
6A
done
clear
View Answer play_arrow
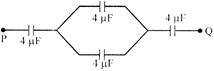
Four condensers each of capacity \[4\mu \] fare connected as shown in figure, with \[{{V}_{P}}-{{V}_{Q}}=15V.\] The energy stored in the system is:
A)
\[2400erg\]
done
clear
B)
\[1800erg\]
done
clear
C)
\[3600erg\]
done
clear
D)
\[5400erg\]
done
clear
View Answer play_arrow
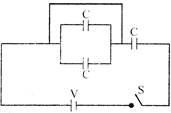
In the given circuit, find the heat generated if switch is closed:
A)
\[\frac{3}{2}C{{V}^{2}}\]
done
clear
B)
\[\frac{1}{2}C{{V}^{2}}\]
done
clear
C)
\[C{{V}^{2}}\]
done
clear
D)
\[\frac{1}{3}C{{V}^{2}}\]
done
clear
View Answer play_arrow
The inward and outward electric flux for a closed surface in units of \[N{{m}^{2}}/c\] arc respectively \[8\times {{10}^{3}}N-{{m}^{2}}/C\] and \[4\times {{10}^{-3}}N-{{m}^{2}}/C\]. Then, the total charge inside the surface is:
A)
\[4\times {{10}^{3}}C\]
done
clear
B)
\[-4\times {{10}^{3}}C\]
done
clear
C)
\[\frac{-4\times {{10}^{3}}}{{{\varepsilon }_{0}}}C\]
done
clear
D)
\[-(4\times {{10}^{3}}){{\varepsilon }_{0}}C\]
done
clear
View Answer play_arrow
The radius of hydrogen atom in its ground state is \[5.3\times {{10}^{-11}}m.\]. After collision with an electron is found to have a radius of \[21.2\times {{10}^{-11}}m\]. What is the principal quantum number n of the final state of atom?
A)
\[n=4\]
done
clear
B)
\[n=2\]
done
clear
C)
\[n=16\]
done
clear
D)
\[n=3\]
done
clear
View Answer play_arrow
In a p - n junction diode, the direction of diffusion current is from:
A)
p - region to n - region
done
clear
B)
n - region to p - region
done
clear
C)
n - region to p - region when biased and vice versa when reverse biased
done
clear
D)
p - region when forward biased and vice-versa when reverse biased
done
clear
View Answer play_arrow
The wavelength of the \[{{K}_{a}}\] line for the uranium atom \[(z=92)\] is \[(R=10{{m}^{-1}})\]
A)
\[1.6\overset{o}{\mathop{A}}\,\]
done
clear
B)
\[0.16\overset{o}{\mathop{A}}\,\]
done
clear
C)
\[0.5\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[2.0\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
The input resistance of a \[C\varepsilon \] amplifier is \[333\Omega \] and the load resistance is \[5K\Omega \]. A change of base current by \[15\mu A\] results in the change of collector current by 1mA. The voltage gain of amplifier is:
A)
550
done
clear
B)
101
done
clear
C)
1001
done
clear
D)
501
done
clear
View Answer play_arrow
A freshly prepared radioactive source of half-life 2h emits radiation of intensity which is 64 times the permissible safe level. The minimum time after which it would be possible to work safely with source is:
A)
\[6h\]
done
clear
B)
\[12h\]
done
clear
C)
\[24h\]
done
clear
D)
\[128h\]
done
clear
View Answer play_arrow
The maximum kinetic energy of photon electrons emitted from a surface when photons of energy \[6eV\] fall on it is\[4eV\]. The stopping potential is:
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
10
done
clear
View Answer play_arrow
To which logic gate, does the truth table given below correspond?
A B X 0 0 0 0 1 1 1 0 1 1 1 1
A)
AND
done
clear
B)
OR
done
clear
C)
NAND
done
clear
D)
XOR
done
clear
View Answer play_arrow
The magnifying power of an astronomical teleshope in normal adjustment is 8 and the distance between the two lenses is 54 cm. The focal length of eye lens and objective lens will be respectively:
A)
6 cm and 48 crn
done
clear
B)
48 cm and 6 cm
done
clear
C)
8 cm and 64 cm
done
clear
D)
64 cm and 8 cm
done
clear
View Answer play_arrow
An object is placed in front of a convex mirror at a dstance of 50cm. A plane mirror is introduced covering the lower half of the convex mirror. If the distance between the object and the plane mirror is 30cm, there is no parallel between the images formed by the two mirrors. The radius of curvature the convex mirror (in cm) is:
A)
60
done
clear
B)
50
done
clear
C)
30
done
clear
D)
25
done
clear
View Answer play_arrow
In Young's double slit experiment when sodium light of wavelength \[5893\overset{o}{\mathop{A}}\,\] is used, then 62 frings are seen in the field of view. Instead of sodium light if violet light of wavelength \[4358\overset{o}{\mathop{A}}\,\] is used then the number of frings that will be seen in the field of view will be:
A)
54
done
clear
B)
64
done
clear
C)
74
done
clear
D)
84
done
clear
View Answer play_arrow
Light wave enters from medium 1 to medium 2. Its velocity in 2nd medium is double from 1st. For total interval reflection, the angle of incidence must be greater than:
A)
\[{{30}^{o}}\]
done
clear
B)
\[{{60}^{o}}\]
done
clear
C)
\[{{45}^{o}}\]
done
clear
D)
\[{{90}^{o}}\]
done
clear
View Answer play_arrow
In an AC circuit, the potential differences across an inductance and a resistance connected in series are respectively 16V and 20V. The total potential difference across the circuit is:
A)
\[20.0\,V\]
done
clear
B)
\[25.6\,V\]
done
clear
C)
\[31.9\,V\]
done
clear
D)
\[53.5\,V\]
done
clear
View Answer play_arrow
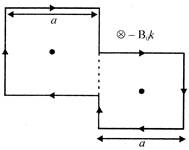
The torque acting on the loop (given), will be:
A)
zero
done
clear
B)
\[\frac{1{{a}^{2}}{{B}_{0}}}{2}\]
done
clear
C)
\[\frac{2l{{a}^{2}}{{B}_{0}}}{3}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A rod length \[l\] is moved with a velocity V in a magnetic field B as shown in figure, the equivalent electrical circuits:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
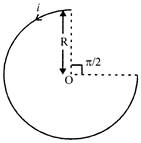
A current of i ampere flows in a circular arc of wire, whose radius is R, which subtend an angle \[3\pi /2\] radian at its centre. The magnetic induction B at the centre is:
A)
\[\frac{{{\mu }_{0}}i}{R}\]
done
clear
B)
\[\frac{{{\mu }_{0}}i}{2R}\]
done
clear
C)
\[\frac{2{{\mu }_{0}}i}{R}\]
done
clear
D)
\[\frac{3{{\mu }_{0}}i}{8R}\]
done
clear
View Answer play_arrow
Which one of the following species is not a pseudo halide?
A)
\[CN{{O}^{-}}\]
done
clear
B)
\[RCO{{O}^{-}}\]
done
clear
C)
\[OC{{N}^{-}}\]
done
clear
D)
\[NN{{N}^{-}}\]
done
clear
View Answer play_arrow
Ammonium dichromate is used in some fire works. The green coloured powder blown in the air is:
A)
\[Cr{{O}_{3}}\]
done
clear
B)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
C)
\[Cr\]
done
clear
D)
\[CrO({{O}_{2}})\]
done
clear
View Answer play_arrow
Which of the following compounds is expected to be coloured
A)
\[A{{g}_{2}}S{{O}_{4}}\]
done
clear
B)
\[CuCl\]
done
clear
C)
\[Cu{{F}_{2}}\]
done
clear
D)
\[Mg{{F}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is an organometallic compound:
A)
Lithium mcthoxide
done
clear
B)
Lithium acetate
done
clear
C)
Lithium dimethylamide
done
clear
D)
Methyl lithium
done
clear
View Answer play_arrow
In the reaction p-chlorotolune with \[KN{{H}_{2}}\] In liq \[N{{H}_{3}},\] the major product is
A)
o-toluidine
done
clear
B)
m-toluidine
done
clear
C)
p-toluidine
done
clear
D)
p-chloroaniline
done
clear
View Answer play_arrow
Which of the following statement(s) is/are correct?
A)
The electronic configuration of \[Cr\] is \[[Ar]3{{d}^{5}}4{{s}^{1}}\].
done
clear
B)
The magnetic quantum number may have a negative value.
done
clear
C)
In silver atom 23 electrons have a spin of one type and 24 of the opposite type.
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following will react with water
A)
\[CHC{{l}_{3}}\]
done
clear
B)
\[C{{l}_{3}}C.CHO\]
done
clear
C)
\[CC{{l}_{4}}\]
done
clear
D)
\[ClC{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
View Answer play_arrow
The pH of \[0.1\text{ }M\] solution of the following salts increases in the order
A)
\[NaCl<N{{H}_{4}}Cl<NaCN<HCl\]
done
clear
B)
\[HCl<N{{H}_{4}}Cl<NaCl<NaCN\]
done
clear
C)
\[NaCN<N{{H}_{4}}Cl<NaCl<HCl\]
done
clear
D)
\[HCl<NaCl<NaCN<N{{H}_{4}}Cl\]
done
clear
View Answer play_arrow
Which of the following is an example of chlorodising roasting?
A)
\[A{{g}_{2}}S+2NaCl\xrightarrow{{}}2AgCl+N{{a}_{2}}S\]
done
clear
B)
\[PbS+2{{O}_{2}}\xrightarrow{{}}PbS{{O}_{4}}\]
done
clear
C)
\[2AgCl+4Hg\xrightarrow{{}}2AgHg+H{{g}_{2}}C{{l}_{2}}\]
done
clear
D)
Both (a) and (c)
done
clear
View Answer play_arrow
The molecule of \[B{{F}_{3}}\] and \[N{{F}_{3}}\] both are covalent compounds but \[B{{F}_{3}}\] is non-polar and \[N{{F}_{3}}\] is polar. The reason is that
A)
boron is a metal and nitrogen is a gas in uncombined state
done
clear
B)
\[B{{F}_{3}}\] bonds have no dipole moment whereas \[N{{F}_{3}}\] bond have dipole moment.
done
clear
C)
Atomic size of boron is smaller than that of nitrogen
done
clear
D)
\[B{{F}_{3}}\] is symmetrical molecule whereas \[N{{F}_{3}}\] is unsymmetrical
done
clear
View Answer play_arrow
\[X\xleftarrow[{{H}_{2}}S{{O}_{4}}.{{H}_{2}}O]{HgS{{O}_{4}}}1-pentyne\xrightarrow[O{{H}^{-}},{{H}_{2}}{{O}_{2}}]{B{{H}_{3}}.THF}Y\] X and Y cannot be distinguished by
A)
Silver mirror test
done
clear
B)
iodoform test
done
clear
C)
\[N{{H}_{2}}OH\]
done
clear
D)
\[LiAl{{H}_{4}}\]
done
clear
View Answer play_arrow
Predominant product is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[AgCl\] is colourless whereas \[AgI\] is yellow because of
A)
\[A{{g}^{+}}\]possess 18 electrons shell to screen the nuclear charge
done
clear
B)
\[A{{g}^{+}}\] shows pseudo inert gas configuration
done
clear
C)
distortion of \[{{I}^{-}}\] is more pronounced than \[C{{l}^{-}}\] ion
done
clear
D)
existence of d-d transition
done
clear
View Answer play_arrow
The osmotic pressure of blood is \[7.65\]atm at \[{{37}^{o}}C\]. How much glucose should be used per litre for an intravenous injection that is to have the same osmotic pressure as blood?
A)
\[180\text{ }g\]
done
clear
B)
\[54.2\text{ }g\]
done
clear
C)
\[86\text{ }g\]
done
clear
D)
\[\text{111 }g\]
done
clear
View Answer play_arrow
The compound ?X? was boiled under reflux for some time with a solution of sodium hydroxide. The solution was cooled, acidified with dilute nitric acid and then silver nitrate solution was added, a thick precipitate was formed which one is incorrect as ?X??
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}C{{H}_{2}}Cl\]
done
clear
C)
\[C{{H}_{2}}ClCOOH\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
View Answer play_arrow
In the reaction sequence \[X\xrightarrow{Ca{{(OH)}_{2}}}Y\xrightarrow{dry\,distillation}Acetone\xrightarrow[{{H}_{2}}S{{O}_{4}}]{conc.}Z\] X, Y and Z are
A)
\[C{{H}_{3}},CHO,\] aldol, phorone
done
clear
B)
\[HCOOH,\,{{(HCOO)}_{2}}Ca,\] mesityl oxide
done
clear
C)
\[C{{H}_{3}}COOH,{{(C{{H}_{3}}COO)}_{2}}Ca,\] mesityl oxide
done
clear
D)
\[C{{H}_{3}}COOH,{{(C{{H}_{3}}COO)}_{2}}Ca,\] pinacol
done
clear
View Answer play_arrow
The process of zone refining is based upon
A)
fractional crystallization
done
clear
B)
fractional distillation
done
clear
C)
magnetic properties of imparities
done
clear
D)
impurities are less fusible than metals
done
clear
View Answer play_arrow
Which of the following is incorrect about thermosetting polymers?
A)
They soften on heating and harden on cooling irreversibly.
done
clear
B)
By heating polymer can be reshaped and reused.
done
clear
C)
They possess three dimensional network structure containing cross links.
done
clear
D)
They are strong, hard and more brittle.
done
clear
View Answer play_arrow
The IIJPAC name of the following compound is \[C{{H}_{3}}-C{{H}_{2}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{N}}\,}}\,-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-H\]
A)
N-formyl-N-methyl ethanamine
done
clear
B)
N-ethyl-N-ixiethyl methanamide
done
clear
C)
N-methyl-N-oxo ethanamine
done
clear
D)
none of these
done
clear
View Answer play_arrow
\[A\xrightarrow{Ca{{(OH)}_{2}}}Y\xrightarrow{dry\,\,distillation}Acetone\xrightarrow[{{H}_{2}}S{{O}_{4}}]{conc.}Z\]\[A+B\xrightarrow{\Delta }Polymer\] Polymer is
A)
Polyacrylonitrile
done
clear
B)
nylon-6
done
clear
C)
nylon-66
done
clear
D)
buna-N
done
clear
View Answer play_arrow
If \[{{r}_{1}}\] is the radius of the first orbit of hydrogen atom, then the radii of second, third and fourth orbitals in terms of \[{{r}_{1}}\] are
A)
\[r_{1}^{2},r_{1}^{3},r_{1}^{4}\]
done
clear
B)
\[8{{r}_{1}},27{{r}_{1}},64{{r}_{1}}\]
done
clear
C)
\[4{{r}_{1}},9{{r}_{1}},16{{r}_{1}}\]
done
clear
D)
\[2{{r}_{1}},6r,8{{r}_{1}}\]
done
clear
View Answer play_arrow
Adsorption occurs not due to
A)
unbalanced force at surface molecules
done
clear
B)
unutilized free valencies at surface
done
clear
C)
vander wall's attraction at surface
done
clear
D)
increased entropy at surface
done
clear
View Answer play_arrow
A mixture of formic acid and oxalic acid is heated with concentrated \[{{H}_{2}}S{{O}_{4}},\] The gaseous product is passed into \[KOH\] solution where the volume decreases by \[\frac{1}{6}\] th. The molecular proportion of the organic acids, formic acid and oxalic acid in the mixture is
A)
\[4:1\]
done
clear
B)
\[1:4\]
done
clear
C)
\[1:2\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
Based on lattice energy and other considerations which one of the following alkali metal chlorides is expected to have the highest melting point?
A)
\[RbCl\]
done
clear
B)
\[KCl\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[LiCl\]
done
clear
View Answer play_arrow
Given the data at \[{{25}^{o}}C\] \[Ag+{{I}^{-}}\xrightarrow{{}}AgI+{{e}^{-}};\] \[{{E}^{o}}=0.152\,V\] \[Ag\xrightarrow{{}}A{{g}^{+}}+{{e}^{-}};\] \[{{E}^{o}}=-0.800V\] What is the vlaue of log \[{{K}_{SP}}\] for \[AgI\]? \[\left( 2.303\frac{RT}{F}=0.059\,V \right)\]
A)
\[-8.12\]
done
clear
B)
\[+8.612\]
done
clear
C)
\[-37.83\]
done
clear
D)
\[-16.13\]
done
clear
View Answer play_arrow
The electrophile involved in the above reaction is
A)
\[\overset{\oplus }{\mathop{C}}\,HC{{l}_{2}}\]
done
clear
B)
\[:CC{{l}_{2}}\]
done
clear
C)
\[\overset{O-}{\mathop{C}}\,C{{l}_{2}}\]
done
clear
D)
\[\overset{\oplus }{\mathop{C}}\,HO\]
done
clear
View Answer play_arrow
The smog is essentially caused by the presence of
A)
\[{{O}_{2}}\] and \[{{O}_{3}}\]
done
clear
B)
Oxides of sulphur and nitrogen
done
clear
C)
\[{{O}_{2}}\] and \[{{N}_{2}}\]
done
clear
D)
\[{{O}_{3}}\] and \[{{N}_{2}}\]
done
clear
View Answer play_arrow
Acetyl bromide reacts with excess of \[C{{H}_{3}}MgI\] followed by treatment with a saturated solution of \[N{{H}_{4}}Cl\] gives
A)
acetone
done
clear
B)
acetamide
done
clear
C)
2-methyl-2-propanol
done
clear
D)
acetyl iodide
done
clear
View Answer play_arrow
The rms velocity of an ideal gas at \[{{27}^{o}}C\] is\[0.3\text{ }m{{s}^{-1}}\].Its rms velocity at \[{{927}^{o}}C\] (in \[{{m}^{-1}}\]) is
A)
\[3.0\]
done
clear
B)
\[2.4\]
done
clear
C)
\[0.9\]
done
clear
D)
\[0.6\]
done
clear
View Answer play_arrow
The volume of \[C{{l}_{2}}\] at S.T.P. obtained on reacting \[4.35\text{ }g\] \[~Mn{{O}_{2}}\]with cone. \[HCl\] (at. wt. of \[Mn=55\]) is
A)
\[4.48\]litre
done
clear
B)
\[2.24\]litre
done
clear
C)
\[1.12\]litre
done
clear
D)
\[0.56\]litre
done
clear
View Answer play_arrow
Which among the following will show maximum osmotic pressure?
A)
\[1\,M\,NaCl\]
done
clear
B)
\[1\,M\,MgC{{l}_{2}}\]
done
clear
C)
\[1\,M\,{{(N{{H}_{4}})}_{3}}P{{O}_{4}}\]
done
clear
D)
\[1\,M\,N{{a}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
In which of the following process of neutralization magnitude of \[\Delta {{H}_{ueutralisation}}\] less than that of \[\Delta {{H}_{ionization}}\] of water?
A)
\[HCl+NaOH\]
done
clear
B)
\[C{{H}_{3}}COOH+NaOH\]
done
clear
C)
\[{{H}_{2}}S{{O}_{4}}+NaOH\]
done
clear
D)
\[HCl{{O}_{4}}+KOH\]
done
clear
View Answer play_arrow
\[0.01\]mole of lime \[(CaO)\] was dissolved in \[100\text{ }c{{m}^{3}}\]of water. Assuming the base is completely ionized in the solution, the pH of the solution will be
A)
\[13\]
done
clear
B)
\[13.3\]
done
clear
C)
\[12.3\]
done
clear
D)
\[12.0\]
done
clear
View Answer play_arrow
In which one of the following reactions, nitrogen is not reduced?
A)
\[N{{O}_{2}}\xrightarrow[{}]{}NO_{2}^{-}\]
done
clear
B)
\[NO_{3}^{-}\xrightarrow[{}]{}NO\]
done
clear
C)
\[NO_{3}^{-}\xrightarrow{{}}NO_{4}^{+}\]
done
clear
D)
\[NO_{4}^{+}\xrightarrow{{}}{{N}_{2}}\]
done
clear
View Answer play_arrow
The value of \[{{E}_{{{H}_{2}}O/{{H}_{2}}\,\,(1atm)\,1\,pt}}\] at 298 K would be
A)
\[-0.207\,V\]
done
clear
B)
\[+\,0.207\,V\]
done
clear
C)
\[-\,0.414\,\,V\]
done
clear
D)
\[+0.414\,V\]
done
clear
View Answer play_arrow
Which of the following is incorrect about transport number?
A)
it decreases with increase in concetration
done
clear
B)
it is different for \[C{{l}^{-}}\] ion in \[0.1\text{ }M\text{ }HCl\]and \[0.1\text{ }M\text{ }NaCl\] solution.
done
clear
C)
it may increase or decrease with increase in temperature
done
clear
D)
it is never zero
done
clear
View Answer play_arrow
If 'a' is the initial concetration of the reactant, the time taken for completion of the reaction, if it is of zero order, will be
A)
\[{}^{a}/{}_{k}\]
done
clear
B)
\[{}^{a}/{}_{2\,k}\]
done
clear
C)
\[{}^{2a}/{}_{k}\]
done
clear
D)
\[{}^{k}/{}_{a}\]
done
clear
View Answer play_arrow
Which of the following elements with atomic numbers 25,30, 48 and 80 has the highest vapour pressure at room temperature?
A)
\[Z=30\]
done
clear
B)
\[Z=25\]
done
clear
C)
\[Z=48\]
done
clear
D)
\[Z=80\]
done
clear
View Answer play_arrow
Which of the following statements is correct?
A)
Lithium carbonate is soluble in water
done
clear
B)
Carbonates of \[Na,\text{ }K,\]and \[NH_{4}^{+}\] are soluble in water
done
clear
C)
Carbonates of \[Ca,\text{ }Sr\] and \[Ba\] are soluble in water
done
clear
D)
Basic carbonates of \[Mg\] and \[Cu\] are soluble in water
done
clear
View Answer play_arrow
A hydride of nitrogen which is acidic is
A)
\[N{{H}_{3}}\]
done
clear
B)
\[{{N}_{2}}{{H}_{4}}\]
done
clear
C)
\[{{N}_{2}}{{H}_{2}}\]
done
clear
D)
\[{{N}_{3}}H\]
done
clear
View Answer play_arrow
Chloramine-T is used as
A)
antipyretic
done
clear
B)
analygesic
done
clear
C)
antiseptic
done
clear
D)
disinfectant
done
clear
View Answer play_arrow
Which has maximum boiling point?
A)
done
clear
B)
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}N\]
done
clear
D)
done
clear
View Answer play_arrow
The major product of the following sequence is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Chloroform, when kept open is oxidised to:
A)
\[C{{O}_{2}}\]
done
clear
B)
\[COC{{l}_{2}}\]
done
clear
C)
\[C{{O}_{2}},C{{l}_{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
Setting of plaster of paris is
A)
oxidation with atmospheric oxygen
done
clear
B)
Combination with atmospheric \[C{{O}_{2}}\]
done
clear
C)
dehydration
done
clear
D)
hydration to yield another hydrate
done
clear
View Answer play_arrow
Which of the following term is not recognized by botanists during nomenclature?
A)
Variety
done
clear
B)
Tautonyms
done
clear
C)
Autonyms
done
clear
D)
Homonyms
done
clear
View Answer play_arrow
In which of the following animals body can be divided into two identical halves by any plane passing through the central axis?
A)
Platyhelminthes, aschelminthes
done
clear
B)
Coe]enterates and platyhelminthes
done
clear
C)
Coelenterates ctenophorans and echinoderms
done
clear
D)
Poriferans, ctenophorans and molluscs
done
clear
View Answer play_arrow
Tiger, rose, fish and roundworm taxon assigned to categories respectively are
A)
Family, genus, species and subspecies
done
clear
B)
Order, family, genus and species
done
clear
C)
Class, order, family and genus
done
clear
D)
Species, genus, class and phylum
done
clear
View Answer play_arrow
Which of the following shows lysogenic cycle?
A)
\[{{T}_{A}}\] phase
done
clear
B)
\[\lambda \] phase
done
clear
C)
Temperate
done
clear
D)
More the one option is correct
done
clear
View Answer play_arrow
Which of the following are common features in locust, scorpion and prawn?
A)
Book gills and Malpighian tubules
done
clear
B)
Antennae and eyes
done
clear
C)
Chitinous exoskeleton and jointed appendages
done
clear
D)
Jointed appendages and tracheal system
done
clear
View Answer play_arrow
The most distinctive feature of echinoderms is presence of water vascular system. This system mainly helps in all except
A)
Locomotion
done
clear
B)
Capture and transport of food
done
clear
C)
Respiration
done
clear
D)
Excretion
done
clear
View Answer play_arrow
Choose the correct match
A)
Crustose lichen - Rhizocarpon
done
clear
B)
Foliose lichen - Usher
done
clear
C)
Bread of heaven - lecanora esculenta
done
clear
D)
Stone mushroom - Dermatocarpon miniatum
done
clear
View Answer play_arrow
Generation time of bacterium is 20 minutes, if initially, there were 50 cells in the culture medium, then what will be the number of cells after 2 hours?
A)
2400
done
clear
B)
2560
done
clear
C)
3200
done
clear
D)
3000
done
clear
View Answer play_arrow
Which of the following phylum includes headless, brainless and heartless animals?
A)
Annelida
done
clear
B)
Mollusca
done
clear
C)
Echinodermata
done
clear
D)
Hemichordata
done
clear
View Answer play_arrow
Osteichthyes fishes can be distinguished from Chondrichthyes fishes by all the following characteristics, except
A)
4 pairs of gills covered by an operculum on each side
done
clear
B)
They can stay at a particular depth without expending energy in swimming
done
clear
C)
Skin is covered with Cycloid/ctenoid scales
done
clear
D)
Male has claspers and fertilization is internal
done
clear
View Answer play_arrow
Find the incorrect matched pair
A)
Gonorrhoea - Salmonella Schottmulleri
done
clear
B)
Soft rot of carrt - Erwinia Cartovora
done
clear
C)
Fire blight of apple - Erwinia amylovora
done
clear
D)
Black rot of cabbage - Streptomyces scabies
done
clear
View Answer play_arrow
Mark the odd one with respect to Halophiles
A)
Are aerobic Chemo heterotrophs
done
clear
B)
Are gram negative
done
clear
C)
Develop becteriorhodopsin in their membrane at high light intensity
done
clear
D)
Possess sap vacuoles, so they cannot get plasmolysed in high salt concentration
done
clear
View Answer play_arrow
Match the following column-I with Column-II
Column-I Column-II A. Pythium debaryanum I. Downy mildew in Mustard B. Peronospora II. Damping off disease in seedling of tomatos C. Puccinia glumarum III. Salmon disease of gills in fishes D. Saprolegnia IV. Yellow rust of wheat
A)
A-IV B-III C-II D-I
done
clear
B)
A-II B-I C-IV D-III
done
clear
C)
A-II B-IV C-I D-III
done
clear
D)
A-II B-I C-III D-IV
done
clear
View Answer play_arrow
Embryophytc which shows siphonogamy is
A)
Riccia
done
clear
B)
Selaginella
done
clear
C)
Pinus
done
clear
D)
Funaria
done
clear
View Answer play_arrow
A structure found in brown algae which is functionally similar to phloem of angiosperm is
A)
Fucosan vesicle
done
clear
B)
Holdfast
done
clear
C)
Stipe
done
clear
D)
Trumpet hyphae
done
clear
View Answer play_arrow
The most unique morphological feature of all mammals is presence of_____
A)
Muscular diaphragm
done
clear
B)
External ear
done
clear
C)
Milk productive giants
done
clear
D)
Seven cervical vertebrae
done
clear
View Answer play_arrow
If Neophron is transferred from \[{{30}^{o}}C\] to \[{{10}^{o}}C,\] their body temperature will change to
A)
\[{{10}^{o}}C\]
done
clear
B)
\[{{30}^{o}}C\]
done
clear
C)
\[{{15}^{o}}C\]
done
clear
D)
Remain unchanged
done
clear
View Answer play_arrow
Which of the following statement is true?
A)
All chordates are vertebrates
done
clear
B)
All vertebrates are chordates
done
clear
C)
Non-chordates have a vertebral column
done
clear
D)
Invertebrates posses tubular nerve cord
done
clear
View Answer play_arrow
Which of the following cannot be considered as parasitic adaptation with respect to platyhelminthes?
A)
Protective tegument around the body
done
clear
B)
Suckers and hooks are present for attachment
done
clear
C)
Highly developed reproductive system
done
clear
D)
Well developed digestive tract and digestive glands
done
clear
View Answer play_arrow
The animals which have a dorsal notochord extended from tail to head region, better developed gill slits but have no paired fins and no vertebral column at any stage may belongs to group
A. Hemichordata B. Urochordata C. Cephalochordata D. Cyclostomata
A)
B only
done
clear
B)
C only
done
clear
C)
C & D
done
clear
D)
All of these
done
clear
View Answer play_arrow
While sitting the entire weight of body falls on
A)
Calcaneum
done
clear
B)
Ilium
done
clear
C)
Pubis
done
clear
D)
Ischium
done
clear
View Answer play_arrow
If the hematocrit value increases the blood viscosity
A)
Increases
done
clear
B)
Remains the same
done
clear
C)
Decreases
done
clear
D)
Viscosity is independent to viscosity
done
clear
View Answer play_arrow
The ear ossicle, malleus is a modification of
A)
Quadrate bone
done
clear
B)
Ethmoidal bone
done
clear
C)
Articular bone
done
clear
D)
Hyomandibular bone
done
clear
View Answer play_arrow
Which of the following is present in medulla oblongata
A)
hunger centre
done
clear
B)
thrust centre
done
clear
C)
vomiting centre
done
clear
D)
thermoregulatory centre
done
clear
View Answer play_arrow
If the velocity of blood increases, blood pressure will
A)
Decreases
done
clear
B)
Increases
done
clear
C)
Remains the same
done
clear
D)
First increase than decrease
done
clear
View Answer play_arrow
Which of the following is secreted in response to high blood pressure and increase in GFR
A)
ANF
done
clear
B)
Renin
done
clear
C)
Angiotensin
done
clear
D)
Rennin
done
clear
View Answer play_arrow
If we say that \[{{P}_{50}}\] value of haemoglobin falls this means the affinity of haemoglobin for \[{{O}_{2}}\]
A)
Rises
done
clear
B)
Falls
done
clear
C)
Remains same
done
clear
D)
Dependence on \[Pc{{o}_{2}}\]
done
clear
View Answer play_arrow
Pitutry dwarf differ from cretinism in lacking
A)
Deaf mutism
done
clear
B)
Stunted growth
done
clear
C)
Better mental ability
done
clear
D)
None of these
done
clear
View Answer play_arrow
Alveolar air volume of healthy person per minute is
A)
2000 ml
done
clear
B)
5600 ml
done
clear
C)
500ml
done
clear
D)
8000 ml
done
clear
View Answer play_arrow
Human dentition is diphyodant. The formula is
A)
\[\frac{0021}{0021}\]
done
clear
B)
\[\frac{2123}{2123}\]
done
clear
C)
\[\frac{212}{212}\]
done
clear
D)
\[\frac{2112}{2112}\]
done
clear
View Answer play_arrow
Widely used herbicide for dicot weeds is
A)
Dalapon
done
clear
B)
NAA
done
clear
C)
Kinetin
done
clear
D)
2,4D
done
clear
View Answer play_arrow
Environmental heterophylly is shown by
A)
Larkspur
done
clear
B)
Cotton
done
clear
C)
Buttercup
done
clear
D)
Coriander
done
clear
View Answer play_arrow
Swelling of piece of wood when placed in water is an example of
A)
Diffusion
done
clear
B)
Imbibitions
done
clear
C)
Differentiation
done
clear
D)
Intrinsic growth
done
clear
View Answer play_arrow
LHC are made up of hundreds of pigments molecules bound to
A)
Proteins
done
clear
B)
Lipids
done
clear
C)
Carbohydrates
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
Number of energy using steps in glycolysis is lane
A)
One
done
clear
B)
Two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
Denitriflcation is carried out by
A)
Rhizobium
done
clear
B)
Thiobarillus
done
clear
C)
Nitrococcus
done
clear
D)
Rhodospirillum
done
clear
View Answer play_arrow
Water absorption by root hair is purely by
A)
Diffusion
done
clear
B)
Osmosis
done
clear
C)
Apoplast
done
clear
D)
Symplast
done
clear
View Answer play_arrow
Diffusion is a \[_{-}{{X}_{-}}\] process and is not dependent on \[_{-}{{Y}_{-}}\]. Select X and Y respectively
A)
Rapid, Pressure
done
clear
B)
Rapid Temperature
done
clear
C)
Slow, living system
done
clear
D)
Slow, gradict of concentration
done
clear
View Answer play_arrow
Which stage of cell division is also called as Diad of cells
A)
Telophase II
done
clear
B)
Telophase I
done
clear
C)
Anaphasel
done
clear
D)
Anaphase II
done
clear
View Answer play_arrow
The chromosome, with the centromere situated close to its end forming one extremely short, and one very long arm is
A)
Telocentric
done
clear
B)
Submetacentric
done
clear
C)
Acrocentric
done
clear
D)
Metacentric
done
clear
View Answer play_arrow
Each centriole is made up of___,_ evenly spaced peripheral fibrils of__ protein.
A)
9, Actin
done
clear
B)
18, Actin and myosin
done
clear
C)
27, Tubulin
done
clear
D)
9, Tubulin
done
clear
View Answer play_arrow
Find out the mismatched pair
A)
Mesophyll - ground tissue of leaf
done
clear
B)
Trichomes - Epidermal hair on stem
done
clear
C)
Radial - Xylem and Phloem are at same radius
done
clear
D)
Subsidiary cells - specialized epidermal cells
done
clear
View Answer play_arrow
Find incorrect match
A)
Monoecious - Cucurbits
done
clear
B)
Isogametes - Cladophora
done
clear
C)
Dioecious - Papaya
done
clear
D)
Unisexual flower - Sweet Potato
done
clear
View Answer play_arrow
Match the following
Column I (Organism) Column II (Mode of propagation) A. Potato I. Tuber B. Agave II. Leaf bud C. Ginger III. Bulbil D. Bryophyllum IV. Rhizome
A)
A-II B-IV C-I D-III
done
clear
B)
A-I B-III C-IV D-II
done
clear
C)
A-I B-IV C-II D-III
done
clear
D)
A-III B-I C-II D-IV
done
clear
View Answer play_arrow
"Terror of Begal"
A)
was introduced in India as an ornamental plant
done
clear
B)
propagates by vegetative means only
done
clear
C)
drains oxygen from water, which leads to death of fishes
done
clear
D)
more than one option is correct
done
clear
View Answer play_arrow
Which of the following plants does not show interflowing period?
A)
Peepal
done
clear
B)
Mango
done
clear
C)
Wheat
done
clear
D)
China Rose
done
clear
View Answer play_arrow
Chromosome number in meiocyte and gamete of drosophila respectively is
A)
18 and 9
done
clear
B)
8 and 4
done
clear
C)
20 and 10
done
clear
D)
12 and 6
done
clear
View Answer play_arrow
Which of the following represents male gametophyte
A)
Anther
done
clear
B)
Stamen
done
clear
C)
Pollen grain
done
clear
D)
Microsporangium
done
clear
View Answer play_arrow
In which of the following cases endosperm is completely consumed by the developing embryo?
A)
Ricinus
done
clear
B)
Coconut
done
clear
C)
Beans
done
clear
D)
Rice
done
clear
View Answer play_arrow
Which of the following is not the case of incomplete dominance?
A)
Flower colour of Mirabilis jalapa
done
clear
B)
Flower colour in shapdragon
done
clear
C)
Starch grain size in pea
done
clear
D)
Blood group in humans
done
clear
View Answer play_arrow
Read the following statements
A. Tall stature B. Overall feminine development C. Presence of barr body D. Development of breast E. Furrowed tongue
How many of the above characters belong to individual with genotype\[44+XXY\]?
A)
Two
done
clear
B)
Three
done
clear
C)
Four
done
clear
D)
Five
done
clear
View Answer play_arrow
Who United the knowledge of chromosomal segregation with Mendelian principles and called if the chromosomal theory of inheritance?
A)
Morgan
done
clear
B)
Henking
done
clear
C)
Sutton
done
clear
D)
John Collins
done
clear
View Answer play_arrow
Which of the following present is semen is used in forensic science?
A)
Litricacid
done
clear
B)
Fibrmogen
done
clear
C)
Lactic acid
done
clear
D)
Fructose
done
clear
View Answer play_arrow
What is the correct sequence in the process of formation of ovum?
A)
Oocyte, Oogonia, Ovum
done
clear
B)
Oogonia, Primary Oocyte, Secondary Oocyte, Ovum
done
clear
C)
Oogonia, Secondary oocyte, primary oocyte, ovum
done
clear
D)
Secondary oocyte, primary oocyte, oogonia, ovum
done
clear
View Answer play_arrow
Which of the following cells synthesise and secrete testicular, hormones called androgens?
A)
Sertoli cells
done
clear
B)
Interstitial cells
done
clear
C)
Leydig cells
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
If a female wants to postpone her mentrual cycle, she should take pills containing
A)
Estrogen
done
clear
B)
Progesterone
done
clear
C)
FSH
done
clear
D)
LH
done
clear
View Answer play_arrow
Which among the following undergoes meiosis-I, leading to the formation of haploid cells with 23 chromosomes?
A)
Spermatogonia
done
clear
B)
Secondary spermatocyte
done
clear
C)
Spermatid
done
clear
D)
Primary spermatocyte
done
clear
View Answer play_arrow
Home use kits for determining a woman's fertile period depend on the detection of a hormone in use this hormone is
A)
FSH
done
clear
B)
Progesterone
done
clear
C)
HCG
done
clear
D)
LH
done
clear
View Answer play_arrow
Vasa efferentia are the ductless leading from
A)
Epididymis to urethra
done
clear
B)
Testicular Lobules to rete testis
done
clear
C)
Rete testis to vas deferens
done
clear
D)
Vas deferens to epididymic
done
clear
View Answer play_arrow
Through which of the following duct milk is sucked out?
A)
Mammary lobes
done
clear
B)
Mammary duct
done
clear
C)
Lactiferous duct
done
clear
D)
Alveoli
done
clear
View Answer play_arrow
Choose the incorrect match/characteristic
A)
Hymen-Presence or absence of hymen is not a reliable indicator of virginity or sexual experience
done
clear
B)
Birth Canal - Cervical canal along with vagina
done
clear
C)
Castration - Choir boys often castrated to maintain high pitch voice
done
clear
D)
Uterus - Bicornuate in human
done
clear
View Answer play_arrow
Identify the odd one from the following
A)
Infundibulum
done
clear
B)
Isthmus
done
clear
C)
Ampulla
done
clear
D)
Labiamajora
done
clear
View Answer play_arrow
Which of the following is not the feature of DNA double helix?
A)
The backbone is constituted by sugar - phosphate and the base project outside
done
clear
B)
A purine always pairs with pyrimidine
done
clear
C)
The pitch of helix is \[3.4\text{ }nm\] and there are roughly 10 base pair in each turn
done
clear
D)
Plane of one base pair stacks over the other in double helix
done
clear
View Answer play_arrow
How many nucleosomes are present in an eubacterial DNA which contains 46200 bp?
A)
15708
done
clear
B)
462
done
clear
C)
Zero
done
clear
D)
231
done
clear
View Answer play_arrow
How many heavy, hybrid and light DNA are obtained respectively if \[^{15}N\] labeled 10 DNA molecules are allowed to replicate in \[{}^{14}N\] solution for three generations?
A)
0, 20, 60
done
clear
B)
10, 20, 50
done
clear
C)
20, 40, 60
done
clear
D)
60, 40, 20
done
clear
View Answer play_arrow
Identify the process marked as A, B and C
A)
A B C Replication Transcription Translation
done
clear
B)
A B C Translation Transcription Transduction
done
clear
C)
A B C Replication Translocation Transduction
done
clear
D)
A B C Replication Transformation Translocation
done
clear
View Answer play_arrow
Choose the incorrect matching:
A)
RNA polymerase III - tRNA, 5srRNA
done
clear
B)
Tailing of RNA- 200 - 300 adenylate residues added at 57 end
done
clear
C)
Splicing-Removal of introns
done
clear
D)
Methyl guanosine triphosphate - Added to 5' end of hn RNA
done
clear
View Answer play_arrow
The sequence of chromosome one of human genome was completed in ________.
A)
May 2006
done
clear
B)
April 2001
done
clear
C)
March 2005
done
clear
D)
May 2003
done
clear
View Answer play_arrow
Match the following Column I with Column II
Column-I (Variety) Column-II (Resistant to) A. PusaKomal (I) White rust B. PusaSawani (II) Aphids C. PusaSwarnim (III) Bacterial blight D. Pusa Gaurav (IV) Shoot and fruit borer
A)
A-III B-I C-IV D-II
done
clear
B)
A-I B-II C-III D-IV
done
clear
C)
A-II B-IV C-III D-I
done
clear
D)
A-III B-IV C-I D-II
done
clear
View Answer play_arrow
Which of considered as the most significant discovery of twentieth century?
A)
Cheese
done
clear
B)
Beverages
done
clear
C)
Antibiotics
done
clear
D)
STP
done
clear
View Answer play_arrow
Greater the BOD of waste water
A)
less is its polluting potential
done
clear
B)
more is its polluting potential
done
clear
C)
more is organic matter in it
done
clear
D)
more than one option is correct
done
clear
View Answer play_arrow
The majority of _____ used as bio-control agents are in the genus Nucleopolyhedrovirus.
A)
Retrovirus
done
clear
B)
Tobacco mosaic virus
done
clear
C)
Reovirus
done
clear
D)
Baculoviruses
done
clear
View Answer play_arrow
What does natural selection means?
A)
Better adaptability
done
clear
B)
Elimination of less adapted
done
clear
C)
Better survival
done
clear
D)
All of these
done
clear
View Answer play_arrow
All of the following statements are related to Hardyweinberg principle, except
A)
It gives geneticists a tool to determine when evolution is occurring
done
clear
B)
Sum total of the allelic frequencies of a gene is equal to one
done
clear
C)
Saltation causes speciation
done
clear
D)
Allelic frequencies in a large random mating population remains constant in absence of mutation and migration.
done
clear
View Answer play_arrow
To prove Oparin's Theory, miller created electric discharge in closed flask containing
A)
\[C{{H}_{4}},{{H}_{2}},N{{H}_{3}}\] and \[{{H}_{2}}O\] vapour at \[{{800}^{o}}C\]
done
clear
B)
\[N{{H}_{3}},C{{H}_{4}}\]and \[{{H}_{2}}\] at \[{{800}^{o}}C\]
done
clear
C)
\[N{{H}_{3}},{{H}_{2}},\] and \[C{{H}_{4}}\] at \[{{500}^{o}}C\]
done
clear
D)
\[C{{H}_{4}},N{{H}_{3}},{{H}_{2}}\] and \[{{H}_{2}}O\] vapour at \[{{500}^{o}}C\]
done
clear
View Answer play_arrow
Which of the following dinosaurs adapted the ability to run on two legs?
A)
Triceratops
done
clear
B)
Tyrannosaurus
done
clear
C)
Stegosaurus
done
clear
D)
Brachiosaurus
done
clear
View Answer play_arrow
Which of the following is richest in fossil?
A)
Basalt
done
clear
B)
Granite
done
clear
C)
Sedimentary rock
done
clear
D)
Lava
done
clear
View Answer play_arrow
The first mammals were like
A)
Birds
done
clear
B)
Lizards
done
clear
C)
Turtles
done
clear
D)
Shrews
done
clear
View Answer play_arrow
Find the odd one out with respect to evolution
A)
Seal's flipper
done
clear
B)
Bat's wing
done
clear
C)
Horse's foot
done
clear
D)
Butterfly's wings
done
clear
View Answer play_arrow
In which organ of the host are sickle shape sporozoites of plasmodium stored?
A)
Salivary glands
done
clear
B)
Intestime
done
clear
C)
Stomach
done
clear
D)
Human brain
done
clear
View Answer play_arrow
Elephantiasis is spread by
A)
Wuchereria bancrofti
done
clear
B)
Wuchereria malayi
done
clear
C)
Female culex
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
Mark the incorrect match with its test
A)
Dengue - Tourniquet test
done
clear
B)
Typhoid - Widal test
done
clear
C)
Scarlet fever - Gravidex test
done
clear
D)
HIV/AIDS - Western blot test
done
clear
View Answer play_arrow
Mark the odd one out with respect to infectious disease which does not get transmitted through food and water
A)
Typhoid
done
clear
B)
Pneumonia
done
clear
C)
Amoebiasis
done
clear
D)
Ascariasis
done
clear
View Answer play_arrow
Both light and heavy chains of immunglobulins are attached with each other through
A)
Ionic bonds
done
clear
B)
Co-ordinate bonds
done
clear
C)
Disulphide bonds
done
clear
D)
Ester bonds
done
clear
View Answer play_arrow
Most cells release histamine and serotonin during allergic response. This can be countered by administration of
A)
Cortisol
done
clear
B)
Thyroxine
done
clear
C)
Calcitriol
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following is a result of autoimmunity
A)
Diptheria
done
clear
B)
Polio
done
clear
C)
Rheumatoid arthritis
done
clear
D)
Hepatitis
done
clear
View Answer play_arrow
Which of the following set of disease is caused by viral pathogens?
A)
Amoebiasis, filariasis, typhoid, pneumonia
done
clear
B)
Diptheria, plague, enteric fever, ascariasis
done
clear
C)
Tuberculosis, chikungzmya, polio, dysentery
done
clear
D)
Hepatitis B, dengue, polio, Chicken pox
done
clear
View Answer play_arrow
A person bit by a snake was administered antivenin. These antivenin are
A)
Antibiotics
done
clear
B)
Preformed antibodies
done
clear
C)
Vaccine with attenuated pathogen
done
clear
D)
Antibodies synthesized by same person bitten by snake
done
clear
View Answer play_arrow
Find out the correct option from below statements about ecological succession
A. Hydrarch succession takes place in wet areas and the successional series progress from hydric to the mesic condition B. Xerarch succession takes place in dry areas and the successional series progress from xeric to mesic condition C. Primary succession begins in areas where natural biotic communities have been destroyed D. The establishment of a new biotic community is generally slow in secondary succession
A)
B & C
done
clear
B)
C & D
done
clear
C)
A & B
done
clear
D)
A & C
done
clear
View Answer play_arrow
Find the correct match with respect to example and type of population interaction
A)
Monarchy butterfly and birds - Competition
done
clear
B)
Sea anemone and clownflsh - Predation
done
clear
C)
Fishes and copepods - Parasitism
done
clear
D)
Mycorrhiza - Amensalism
done
clear
View Answer play_arrow

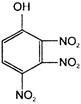
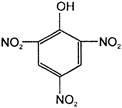
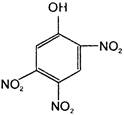
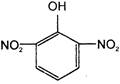
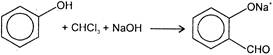 The electrophile involved in the above reaction is
The electrophile involved in the above reaction is