The dimensions of \[a\times b\] in the relation \[E=\frac{b-{{x}^{2}}}{at}\] where E is the energy, x is the displacement and t is time are
A)
\[M{{L}^{2}}T\]
done
clear
B)
\[{{M}^{-1}}{{L}^{2}}T\]
done
clear
C)
\[M{{L}^{2}}{{T}^{-2}}\]
done
clear
D)
\[ML{{T}^{-2}}\]
done
clear
View Answer play_arrow
A particle starting with certain initial velocity and uniform acceleration covers a distance of 12 m in first 3 seconds and a distance of 30 m in next 3 seconds. The initial velocity of the particle is
A)
\[3\,\,m{{s}^{-1}}\]
done
clear
B)
\[2.5\,\,m{{s}^{-1}}\]
done
clear
C)
\[2\,\,m{{s}^{-1}}\]
done
clear
D)
\[1\,\,\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
A particle undergoes simple harmonic motion having time period T. The time taken in 3/8th oscillation is
A)
\[\frac{3}{8}T\]
done
clear
B)
\[\frac{5}{8}T\]
done
clear
C)
\[\frac{5}{12}T\]
done
clear
D)
\[\frac{7}{12}T\]
done
clear
View Answer play_arrow
When current in a coil changes from 5 A to 2 A in 0.1 s, average voltage of 50 V is produced. The self-inductance of the coil is:
A)
6 H
done
clear
B)
0.67 H
done
clear
C)
3 H
done
clear
D)
1.67 H
done
clear
View Answer play_arrow
The given electrical network is equivalent to:
A)
OR gate
done
clear
B)
NOR gate
done
clear
C)
NOT gate
done
clear
D)
AND gate
done
clear
View Answer play_arrow
The work function of aluminium is 4.2 eV. If two photons each of energy 3.5 eV strike an electron of aluminium, then emission of electron will
A)
depend upon the density of the surface
done
clear
B)
possible
done
clear
C)
not possible
done
clear
D)
None of these
done
clear
View Answer play_arrow
A doubly ionised Li atom is excited from its ground state \[(n\,\,=\,\,1)\] to \[n\,\,=\,\,3\] state. The wavelengths of the spectral lines are given by \[{{\lambda }_{32,}}{{\lambda }_{31}}\]and\[{{\lambda }_{21}}\]. The ratio a \[{{\lambda }_{32}}{{\lambda }_{31}}\]and \[{{\lambda }_{21}}{{\lambda }_{31}}\]are, respectively
A)
8.1, 0.67
done
clear
B)
8.1, 1.2
done
clear
C)
6.4, 1.2
done
clear
D)
6.4, 0.67
done
clear
View Answer play_arrow
Two wires A and B of the same material, having radii in the ratio 1 : 2 and carry currents in the ratio 4 : 1. The ratio of drift speed of electrons in A and B is
A)
16 : 1
done
clear
B)
1 : 16
done
clear
C)
1 : 4
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
A stone is thrown with a velocity u making an angle \[\theta \] with the horizontal. The horizontal distance covered by its fall to ground is maximum when the angle \[\theta \] is equal to
A)
\[0{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[90{}^\circ \]
done
clear
View Answer play_arrow
An object undergoing SHM takes 0.5 s to travel from one point of zero velocity to the next such point. The distance between those points is 50 cm. The period, frequency and amplitude of the motion is
A)
1s, 1Hz, 25cm
done
clear
B)
2s, 1Hz, 50cm
done
clear
C)
1s, 2Hz, 25cm
done
clear
D)
2s, 2Hz, 50cm
done
clear
View Answer play_arrow
ABC is an equilateral triangle. Charges +q are placed at each comer as shown in fig. The electric intensity at centre O will be
A)
\[\frac{1}{4\pi {{\in }_{o}}}\frac{q}{r}\]
done
clear
B)
\[\frac{1}{4\pi {{\in }_{o}}}\frac{q}{{{r}^{2}}}\]
done
clear
C)
\[\frac{1}{4\pi {{\in }_{o}}}\frac{3q}{{{r}^{2}}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
The figure shows the path of a positively charged particle 1 through a rectangular region of uniform electric field as shown in the figure. What is the direction of electric field and the direction of particles 2, 3 and 4?
A)
Top, down, top, down
done
clear
B)
Top, down, down, top
done
clear
C)
Down, top, top, down
done
clear
D)
Down; top, down, down
done
clear
View Answer play_arrow
A circular disc A of radius r is made from an iron plate of thickness t and another circular disc B of radius 4r is made from an iron plate of thickness t/4. The relation between the moments of inertia \[{{I}_{A}}\]and \[{{I}_{B}}\] is
A)
\[{{I}_{A}}>{{I}_{B}}\]
done
clear
B)
\[{{I}_{A}}={{I}_{B}}\]
done
clear
C)
\[{{I}_{A}}<{{I}_{B}}\]
done
clear
D)
depends on the actual value of t and r
done
clear
View Answer play_arrow
The threshold frequency for a photosensitive metal is \[3.3\times {{10}^{14}}Hz\]. If light of frequency \[8.2\times {{10}^{14}}Hz\] is incident on this metal, the cut-off voltage for the photoelectric emission is nearly
A)
2 V
done
clear
B)
3 V
done
clear
C)
5 V
done
clear
D)
1 V
done
clear
View Answer play_arrow
A metallic bar is heated from \[0{}^\circ C\] to\[100{}^\circ C\]. The coefficient of linear expansion is \[{{10}^{-5}}{{K}^{-1}}\]. What will be the percentage increase in length?
A)
0.01%
done
clear
B)
0.1%
done
clear
C)
1%
done
clear
D)
10%
done
clear
View Answer play_arrow
A rough vertical board has an acceleration a along the horizontal so that a block of mass M pressing against it does not fall. The coefficient of friction between block and the board is
A)
\[>\frac{a}{g}\]
done
clear
B)
\[<\frac{g}{a}\]
done
clear
C)
\[=\frac{a}{g}\]
done
clear
D)
\[>\frac{g}{a}\]
done
clear
View Answer play_arrow
A light ray falls on a rectangular glass slab as shown. The index of refraction of the glass, if total internal reflection is to occur at the vertical face, is
A)
\[\sqrt{3/2}\]
done
clear
B)
\[\frac{(\sqrt{3}+1)}{2}\]
done
clear
C)
\[\frac{(\sqrt{2}+1)}{2}\]
done
clear
D)
\[\sqrt{5}/2\]
done
clear
View Answer play_arrow
Plates of area A are arranged as shown. The distance between each plate is d, the net capacitance is
A)
\[\frac{{{\varepsilon }_{0}}A}{d}\]
done
clear
B)
\[\frac{7{{\varepsilon }_{0}}A}{d}\]
done
clear
C)
\[\frac{6{{\varepsilon }_{0}}A}{d}\]
done
clear
D)
\[\frac{5{{\varepsilon }_{0}}A}{d}\]
done
clear
View Answer play_arrow
A plane wave of wavelength 6250 A is incident normally on a slit of width \[2\times {{10}^{-2}}\]cm. The width of the principal maximum on a screen distant 50 cm will be
A)
\[312.5\times {{10}^{-3}}\]cm
done
clear
B)
\[312.5\times {{10}^{-6}}\]m
done
clear
C)
\[312.5\times {{10}^{-3}}\]m
done
clear
D)
\[312.5\times {{10}^{-6}}\]cm
done
clear
View Answer play_arrow
The heat radiated per unit area in 1 hour by a furnace whose temperature is 3000 K is \[(\sigma =5.7\times {{10}^{-8}}W{{m}^{-2}}{{K}^{-4}})\]
A)
\[1.7\times {{10}^{10}}\]J
done
clear
B)
\[1.1\times {{10}^{12}}\]J
done
clear
C)
\[2.8\times {{10}^{8}}\]J
done
clear
D)
\[4.6\times {{10}^{6}}\]J
done
clear
View Answer play_arrow
A gun fires two bullets at \[60{}^\circ \] and \[30{}^\circ \] with horizontal. The bullets strike at some horizontal distance. The ratio of maximum height for the two bullets is in the ratio of
A)
2 : 1
done
clear
B)
3 : 1
done
clear
C)
4 : 1
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
A generator has an e.m.f. of 440 Volt and internal resistance of 400 Ohm. Its terminals are connected to a load of 4000 Ohm the voltage across the load is
A)
220 volt
done
clear
B)
440 volt
done
clear
C)
200 volt
done
clear
D)
400 volt
done
clear
View Answer play_arrow
Electric field inside a copper wire of length 10 metres, resistance 2 ohm connected to a 10 volt battery is
A)
\[1\,V{{M}^{-1}}\]
done
clear
B)
\[0.5\,V{{M}^{-1}}\]
done
clear
C)
\[10\,\,V{{M}^{-1}}\]
done
clear
D)
\[\,5\,V{{M}^{-1}}\]
done
clear
View Answer play_arrow
A large number of liquid drops each of radius r coalesce to from a single drop of radius R. The energy released in the process is converted into kinetic energy of the big drop so formed. The speed of the big drop is (given, surface tension of liquid T, density r)
A)
\[\sqrt{\frac{T}{p}\left( \frac{1}{r}-\frac{1}{R} \right)}\]
done
clear
B)
\[\sqrt{\frac{2T}{p}\left( \frac{1}{r}-\frac{1}{R} \right)}\]
done
clear
C)
\[\sqrt{\frac{4T}{p}\left( \frac{1}{r}-\frac{1}{R} \right)}\]
done
clear
D)
\[\sqrt{\frac{6T}{p}\left( \frac{1}{r}-\frac{1}{R} \right)}\]
done
clear
View Answer play_arrow
The path difference between the two waves : \[{{y}_{1}}={{a}_{1}}\sin \left( \omega t-\frac{2\pi x}{\lambda } \right)\]and\[{{y}_{2}}={{a}_{2}}\sin \left( \omega t-\frac{2\pi x}{\lambda }+\phi \right)\]will be
A)
\[\frac{2\pi }{\lambda }\phi \]
done
clear
B)
\[\frac{2\pi }{\lambda }\left( \phi -\frac{\pi }{2} \right)\]
done
clear
C)
\[\frac{\lambda }{2\pi }\phi \]
done
clear
D)
\[\frac{2\pi }{\lambda }\left( \phi +\frac{\pi }{2} \right)\]
done
clear
View Answer play_arrow
The diagram shows the energy levels for an electron in a certain atom. Which transition shown represents the emission of a photon with the most energy?
A)
IV
done
clear
B)
III
done
clear
C)
II
done
clear
D)
I
done
clear
View Answer play_arrow
During an adiabatic compression, 830 J of work is done on 2 moles of a diatomic ideal gas to reduce its volume by 50%. The change in its temperature is nearly:\[(R=8.3\,\,J{{K}^{-1}}mo{{l}^{-1}})\]
A)
40 K
done
clear
B)
33 K
done
clear
C)
20 K
done
clear
D)
14 K
done
clear
View Answer play_arrow
A body of mass 10 kg and velocity 10 m/s collides with a stationary body of mass 5 kg. After collision both bodies stick to each other, velocity of the bodies after collision will be
A)
\[\frac{3}{10}\,m/s\]
done
clear
B)
\[\frac{18}{3}\,m/s\]
done
clear
C)
\[\frac{9}{20}\,m/s\]
done
clear
D)
\[\frac{20}{3}\,m/s\]
done
clear
View Answer play_arrow
Two particles of mass m\[_{1}\]and m\[_{2}\](m\[_{1}\]>m\[_{2}\]) attract each other with a force inversely proportional to the square of the distance between them. If the particles are initially held at rest and then released, the centre of mass will
A)
move towards m\[_{1}\]
done
clear
B)
move towards m\[_{2}\]
done
clear
C)
remain at rest
done
clear
D)
Nothing can be said
done
clear
View Answer play_arrow
Which of the given graphs proves Newton's law of cooling?
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of these
done
clear
View Answer play_arrow
The r.m.s. velocity of oxygen molecule at \[16{}^\circ C\] is 474 m/sec. The r.m.s. velocity in m/s of hydrogen molecule at \[127{}^\circ C\]is
A)
1603
done
clear
B)
1896
done
clear
C)
2230.59
done
clear
D)
2730
done
clear
View Answer play_arrow
When the rms voltages \[{{V}_{L}},\] \[{{V}_{C}}\] and \[{{V}_{R}}\]are measured respectively across the inductor L, the capacitor C and the resistor R in a series LCR circuit connected to an AC source, it is found that the ratio\[{{V}_{L}}:{{V}_{C}}:{{V}_{R}}=1:2:3.\] If the rms voltage of the AC sources is 100 V, the \[{{V}_{R}}\] is close to:
A)
50 V
done
clear
B)
70 V
done
clear
C)
90 V
done
clear
D)
100 V
done
clear
View Answer play_arrow
The oscillating electric and magnetic field vectors of electromagnetic wave are oriented along
A)
the same direction and in phase
done
clear
B)
the same direction but have a phase difference of\[90{}^\circ \]
done
clear
C)
mutually perpendicular directions and are in same phase
done
clear
D)
mutually perpendicular directions but has a phase difference of \[90{}^\circ \]
done
clear
View Answer play_arrow
The gravitational field in a region is given by \[\to \]\[g=5N/kg\,\hat{i}+12N/kg\,\hat{j}.\] The change in the gravitational potential energy of a particle of mass 1 kg when it is taken from the origin to a \[\left( 7\,m,-3\,m \right)\] is:
A)
71 J
done
clear
B)
\[13\sqrt{58}\]J
done
clear
C)
\[-71\]J
done
clear
D)
1 J
done
clear
View Answer play_arrow
The counting rate observed from a radioactive source at t = 0 was 1600 count \[{{S}^{-1}}\], and t = 8 s, it was 100 counts \[{{S}^{-1}}\]. The counting rate observed as counts \[{{S}^{-1}}\]at t = 6s will be
A)
250
done
clear
B)
400
done
clear
C)
300
done
clear
D)
200
done
clear
View Answer play_arrow
At \[0{}^\circ \]K which of the following properties of a gas will be zero?
A)
Kinetic energy
done
clear
B)
Potential energy
done
clear
C)
Vibrational energy
done
clear
D)
Density
done
clear
View Answer play_arrow
A galvanometer coil has a resistance of\[{{\lambda }_{21}}=\frac{4}{3}\]and gives full scale deflection for a current of 4 mA. To convert it to an ammeter of range 0 to 6 A
A)
\[10\,\,m\Omega \] resistance is to be connected in parallel to the galvanometer
done
clear
B)
\[10\,\,m\Omega \] resistance is to be connected in series with the galvanometer
done
clear
C)
\[0.1\,\,\Omega \] resistance is to be connected in parallel to the galvanometer
done
clear
D)
\[0.1\,\,\Omega \] resistance is to be connected in series with the galvanometer
done
clear
View Answer play_arrow
A uniform rod of mass m, length \[\ell \], area of cross- section A has Young's modulus Y. If it is hanged vertically, elongation under its own weight will be
A)
\[\frac{mgl}{2AY}\]
done
clear
B)
\[\frac{2mgl}{AY}\]
done
clear
C)
\[\frac{mgl}{AY}\]
done
clear
D)
\[\frac{mgY}{A\ell }\]
done
clear
View Answer play_arrow
The magnifying power of a telescope is 9. When it is adjusted for parallel rays, the distance between the objective and the eye piece is found to be 20 cm. The focal length of lenses are
A)
18 cm, 2 cm
done
clear
B)
11 cm, 9 cm
done
clear
C)
10 cm, 10 cm
done
clear
D)
15 cm, 5 cm
done
clear
View Answer play_arrow
If two soap bubbles of different radii are connected by a tube. Then
A)
air flows from the smaller bubble to the bigger bubble
done
clear
B)
air flows from bigger bubble to the smaller bubble till the sizes are interchanged
done
clear
C)
air flows from the bigger bubble to the smaller bubble till the sizes become equal
done
clear
D)
there is no flow of air.
done
clear
View Answer play_arrow
In a transistor
A)
both emitter and collector have same length
done
clear
B)
length of emitter is greater than that of collector
done
clear
C)
length of collector is greater than that of emitter
done
clear
D)
any one of emitter and collector can have greater length
done
clear
View Answer play_arrow
A brass scale of a barometer gives correct reading at \[0{}^\circ C.\] 0 The barometer reads 75 cm at \[27{}^\circ C.\] the atmospheric pressure at \[0{}^\circ C\]is
A)
74.20 cm
done
clear
B)
74.62 cm
done
clear
C)
74.92 cm
done
clear
D)
75.04 cm
done
clear
View Answer play_arrow
The total length of a sonometer wire between fixed ends is 110 cm. Two bridges are placed to divide the length of wire in ratio 6 : 3 : 2. The tension in the wire is 400 N and the mass per unit length is 0.01 kg/m. What is the minimum common frequency with which three parts can vibrate?
A)
1100 Hz
done
clear
B)
1000 Hz
done
clear
C)
166 Hz
done
clear
D)
100 Hz
done
clear
View Answer play_arrow
For the velocity time graph shown in the figure below the distance covered by the body in the last two seconds of its motion is what fraction of the total distance travelled by it in all the seven seconds?
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{1}{4}\]
done
clear
C)
\[\frac{2}{3}\]
done
clear
D)
\[\frac{1}{3}\]
done
clear
View Answer play_arrow
A 25 cm long solenoid has radius 2 cm and 500 total number of turns. It carries a current of 15 A. If it is equivalent to a magnet of the same size and magnetization \[\vec{M}\] (magnetic moment/ volume), then \[\left| \overrightarrow{M} \right|\] is
A)
\[30000\pi A{{m}^{-1}}\]
done
clear
B)
\[3\pi A{{m}^{-1}}\]
done
clear
C)
\[30000A{{m}^{-1}}\]
done
clear
D)
\[300A{{m}^{-1}}\]
done
clear
View Answer play_arrow
The enolic form of a acetone contains
A)
9 sigma bonds, 1 pi bond and 2 lone pairs
done
clear
B)
8 sigma bonds, 2 pi bonds and 2 lone pairs
done
clear
C)
10 sigma bonds, 1 pi bond and 1 lone pair
done
clear
D)
9 sigma bonds, 2 pi bonds and 1 lone pair
done
clear
View Answer play_arrow
Following reaction occurrs in an automobile \[2{{C}_{8}}{{H}_{18}}(g)+25{{O}_{2\,\,}}\,\,(g)\to \] \[16C{{O}_{2}}(g)+18{{H}_{2\,\,}}O(g)\] The sign of \[\Delta H\], \[\Delta S\] and \[\Delta G\]would be
A)
\[+,\]\[-,\]\[+\]
done
clear
B)
\[-,\]\[+,\]\[-\]
done
clear
C)
\[-,\]\[+,\]\[+\]
done
clear
D)
\[+,\]\[+,\]\[-\]
done
clear
View Answer play_arrow
What is Z in the following sequence of reactions? \[Z\xrightarrow{PC{{I}_{5}}}X\xrightarrow{ALC.KOH}Y\xrightarrow[(ii){{H}_{2}}O;\,boil]{(i)\,Conc.\,{{H}_{2}}S{{O}_{4}}}Z\]
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{3}}CH(OH)C{{H}_{3}}\]
done
clear
C)
\[{{(C{{H}_{3}}C{{H}_{2}})}_{2}}CHOH\]
done
clear
D)
\[C{{H}_{3}}CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Complexes formed in the following methods are
I. Mond's process for purification of nickel. II. Removal of lead poisoning from the body. III. Cyanide process for extraction of silver. IV. Froth flotation process for separation of ZnS from galena ore by using depressant.
A)
I - \[\text{Ni(CO}{{\text{)}}_{\text{4}\,}}\] II - \[[\text{Pb}{{(EDTA]}^{2-}}\] III - \[{{\text{ }\!\![\!\!\text{ Ag(CN}{{\text{)}}_{\text{2}}}\text{ }\!\!]\!\!\text{ }}^{-}}\] IV - \[\text{ }\!\![\!\!\text{ Zn(CN}{{\text{)}}_{\text{2}}}\text{ }\!\!]\!\!\text{ }\]
done
clear
B)
I - \[\text{Ni(CO}{{\text{)}}_{\text{4}\,}}\] II - \[\text{ }\!\![\!\!\text{ Pb(EDTA}{{]}^{2-}}\] III - \[{{\text{ }\!\![\!\!\text{ Ag(CN}{{\text{)}}_{\text{2}}}\text{ }\!\!]\!\!\text{ }}^{-}}\] IV - \[{{\text{ }\!\![\!\!\text{ Zn(CN}{{\text{)}}_{\text{4}}}\text{ }\!\!]\!\!\text{ }}^{2-}}\]
done
clear
C)
I - \[\text{Ni(CO}{{\text{)}}_{\text{6}\,}}\] II - \[\text{ }\!\![\!\!\text{ Pb(EDTA}{{]}^{4-}}\] III - \[{{[\text{Ag(CN}{{\text{)}}_{\text{2}}}\text{ }\!\!]\!\!\text{ }}^{-}}\] IV - \[{{\text{ }\!\![\!\!\text{ Zn(CN}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{4-}}\]
done
clear
D)
I - \[\text{Ni(CO}{{\text{)}}_{\text{4}}}\] II - \[\text{ }\!\![\!\!\text{ Pb(EDTA}{{]}^{2-}}\] III - \[{{\text{ }\!\![\!\!\text{ Ag(CN}{{\text{)}}_{\text{4}}}\text{ }\!\!]\!\!\text{ }}^{3-}}\] IV - \[{{\text{ }\!\![\!\!\text{ Zn(CN}{{\text{)}}_{\text{4}}}\text{ }\!\!]\!\!\text{ }}^{2-}}\]
done
clear
View Answer play_arrow
An example of electrophilic substitution reaction is:
A)
Chlorination of methane
done
clear
B)
Conversion of methyl chloride to methyl alcohol
done
clear
C)
Nitration of benzene
done
clear
D)
Formation of ethylene from ethyl alcohol.
done
clear
View Answer play_arrow
Structurally biodegradable detergents, should contain
A)
normal alkyl chain
done
clear
B)
branched alkyl chain
done
clear
C)
phenyl side chain
done
clear
D)
cyclohexyl side chain
done
clear
View Answer play_arrow
Ozone hole refers to:
A)
Increase in concentration of ozone
done
clear
B)
Hole in ozone layer
done
clear
C)
Reduction in thickness of ozone layer in troposphere
done
clear
D)
Reduction in thickness of ozone layer in Stratosphere
done
clear
View Answer play_arrow
Which of the following does not represent the correct order of the properties indicated?
A)
\[N{{i}^{2+}}>C{{r}^{2+}}>F{{e}^{2+}}>M{{n}^{2+}}\]
done
clear
B)
\[Sc>Ti>Cr>Mn\]
done
clear
C)
\[M{{n}^{2+}}>N{{i}^{2+}}<C{{o}^{2+}}<F{{e}^{2+}}\] (unpaired electron)
done
clear
D)
\[F{{e}^{2+}}>C{{o}^{2+}}>N{{i}^{2+}}>C{{u}^{2+}}\] (unpaired electron)
done
clear
View Answer play_arrow
Match the columns
Column - I Column - II \[{{C}_{2}}{{H}_{6}}\xrightarrow{C{{l}_{2}}/UV\,\,light}{{C}_{2}}{{H}_{5}}Cl\] (p) Finkelstein reaction \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\xrightarrow[273-278K]{NaN{{O}_{2}}+HCL/C{{u}_{2}}C{{l}_{2}}}{{C}_{6}}{{H}_{5}}Cl\] (q) Free radical substitution \[C{{H}_{3}}Cl+NaI\to C{{H}_{3}}I+NaCl\] (r) Swarts reaction \[C{{H}_{3}}-Br+AgF\to C{{H}_{3}}F+AgBr\] (s) Sandmeyer?s reaction
A)
A\[\to \](q), B\[\to \](s), C\[\to \](p), D\[\to \](r)
done
clear
B)
A\[\to \](q), B\[\to \](r), C\[\to \](p), D\[\to \](s)
done
clear
C)
A\[\to \](r), B\[\to \](p), C\[\to \](s), D\[\to \](q)
done
clear
D)
A\[\to \](s), B\[\to \](r), C\[\to \](p), D\[\to \](q)
done
clear
View Answer play_arrow
The electrons, identified by quantum numbers n and 1(i) n = 4, 1 = 1 (ii) n= 4 , 1 = 0 (iii) n = 3, 1 = 2 (iv) n = 3, 1 = 1 can be placed in order of increasing energy, from the lowest to highest, as
A)
(iv)<(ii)<(iii)<(i)
done
clear
B)
(ii)<(iv)<(i)<(iii)
done
clear
C)
(i)<(iii)<(ii)<(iv)
done
clear
D)
(iii)<(i)<(iv)<(ii)
done
clear
View Answer play_arrow
In an adiabatic process, no transfer of heat takes place between system and surroundings. Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following.
A)
\[q=0,\]\[\Delta T\ne 0,\]\[w=0\]
done
clear
B)
\[q\ne 0,\]\[\Delta T=0,\]\[w=0\]
done
clear
C)
\[q=0,\]\[\Delta T=0,\]\[w=0\]
done
clear
D)
\[q=0,\]\[\Delta T<0,\]\[w\ne 0\]
done
clear
View Answer play_arrow
In which of the following cases, the stability of two oxidation states is correctly represented
A)
\[T{{i}^{3+}}>T{{i}^{4+}}\]
done
clear
B)
\[M{{n}^{2+}}>M{{n}^{3+}}\]
done
clear
C)
\[F{{e}^{2+}}>F{{e}^{3+}}\]
done
clear
D)
\[C{{u}^{+}}>C{{u}^{2+}}\]
done
clear
View Answer play_arrow
Which is not the disproportionation reaction?
A)
\[3{{H}_{3}}P{{O}_{2}}\to 2{{H}_{3}}P{{O}_{2}}+P{{H}_{3}}\]
done
clear
B)
\[HCHO+O{{H}^{-}}\to HCO{{O}^{-}}+C{{H}_{3}}OH\]
done
clear
C)
\[N{{H}_{4}}N{{O}_{3}}\to {{N}_{2}}O+2{{H}_{2}}O\]
done
clear
D)
\[3C{{l}_{2}}+6O{{H}^{-}}\to 5C{{l}^{-}}+Cl{{O}^{-}}_{3}+3{{H}_{2}}O\]
done
clear
View Answer play_arrow
The major organic product in the reaction, \[C{{H}_{3}}-O-CH{{(C{{H}_{3}})}_{2}}+HI\to \]Product is
A)
\[IC{{H}_{2}}OCH{{(C{{H}_{3}})}_{2}}\]
done
clear
B)
\[\begin{align} & C{{H}_{3}}OC{{(C{{H}_{3}})}_{2}} \\ & | \\ & I \\ \end{align}\]
done
clear
C)
\[C{{H}_{3}}I+{{(C{{H}_{3}})}_{2}}CHOH\]
done
clear
D)
\[C{{H}_{3}}OH+{{(C{{H}_{3}})}_{2}}CHI\]
done
clear
View Answer play_arrow
The rate constant of a zero order reaction is \[2.0\times {{10}^{-2}}\,\text{mol}\,\,{{L}^{-1}}{{s}^{-1}}\]. If the concentration of the reactant after 25 seconds is 0.5 M. What is the initial concentration?
A)
0.5 M
done
clear
B)
1.25 M
done
clear
C)
12.5 M
done
clear
D)
1.0 M
done
clear
View Answer play_arrow
The correct order of solubility in water for He, Ne, Ar, Kr, Xe is
A)
He > Ne > Ar > Kr > Xe
done
clear
B)
Xe > Kr > Ar > Ne > He
done
clear
C)
Ne > Ar > Kr > He > Xe
done
clear
D)
Ar > Ne > He > Kr > Xe
done
clear
View Answer play_arrow
The de Broglie wavelength of a car of mass 1000 kg and velocity 3 6 km/hr is:
A)
\[6.626\times {{10}^{-34}}m\]
done
clear
B)
\[6.626\times {{10}^{-38}}m\]
done
clear
C)
\[6.626\times {{10}^{-31}}m\]
done
clear
D)
\[6.626\times {{10}^{-30}}m\]
done
clear
View Answer play_arrow
The correct IUPAC name for
A)
5 - Methyl-4 -(1?-2'-dimethylpropyl) heptane
done
clear
B)
3-Methyl-4 -(1?, 2?-dimethyl propyl) heptane
done
clear
C)
2, 3, 5-Trimethyl-4 -propyl heptane
done
clear
D)
4 -Propyl-2, 3, 5-trimethylpeptane
done
clear
View Answer play_arrow
Which of the following shows iso-structural species?
A)
\[NH_{4}^{+}\] and \[NH_{2}^{-}\]
done
clear
B)
\[CH_{3}^{-}\] and \[CH_{3}^{+}\]
done
clear
C)
\[SO_{4}^{2-}\],\[PO_{4}^{3-}\] and \[{{[B{{F}_{4}}]}^{-}}\]
done
clear
D)
\[NH_{4}^{+}\] and \[N{{H}_{3}}\]
done
clear
View Answer play_arrow
When dihydroxyacetone reacts with \[HI{{O}_{4}}\], the product is/are :
A)
HCHO
done
clear
B)
HCOOH
done
clear
C)
HCHO and HCOOH
done
clear
D)
HCHO and \[C{{O}_{2}}\]
done
clear
View Answer play_arrow
Consider the reactions
[A] \[{{H}_{2}}{{O}_{2}}+2HI\to {{I}_{2}}+2{{H}_{2}}O\] [B] \[HOCL+{{H}_{2}}{{O}_{2}}\to {{H}_{3}}{{O}^{+}}+C{{l}^{-}}+{{O}_{2}}\] Which of the following statements is correct about\[{{H}_{2}}{{O}_{2}}\], with reference to these reactions? Hydrogen peroxide is ____.
A)
an oxidising agent in both [A] and [B]
done
clear
B)
an oxidising agent in [A] and reducing agent in [B]
done
clear
C)
a reducing agent in [A] and oxidising agent in [B]
done
clear
D)
a reducing agent in both [A] and [B]
done
clear
View Answer play_arrow
A certain compound (X) when treated with copper sulphate solution yields a brown precipitate. On adding hypo solution, the precipitate turns white. The compound is
A)
\[{{K}_{2}}C{{O}_{3}}\]
done
clear
B)
KI
done
clear
C)
KBr
done
clear
D)
\[{{K}_{3}}P{{O}_{4}}\]
done
clear
View Answer play_arrow
Consider the following reactions:
(i) \[{{H}^{+}}(aq)+O{{H}^{-}}(aq)\to {{H}_{2}}O(1),\]\[\Delta H=-{{X}_{1}}\,kJ\,mo{{l}^{-1}}\] (ii) \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\to {{H}_{2}}O(1)\],\[\Delta H=-{{X}_{2}}\,kJ\,mo{{l}^{-1}}\] (iii) \[C{{O}_{2}}(g)+{{H}_{2}}(g)\to CO(g)+{{H}_{2}}O,\]\[\Delta H=-{{X}_{3}}kJ\,mo{{l}^{-1}}\] (iv) \[{{C}_{2}}{{H}_{2}}(g)+\frac{5}{2}{{O}_{2}}(g)\to 2C{{O}_{2}}(g)+{{H}_{2}}O(1),\]\[\Delta H=+\,\,4{{X}_{4}}kJ\,mo{{l}^{-1}}\]
Enthalpy of formation of \[{{H}_{2}}O\] is
A)
\[+\,{{X}_{3}}\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[-\,{{X}_{4}}\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[+\,{{X}_{1}}\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-\,{{X}_{2}}\,kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
Which of the following possesses a sp-carbon in its structure ?
A)
\[C{{H}_{2}}=CCl\,-CH=C{{H}_{2}}\]
done
clear
B)
\[CC{{l}_{2}}\,=\,CC{{l}_{2}}\]
done
clear
C)
\[C{{H}_{2}}=C=C{{H}_{2}}\]
done
clear
D)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Aballoon has maximum capacity of 20 L. At one atmospheric pressure 10 L of air is filled in the balloon. It will burst when pressure is (assuming isothermal condition)
A)
\[>0.5\,atm\]
done
clear
B)
\[<0.5\,atm\]
done
clear
C)
\[=0.5\,atm\]
done
clear
D)
\[\ge \,0.5\,atm\]
done
clear
View Answer play_arrow
Which one of the following complexes will have four different isomers?
A)
\[[Co{{(en)}_{2}}C{{l}_{2}}]\,Cl\]
done
clear
B)
\[[Co(en){{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\,Cl\]
done
clear
C)
\[[Co{{(PP{{h}_{3}})}_{2}}C{{l}_{2}}]\,Cl\]
done
clear
D)
\[[Co{{(en)}_{3}}]\,C{{l}_{3}}\]
done
clear
View Answer play_arrow
Solubility product of a salt AB is \[1\,\,\times \,\,{{10}^{-8}}\] in a solution in which the concentration of \[{{A}^{+}}\]ions is\[{{10}^{-3}}\,\,M\]. The salt will precipitate when the concentration of \[{{B}^{-}}\] ions is kept
A)
between \[{{10}^{-8}}\] M to \[{{10}^{-7}}\]M
done
clear
B)
between \[{{10}^{-7}}\] M to \[{{10}^{-8}}\]M
done
clear
C)
\[\,>\,{{10}^{-5}}\]M
done
clear
D)
\[\,<\,{{10}^{-8}}\]M
done
clear
View Answer play_arrow
Sucrose in water is dextro-rotatory, \[{{[\alpha ]}_{D}}=66.4{}^\circ .\]When boiled with dilute HCl, the solution becomes leavo-rotatory, \[{{[\alpha ]}_{D}}=-\,\,20{}^\circ .\] In this process the sucrose molecule breaks into
A)
L-glucose + D-fructose
done
clear
B)
L-glucose + L-fructose
done
clear
C)
D-glucose + D-fructose
done
clear
D)
D-glucose + L-fructose
done
clear
View Answer play_arrow
Select correct statement(s).
A)
Cyanamide ion \[(C{{N}_{2}}^{2-})\] is isoelectronic with\[C{{O}_{2}}\] and has the same linear structure
done
clear
B)
\[M{{g}_{2}}{{C}_{3}}\] reacts with water to form propyne
done
clear
C)
\[Ca{{C}_{2}}\] has NaCl type lattice
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which of the following statement is not true about secondary structure of protein?
A)
The alpha helix, beta pleated sheet and beta turns are examples of secondary structure of protein.
done
clear
B)
The ability of peptide bonds to form intramolecular hydrogen bonds is important to secondary structure.
done
clear
C)
The steric influence of amino acid residues is important to secondary structure.
done
clear
D)
The hydrophilic/ hydrophobic character of amino acid residues is important to secondary structure.
done
clear
View Answer play_arrow
Vapour pressure of benzene at \[30{}^\circ C\] is 121.8 mm. When 15 g of a non-volatile solute is dissolved in 250 g of benzene its vapour pressure decreased to 120.2mm. The molecular weight of the solute (Mo. wt. of solvent = 78)
A)
356.2
done
clear
B)
456.8
done
clear
C)
530.1
done
clear
D)
656.7
done
clear
View Answer play_arrow
Which of the following statements is not true?
A)
\[N{{O}_{2}}\] can be prepared by heating \[Pb{{(N{{O}_{3}})}_{2}}\]
done
clear
B)
\[N{{O}_{2}}\] is red - brown gas
done
clear
C)
\[N{{O}_{2}}\] is diamagnetic
done
clear
D)
\[N{{O}_{2}}\] readily dimerises to \[{{N}_{2}}{{O}_{4}}\]
done
clear
View Answer play_arrow
For reaction \[a\,A\to x\,P\], when [A] =2.2 mM, the rate was found to be \[2.4\,\,mM{{s}^{-1}}\]. On reduce concentration of A to half, the rate changes to. The order of reaction with respect to A is:
A)
1.5
done
clear
B)
2.0
done
clear
C)
2.5
done
clear
D)
3.0
done
clear
View Answer play_arrow
The tendency of \[B{{F}_{3'}}\,BC{{l}_{3}}\] and \[BB{{r}_{3}}\] to behave as Lewis acid decreases in the sequence:
A)
\[BC{{l}_{3}}\,\,>\,\,B{{F}_{3}}\,\,>\,\,BB{{r}_{3}}\]
done
clear
B)
\[BB{{r}_{3}}\,\,>\,\,BC{{l}_{3}}\,\,>\,\,B{{F}_{3}}\]
done
clear
C)
\[BB{{r}_{3}}\,\,>\,\,B{{F}_{3}}\,\,>\,\,BC{{l}_{3}}\]
done
clear
D)
\[B{{F}_{3}}\,\,>\,\,BC{{l}_{3}}\,\,>\,\,BB{{r}_{3}}\]
done
clear
View Answer play_arrow
If 'a' stands for the edge length of the cubic systems : simple cubic, body centred cubic and face centred cubic, then the ratio of radii of the spheres in these systems will be respectively,
A)
\[\frac{1}{2}a\,\, & :\frac{\sqrt{3}}{4}a\,\,:\frac{1}{2\sqrt{2}}a\]
done
clear
B)
\[\frac{1}{2}a\,\, & :\,\sqrt{3}a:\frac{1}{\sqrt{2}}a\]
done
clear
C)
\[\frac{1}{2}a\,\, & :\,\frac{\sqrt{3}}{2}a\,\,:\frac{\sqrt{3}}{2}a\]
done
clear
D)
\[1a:\sqrt{3}a:\sqrt{2}a\]
done
clear
View Answer play_arrow
Which of the following is correct order of acidity?
A)
\[HCOOH>C{{H}_{3}}COOH>ClC{{H}_{2}}COOH\]\[>{{C}_{2}}{{H}_{5}}COOH\]
done
clear
B)
\[ClC{{H}_{2}}COOH>HCOOH>C{{H}_{3}}COOH\]\[>{{C}_{2}}{{H}_{5}}COOH\]
done
clear
C)
\[C{{H}_{3}}COOH>HCOOH>ClC{{H}_{2}}COOH\]\[>{{C}_{2}}{{H}_{5}}COOH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}COOH>C{{H}_{3}}COOH>HCOOH\]\[>ClC{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
Penicillin is:
A)
Analgesic
done
clear
B)
Antipyretic
done
clear
C)
Antimalarial
done
clear
D)
Antibiotic
done
clear
View Answer play_arrow
Decomposition of \[{{H}_{2}}{{O}_{2}}\] follows a first order reaction. In fifty minutes the concentration of \[{{H}_{2}}{{O}_{2}}\] decreases from 0.5 to 0.125 M in one such decomposition. When the concentration of \[{{H}_{2}}{{O}_{2}}\]reaches 0.05 M, the rate of formation of \[{{O}_{2}}\] will be:
A)
\[2.66\,L\,\,{{\min }^{-1}}\,\text{at}\,\,\text{STP}\]
done
clear
B)
\[1.34\times {{10}^{-2\,}}\,\text{mol}\,\,{{\min }^{-1}}\]
done
clear
C)
\[6.96\times {{10}^{-2}}\,\text{mol}\,\,{{\min }^{-1}}\]
done
clear
D)
\[6.93\times {{10}^{-4}}\,\text{mol}\,\,{{\min }^{-1}}\]
done
clear
View Answer play_arrow
Which of the following reactions can produce aniline as main product?
A)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}+Zn/KOH\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}+Zn/N{{H}_{4}}Cl\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}+Li/Al{{H}_{4}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}+Zn/HCl\]
done
clear
View Answer play_arrow
What is the standard reduction potential \[(E{}^\circ )\] for \[F{{e}^{3+}}\to Fe\]? Given that: \[F{{e}^{2+}}2{{e}^{-}}\to Fe;\,\,\,\,\,E{}^\circ {{\,}_{F{{e}^{2+}}/Fe}}=-0.47\,V\] \[F{{e}^{3+}}+{{e}^{-}}\to F{{e}^{2+}};\,\,E{{{}^\circ }_{F{{e}^{3+}}/F{{e}^{2+}}}}=+0.77\,V\]
A)
\[-\,\,0.057\,\,V\]
done
clear
B)
\[+\,\,0.057\,\,V\]
done
clear
C)
\[+\,\,0.30\,\,V\]
done
clear
D)
\[-\,\,0.30\,\,V\]
done
clear
View Answer play_arrow
Which does not exist?
A)
\[{{\text{ }\!\![\!\!\text{ SiC}{{\text{l}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{2-}}\]
done
clear
B)
\[{{[Ge{{F}_{6}}]}^{2-}}\]
done
clear
C)
\[{{[CC{{l}_{6}}]}^{2-}}\]
done
clear
D)
\[{{[SnC{{l}_{6}}]}^{2-}}\]
done
clear
View Answer play_arrow
In which case, van't Hoff factor i remains unchanged?
A)
\[PtC{{l}_{4}}\] reacts with aq. KCl
done
clear
B)
aq.\[ZnC{{l}_{2}}\] reacts with aq. \[N{{H}_{3}}\]
done
clear
C)
aq.\[FeC{{l}_{3}}\] reacts with aq. \[{{K}_{4}}{{[Fe(CN)]}_{6}}\]
done
clear
D)
\[KMn{{O}_{4}}\] reduced to \[Mn{{O}_{2}}\] in alkaline Medium
done
clear
View Answer play_arrow
The unit of equivalent conductivity is
A)
\[S\,\,c{{m}^{-2}}\]
done
clear
B)
\[ohm\,\,c{{m}^{2}}\] (g equivalent)
done
clear
C)
\[ohm\,\,cm\]
done
clear
D)
\[oh{{m}^{-1}}\,\,c{{m}^{2}}{{(g\,\,equivalent)}^{-1}}\]
done
clear
View Answer play_arrow
Perlon is:
A)
Terylene
done
clear
B)
Rubber
done
clear
C)
Nylon\[-\,\,6\]
done
clear
D)
Polyester
done
clear
View Answer play_arrow
Tertiary nitro compounds do not tautomerise because
A)
there is no double bond.
done
clear
B)
there is no a-hydrogen.
done
clear
C)
oxygen is more electronegative thanhydrogen.
done
clear
D)
all of the above.
done
clear
View Answer play_arrow
Choose correct option w.r.t origin and position Of meristem responsible for the regeneration of parts removed by the grazing herbivores.
A)
Origin - Secondary Position - Lateral
done
clear
B)
Origin - Primary Position - A Pical
done
clear
C)
Origin - Secondary Position - A Pical
done
clear
D)
Origin - Primary Position - Intercalated
done
clear
View Answer play_arrow
Which of the following changes occur in diaphragm and intercostal muscles when expiration of air takes place?
A)
External intercostal muscles relax and diaphragm contracts
done
clear
B)
External intercostal muscles contract and diaphragm relaxes
done
clear
C)
External intercostal muscles and diaphragm relax
done
clear
D)
External intercostal muscles and diaphragm Contract
done
clear
View Answer play_arrow
Which one of the following is a long day plant?
A)
Bajra
done
clear
B)
Soyabean
done
clear
C)
Tobacco
done
clear
D)
Wheat
done
clear
View Answer play_arrow
Life cycle of Ulothrix is shown in the diagram. The correct policy levels at the four stages A, B, C and D are:
A)
A : n B : n C : 2n D : n
done
clear
B)
A : n B : n C : 2n D : 2n
done
clear
C)
A : 2n B : n C : 2n D : n
done
clear
D)
A : n B : n C : n D : n
done
clear
View Answer play_arrow
Connecting link between glycolysis and Krebs cycle is/before entering Krebs cycle pyruvate is changed to
A)
oxaloacetate
done
clear
B)
phosphoenol pyruvate
done
clear
C)
pyruvate
done
clear
D)
acetyl CoA
done
clear
View Answer play_arrow
Differentiation of shoot is controlled by
A)
high gibberellin : cytokinin ratio
done
clear
B)
high auxin : cytokinin ratio
done
clear
C)
high cytokinin : auxin ratio
done
clear
D)
high gibberellin : auxin ratio
done
clear
View Answer play_arrow
Match Column-I with Column-II and select the correct option from the codes given below.
Column-I Column-II A. Disintegration of nuclear membrane (i) Anaphase B. Appearance of nucleolus (ii) Prophase C. Division of centrometer (iii) Telophase D. Replication of DNA (iv) S - Phase
A)
A\[\to \](ii), B\[\to \](iii), C\[\to \](i), D\[\to \](iv)
done
clear
B)
A\[\to \](ii), B\[\to \](iii), C\[\to \](iv), D\[\to \](i)
done
clear
C)
A\[\to \](iii), B\[\to \](ii), C\[\to \](i), D\[\to \](iv)
done
clear
D)
A\[\to \](iii), B\[\to \](ii), C\[\to \](iv), D\[\to \](i)
done
clear
View Answer play_arrow
The enzyme used for joining two DNA fragment is called:
A)
tigase
done
clear
B)
restriction endonuclease
done
clear
C)
DNA polymerase
done
clear
D)
gyrase
done
clear
View Answer play_arrow
Thorn is a modification of
A)
stem
done
clear
B)
leaf
done
clear
C)
stipule
done
clear
D)
root
done
clear
View Answer play_arrow
During double fertilization in plants, one sperm fuses with the egg cell and the other sperm fuses with
A)
synergids cell
done
clear
B)
central cell
done
clear
C)
antipodal cell
done
clear
D)
nucellar cell
done
clear
View Answer play_arrow
Drawback of DDT as pesticide is that
A)
it becomes ineffective after sometime.
done
clear
B)
it is less effective than others.
done
clear
C)
it is not easily/rapidly degraded in nature.
done
clear
D)
its high cost.
done
clear
View Answer play_arrow
Nitrogen fixation is a process of
A)
converting nitrogen in the air to form a usable form by plants.
done
clear
B)
recycling nitrogen from organic matter in the soil.
done
clear
C)
absorbing nitrogen from the soil.
done
clear
D)
conversion of\[N{{O}_{3}}\]to\[{{N}_{2}}\].
done
clear
View Answer play_arrow
The diagram below is a model demonstrating the mass flow hypothesis of translocation.
What are the structures represented by W, X, Y and Z and what is the direction of flow of solution along W?
A)
Phloem - W Xylem - X Roots - Y Leaves - Z Direction of flow along W -from Z to Y
done
clear
B)
Phloem - W Xylem - X Roots - Z Leaves - Y Direction of flow along W -from Y to Z
done
clear
C)
Phloem - X Xylem - W Roots - Z Leaves - Y Direction of flow along W -from Z to Y
done
clear
D)
Phloem - X Xylem - W Roots - Z Leaves - Y Direction of flow along W -from Y to Z
done
clear
View Answer play_arrow
Female gametophyte of angiosperms is represented by
A)
Ovule
done
clear
B)
Megaspore mother cell
done
clear
C)
Embryo sac
done
clear
D)
Nucellus
done
clear
View Answer play_arrow
Given below are the diagrammatic representation of position of floral parts on thalamus, condition of ovary and example. Find the correctly matched combination?
A)
Position of floral parts on thalamus -
Condition of ovary - \[\overline{G}\] Example - Cucumber
done
clear
B)
Position of floral parts on thalamus -
Condition of ovary - \[G-\] Example - Brinjal
done
clear
C)
Position of floral parts on thalamus -
Condition of ovary - \[\underline{G}\] Example - Plum
done
clear
D)
Position of floral parts on thalamus -
Condition of ovary - \[\overline{G}\] Example - Rose
done
clear
View Answer play_arrow
Today, concentration of greenhouse gases is high because of
A)
Use of refrigerator
done
clear
B)
Increased combustion of oil and coal
done
clear
C)
Deforestation
done
clear
D)
All the above
done
clear
View Answer play_arrow
Cross between AaBB and aaBB will form
A)
1 AaBB : 1aaBB
done
clear
B)
All AaBB
done
clear
C)
3 AaBB : 1aaBB
done
clear
D)
1 AaBB : 3aaBB
done
clear
View Answer play_arrow
Which of the following is the most stable ecosystem?
A)
Forest
done
clear
B)
Desert
done
clear
C)
Mountain
done
clear
D)
Ocean
done
clear
View Answer play_arrow
Parthenocarpic tomato fruits can be produced by
A)
treating the plants with low concentrations of gibberellic acid and auxins
done
clear
B)
raising the plants from vernalized seeds
done
clear
C)
treating the plants with phenylmercuric acetate
done
clear
D)
removing androecium of flowers before pollen grains are released
done
clear
View Answer play_arrow
A gene pair hides the effect of another gene. The phenomenon is
A)
epistasis
done
clear
B)
dominance
done
clear
C)
mutation
done
clear
D)
None of the above
done
clear
View Answer play_arrow
What would happen if in a gene encoding a polypeptide of 50 amino acids, 25th codon (UAU) is mutated to UAA?
A)
A polypeptide of 24 amino acids will be formed.
done
clear
B)
Two polypeptides of 24 and 25 amino acids will be formed
done
clear
C)
A polypeptide of 49 amino acids will be formed
done
clear
D)
A polypeptide of 25 amino acids will be Formed
done
clear
View Answer play_arrow
The rate at which light energy is converted to the chemical energy of organic molecules in the ecosystem's is
A)
net primary productivity
done
clear
B)
gross primary productivity
done
clear
C)
net secondary productivity
done
clear
D)
gross secondary productivity
done
clear
View Answer play_arrow
Consider the following statements concerning food chains :
(i) Removal of 80% tigers from an area resulted in greatly increased growth of vegetation. (ii) Removal of most of the carnivores resulted in an increased population of deers. (iii) The length of food chains is generally limited to 3-4 trophic-levels due to energy loss. (iv) The length of food chains may vary from 2 to 8 trophic levels. Which two of the above statements are correct?
A)
(ii) and (iii)
done
clear
B)
(iii) and (iv)
done
clear
C)
(i) and (iv)
done
clear
D)
(i) and (ii)
done
clear
View Answer play_arrow
Which of the following pairs of the cell structures are important for determining the movement of molecules in or out of the plant cell?
A)
Tonoplast + Vacuolar membrane
done
clear
B)
Tonoplast + Cell membrane
done
clear
C)
Cell wall + Cell membrane
done
clear
D)
Cell wall + Tonoplasts
done
clear
View Answer play_arrow
Binomial nomenclature was introduced by
A)
Carolus Linnaeus
done
clear
B)
Charles Darwin
done
clear
C)
Bentham and Hooker
done
clear
D)
Aristotle
done
clear
View Answer play_arrow
Pteridophytes differ from mosses/bryophytes in possessing
A)
independent gametophyte
done
clear
B)
well developed vascular system
done
clear
C)
archegonia structure
done
clear
D)
flagellate spermatozoids
done
clear
View Answer play_arrow
Centrioles and centrosomes occur in the cells of
A)
green plants
done
clear
B)
animals
done
clear
C)
bacteria and cyanobacteria
done
clear
D)
both [b] and [c]
done
clear
View Answer play_arrow
Mycorrhiza is
A)
a symbiotic association of plant roots and certain fungi.
done
clear
B)
an association of algae with fungi.
done
clear
C)
a fungus parasitising root system of higher plants.
done
clear
D)
an association of Rhizobium with the roots of leguminous plants.
done
clear
View Answer play_arrow
Laminaria (kelp) and Fucus (rock weed) are the examples of
A)
red algae
done
clear
B)
brown algae
done
clear
C)
green algae
done
clear
D)
golden brown algae
done
clear
View Answer play_arrow
The outer layer ofvacuole is called
A)
cell wall
done
clear
B)
tonoplast
done
clear
C)
plasma layer
done
clear
D)
leucoplast
done
clear
View Answer play_arrow
A narrow layer of thin walled cells found between phloem/bark and wood of a dicot is
A)
cork cambium
done
clear
B)
vascular cambium
done
clear
C)
endodermis
done
clear
D)
pericycle
done
clear
View Answer play_arrow
Bulk flow of substances over the longer distances through the vascular tissue is called
A)
simple diffusion
done
clear
B)
facilitated diffusion
done
clear
C)
active transport
done
clear
D)
translocation
done
clear
View Answer play_arrow
The linking of antibiotic resistance gene with the plasmid vector became possible with
A)
DNA ligase
done
clear
B)
En donucl eases
done
clear
C)
DNA polymerase
done
clear
D)
Exonucleases
done
clear
View Answer play_arrow
Which one of the following is not included under in-situ conservation?
A)
Botanical garden
done
clear
B)
Biosphere reserve
done
clear
C)
National park
done
clear
D)
Sanctuary
done
clear
View Answer play_arrow
Monascus purpureus is a yeast used commercially in the production of:
A)
ethanol
done
clear
B)
streptokinase for removing clots from the blood vessels.
done
clear
C)
Citric acid
done
clear
D)
blood cholesterol lowering statins
done
clear
View Answer play_arrow
Gel electrophoresis is used for
A)
cutting of DNA into fragments
done
clear
B)
separation of DNA fragments according to their size
done
clear
C)
construction of recombinant DNA by joining with cloning vectors
done
clear
D)
isolation of DNA molecule
done
clear
View Answer play_arrow
DNA or RNA segment tagged with a radioactive molecule is called
A)
Vector
done
clear
B)
Probe
done
clear
C)
Clone
done
clear
D)
Plasmid
done
clear
View Answer play_arrow
Which of the following is considered a hot-spot, of biodiversity in India?
A)
Indo-Gangetic Plain
done
clear
B)
Eastern Ghats
done
clear
C)
Aravalli Hills
done
clear
D)
Western Ghats
done
clear
View Answer play_arrow
Which one of the following shows concept of species-area relationship?
A)
The number of species in an area increases with the size of the area.
done
clear
B)
Larger species require larger habitat areas than do smaller species.
done
clear
C)
Most species within any given area are endemic.
done
clear
D)
The larger the area, the greater the extinction rate.
done
clear
View Answer play_arrow
The movement of mineral ions into plant root cells as a result of diffusion is called
A)
osmosis
done
clear
B)
active absorption
done
clear
C)
passive absorption
done
clear
D)
endocytosis
done
clear
View Answer play_arrow
The rate of transpiration in plants is dependent upon
A)
temperature and soil
done
clear
B)
light and temperature
done
clear
C)
wind, temperature and light
done
clear
D)
light, temperature, atmospheric humidity and wind
done
clear
View Answer play_arrow
Stomata open and close due to
A)
circadian rhythm
done
clear
B)
genetic clock
done
clear
C)
pressure of gases inside the leaves
done
clear
D)
turgor pressure of guard cells
done
clear
View Answer play_arrow
Match Column-I with Column-II and select the correct answer from the codes given below.
Column-I Column-II A. Trichoderma (i) Nitrification B. Streptomyces (ii) Biocontrol agent C. Nitrosomonas (iii) Lactic acid D. Lactobacillus (iv) Source of antibiotic
A)
A\[\to \](ii), B\[\to \](iii), C\[\to \](iv), D\[\to \](i)
done
clear
B)
A\[\to \](ii), B\[\to \](iv), C\[\to \](i), D\[\to \](iii)
done
clear
C)
A\[\to \](iii), B\[\to \](i), C\[\to \](ii), D\[\to \](iv)
done
clear
D)
A\[\to \](iv), B\[\to \](ii), C\[\to \](i), D\[\to \](iii)
done
clear
View Answer play_arrow
The term 'keel' is used for special type of
A)
sepals
done
clear
B)
petals
done
clear
C)
stamens
done
clear
D)
carpels
done
clear
View Answer play_arrow
Chemiosmotic hypothesis given by Peter Mitchell proposes the mechanism of
A)
synthesis of ATP
done
clear
B)
synthesis of FADFL,
done
clear
C)
synthesis of NADH
done
clear
D)
synthesis of NADPH
done
clear
View Answer play_arrow
Which ion is essential for muscle contraction?
A)
\[N{{a}^{+}}\]
done
clear
B)
\[{{K}^{+}}\]
done
clear
C)
\[C{{a}^{2+}}\]
done
clear
D)
\[C{{l}^{-}}\]
done
clear
View Answer play_arrow
Which one of the following organs in the human body is most affected due to shortage of oxygen?
A)
Intestine
done
clear
B)
Skin
done
clear
C)
Kidney
done
clear
D)
Brain
done
clear
View Answer play_arrow
A cricket player is fast chasing a ball in the field. Which one of the following groups of bones is directly contributing in this movement?
A)
Femur, malleus, tibia, metatarsals
done
clear
B)
Pelvis, ulna, patella, tarsals
done
clear
C)
Sternum, femur, tibia, fibula
done
clear
D)
Tarsals, femur, metatarsals, tibia
done
clear
View Answer play_arrow
Choose the correct names of the different bacteria according to their shapes.
A)
A - Cocci, B - Bacilli, C - Spirilla, D - Vibrio
done
clear
B)
A - Bacilli, B - Cocci, C - Spirilla, D - Vibrio
done
clear
C)
A - Spirilla, B - Bacilli, C - Cocci, D - Vibrio
done
clear
D)
A - Spirilla, B - Vibrio, C - Coed, D - Bacilli
done
clear
View Answer play_arrow
Match the source gland with respective hormone as well as the function correctly.
A)
Source gland - Anterior pituitary Hormone - Oxytocin Function - Contraction of uterus muscles during child birth
done
clear
B)
Source gland - Posterior pituitary Hormone - Vasopress in Function - Stimulates reabsorption of water in the distal tubules in the nephron
done
clear
C)
Source gland - Corpus luteum Hormone - Estrogen Function - Supports pregnancy
done
clear
D)
Source gland - Thyroid Hormone - Thyroxine Function - Regulates blood calcium
done
clear
View Answer play_arrow
The given diagram shows microsporangium of a mature anther. Identify A, B and C.
A)
A\[\to \]Middle layer, B\[\to \]Endothecium, C\[\to \]Tapetum
done
clear
B)
A\[\to \]Endothecium, B\[\to \]Tapetum, C\[\to \]Middle layer
done
clear
C)
A\[\to \]Endothecium, B\[\to \]Middle layer, C\[\to \] Tapetum
done
clear
D)
A\[\to \]Tapetum, B\[\to \]Middle layer, C\[\to \] Endothedum
done
clear
View Answer play_arrow
In the chemistry of vision in mammals, the photosensitive substance is called
A)
sclerotin
done
clear
B)
retinol
done
clear
C)
rhodopsin
done
clear
D)
melanin
done
clear
View Answer play_arrow
Vaccine against polio viruses is an example of
A)
auto-immunization
done
clear
B)
passive immunization
done
clear
C)
active immunization.
done
clear
D)
simple immunization
done
clear
View Answer play_arrow
Egg is liberated from ovary in
A)
secondary oocyte stage
done
clear
B)
primary oocyte stage
done
clear
C)
oogonial stage
done
clear
D)
mature ovum stage
done
clear
View Answer play_arrow
Animals that can tolerate a narrow range of salinity are
A)
stenohaline
done
clear
B)
euryhaline
done
clear
C)
anadromous
done
clear
D)
catadromous
done
clear
View Answer play_arrow
The most important component of the oral contraceptive pills is
A)
Progesterone
done
clear
B)
Growth hormone
done
clear
C)
Thyroxine
done
clear
D)
Luteinizing hormone
done
clear
View Answer play_arrow
The following graph of relative concentrations of the four hormones present in the blood plasma of a woman during her menstrual cycle. Identify the hormones.
A)
A - FSH B - Progesterone C - LH D - Oestrogen
done
clear
B)
A - LH B - Progesterone C - FSH D - Oestrogen
done
clear
C)
A - FSH B - Oestrogen C - LH D - Progesterone
done
clear
D)
A - LH B - Oestrogen C - FSH D - Progesterone
done
clear
View Answer play_arrow
Reproductive health in society can be improved by-
(i) Introduction of sex education in schools. (ii) Increased medical assistance. (iii) Awareness about contraception and STDs. (iv) Equal opportunities to male and female child. (v) Ban on aminocentesis. (vi) Encouraging myths and misconceptions.
A)
All of these
done
clear
B)
(i), (ii), (iv) & (vi)
done
clear
C)
(i), (ii), (iii), (iv) & (v)
done
clear
D)
(ii) and (v)
done
clear
View Answer play_arrow
Foetal ejection reflex in human female is induced by
A)
release oxytocin from pituitary
done
clear
B)
fully developed foetus and placenta
done
clear
C)
differentiation of mammary glands
done
clear
D)
pressure exerted by amniotic fluid
done
clear
View Answer play_arrow
Natality is the characteristic of a population which means
A)
the total number of individuals present per unit area at a given time
done
clear
B)
the increase in number of individuals in a population under given environmental conditions
done
clear
C)
loss of individuals due to death in a population under given environmental conditions
done
clear
D)
the movement of individuals into and out of population
done
clear
View Answer play_arrow
In human female the blastocyst
A)
Forms placenta even before implantation
done
clear
B)
Gets implanted into uterus 3 days after ovulation
done
clear
C)
Gets nutrition from uterine endometrial secretion only after implantation
done
clear
D)
Gets implanted in endometrium by the trophoblast cells
done
clear
View Answer play_arrow
Choose the wrong statement regarding urine formation
A)
Filtration is non-selective process performed by glomerulus
done
clear
B)
The glomerular capillary blood pressure causes filtration of blood through three layers
done
clear
C)
GFR in a healthy individual is approximately 125ml/min
done
clear
D)
The ascending limb of the Henle's loop is permeable to water but allows transport of electrolytes actively or passively
done
clear
View Answer play_arrow
Which one of the following is an exotic Indian fish?
A)
Catla catla
done
clear
B)
Heteropneustes fossilis
done
clear
C)
Cyprinus carpio
done
clear
D)
Labeo rohita
done
clear
View Answer play_arrow
Which of the following primate is the closest relative of humans?
A)
Rhesus monkey
done
clear
B)
Orangutan
done
clear
C)
Gorilla
done
clear
D)
Gibbon
done
clear
View Answer play_arrow
Adaptive radiation refers to
A)
evolution of different species from a common ancestor
done
clear
B)
migration of members of a species to different geographical areas
done
clear
C)
power of adaptation in an individual to a variety of environments
done
clear
D)
adaptations due to geographical isolation.
done
clear
View Answer play_arrow
What was the most significant trend in evolution of modern man (Homo sapiens) from his ancestors?
A)
Upright posture
done
clear
B)
Shortening of jaws
done
clear
C)
Binocular-vision
done
clear
D)
Increasing brain capacity
done
clear
View Answer play_arrow
The given figure shows the regions of root tip with labelling as A, B and C. Choose the option which shows the correct labelling of A, B and C. Region of mature cells
A)
A - Zone of elongation, B - Zone of meiosis, C - Zone of mitosis.
done
clear
B)
A - Zone of maturation, B - Zone of meristematic activity, C - Zone of elongation.
done
clear
C)
A - Zone of mitosis, B - Zone of elongation, C - Zone of root cap.
done
clear
D)
A - Region of maturation, B - Region of elon-gation, C - Zone of meristematic activity.
done
clear
View Answer play_arrow
Select the incorrect statement from the following.
A)
Apiculture provides generating additional income industry for the farmers.
done
clear
B)
Bee keeping is labour intensive process.
done
clear
C)
Bee venom is used to cure certain diseases like gout and arthritis.
done
clear
D)
Honey is used as laxative, antiseptic and sedative.
done
clear
View Answer play_arrow
Classification of Porifera is based on
A)
branching
done
clear
B)
spicules
done
clear
C)
reproduction
done
clear
D)
symmetry
done
clear
View Answer play_arrow
The kind of epithelium which forms the inner walls of blood vessels is
A)
cuboidal epithelium
done
clear
B)
columnar epithelium
done
clear
C)
ciliated columnar epithelium
done
clear
D)
squamous epithelium
done
clear
View Answer play_arrow
Myoglobin is present in
A)
all muscle fibres
done
clear
B)
white muscle fibres only
done
clear
C)
red muscle fibres only
done
clear
D)
[d] both white and red muscle fibres
done
clear
View Answer play_arrow
Mast cells are associated with
A)
exocrine glands
done
clear
B)
endocrine glands
done
clear
C)
areolar connective tissue
done
clear
D)
neural tissue
done
clear
View Answer play_arrow
Which of the following is incorrect match of animal group/life style/structure/function?
A)
Animal group - Sponges Lifestyle - Sessile filter feeders Structure/function - Amoebocytes/ carry food and wastes Spicules/Support and Protection sponging / support
done
clear
B)
Animal group - Cnidarians Lifestyle - Free floating or attached Structure/function - Gastrovascular cavity / digestion Cnidocytes / protection and getting
done
clear
C)
Animal group - Flatworms Lifestyle - Free living or parasite Structure/function - Flame cells/excretion Tegument/protection
done
clear
D)
Animal group - Molluscs Lifestyle - Terrestrial marine, fresh Structure/function - Radula/feeding mantle/motility water inhabitants
done
clear
View Answer play_arrow
Man, in the life cycle of Plasmodium, is
A)
primary host
done
clear
B)
secondary host
done
clear
C)
intermediate host
done
clear
D)
None of these
done
clear
View Answer play_arrow
The most active phagocytic white blood cells are
A)
neutrophils and monocytes
done
clear
B)
neutrophils and eosinophils
done
clear
C)
lymphocytes and macrophages
done
clear
D)
eosinophils and lymphocytes
done
clear
View Answer play_arrow
Human insulin is being commercially produced from a transgenic species of
A)
Escherichia
done
clear
B)
Mycobacterium
done
clear
C)
Rhizobium
done
clear
D)
Saccharomyces
done
clear
View Answer play_arrow
Obstacle to large scale transplantation of organs is
A)
insufficiency of organ donors
done
clear
B)
immunological rejection of foreign bodies
done
clear
C)
religious or ethnic considerations
done
clear
D)
lack of effective surgical techniques
done
clear
View Answer play_arrow
Which one of the following is an Indian medicinal plant?
A)
Saccharum officinarum
done
clear
B)
Rauwolfia serpentine
done
clear
C)
Oryza sativa
done
clear
D)
Solarium melongena
done
clear
View Answer play_arrow
Which of the following bacteria carry out oxygenic photosynthesis by means of a photosynthetic apparatus similar to the eukaryotes?
A)
Purple sulphur bacteria
done
clear
B)
Green sulphur bacteria
done
clear
C)
Cyanobacteria
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
It is commonly said that ladies are protected from heart attacks in reproductive period i.e., from puberty to menopause because:
A)
Level of HDL is high due to oestrogen production
done
clear
B)
Level of HDL remains lower during this period
done
clear
C)
Level of LDL and HDL remain in balance
done
clear
D)
Level of LDL is high due to oestrogen Production
done
clear
View Answer play_arrow
Dolly sheep was obtained by -
A)
Cloning the udder cell (somatic cell) fused with enucleated oocyte
done
clear
B)
Cloning of gametes
done
clear
C)
Tissue culture
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Which of the following forms the basis of DNA finger printing?
A)
The relative proportions of purines and pyrimidines in DNA.
done
clear
B)
Satellite DNA occurring as highly repeated short DNA segments.
done
clear
C)
The relative' difference in the DNA occurrence in blood, skin and saliva.
done
clear
D)
The relative amount of DNA in the ridges and grooves of the fingerprints.
done
clear
View Answer play_arrow
Which one ofthe following correctly represents the normal adult human dental formula?
A)
\[\frac{3}{3},\]\[\frac{1}{1},\]\[\frac{3}{2},\]\[\frac{1}{1}\]
done
clear
B)
\[\frac{2}{2},\]\[\frac{1}{1},\]\[\frac{3}{2},\]\[\frac{3}{3}\]
done
clear
C)
\[\frac{2}{2},\]\[\frac{1}{1},\]\[\frac{2}{2},\]\[\frac{3}{3}\]
done
clear
D)
\[\frac{3}{3},\]\[\frac{1}{1},\]\[\frac{3}{3},\]\[\frac{3}{3}\]
done
clear
View Answer play_arrow
Toxic substances are detoxified in human body in
A)
kidney
done
clear
B)
lungs
done
clear
C)
liver
done
clear
D)
stomach
done
clear
View Answer play_arrow
The function of rennin is
A)
vasodilation
done
clear
B)
reduce blood pressure
done
clear
C)
degradation of angiotensinogen
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Heart is covered by
A)
Peritoneum
done
clear
B)
Pleural membrane
done
clear
C)
Pericardium
done
clear
D)
Visceral membrane
done
clear
View Answer play_arrow
Which of the following is incorrect?
A)
Iodine is needed for thyroxine formation.
done
clear
B)
Calcium regulates the excitibility of nerve fibres.
done
clear
C)
Potassium plays an important role in the regulation of acid base balance in cell.
done
clear
D)
Phosphorus helps to maintain the osmotic pressure of the body fluids.
done
clear
View Answer play_arrow
Diagram represents one cardiac cycle lasting 0.8 s and to the possible answers that follow it.
Which answer describes the events that occur during period X?
A)
atrial diastole and ventricular systole
done
clear
B)
atrial diastole and ventricular diastole
done
clear
C)
atrial systole and ventricular systole
done
clear
D)
atrial systole and ventricular diastole
done
clear
View Answer play_arrow
Diabetes insipidus is a condition in which a per -son is unable to produce sufficient levels of the hormone ADH. The hormone increases the permeability to water of the second (distal) convoluted tubule and collecting duct in the kidney nephrons. What is produced as a result?
A)
large volumes of concentrated urine
done
clear
B)
large volumes of dilute urine
done
clear
C)
small volumes of concentrated urine
done
clear
D)
small volumes of dilute urine
done
clear
View Answer play_arrow
Dark purplish gland lying on the left side of abdomen is called:
A)
liver
done
clear
B)
spleen
done
clear
C)
gall bladder
done
clear
D)
appendix
done
clear
View Answer play_arrow
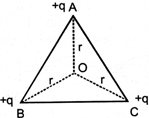
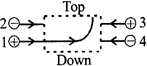
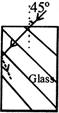
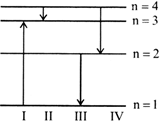
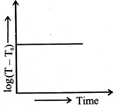
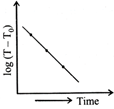
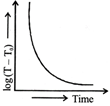
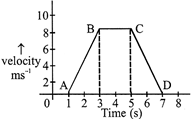
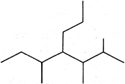
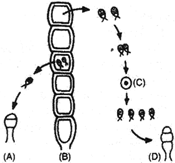
 What are the structures represented by W, X, Y and Z and what is the direction of flow of solution along W?
What are the structures represented by W, X, Y and Z and what is the direction of flow of solution along W?  Condition of ovary - \[\overline{G}\] Example - Cucumber
Condition of ovary - \[\overline{G}\] Example - Cucumber  Condition of ovary - \[G-\] Example - Brinjal
Condition of ovary - \[G-\] Example - Brinjal  Condition of ovary - \[\underline{G}\] Example - Plum
Condition of ovary - \[\underline{G}\] Example - Plum  Condition of ovary - \[\overline{G}\] Example - Rose
Condition of ovary - \[\overline{G}\] Example - Rose 
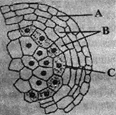
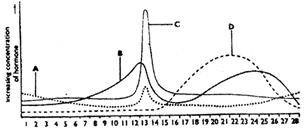
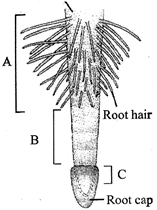
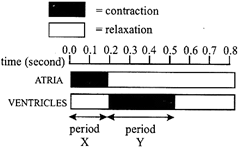 Which answer describes the events that occur during period X?
Which answer describes the events that occur during period X? 