The change in the value of 'g' at a height 'h' above the surface of the earth is the same as at a depth 'd' below the surface of the earth. When both ?d? and 'h' are much smaller than the radius of earth, then which one of the following relation is correct?
A)
\[d=\frac{3h}{2}\]
done
clear
B)
\[d=\frac{h}{2}\]
done
clear
C)
\[d=h\]
done
clear
D)
\[d=2h\]
done
clear
View Answer play_arrow
An electron of a stationary hydrogen atom passes from the fifth energy level to the ground level. The velocity that the atom acquired as a result of photon emission will be
A)
\[\frac{24hR}{25m}\]
done
clear
B)
\[\frac{25hR}{24m}\]
done
clear
C)
\[\frac{25m}{24hR}\]
done
clear
D)
\[\frac{24m}{25hR}\]
done
clear
View Answer play_arrow
A particle of mass of 4 kg suspended from a spring of force constant \[800\,\,N\,\,{{m}^{-1}}\] executes simple harmonic oscillations. If the total energy of the oscillator is 4 J, the maximum acceleration \[(in\,\,m\,{{s}^{-2}})\] of the particle is
A)
5
done
clear
B)
15
done
clear
C)
45
done
clear
D)
20
done
clear
View Answer play_arrow
A body of mass \[2\,\,kg\] is rotating on a circular path of radius \[0.8\,\,m\] with an angular velocity \[44\,\,red/\sec .\] If radius of path becomes \[1\,\,m\] then value of angular velocity will be
A)
35.28 rad/sec
done
clear
B)
14.08 rad/sec
done
clear
C)
28.16 rad/sec
done
clear
D)
24.08 rad/sec
done
clear
View Answer play_arrow
The motion of particle is described by the equation \[x=a+b{{t}^{2}},\] where \[a=15\,\,cm\] and \[b=3\,\,cm/{{\sec }^{2}}.\] Its instant velocity at time \[3\,\,\sec \] will be
A)
3 6 cm/sec
done
clear
B)
9 cm/sec
done
clear
C)
4.5 cm/sec
done
clear
D)
18 cm/sec
done
clear
View Answer play_arrow
An observer moves towards a stationary source of sound with a speed 1/5th of the speed of sound. The wavelength and frequency of the source emitted by any source be\[\lambda \] and f respectively. The apparent frequency and wavelength recorded by the observer are respectively
A)
\[0.8f,\]\[0.8\lambda \]
done
clear
B)
\[1.2f,\]\[1.2\lambda \]
done
clear
C)
\[1.2f,\]\[\lambda \]
done
clear
D)
\[f,\]\[1.2\lambda \]
done
clear
View Answer play_arrow
A coil of insulated wire is connected to a battery. If it is taken to galvanometer, its pointer is deflected, because
A)
induced current is produced
done
clear
B)
the coil acts like a magnet
done
clear
C)
the number of turns in the coil of the galvanometer are changed
done
clear
D)
None of these
done
clear
View Answer play_arrow
A heater of 220 V heats a volume of water in 5 minutes. A heater of 110 V heats the same volume of water in
A)
5 minutes
done
clear
B)
8 minutes
done
clear
C)
10 minutes
done
clear
D)
20 minutes
done
clear
View Answer play_arrow
A mass of m kg moving with a speed of 1.5 m/s on a horizontal smooth surface, collides with a nearly weightless spring of force constant k= 50 N/m. If the maximum compression of the spring is 0.15 m, the value of mass m is
A)
0.5 kg
done
clear
B)
0.15 kg
done
clear
C)
0.12 kg
done
clear
D)
1.5 kg
done
clear
View Answer play_arrow
The perfect gas equation for 4 gram of hydrogen gas is
A)
\[PV=RT\]
done
clear
B)
\[PV=2RT\]
done
clear
C)
\[PV=\frac{1}{2}RT\]
done
clear
D)
\[PV=4RT\]
done
clear
View Answer play_arrow
A parallel monochromatic beam of light is incident normally on a narrow slit. A dim-action pattern is formed on a screen placed perpendicular to the direction of the incident beam. At the first minimum of the diffraction pattern, the phase difference between the rays coming from the two edges of the slit is
A)
\[0\]
done
clear
B)
\[\pi /2\]
done
clear
C)
\[\pi \]
done
clear
D)
\[2\pi \]
done
clear
View Answer play_arrow
According to Einstein's photoelectric equation, the plot of the kinetic energy of the emitted photoelectrons from a metal vs the frequency of the incident radiation gives a straight line whose slope
A)
depends on the intensity of the radiation
done
clear
B)
depends on the nature of the metal used
done
clear
C)
depends both on the intensity of the radiation and the metal used
done
clear
D)
is the same for all metals and independent of the intensity of the radiation
done
clear
View Answer play_arrow
If a body of mass 3 kg is dropped from the top of a tower of height 45 m. Then its kinetic energy after 3 sec is \[[take\,\,g=10m/{{s}^{2}}]\]
A)
557 J
done
clear
B)
1450 J
done
clear
C)
1050 J
done
clear
D)
1296 J
done
clear
View Answer play_arrow
In a nuclear fission, 0.1% mass is converted into energy. The energy released by the fission of 1 kg mass will be
A)
\[9\times {{10}^{19}}\,J\]
done
clear
B)
\[9\times {{10}^{17}}\,J\]
done
clear
C)
\[9\times {{10}^{16}}\,J\]
done
clear
D)
\[9\times {{10}^{13}}\,J\]
done
clear
View Answer play_arrow
If the coefficient of cubical expansion is x times of the coefficient of superficial expansion, then value of x is
A)
3
done
clear
B)
2.5
done
clear
C)
1.5
done
clear
D)
2
done
clear
View Answer play_arrow
A projectile is thrown in the upward direction making an angle of \[60{}^\circ \] with the horizontal direction, with a velocity of \[147\,m{{s}^{-1}}.\] Then the time after which inclination with the horizontal is \[45{}^\circ ,\] is
A)
\[15(\sqrt{3}-1)\,s\]
done
clear
B)
\[15(\sqrt{3}+1)\,s\]
done
clear
C)
\[7.5(\sqrt{3}-1)\,s\]
done
clear
D)
\[7.5(\sqrt{3}+1)\,s\]
done
clear
View Answer play_arrow
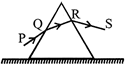
An equilateral prism is placed on a horizontal surface. A ray PQ is incident onto it. For minimum deviation
A)
PQ is horizontal
done
clear
B)
QR is horizontal
done
clear
C)
RS is horizontal
done
clear
D)
any one will be horizontal
done
clear
View Answer play_arrow
A tin nucleus (atomic number Z = 50) has a radius of \[6.6\times {{10}^{-15}}m.\] The potential on its surface will be (the charge on the proton \[=1.6\times {{10}^{-19}}C\])
A)
\[1.1\times {{10}^{7}}\,V\]
done
clear
B)
\[2.1\times {{10}^{7}}\,V\]
done
clear
C)
\[3.1\times {{10}^{7}}\,V\]
done
clear
D)
\[0.15\times {{10}^{7}}\,V\]
done
clear
View Answer play_arrow
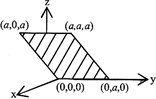
Consider an electric field \[\overrightarrow{E}={{E}_{0}}\hat{x}\] where \[{{E}_{0}}\] is a constant. The flux through the shaded area (as shown in the figure) due to this field is
A)
\[2{{E}_{0}}{{a}^{2}}\]
done
clear
B)
\[\sqrt{2}{{E}_{0}}{{a}^{2}}\]
done
clear
C)
\[{{E}_{0}}\,{{a}^{2}}\]
done
clear
D)
\[\frac{{{E}_{0}}{{a}^{2}}}{\sqrt{2}}\]
done
clear
View Answer play_arrow
Two rods of the same length and areas of cross- section \[{{A}_{1}}\] and \[{{A}_{2}}\] have their ends at the same temperature \[{{K}_{1}}\] and \[{{K}_{2}}\] are the thermal conductivities of the two rods. The rate of flow of heat is same in both rods if
A)
\[\frac{{{A}_{1}}}{{{A}_{2}}}=\frac{{{K}_{1}}}{{{K}_{2}}}\]
done
clear
B)
\[\frac{{{A}_{1}}}{{{A}_{2}}}=\frac{{{K}_{2}}}{{{K}_{1}}}\]
done
clear
C)
\[{{A}_{1}}{{A}_{2}}={{K}_{1}}{{K}_{2}}\]
done
clear
D)
\[{{A}_{1}}{{K}_{1}}^{2}={{A}_{2}}{{K}_{2}}^{2}\]
done
clear
View Answer play_arrow
A photon collides with a stationary hydrogen atom in ground state in elastically. Energy of the colliding photon is 10.2 eV. After a time interval of the order of micro second another photon collides with same hydrogen atom in elastically with an energy of 15 eV. What will be observed by the detector?
A)
2 photon of energy 10.2 eV
done
clear
B)
2 photon of energy 1.4 eV
done
clear
C)
One photon of energy 10.2 eV and an electron of energy 1.4 eV
done
clear
D)
One photon of energy 10.2 eV and another photon of 1.4 eV
done
clear
View Answer play_arrow
A cylindrical resonance tube, open at both ends, has a fundamental frequency fin air. If half of the length is dipped vertically in water, the fundamental frequency of the air column will be
A)
\[\frac{3f}{2}\]
done
clear
B)
\[2f\]
done
clear
C)
\[f\]
done
clear
D)
\[\frac{f}{2}\]
done
clear
View Answer play_arrow
The ionisation energy of hydrogen atom is 13.6 eV. Following Bohr's theory, the energy corresponding to a transition between the 3rd and the 4th orbit is
A)
\[3.40\,eV\]
done
clear
B)
\[1.51\,eV\]
done
clear
C)
\[0.85\,eV\]
done
clear
D)
\[0.66\,eV\]
done
clear
View Answer play_arrow
The kinetic energy of a body becomes four times its initial value. The new momentum will be
A)
same as initial value
done
clear
B)
twice the initial value
done
clear
C)
thrice the initial value
done
clear
D)
four times the initial value
done
clear
View Answer play_arrow
Error in the measurement of radius of a sphere is 1%. Then error in the measurement of volume is
A)
1%
done
clear
B)
5%
done
clear
C)
3%
done
clear
D)
8%
done
clear
View Answer play_arrow
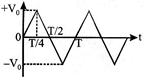
The voltage time (V-t) graph for triangular wave having peak value \[{{V}_{0}}\] is as shown in figure. The rms value of V in time interval from t = 0 to T/4 is \[\frac{{{V}_{0}}}{\sqrt{x}}\]then find the value of x.
A)
5
done
clear
B)
4
done
clear
C)
7
done
clear
D)
3
done
clear
View Answer play_arrow
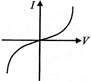
The \[I-V\]characteristics shown in figure represents
A)
ohmic conductors
done
clear
B)
non-ohmic conductors
done
clear
C)
insulators
done
clear
D)
superconductors
done
clear
View Answer play_arrow
A 2 kg copper block is heated to \[500{}^\circ C\] and then it is placed on a large block of ice at \[0{}^\circ C.\] If the specific heat capacity of copper is \[400\,J/kg{}^\circ C\] and latent heat of fusion of water is \[3.5\times {{10}^{5}}J/kg,\]the amount of ice that can melt is
A)
(7/8) kg
done
clear
B)
(7/5) kg
done
clear
C)
(8/7) kg
done
clear
D)
(5/7) kg
done
clear
View Answer play_arrow
A gymnast takes turns- with her arms & legs stretched. When she pulls her arms and legs in
A)
the angular velocity decreases
done
clear
B)
the moment of inertia decreases
done
clear
C)
the angular velocity stays constant
done
clear
D)
the angular momentum increases
done
clear
View Answer play_arrow
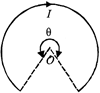
A current of I ampere flows in a wire forming a circular arc of radius r metres subtending an angle \[\theta \]at the centre as shown. The magnetic field at the centre O in tesla is
A)
\[\frac{{{\mu }_{0}}I\theta }{4\pi r}\]
done
clear
B)
\[\frac{{{\mu }_{0}}\,I\theta }{2\pi r}\]
done
clear
C)
\[\frac{{{\mu }_{0}}\,I\theta }{2r}\]
done
clear
D)
\[\frac{{{\mu }_{0}}\,I\theta }{4r}\]
done
clear
View Answer play_arrow
The number of degrees of freedom for each atom of a monoatomic gas is
A)
3
done
clear
B)
5
done
clear
C)
6
done
clear
D)
1
done
clear
View Answer play_arrow
A radioactive nuclei with decay constant 0.5/s is being produced at a constant rate of 100 nuclei/s. If at t = 0 there were no nuclei, the time when there are 50 nuclei is:
A)
\[1\,s\]
done
clear
B)
\[2\ln \left( \frac{4}{3} \right)s\]
done
clear
C)
\[\ln \,2\,s\]
done
clear
D)
\[\ln \left( \frac{4}{3} \right)s\]
done
clear
View Answer play_arrow
The velocity of electromagnetic radiation in a medium of permittivity \[{{\varepsilon }_{0}}\] and permeability \[{{\mu }_{0}}\] is given by
A)
\[\sqrt{\frac{{{\in }_{0}}}{{{\mu }_{0}}}}\]
done
clear
B)
\[\sqrt{{{\mu }_{0}}\,\,{{\in }_{0}}}\]
done
clear
C)
\[\frac{1}{\sqrt{{{\mu }_{0}}\,\,{{\in }_{0}}}}\]
done
clear
D)
\[\sqrt{\frac{{{\mu }_{0}}}{{{\in }_{0}}}}\]
done
clear
View Answer play_arrow
A uniform solid cylindrical roller of mass 'm' is being pulled on a horizontal surface with force F parallel to the surface and applied at its centre. If the acceleration of the cylinder is 'a' and it is rolling without slipping then the value of 'F' is:
A)
\[ma\]
done
clear
B)
\[\frac{5}{3}\,ma\]
done
clear
C)
\[\frac{3}{2}\,ma\]
done
clear
D)
\[2\,ma\]
done
clear
View Answer play_arrow
If L denotes the inductance of an inductor through which a current i is flowing, the dimensions of L \[{{i}^{2}}\] are
A)
\[[M{{L}^{2}}{{T}^{-2}}]\]
done
clear
B)
\[[ML{{T}^{-2}}]\]
done
clear
C)
\[[{{M}^{2}}{{L}^{2}}{{T}^{-2}}]\]
done
clear
D)
Not expressible in \[M,\,L,\,T\]
done
clear
View Answer play_arrow
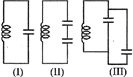
Figure shows three oscillating LC circuit with identical inductors and capacitors. If \[{{t}_{1}},{{t}_{2}},{{t}_{3}}\] are the time taken by the circuits I, II, III for fully discharge, then
A)
\[{{t}_{1}}>{{t}_{2}}>{{t}_{3}}\]
done
clear
B)
\[{{t}_{1}}<{{t}_{2}}<{{t}_{3}}\]
done
clear
C)
\[{{t}_{2}}<{{t}_{1}}<{{t}_{3}}\]
done
clear
D)
\[{{t}_{3}}=\sqrt{{{t}_{1}}{{t}_{2}}}\]
done
clear
View Answer play_arrow
The internal resistance of a 2.1V cell which gives a current of 0.2 A through a resistance of \[10\Omega \]is
A)
\[0.5\,\Omega \]
done
clear
B)
\[0.8\,\Omega \]
done
clear
C)
\[1.0\,\Omega \]
done
clear
D)
\[0.2\,\Omega \]
done
clear
View Answer play_arrow
A spherical ball of iron of radius 2 mm is falling through a column of glycerine. If densities of glycerine and iron are respectively \[1.3\times {{10}^{3}}kg/{{m}^{3}}\] and \[8\times {{10}^{3}}kg/{{m}^{3}},\]\[\eta \]for glycerine \[=0.83N{{m}^{-2}}\sec ,\] then the terminal velocity is
A)
0.7 m/s
done
clear
B)
0.07 m/s
done
clear
C)
0.007 m/s
done
clear
D)
0.0007 m/s
done
clear
View Answer play_arrow
The time taken by light to pass through 4 mm thick glass slab of refractive index 1.5 will be (velocity of light in air \[=3\times {{10}^{8}}\,m/s\])
A)
\[8\times {{10}^{-11}}s\]
done
clear
B)
\[2\times {{10}^{-11}}s\]
done
clear
C)
\[3\times {{10}^{-11}}s\]
done
clear
D)
\[4\times {{10}^{-11}}s\]
done
clear
View Answer play_arrow
Surface tension of a solution is \[30\times {{10}^{-2}}N/m.\] The radius of the soap bubble is 5 cm. The surface energy of soap bubble is
A)
\[1.8\times {{10}^{1}}\,J\]
done
clear
B)
\[1.8\times {{10}^{2}}\,J\]
done
clear
C)
\[1.0\times {{10}^{-1}}\,J\]
done
clear
D)
\[1.8\times {{10}^{-2}}\,J\]
done
clear
View Answer play_arrow
The half-life \[o{{f}^{215}}\] At is\[100\mu s.\] The time taken for the radioactivity of a sample \[o{{f}^{215}}\]At to decay to\[1/{{16}^{th}}\]of its initial value is
A)
\[400\mu s\]
done
clear
B)
\[6.3\mu s\]
done
clear
C)
\[40\mu s\]
done
clear
D)
\[300\mu s\]
done
clear
View Answer play_arrow
In household electric circuit, which of the following statements is/are incorrect
A)
all electric appliances drawing power are joined in parallel
done
clear
B)
a switch may be either in series or in parallel with the appliance which it controls
done
clear
C)
if a switch is in parallel with an appliance, it will draw power when the switch is in the 'off position (open)
done
clear
D)
if a switch is in parallel with an appliance, the fuse will blow (burn out) when the switch is put 'on' closed.
done
clear
View Answer play_arrow
In an p-n-p transistor working as a common base amplifier current gain is 0.96 and emitter current is 7.2 mA. The base current is
A)
0.5 mA
done
clear
B)
0.29 mA
done
clear
C)
0.35 mA
done
clear
D)
0.4 mA
done
clear
View Answer play_arrow
An intersteller spacecraft far away from the influence of any star or planet is moving at high speed under the influence of fusion rockets (due to thrust exerted by fusion rockets, the space crafts is accelerating). Suddenly the engine malfunctions and stops. The spacecraft will
A)
immediately stops, throwing all of the occupants to the front
done
clear
B)
begins slowing down and eventually comes to rest
done
clear
C)
keep moving at constant speed for a while, and then begins to slow down
done
clear
D)
keeps moving forever with constant speed
done
clear
View Answer play_arrow
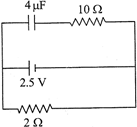
A capacitor of \[4\,\mu F\] is connected as shown in the circuit. The internal resistance of the battery is \[0.5\Omega .\] The amount of charge on the capacitor plates will be
A)
\[0\mu C\]
done
clear
B)
\[4\mu C\]
done
clear
C)
\[16\mu C\]
done
clear
D)
\[8\mu C\]
done
clear
View Answer play_arrow
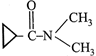
IUPAC name of the following compound:
A)
N, N-dimethylcyclopropanecarboxamide
done
clear
B)
N-methylcyclopropanamide
done
clear
C)
cyclopropionamide
done
clear
D)
none of the above
done
clear
View Answer play_arrow
Match the columns
Column-I Column-II [A] Argyrol (p) Kalazar [B] Antimony (q) Intramuscular injection [C] Colloidal gold (r) Stomach disorders [D] Milk of magnesia (s) Eye lotion
A)
A-(r), B-(p), C-(s), D-(q)
done
clear
B)
A-(r), B-(p), C-(q), D-(s)
done
clear
C)
A-(s), B-(q), C-(p) D-(s)
done
clear
D)
A-(s), B-(p), C-(q), D-(r)
done
clear
View Answer play_arrow
A is a lighter phenol and B is an aromatic carboxylic acid. Separation of a mixture of A and B can be carried out easily by using a solution of:
A)
Sodium hydroxide
done
clear
B)
Sodium sulphate
done
clear
C)
Calcium chloride
done
clear
D)
Sodium bicarbonate
done
clear
View Answer play_arrow
\[(Ag+Pb)\,alloy\xrightarrow[Zinc\,\,is\,\,added]{Melt\,\,and}\] \[(Ag+Pb+Zn)\,melt\xrightarrow{Cool}\,\,\xrightarrow[Layer\,\,Y]{Layer\,\,X}\] Select correct statement based on above scheme.
A)
Layer X contains Zn and Ag
done
clear
B)
Layer Y contains Pb and Ag but amount of silver in this layer is smaller than in layer X
done
clear
C)
X and Y are immiscible layers
done
clear
D)
All are correct statements
done
clear
View Answer play_arrow
The rate of a reaction A doubles on increasing the temperature from 300 to 310 K. By how much, the temperature of reaction B should be increased from 300 K so that rate doubles if activation energy of the reaction B is twice to that of reaction A.
A)
9.84 K
done
clear
B)
4.92 K
done
clear
C)
2.45 K
done
clear
D)
19.67 K
done
clear
View Answer play_arrow
Which of the following is not correct? Silent electric
A)
\[3{{O}_{2}}\underset{disch\arg e}{\overset{Silent\,\,electric}{\longleftrightarrow}}2{{O}_{3}};\,\,\Delta H=+284.5\,kJ.\]
done
clear
B)
Ozone undergoes addition reaction with unsaturated carbon compounds.
done
clear
C)
Sodium thiosulphate reacts with L to form sodium tetrathionate and sodium iodide.
done
clear
D)
Ozone oxidises lead sulphide to lead sulphate.
done
clear
View Answer play_arrow
(I) \[n=3,\] \[l=2,\]\[{{m}_{1}}=-2\] (II) \[n=3,\] \[l=1,\]\[{{m}_{1}}=0\] (III) \[n=3,\] \[l=0,\] \[{{m}_{1}}=-1\] (IV) \[n=3,\] \[l=2,\] \[{{m}_{1}}=0\] (V) \[n=3,\] \[l=3,\] \[{{m}_{1}}=-2\] Of these question state designation which does not describe an allowed state for an electron in an atom?
A)
I and IV
done
clear
B)
III and V
done
clear
C)
II and V
done
clear
D)
IV and V
done
clear
View Answer play_arrow
Which of the following compounds is a good conductor of electricity in solution state?
A)
covalent
done
clear
B)
molecular solid
done
clear
C)
metallic solid
done
clear
D)
ionic compounds
done
clear
View Answer play_arrow
Which is the most suitable reagent among the following to distinguish compound (3) from rest of the compounds? 1. \[C{{H}_{3}}-C\equiv C-C{{H}_{3}}\] 2. \[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\] 3. \[C{{H}_{3}}-C{{H}_{2}}C\equiv CH\] 4. \[C{{H}_{3}}-CH=C{{H}_{2}}.\]
A)
Bromine in carbon tetrachloride
done
clear
B)
Bromine in acetic acid
done
clear
C)
Alk\[KMn{{O}_{4}}\]
done
clear
D)
Ammonical silver nitrate.
done
clear
View Answer play_arrow
\[{{N}_{2}}+3{{H}_{2}}\xrightarrow{{}}2N{{H}_{3}}.\] In this reaction equivalent weight of\[{{N}_{2}}\] is
A)
4.67
done
clear
B)
28
done
clear
C)
14
done
clear
D)
2.33
done
clear
View Answer play_arrow
Which of the following is/are the hazardous pollutant(s) present in automobile exhaust gases?
(i) \[{{N}_{2}}\] (ii) \[CO\] (iii) \[C{{H}_{4}}\] (iv) Oxides of nitrogen
A)
(ii) and (iii)
done
clear
B)
(i) and (ii)
done
clear
C)
(ii) and (iv)
done
clear
D)
(i) and (iii)
done
clear
View Answer play_arrow
\[Z{{n}^{2+}}\to Z{{n}_{(s)}};\,\,{{E}^{0}}=-0.76\,\,V\] \[C{{u}^{2+}}\to C{{u}_{(s)}};\,\,{{E}^{0}}=-0.34\,\,V\] Which of the following is spontaneous?
A)
\[Z{{n}^{2+}}+Cu\to Zn+C{{u}^{2+}}\]
done
clear
B)
\[C{{u}^{2+}}+Zn\to Cu+Z{{n}^{2+}}\]
done
clear
C)
\[Z{{n}^{2+}}+C{{u}^{2+}}\to Zn+Cu\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
In the form of dichromate, Cr (VI) is a strong oxidising agent in acidic medium but Mo (VI) in \[Mo{{O}_{3}}\] and W (VI) in\[W{{O}_{3}}\]are not because\[\_\_\_\_\_\_\_\_.\]
(i) Cr (VI) is more stable than Mo (VI) and W (VI). (u) Mo (VI) and W (VI) are more stable than Cr (VI). (iii) Higher oxidation states of heavier members of group-6 of transition series are more stable. (iv) Lower oxidation states of heavier members of group-6 of transition series are more stable.
A)
(i) and (ii)
done
clear
B)
(ii) and (iii)
done
clear
C)
(i) and (iv)
done
clear
D)
(ii) and (iv)
done
clear
View Answer play_arrow
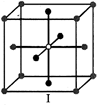
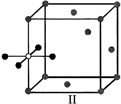
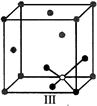
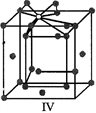
A)
I and II represent tetrahedral holes
done
clear
B)
II, III and IV represent tetrahedral holes
done
clear
C)
I and II represent octahedral holes
done
clear
D)
I, II and IV represent octahedral holes
done
clear
View Answer play_arrow
An alkene having molecular formula\[{{C}_{7}}{{H}_{14}}\]was subjected to ozonolysis in the presence of zinc dust. An equimolar amount of the following two compounds was obtained
and
The IUPAC name of the alkene is .
A)
3, 4-dimethyl-3-pentene
done
clear
B)
3, 4-dimethyl-2-pentene
done
clear
C)
2, 3-dimethyl-3-pentene
done
clear
D)
2, 3-dimethyl-2-pentene
done
clear
View Answer play_arrow
At a particular temperature, the vapour pressures of two liquids A and B are respectively 120 and 180 mm of mercury. If 2 moles of A and 3 moles of B are mixed to form an ideal solution, the vapour pressure of the solution at the same temperature will be (in mm of mercury)
A)
156
done
clear
B)
145
done
clear
C)
150
done
clear
D)
108
done
clear
View Answer play_arrow
Which of the following complex ions is expected to absorb visible light?
A)
\[{{[Ti\,\,{{(en)}_{2}}{{(N{{H}_{3}})}_{2}}]}^{4+}}\]
done
clear
B)
\[{{[Cr\,\,{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
C)
\[{{[Zn\,\,{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
done
clear
D)
\[{{[Sc\,\,{{({{H}_{2}}O)}_{3}}{{(N{{H}_{3}})}_{3}}]}^{3+}}\] (At. no. Zn = 30, Sc = 21, Ti = 22, Cr = 24)
done
clear
View Answer play_arrow
According to the adsorption theory of catalysis, the speed of the reaction increases because-
A)
Adsorption lowers the activation energy of the reaction.
done
clear
B)
The concentration of reactant molecules at the active centers of the catalyst becomes high due to strong adsorption.
done
clear
C)
In the process of adsorption, the activation energy of the molecules becomes large.
done
clear
D)
Adsorption produces heat which increases the speed of the reaction.
done
clear
View Answer play_arrow
A pressure cooker reduces cooking time for food because
A)
boiling point of water involved in cooking is increased.
done
clear
B)
the higher pressure inside the cooker crushes the food material.
done
clear
C)
cooking involves chemical changes helped by a rise in temperature.
done
clear
D)
heat is more evenly distributed in the cooking space.
done
clear
View Answer play_arrow
Most reactive halide towards\[{{S}_{N}}1\] reaction is
A)
n-Butyl chloride
done
clear
B)
sec-Butyl chloride
done
clear
C)
tert-Butyl chloride
done
clear
D)
Allyl chloride
done
clear
View Answer play_arrow
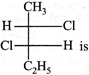
The absolute configuration of the following :
A)
2S, 3R
done
clear
B)
2S, 3S
done
clear
C)
2R, 3S
done
clear
D)
2R, 3R
done
clear
View Answer play_arrow
The rate constant k, for the reaction \[{{N}_{2}}{{O}_{5}}(g)\xrightarrow{{}}2N{{O}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\] is\[1.3\times {{10}^{-2}}{{s}^{-1}}.\] Which equation given below describes the change of\[[{{N}_{2}}{{O}_{5}}]\]with time? \[{{[{{N}_{2}}{{O}_{5}}]}_{0}}\]and\[{{[{{N}_{2}}{{O}_{5}}]}_{t}}\]correspond to concentration \[{{N}_{2}}{{O}_{5}}\]of initially and at time t.
A)
\[[{{N}_{2}}{{O}_{5}}]t={{[{{N}_{2}}{{O}_{5}}]}_{0}}+kt\]
done
clear
B)
\[{{[{{N}_{2}}{{O}_{5}}]}_{0}}={{[{{N}_{2}}{{O}_{5}}]}_{t}}{{e}^{kt}}\]
done
clear
C)
\[log{{[{{N}_{2}}{{O}_{5}}]}_{t}}=\log {{[{{N}_{2}}{{O}_{5}}]}_{0}}+kt\]
done
clear
D)
In \[\frac{{{[{{N}_{2}}{{P}_{5}}]}_{0}}}{{{[{{N}_{2}}{{P}_{5}}]}_{t}}}=kt\]
done
clear
View Answer play_arrow
Among the following the most stable compound is :
A)
cis-1, 2-cyclohexanediol
done
clear
B)
trans-1, 2-cyclohexanediol
done
clear
C)
cis-1, 3 ?cyclohexanediol
done
clear
D)
trans-1, 3-cyclohexanediol
done
clear
View Answer play_arrow
Lead is not affected by dil. HCl in cold because
A)
Pb is less electronegative than H.
done
clear
B)
PbO film is formed which resists chemical attack by acid.
done
clear
C)
\[PbC{{L}_{2}}\] protective coating gets formed on Pb surface.
done
clear
D)
\[Pb{{O}_{2}}\]film is always present on Pb surface, which resist chemical attack.
done
clear
View Answer play_arrow
Aniline is reacted with bromine water and the resulting product is treated with an aqueous solution of sodium nitrite in presence of dilute hydrochloric acid. The compound so formed is converted into a tetrafluoroborate which is subsequently heated dry. The final product is
A)
1,3,5-tribromobenzene
done
clear
B)
p-bromofluorobenzene
done
clear
C)
p-bromoaniline
done
clear
D)
2, 4, 6-tribromofluorobenzene
done
clear
View Answer play_arrow
With which one of the following elements silicon should be doped so as to give p-type of semiconductor?
A)
Germanium
done
clear
B)
Arsenic
done
clear
C)
Selenium
done
clear
D)
Boron
done
clear
View Answer play_arrow
X is identified as
A)
2, 4, 6-tribromophenol
done
clear
B)
2-bromo-4-hydroxylbenzene sulphonic acid
done
clear
C)
3, 5-dibromo-4-hydroxybenzene sulphonic acid
done
clear
D)
2-bromophenol
done
clear
View Answer play_arrow
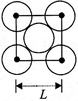
The packing efficiency of the two-dimensional square unit cell shown below is:
A)
39.27%
done
clear
B)
68.02%
done
clear
C)
74.05%
done
clear
D)
78.54%
done
clear
View Answer play_arrow
Aminoglycosides are usually used as:
A)
Antibiotic
done
clear
B)
Analgesic
done
clear
C)
Hypnotic
done
clear
D)
Antifertility
done
clear
View Answer play_arrow
Aluminium displaces hydrogen from acids but copper does not. A galvanic cell prepared by combining \[Cu/C{{u}^{2+}}\]and\[Al/A{{l}^{3+}}\]has an e.m.f of 2.0 V at \[298{}^\circ K.\] If the potential of copper electrode is \[+0.34\,\,V\] that of aluminium is
A)
\[+1.66\,\,V\]
done
clear
B)
\[-1.66\,\,V\]
done
clear
C)
\[+2.34\,\,V\]
done
clear
D)
\[-2.3\,\,V\]
done
clear
View Answer play_arrow
An organic compound X on treatment with pyridinium chlorochromate in dichloromethane gives compound Y. Compound Y reacts with \[{{I}_{2}}\]and alkali to form triiodomethane. The compound 'X' is
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
A sudden large jumps between the values of second and third ionization energies of an element, would be associated with which of the following electronic configuration?
A)
\[1{{s}^{2}},\]\[2{{s}^{2}},\]\[2{{p}^{6}},\]\[3{{s}^{1}}\]
done
clear
B)
\[1{{s}^{2}},\]\[2{{s}^{2}},\]\[2{{p}^{6}},\]\[3{{s}^{1}},\]\[3{{p}^{1}}\]
done
clear
C)
\[1{{s}^{2}},\]\[2{{s}^{2}},\]\[2{{p}^{6}},\]\[3{{s}^{2}}\]
done
clear
D)
\[1{{s}^{2}},\]\[2{{s}^{2}},\]\[2{{p}^{6}},\]\[3{{s}^{2}},\]\[3{{p}^{2}}\]
done
clear
View Answer play_arrow
Which is major product formed when acetone is heated with iodine and potassium hydroxide?
A)
lodoacetone
done
clear
B)
Acetic acid
done
clear
C)
lodoform
done
clear
D)
Acetophenone
done
clear
View Answer play_arrow
On increasing the pressure, the gas phase reaction proceed forward to re-establish equilibrium, as predicted by applying the Le-Chatelier's principle. Consider the reaction. \[{{N}_{2}}(g)+3{{H}_{2}}(g)2N{{H}_{3}}(g)\] Which of the following is correct, if the total pressure at which the equilibrium is established, is increased without changing the temperature?
A)
K will remain same
done
clear
B)
K will decrease
done
clear
C)
K will increase
done
clear
D)
K will increase initially and decrease when pressure is very high
done
clear
View Answer play_arrow
Ethanol can be prepared more easily by which reaction?
(i) \[C{{H}_{3}}C{{H}_{2}}Br+{{H}_{2}}O\xrightarrow{{}}C{{H}_{3}}C{{H}_{2}}OH\] (ii) \[C{{H}_{3}}C{{H}_{2}}Br+A{{g}_{2}}O\](in boiling water)\[\xrightarrow{{}}C{{H}_{3}}C{{H}_{2}}OH\]
A)
by (i) reaction
done
clear
B)
by (ii) reaction
done
clear
C)
Both reactions proceed at same rate
done
clear
D)
by none
done
clear
View Answer play_arrow
The oxidation states of iodine in \[HI{{O}_{4}},\]\[{{H}_{3}}I{{O}_{5}}\] and \[{{H}_{5}}I{{O}_{6}}\] are respectively
A)
+1, +3, +7
done
clear
B)
+7, +7, +3
done
clear
C)
+7, +7, +7
done
clear
D)
+7, +5, +3
done
clear
View Answer play_arrow
Which is correct statement?
A)
Starch is a polymer of \[\alpha \text{-}\]glucose.
done
clear
B)
In cyclic structure of fructose, there are four carbons and one oxygen atom.
done
clear
C)
Amylose is a component of cellulose.
done
clear
D)
Proteins are composed of only one type of amino acids.
done
clear
View Answer play_arrow
Regarding \[{{F}^{-}}\] and \[C{{l}^{-}}\] which of the following statements is/are correct?
(i) \[C{{l}^{-}}\]can give up an electron more easily than\[{{F}^{-}}.\] (ii) \[C{{l}^{-}}\]is a better reducing agent than \[{{F}^{-}}.\] (iii) \[C{{l}^{-}}\]is smaller in size than \[{{F}^{-}}.\] (iv) \[{{F}^{-}}\]can be oxidized more readily than \[C{{l}^{-}}.\]
A)
(i) and (ii)
done
clear
B)
(i), (ii) and (iv)
done
clear
C)
(iii) and (iv)
done
clear
D)
only (i)
done
clear
View Answer play_arrow
Among the following, the species having square planar geometry for central atom are
(i) \[Xe{{F}_{4}}\] (ii) \[S{{F}_{4}}\] (iii) \[{{[NiC{{l}_{4}}]}^{2-}}\] (iv) \[{{[PtC{{l}_{4}}]}^{2-}}\]
A)
(i) and (iv)
done
clear
B)
(i) and (ii)
done
clear
C)
(ii) and (iii)
done
clear
D)
(iii) and (iv)
done
clear
View Answer play_arrow
Carbon and silicon belong to group 14. The maximum coordination number of carbon in commonly occurring compounds is 4, whereas that of silicon is 6. This is due to
A)
large size of silicon.
done
clear
B)
more electropositive nature of silicon.
done
clear
C)
availability of d-orbitals in silicon.
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
Which one of the following statement is not true?
A)
In vulcanization the formation of sulphur bridges between different chains make rubber harder and stronger.
done
clear
B)
Natural rubber has the trans ?configuration at every double bond.
done
clear
C)
Buna-S is a copolymer of butadiene and styrene.
done
clear
D)
Natural rubber is a 1, 4 - polymer of isoprene.
done
clear
View Answer play_arrow
For a first order reaction, to obtain a positive slope, we need to plot {where [A] is the concentration of reactant A}
A)
\[-{{\log }_{10}}[A]\,\,vs\,\,t\]
done
clear
B)
\[-{{\log }_{e}}[A]\,\,vs\,\,t\]
done
clear
C)
\[{{\log }_{10}}[A]\,\,vs\,\,\log \,\,t\]
done
clear
D)
\[[A]\,\,vs\,\,t\]
done
clear
View Answer play_arrow
Among the following molecules
(i) \[Xe{{O}_{3}}\] (ii) \[XeO{{F}_{4}}\] (iii) \[Xe{{F}_{6}}\] Those having same number of lone pairs on Xe are
A)
(i) and (ii) only
done
clear
B)
(i) and (iii) only
done
clear
C)
(ii) and (iii) only
done
clear
D)
(i), (ii) and (iii)
done
clear
View Answer play_arrow
If the energy difference between the ground state of an atom and its excited state is\[4.4\times {{10}^{-4}}J,\] the wavelength of photon required to produce the transition :
A)
\[2.26\times {{10}^{-22}}m\]
done
clear
B)
\[1.13\times {{10}^{-22}}m\]
done
clear
C)
(e) \[4.52\times {{10}^{-26}}m\]
done
clear
D)
\[4.52\times {{10}^{-22}}m\]
done
clear
View Answer play_arrow
Which of the following is not a fat soluble vitamin?
A)
Vitamin B complex
done
clear
B)
Vitamin D
done
clear
C)
Vitamin E
done
clear
D)
Vitamin A
done
clear
View Answer play_arrow
The first organisms to appear on earth were
A)
photoautotrophs
done
clear
B)
chemoautotrophs
done
clear
C)
chemoheterotrophs
done
clear
D)
heterotrophs
done
clear
View Answer play_arrow
Which of the following bacteria is/are photosynthetic autotrophs?
A)
Purple sulphur bacteria
done
clear
B)
Green sulphur bacteria
done
clear
C)
Cyanobacteria
done
clear
D)
All of the above
done
clear
View Answer play_arrow
A water fern which is used as a green manure in rice fields is
A)
Salvinia
done
clear
B)
Mucor
done
clear
C)
Aspergillus
done
clear
D)
Azolla
done
clear
View Answer play_arrow
Which of the following does not have stomata?
A)
Hydrophytes
done
clear
B)
Mesophytes
done
clear
C)
Xerophytes
done
clear
D)
Submerged hydrophytes
done
clear
View Answer play_arrow
Which is correct about transport or conduction of substances?
A)
Organic food moves up through phloem
done
clear
B)
Organic food moves up through xylem
done
clear
C)
Inorganic food moves upwardly and downwardly through xylem
done
clear
D)
Organic food moves upwardly and downwardly through phloem
done
clear
View Answer play_arrow
The quiescent centre in root meristem serves as a
A)
site for storage of food which is utilized during maturation.
done
clear
B)
reservoir of growth hormones.
done
clear
C)
reserve for replenishment of damaged cells of the meristem.
done
clear
D)
region for absorption of water.
done
clear
View Answer play_arrow
This plant was introduced in India because of its beautiful flowers and shape of leaves but it became a notorious weed in Indian water bodies. Identify this plant.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
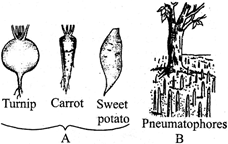
The given figures (A and B) show the modification of roots. Which of the following statements regarding the figures is correct?
A)
Tap roots of carrot, turnip and adventitious root of sweet potato, get swollen and store food.
done
clear
B)
Pneumatophores conducts water, minerals & photosynthesis
done
clear
C)
Pneumatophore is found in the plants that grow in sandy soil.
done
clear
D)
Turnip & carrot shows adventitious roots and sweet potato shows tap root.
done
clear
View Answer play_arrow
Conifers are especially sensitive to acid rain because their needles literally take moisture out of clouds. Because of acid rain, Mt. Mitchell, the highest peak in the eastern Appalachians, has seen dramatic loss of pine forest. The damage on this mountain most likely stems from
A)
drought-like conditions in the region.
done
clear
B)
excessive sulphur in the atmosphere due to industrialization along the eastern seaboard.
done
clear
C)
loss of biomass throughout the Appalachians.
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which of the following steps is not taken for reducing vehicular pollution?
A)
Use of unleaded petrol.
done
clear
B)
Use of high-sulphur petrol and diesel.
done
clear
C)
Use of catalytic converters in vehicles.
done
clear
D)
Application of stringent pollution-level norms for vehicles.
done
clear
View Answer play_arrow
Stirred tank bioreactors have been designed for
A)
addition of preservatives to the product
done
clear
B)
purification of theproduct
done
clear
C)
ensuring anaerobic conditions in the culture vessel
done
clear
D)
availability of oxygen throughout the process
done
clear
View Answer play_arrow
Which of the following pair has haploid structures?
A)
Nucellus and antipodal cells
done
clear
B)
Antipodal cells and egg cell
done
clear
C)
Antipodal cells and megaspore mother cell
done
clear
D)
Nucellus and primary endosperm nucleus
done
clear
View Answer play_arrow
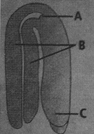
Identify the different parts of a typical dicot embryo labelled as A, B and C and select the correct option.
A)
A - Plumule B - Cotyledons C ? Radicle
done
clear
B)
A - Radicle B - Cotyledons C ? Plumule
done
clear
C)
A - Cotyledons B - Plumule C ? Radicle
done
clear
D)
A - Cotyledons B - Radicle C - Plumule
done
clear
View Answer play_arrow
Double fertilization is fusion of
A)
two eggs
done
clear
B)
two eggs and polar nuclei with pollen nuclei
done
clear
C)
one male gamete with egg and other with synergid
done
clear
D)
one male gamete with egg and other with secondary nucleus
done
clear
View Answer play_arrow
Acacia trees endure a 7-month dry season in the African savanna by dropping their leaves. This adaptation allows the trees to
A)
conserve valuable water.
done
clear
B)
trap extra carbon dioxide.
done
clear
C)
increase the rate of photosynthesis.
done
clear
D)
move water and nutrients upward from the roots.
done
clear
View Answer play_arrow
'Whip-tail' disease in cauliflower is noted due to deficiency of
A)
manganese
done
clear
B)
magnesium
done
clear
C)
molybdenum
done
clear
D)
nitrogen
done
clear
View Answer play_arrow
Six tubes containing preparations from animal tis- sue were set up as shown in the table. Tube con- tents
(i) glucose + homogenised cells (ii) glucose + mitochondria (iii) glucose + cytoplasm lacking organelles (iv) pyruvic acid + homogenised cells (v) pyruvic acid + mitochondria (vi) pyruvic add + cytoplasm lacking organelles After incubation, in which three tubes would carbon dioxide be produced?
A)
(i), (ii) and (iii)
done
clear
B)
(i), (iv) and (v)
done
clear
C)
(iii), (iv) and (vi)
done
clear
D)
(iii), (v) and (vi)
done
clear
View Answer play_arrow
Carbon dioxide labelled with \[^{14}C\] has been used to identify the intermediate compounds in the Calvin cycle, the light independent stage in photosynthesis. Which compound would be the first to contain the \[^{14}C?\]
A)
glucose
done
clear
B)
starch
done
clear
C)
GP(PGA)
done
clear
D)
triose phosphate
done
clear
View Answer play_arrow
Which one of the following is essential for photolysis of water?
A)
Manganese
done
clear
B)
Zinc
done
clear
C)
Copper
done
clear
D)
Boron
done
clear
View Answer play_arrow
Three or four of the following elements are absorbed by terrestrial plants as ions or components of ions. Which, if any, of the following is NOT absorbed by plants as an ion? If all are absorbed in ionic form, choose answer :
A)
Phosphorus
done
clear
B)
Carbon
done
clear
C)
Nitrogen
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
The graph below shows the relationship between diameter of stomatal aperture and rate of transpiration in still air and in moving air.
At which point on the graph is the rate of transpiration independent of stomatal aperture?
A)
A or E
done
clear
B)
C
done
clear
C)
D
done
clear
D)
B
done
clear
View Answer play_arrow
The dihybrid ratio in \[{{F}_{2}}\] - generation is
A)
1 : 1 : 1 : 1
done
clear
B)
2 : 1 : 2 : 1
done
clear
C)
3 : 1
done
clear
D)
9 : 3 : 3 : 1
done
clear
View Answer play_arrow
The zone of atmosphere that lies near the ground is
A)
troposphere
done
clear
B)
stratosphere
done
clear
C)
homosphere
done
clear
D)
ionosphere
done
clear
View Answer play_arrow
Reverse transcriptase is
A)
RNA dependent RNA polymerase
done
clear
B)
DNA dependent RNA polymerase
done
clear
C)
DNA dependent DNA polymerase
done
clear
D)
RNA dependent DNA polymerase
done
clear
View Answer play_arrow
Which of the following is a variety of Brassica resistant to white rust disease?
A)
Himgiri
done
clear
B)
Pusa komal
done
clear
C)
Pusa swamim (Karan rai)
done
clear
D)
Pusa Sadabahar
done
clear
View Answer play_arrow
A lake near a village suffered heavy mortality of fishes within a few days. Consider the following reasons for this.
(i) Lots of urea and phosphate fertilizer were used in the crops in the vicinity (ii) The area was sprayed with DDT by an aircraft (iii) The lake water turned green and stinky (iv) Phytoplankton populations in the lake declined initially thereby greatly reducing photosynthesis.
A)
(i) and (iii)
done
clear
B)
(i), (ii) and (iv)
done
clear
C)
(iii) and (iv)
done
clear
D)
(ii), (iii) and (iv)
done
clear
View Answer play_arrow
Which of the following features would be characteristic of the stratosphere but not of the troposphere?
A)
Most ozone resides here
done
clear
B)
Most water vapour resides here
done
clear
C)
Most of the mass of the atmosphere lies here
done
clear
D)
Circulation of this layer influences oceans currents
done
clear
View Answer play_arrow
Glycogen is a polymer of
A)
galactose
done
clear
B)
glucose
done
clear
C)
fructose
done
clear
D)
sucrose
done
clear
View Answer play_arrow
What is the end product of glycolysis of a glucose molecule?
A)
Fructose 1, 6-diphosphate
done
clear
B)
Pyruvate and ATP
done
clear
C)
Phosphoglyceraldehyde
done
clear
D)
Lactic acid and ATP
done
clear
View Answer play_arrow
In some succulent plants like Opuntia the R. Q. is zero because
A)
\[C{{O}_{2}}\] is released without any absorption of \[{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}}\] is absorbed but \[C{{O}_{2}}\] is not released
done
clear
C)
There is often compensation point in these plants due to thick phylloclades
done
clear
D)
None of these
done
clear
View Answer play_arrow
Electrons are picked up by an electron acceptor which passes them to an electron transport system consisting of
A)
phytochromes
done
clear
B)
cytochromes
done
clear
C)
Z-scheme
done
clear
D)
redox potential scale
done
clear
View Answer play_arrow
In humans, the hormone testosterone enters cells and binds to specific proteins, which in turn bind to specific sites on the cells' DNA. These proteins probably act to
A)
help KNA polymerase transcribe certain genes.
done
clear
B)
alter the pattern of DNA splicing.
done
clear
C)
stimulate protein synthesis.
done
clear
D)
unwind the DNA so that its genes can be transcribed.
done
clear
View Answer play_arrow
Given below is the pathway (2-scheme) of light reaction. Identify the blanks indicated by A, B, C and D.
A)
A - P 700 B - \[{{H}^{+}}\] acceptor C - P680 D - \[NAD{{P}^{+}}\]
done
clear
B)
A ? Photosystem I B - \[{{e}^{-}}\] acceptor C - Photosystem II D - \[NADP{{H}_{2}}+ATP\]
done
clear
C)
A ? Photosystem I B - \[{{H}^{+}}\] acceptor C - P700 D - \[NADPH\]
done
clear
D)
A ? Photosystem III B - \[{{e}^{-}}\] acceptor C - Photosystem I D - \[NADPH+{{H}^{+}}\]
done
clear
View Answer play_arrow
Consumption of which one of the following foods can prevent the kind of blindness associated with vitamin 'A' deficiency?
A)
'Flavr Savr' tomato
done
clear
B)
Canola
done
clear
C)
Golden rice
done
clear
D)
Bt-Brinjal
done
clear
View Answer play_arrow
Biodiversity of a geographical region represents
A)
endangered species found in the region.
done
clear
B)
the diversity in the organisms living in the region.
done
clear
C)
genetic diversity present in the dominant species of the region.
done
clear
D)
species endemic to the region.
done
clear
View Answer play_arrow
Use of transgenic plants as biological factories for the production of special chemicals is called-
A)
Molecular farming
done
clear
B)
Molecular genetics
done
clear
C)
Molecular mapping
done
clear
D)
Dry farming
done
clear
View Answer play_arrow
Which of the following sets consists of endangered wild life species of India?
A)
Egret, black boar and spotted deer
done
clear
B)
Himalayan musk deer, black buck, Indian lion and rhino
done
clear
C)
Himalayan musk deer, Indian lion, rhino and wild buffalo
done
clear
D)
Himalayan musk deer, great Indian bustard, wild ass and Kashmir stag
done
clear
View Answer play_arrow
Agarose extracted from sea weeds finds use in:
A)
Spectrophotometry
done
clear
B)
Tissue culture
done
clear
C)
PCR
done
clear
D)
Gel electrophoresis
done
clear
View Answer play_arrow
Select the wrongly matched pair of antibiotic to its source
A)
Terramycin - Streptomyces rimosus
done
clear
B)
Erythromycin - Streptomyces griseus
done
clear
C)
Neomycin - Streptomyces fradiae
done
clear
D)
Gramicidin - Bacillus brevis
done
clear
View Answer play_arrow
Which organelle is surrounded by a double phospholipid bilayer with many large pores?
A)
Nuclear envelope
done
clear
B)
Plasma membrane
done
clear
C)
Golgi apparatus
done
clear
D)
Mitochondrion
done
clear
View Answer play_arrow
Niche overlap indicates
A)
two different parasites on the same host
done
clear
B)
sharing of one or more resources between the two species
done
clear
C)
mutualism between two species
done
clear
D)
active cooperation between two species
done
clear
View Answer play_arrow
Coconut milk (coconut water) is widely used in tissue culture because it contains
A)
auxins
done
clear
B)
ethylene
done
clear
C)
cytokinin
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
The okazaki fragments in DNA chain growth
A)
polymerize in the 3' - to - 5' direction and forms replication fork
done
clear
B)
proves semi-conservative nature of DNA replication
done
clear
C)
polymerize in the 5' - to - 3' direction and explain 3' - to - 5' DNA replication
done
clear
D)
result in transcription
done
clear
View Answer play_arrow
Leghaemoglobin helps in
A)
nitrogen fixation
done
clear
B)
protecting nitrogenase from \[{{O}_{2}}\]
done
clear
C)
destroys bacteria
done
clear
D)
transport of food in plants
done
clear
View Answer play_arrow
Important objective of biotechnology in agriculture section is -
A)
To produce pest resistant varieties of plants
done
clear
B)
To increase the nitrogen content
done
clear
C)
To decrease the seed number
done
clear
D)
To increase the plant weight
done
clear
View Answer play_arrow
The combination of a true coelom and repeating body segmentation allows the annelids to do which of the following that anatomically "simpler" worms cannot?
A)
Move through loose marine sediments
done
clear
B)
Attain complex body shapes and thus locomote more precisely
done
clear
C)
Feed as external parasites on other animals
done
clear
D)
Be hermaphroditic
done
clear
View Answer play_arrow
Which bacteria is utilized in gober gas plant?
A)
Methanogens
done
clear
B)
Nitrifying bacteria
done
clear
C)
Ammonifying bacteria
done
clear
D)
Denitrifying bacteria
done
clear
View Answer play_arrow
Excretory organs of Cockroach are
A)
flame cells
done
clear
B)
nephridia
done
clear
C)
green glands
done
clear
D)
malpighian tubules
done
clear
View Answer play_arrow
What is common among silverfish, scorpion, crab and honey bee?
A)
Compound eyes
done
clear
B)
Poison gland
done
clear
C)
Jointed legs
done
clear
D)
Metamorphosis
done
clear
View Answer play_arrow
Tendons and ligaments are the example of
A)
areolar connective tissue
done
clear
B)
adipose tissue
done
clear
C)
dense regular connective tissue
done
clear
D)
loose connective tissue
done
clear
View Answer play_arrow
The type of epithelial cells which line inner surface of fallopian tubes, bronchioles and small bronchi are known as
A)
squamous epithelium
done
clear
B)
columnar epithelium
done
clear
C)
ciliated epithelium
done
clear
D)
cubical epithelium
done
clear
View Answer play_arrow
The graph below shows how gaseous exchange in an amphibian varies with temperature.
The graph shows that at all temperatures the lungs:
A)
absorb more oxygen than the skin
done
clear
B)
absorb less oxygen than the skin
done
clear
C)
release more carbon dioxide than the skin
done
clear
D)
release less carbon dioxide than the skin
done
clear
View Answer play_arrow
Mark the unmatched one
A)
Neutrophils ? Phagocytic
done
clear
B)
Basophils - Secrete histamine
done
clear
C)
Acidophils - Secrete heparin
done
clear
D)
Lymphocytes - Secrete antibodies
done
clear
View Answer play_arrow
Match Column-1 with Column-11 and select the correct option from the codes given below.
Column-I Column-II A Cleavage (I) Fertilization B Morula (II) Mitotic divisions C Polyspermy (III) Endometric D Implantation (IV) Little mulberry
A)
A\[\to \](II), B\[\to \](I), C\[\to \](IV), D\[\to \](III)
done
clear
B)
A\[\to \](I), B\[\to \](IV), C\[\to \](II), D\[\to \](III)
done
clear
C)
A\[\to \](IV), B\[\to \](II), C\[\to \](I), D\[\to \](III)
done
clear
D)
A\[\to \](II), B\[\to \](IV),C\[\to \](III), D\[\to \](I)
done
clear
View Answer play_arrow
If for some reason, the vasa efferentia in the human reproductive system get blocked, the gametes will not be transported from
A)
testes to epididymis
done
clear
B)
epididymis to vas deferens
done
clear
C)
ovary to uterus
done
clear
D)
vagina to uterus
done
clear
View Answer play_arrow
Where is one least likely to find complete semen (all components are present)?
A)
At the junction of the bulbourethral duct and the urethra
done
clear
B)
In the epididymis
done
clear
C)
At the male urethral orifice
done
clear
D)
In the vagina of a woman whose trying to become pregnant
done
clear
View Answer play_arrow
Plants of salty sea shore wet lands are :
A)
heliophytes
done
clear
B)
hydrophytes
done
clear
C)
halophytes
done
clear
D)
saprophytes
done
clear
View Answer play_arrow
Colostrum
A)
is a hormone essential for milk secretion.
done
clear
B)
can be synthesized by the newborn infant but not by a foetus.
done
clear
C)
stimulates further secretion of oxytocin for uterine contraction.
done
clear
D)
is a source of antibodies essential to develop resistance against diseases in new born babies.
done
clear
View Answer play_arrow
The diagram below shows a mammalian nephron and indicate how it functions in three parts, x, y and z. In x the adrenal glands have been removed In y the adrenal glands have been left intact (control). In z the adrenal glands have been removed and an extract of adrenal hormone produced which was then injected back into the same animal.
Which one of the following accounts for the higher rate of reabsorption of \[N{{a}^{+}}\] in z?
A)
The hormone present was purer in the extract.
done
clear
B)
The extract may have contained impurities.
done
clear
C)
The amount of hormone injected was greater than the amount of hormone in y.
done
clear
D)
The \[N{{a}^{+}}\] content of the body fluids was low in the experimental animals.
done
clear
View Answer play_arrow
The logistic population growth is expressed by the equation :
A)
\[dt/dN=Nr\left( \frac{K-N}{K} \right)\]
done
clear
B)
\[dN/dt=rN\left( \frac{K-N}{K} \right)\]
done
clear
C)
\[dN/dt=rN\]
done
clear
D)
\[dN/dt=rN\left( \frac{N-K}{N} \right)\]
done
clear
View Answer play_arrow
Which one of the following pairs of food components in humans reaches the stomach totally undigested?
A)
Starch and cellulose
done
clear
B)
Protein and starch
done
clear
C)
Starch and fat
done
clear
D)
Fat and cellulose
done
clear
View Answer play_arrow
Which one of the following is a correct matching pair?
A)
Lubb - Sharp closure of AV valves at the beginning of ventricular systole.
done
clear
B)
Dup - Sudden opening of semilunar valves at the beginning of ventricular diastole.
done
clear
C)
Pulsation of the radial artery valves in the blood vessels.
done
clear
D)
Purkinje fibers - Initiation of the heartbeat.
done
clear
View Answer play_arrow
Proximal and distal convoluted tubules are parts of
A)
Seminiferous tubules
done
clear
B)
Nephron
done
clear
C)
Oviduct
done
clear
D)
Vas deferens
done
clear
View Answer play_arrow
Which one of the components of ECG in human is correctly interpreted below?
A)
Complex QRS-One complete Pulse
done
clear
B)
Peak T - Initiation of total cardiac contraction
done
clear
C)
Peak P and Peak R together-Systolic and diastolic blood pressures
done
clear
D)
Peak P- Initiation of left atrial contraction only
done
clear
View Answer play_arrow
Which tube in the accompanying diagram of a heart correctly represents the result of a successful coronary bypass operation?
A)
A
done
clear
B)
D
done
clear
C)
B
done
clear
D)
C
done
clear
View Answer play_arrow
Deposition of uric acid crystals within the synovial joint causes:
A)
Osteoarthritis
done
clear
B)
Rheumatoid arthritis
done
clear
C)
Gout
done
clear
D)
Paralysis
done
clear
View Answer play_arrow
Homologous organs are
A)
Wings of Insects and Bat
done
clear
B)
Gills of Fish and lungs of Rabbit
done
clear
C)
Pectoral fins of Fish and fore limbs of Horse
done
clear
D)
Wings of Grosshopper and Crow
done
clear
View Answer play_arrow
In down's syndrome of a male child, the sex complement is
A)
XO
done
clear
B)
45XY
done
clear
C)
45 XX
done
clear
D)
XXY
done
clear
View Answer play_arrow
Which one of the following is a freshwater fish?
A)
Catla
done
clear
B)
Rohu
done
clear
C)
Common carp
done
clear
D)
All of these
done
clear
View Answer play_arrow
Lack of independent assortment of two genes A and B in fruit fly Drosophila is due to
A)
repulsion
done
clear
B)
recombination
done
clear
C)
linkage
done
clear
D)
crossing over
done
clear
View Answer play_arrow
Jurassic period of the mesozoic era was characterised by
A)
Radiation of reptiles and origin of mammal- like reptiles
done
clear
B)
Dinosaurs become extinct and angiosperms appeared
done
clear
C)
Flowering plants and first dinosaurs appeared
done
clear
D)
Gymnosperms were dominant plants and first birds appeared
done
clear
View Answer play_arrow
The "Cri-du-Chat" syndrome is caused by change in chromosome structure involving
A)
deletion
done
clear
B)
duplication
done
clear
C)
inversion
done
clear
D)
translocation
done
clear
View Answer play_arrow
In history of biology, human genome project led to the development of:
A)
biotechnology
done
clear
B)
biomonitoring
done
clear
C)
bioinformatics
done
clear
D)
biosystematics
done
clear
View Answer play_arrow
Look at the diagram given below.
Parts labelled as 'A', 'B', 'C', 'D' and ' E 'respectively indicate
A)
ilium, femur, tibia, pubis and sacrum
done
clear
B)
pubis, tibia, femur, ilium and sacrum
done
clear
C)
ilium, femur, tibia, pubis, and sacrum
done
clear
D)
pubis, femur, tibia, ilium and sacrum
done
clear
View Answer play_arrow
Oxygen dissociation curve of haemoglobin is
A)
Sigmoid
done
clear
B)
Hyperbolic
done
clear
C)
Linear
done
clear
D)
Hypobolic
done
clear
View Answer play_arrow
Which of the following is made up of a single bone in mammal?
A)
Dentary
done
clear
B)
Hyoid
done
clear
C)
Upper jaw
done
clear
D)
All of these
done
clear
View Answer play_arrow
Intercostal muscles are found attached with
A)
diaphragm
done
clear
B)
ribs
done
clear
C)
pleura
done
clear
D)
lungs
done
clear
View Answer play_arrow
Eustachian tube connects
A)
External ear with middle ear
done
clear
B)
External ear with internal ear
done
clear
C)
Middle ear with pharynx
done
clear
D)
Internal pharynx
done
clear
View Answer play_arrow
Match Column-1 with Column-11 and select the correct option from the codes given below.
Colunm-I Column-II A ANF (I) Regulatates blood calcium levels B MSH (II) Decreases blood pressure C GIP (III) Pigmentation D CT (IV) Inhibits gastric secretion
A)
A\[\to \](IV), B\[\to \](I), C\[\to \](II), D\[\to \](III)
done
clear
B)
A\[\to \](II), B\[\to \](I), C\[\to \](IV), D\[\to \](III)
done
clear
C)
A\[\to \](IV), B\[\to \](I), C\[\to \](III), D\[\to \](II)
done
clear
D)
A\[\to \](II), B\[\to \](III), C\[\to \](IV), D\[\to \](I)
done
clear
View Answer play_arrow
Which one of the following is not a second messenger in hormone action?
A)
Calcium
done
clear
B)
Sodium
done
clear
C)
cAMP
done
clear
D)
cGMP
done
clear
View Answer play_arrow
A genetically engineered bacteria used for clearing oil spills is:
A)
Escherisechia coli
done
clear
B)
Bacillus subtilis
done
clear
C)
Agrobacterium tumifaciens
done
clear
D)
Pseudomonas putida
done
clear
View Answer play_arrow
What is true about T-lymphocytes in mammals?
A)
These are produced in thyroid
done
clear
B)
There are three main types - cytotoxic T-cells, helper T-cells and suppressor T-cells
done
clear
C)
These originate in lymphoid tissues
done
clear
D)
They scavenge damaged cells and cellular Debris
done
clear
View Answer play_arrow
The use of Cannabis products (bhang, ganja, charas, marijuana or hashish) causes
A)
depression of brain activity and feeling of calmness
done
clear
B)
alters thoughts, perceptions and feelings
done
clear
C)
suppresses brain function and relieves pain
done
clear
D)
stimulates the nervous system and increases alterness and activity.
done
clear
View Answer play_arrow
During anaerobic digestion of organic waste, such as in producing biogas, which one of the following is left undegraded?
A)
Lipids
done
clear
B)
Lignin
done
clear
C)
Hemi-cellulose
done
clear
D)
Cellulose
done
clear
View Answer play_arrow
Carcinoma refers to
A)
benign tumours of the connective tissue
done
clear
B)
malignant tumours of the connective tissue
done
clear
C)
malignant tumours of the skin or mucous membrane
done
clear
D)
malignant tumours of the colon
done
clear
View Answer play_arrow
Atropine is a drug that causes relaxation of the smooth muscle of the digestive tract, a dry mouth sensation, and dilated pupils. Therefore, atropine is a(n)______ drug and mimics the effect of the ______ nervous system.
A)
Adrenergic, parasympathetic
done
clear
B)
Adrenergic, sympathetic
done
clear
C)
Cholinergic, parasympathetic
done
clear
D)
Cholinergic, sympathetic
done
clear
View Answer play_arrow
African sleeping sickness is due to
A)
Plasmodium vivax transmitted by Tsetse fly
done
clear
B)
Trypanosoma lewsii transmitted by Bed Bug
done
clear
C)
Trypanosoma gambiense transmitted by Glossina palpalis
done
clear
D)
Entamoeba gingivalis spread by Housefly
done
clear
View Answer play_arrow
Which one of the following are rich in fructose, calcium and some enzymes?
A)
Male accessory glands
done
clear
B)
Liver
done
clear
C)
Pancreas
done
clear
D)
Salivary glands
done
clear
View Answer play_arrow
The number of floating ribs, in the human body, is
A)
6 pairs
done
clear
B)
5 pairs
done
clear
C)
3 pairs
done
clear
D)
2 pairs
done
clear
View Answer play_arrow
Which one of the following statements is correct?
A)
Cro-Magnon man's fossil has been found in Ethiopia
done
clear
B)
Homo erectus is the ancestor of man
done
clear
C)
Neanderthal man is the direct ancestor of
done
clear
D)
Homo sapiens Australopithecus is the real ancestor of modern man
done
clear
View Answer play_arrow



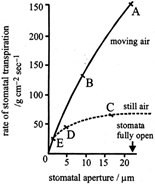 At which point on the graph is the rate of transpiration independent of stomatal aperture?
At which point on the graph is the rate of transpiration independent of stomatal aperture? 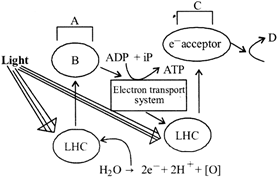
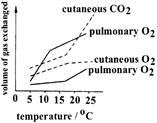 The graph shows that at all temperatures the lungs:
The graph shows that at all temperatures the lungs: 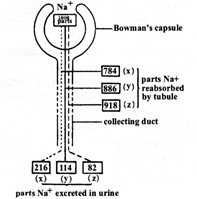 Which one of the following accounts for the higher rate of reabsorption of \[N{{a}^{+}}\] in z?
Which one of the following accounts for the higher rate of reabsorption of \[N{{a}^{+}}\] in z? 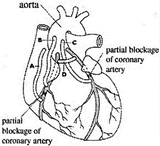
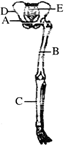 Parts labelled as 'A', 'B', 'C', 'D' and ' E 'respectively indicate
Parts labelled as 'A', 'B', 'C', 'D' and ' E 'respectively indicate 