Answer:
Aim To show reaction
between iron and steam.
Material Burner, stand,
boiling tube, cork, delivery tube, water, glass tube, glass wool an iron
sample.
Procedure
(i) Set up the apparatus as
shown in the figure.
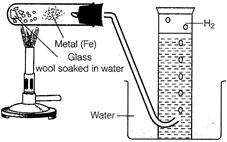 (2)
(ii) Heat the metal by using
burner
(iii) When the metal gets hot,
heat the glass wool soaked in water. The water present in glass wool forms
steam on heating which passes over the hot metal.
(iv) The hot metal reacts with
steam forming metal oxide and hydrogen gas.
(v) Hydrogen gas collects in the
glass tube over the water. When a lighted match stick is applied to the gas
collected in glass tube, the gas burns with a 'pop' sound, indicating that it
is hydrogen. (2)
(2)
(ii) Heat the metal by using
burner
(iii) When the metal gets hot,
heat the glass wool soaked in water. The water present in glass wool forms
steam on heating which passes over the hot metal.
(iv) The hot metal reacts with
steam forming metal oxide and hydrogen gas.
(v) Hydrogen gas collects in the
glass tube over the water. When a lighted match stick is applied to the gas
collected in glass tube, the gas burns with a 'pop' sound, indicating that it
is hydrogen. (2)
![]() (1)
Or
(i) Reactive metals react with
cold water and reaction results in fire.
(1)
Or
(i) Reactive metals react with
cold water and reaction results in fire.
![]() (1)
(ii) Less reactive metals also
release hydrogen, but nofire occurs.
(1)
(ii) Less reactive metals also
release hydrogen, but nofire occurs.
![]() (1)
(iii) Metals such as magnesium
react with hot water.
(1)
(iii) Metals such as magnesium
react with hot water.
![]() (1)
(iv) Less reactive metals like
Al, Zn, etc react with steam.
(1)
(iv) Less reactive metals like
Al, Zn, etc react with steam.
![]() (1)
(v) Least reactive metals like
Cu, Pt, etc do not reacteven with steam. (1)
(1)
(v) Least reactive metals like
Cu, Pt, etc do not reacteven with steam. (1)
You need to login to perform this action.
You will be redirected in
3 sec
