A) \[s{{p}^{3}}-s{{p}^{3}}\] bond
B) \[s{{p}^{3}}-p\] bond
C) \[s{{p}^{3}}-s{{p}^{2}}\] bond
D) \[s{{p}^{2}}-p\] bond
Correct Answer: B
Solution :
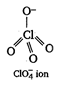 In perchlorate ion, Cl is \[s{{p}^{3}}\] hybridised therefore, all the four sigma bonds are \[s{{p}^{3}}-n\]bond.
In perchlorate ion, Cl is \[s{{p}^{3}}\] hybridised therefore, all the four sigma bonds are \[s{{p}^{3}}-n\]bond.
You need to login to perform this action.
You will be redirected in
3 sec
