question_answer 1) In which process the\[pV\]indicator diagram is a straight line parallel to volume axis
A)
isothermal
done
clear
B)
isobaric
done
clear
C)
irreversible
done
clear
D)
adiabatic
done
clear
View Answer play_arrow
question_answer 2) A body executes simple harmonic motion under the action of force\[{{F}_{1}}\]with a time period 4/5 s. If the force is changed to\[{{F}_{2}}\]it executes simple harmonic motion with time period 3/5 s. If both forces\[{{F}_{1}}\]and\[{{F}_{2}}\]act simultaneously in the same direction on the body, its time period will be
A)
\[\frac{12}{25}s\]
done
clear
B)
\[\frac{24}{25}s\]
done
clear
C)
\[\frac{35}{24}s\]
done
clear
D)
\[\frac{15}{12}s\]
done
clear
View Answer play_arrow
question_answer 3) A diatomic gas is heated at constant pressure. What fraction of the heat energy is used to increase the internal energy?
A)
\[\frac{3}{5}\]
done
clear
B)
\[\frac{3}{7}\]
done
clear
C)
\[\frac{5}{7}\]
done
clear
D)
\[\frac{5}{9}\]
done
clear
View Answer play_arrow
question_answer 4) In interference pattern, the energy is
A)
created at the maximum
done
clear
B)
destroyed at the minimum
done
clear
C)
conserved but redistributed
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 5) A red flower kept in green light will appear
A)
red
done
clear
B)
yellow
done
clear
C)
black
done
clear
D)
white
done
clear
View Answer play_arrow
question_answer 6) A band playing music at a frequency/ is moving towards a wall at a speed\[{{v}_{b}}\]. A motorist is following the band with a speed\[{{v}_{m}}\]. If v be the speed of the sound, the expression for beat frequency heard by motorist is
A)
\[\frac{v+{{v}_{m}}}{v+{{v}_{b}}}f\]
done
clear
B)
\[\frac{v+{{v}_{m}}}{v-{{v}_{b}}}f\]
done
clear
C)
\[\frac{2{{v}_{b}}(v+{{v}_{m}})}{{{v}^{2}}-v_{b}^{2}}f\]
done
clear
D)
\[\frac{2{{v}_{m}}(v+{{v}_{b}})}{{{v}^{2}}-v_{m}^{2}}f\]
done
clear
View Answer play_arrow
question_answer 7) An eye specialist prescribes spectacles having a combination of a convex lens of focal length 40 cm in contact with a concave lens of focal length 25 cm. The power of this lens combination will be
A)
\[+1.5\text{ }D\]
done
clear
B)
\[-1.5\text{ }D\]
done
clear
C)
\[+6.67\text{ }D\]
done
clear
D)
\[-6.67D\]
done
clear
View Answer play_arrow
question_answer 8) When light wave suffers reflection at the interface between air and glass, the change of phase of reflected wave is equal to
A)
zero
done
clear
B)
n/2
done
clear
C)
n
done
clear
D)
2n
done
clear
View Answer play_arrow
question_answer 9) A lens behaves as a converging lens in air and diverging lens in water. The refractive index of the material of the lens is
A)
equal to that of water
done
clear
B)
less than that of water
done
clear
C)
greater than that of water
done
clear
D)
Nothing can be predicted
done
clear
View Answer play_arrow
question_answer 10) The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photoelectron emission from this substance is approximately
A)
540 nm
done
clear
B)
400 nm
done
clear
C)
310 nm
done
clear
D)
220 nm
done
clear
View Answer play_arrow
question_answer 11) The electron emitted in beta radiation originates from
A)
inner orbits of atoms
done
clear
B)
free electron existing in nuclei
done
clear
C)
decay of neutron in the nucleus
done
clear
D)
photon escaping from the nucleus
done
clear
View Answer play_arrow
question_answer 12) If elements with principal quantum number \[n>4\]were not allowed in nature, then the number of possible elements would be
A)
32
done
clear
B)
60
done
clear
C)
18
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 13) The magnifying power of telescope is high if
A)
both objective and eye-piece have short focal length
done
clear
B)
both objective and eye-piece have long focal length
done
clear
C)
c) the objective has a long focal length and the eye-piece has a short focal length
done
clear
D)
the objective has a short focal length and the eye piece has a long focal length
done
clear
View Answer play_arrow
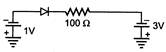
question_answer 14)
What is the current through an ideal pn-junction diode shown in figure below?
A)
Zero
done
clear
B)
10mA
done
clear
C)
20mA
done
clear
D)
50mA
done
clear
View Answer play_arrow
question_answer 15) The output form of a full wave rectifier is
A)
an AC voltage
done
clear
B)
a DC voltage
done
clear
C)
zero
done
clear
D)
a pulsating unidirectional voltage
done
clear
View Answer play_arrow
question_answer 16) Suitable impurities are added to a semiconductor depending on its use. This is done to
A)
increase its life
done
clear
B)
enable it to withstand high voltage
done
clear
C)
c) increase its electrical conductivity
done
clear
D)
increase its electrical resistivity
done
clear
View Answer play_arrow
question_answer 17) Absorption of X-rays is maximum in which of the following material sheets of same thickness?
A)
Cu
done
clear
B)
Au
done
clear
C)
Be
done
clear
D)
\[Pb\]
done
clear
View Answer play_arrow
question_answer 18) Lenz's law is a consequence of the law of conservation of
A)
charge
done
clear
B)
mass
done
clear
C)
momentum
done
clear
D)
energy
done
clear
View Answer play_arrow
question_answer 19) A magnetic needle is kept in a non-uniform magnetic field. It experience
A)
a force only but not a torque
done
clear
B)
a force and torque both
done
clear
C)
a torque only but not a force
done
clear
D)
neither a torque nor a force
done
clear
View Answer play_arrow
question_answer 20) The magnitude of magnetic induction for a current carrying toroid of uniform cross-section is
A)
uniform over the whole cross-section
done
clear
B)
maximum on the outer edge
done
clear
C)
maximum on the inner edge
done
clear
D)
maximum at the centre of cross-section
done
clear
View Answer play_arrow
question_answer 21) Isogonic lines are those for which
A)
declination is the same at all places on the line
done
clear
B)
angle of dip is the same at the place on the line
done
clear
C)
the value of horizontal component of earth's magnetic field is the same
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 22) An electric current passes through a long straight wire. At a distance 5 cm from the wire, the magnetic field is B. The field at 20 cm from the wire would be
A)
2B
done
clear
B)
B/4
done
clear
C)
B/2
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 23) An ammeter and a voltmeter of resistance R are connected in series to an electric cell of negligible internal resistance. Their readings are A and V respectively. If another resistance R is connected in parallel with the voltmeter, then
A)
both A and V will increase
done
clear
B)
both A and V will decrease
done
clear
C)
A will decrease and V will increase
done
clear
D)
A will increase and V will decrease
done
clear
View Answer play_arrow
question_answer 24) The core of transformer is laminated to reduce the effect of
A)
copper losses
done
clear
B)
flux leakage
done
clear
C)
hysteresis loss
done
clear
D)
eddy current
done
clear
View Answer play_arrow
question_answer 25) The average power dissipation in pure inductance is
A)
\[\frac{1}{2}L{{I}^{2}}\]
done
clear
B)
\[2L{{I}^{2}}\]
done
clear
C)
\[\frac{1}{4}L{{I}^{2}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 26) The charge given to any conductor resides on its outer surface, because
A)
the free charge tends to be in its minimum potential energy state
done
clear
B)
the free charge tends to be in its minimum kinetic energy state
done
clear
C)
the free charge tends to be in its maximum potential energy state
done
clear
D)
the free charge tends to be in its maximum kinetic energy state
done
clear
View Answer play_arrow
question_answer 27) \[n\]identical mercury droplets charged to the same potential V coalesce to form a single bigger drop. The potential of new drop will be
A)
\[\frac{V}{n}\]
done
clear
B)
\[nV\]
done
clear
C)
\[n{{V}^{2}}\]
done
clear
D)
\[{{n}^{2/3}}V\]
done
clear
View Answer play_arrow
question_answer 28) For protecting sensitive equipment from external magnetic field, it should be
A)
wrapped with insulation around it when passing current through it
done
clear
B)
placed inside an iron can
done
clear
C)
surrounded with Cu sheet
done
clear
D)
placed inside aluminium can
done
clear
View Answer play_arrow
question_answer 29) The potential difference across the terminals of a battery is 50 V when 11A current is drawn and 60 V when 1 A current is drawn. The emf and the internal resistance of the battery are
A)
\[62\text{ }V,\text{ }2\,\Omega \]
done
clear
B)
\[63\text{ }V,\text{1}\,\Omega \]
done
clear
C)
\[61\text{ }V,\text{ 1}\,\Omega \]
done
clear
D)
\[64\text{ }V,2\,\Omega \]
done
clear
View Answer play_arrow
question_answer 30) Four resistances\[10\,\Omega ,5\Omega ,7\Omega \]and\[3\,\Omega \]are connected so that they form the sides of a rectangle AB, BC, CD, and DA respectively. Another resistance of 10 0 is connected across the diagonal AC. The equivalent resistance between A and B is
A)
\[2\,\Omega \]
done
clear
B)
\[5\,\Omega \]
done
clear
C)
\[7\,\Omega \]
done
clear
D)
\[10\,\Omega \]
done
clear
View Answer play_arrow
question_answer 31) The potential energy of a charged parallel plate capacitor is\[{{U}_{0}}\]. If a slab of dielectric constant K is inserted between the plates, then the new potential energy will be
A)
\[\frac{{{U}_{0}}}{K}\]
done
clear
B)
\[{{U}_{0}}{{K}^{2}}\]
done
clear
C)
\[\frac{{{U}_{0}}}{{{K}^{2}}}\]
done
clear
D)
\[U_{0}^{2}\]
done
clear
View Answer play_arrow
question_answer 32) Two similar heater coils separately take 10 min to boil a certain amount of water. If both coils are connected in series, time taken to boil the same amount of water will be
A)
15 min
done
clear
B)
20 min
done
clear
C)
7.5 min
done
clear
D)
25 min
done
clear
View Answer play_arrow
question_answer 33) Same current is being passed through a copper voltameter and a silver voltameter. The rate of increase in weights of the cathode of the two voltameters will be proportional to
A)
atomic masses
done
clear
B)
atomic number
done
clear
C)
relative densities
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 34) Two equal and opposite charge (+ q and - q) are situated at x distance from each other, the value of potential at very far point will depend upon
A)
only on q
done
clear
B)
only on \[x\]
done
clear
C)
on\[qx\]
done
clear
D)
on \[\frac{q}{x}\]
done
clear
View Answer play_arrow
question_answer 35) In a potentiometer of one metre length, an unknown emf voltage source is balanced at 60 cm length of potentiometer wire, while a 3 V battery is balanced at 45 cm length. Then the emf of the unknown voltage source is
A)
3V
done
clear
B)
2.25V
done
clear
C)
4V
done
clear
D)
4.5V
done
clear
View Answer play_arrow
question_answer 36) A car travelling on a straight path moves with uniform velocity\[{{v}_{1}}\]for some time and with velocity\[{{v}_{2}}\]for next equal time, the average velocity is given by
A)
\[\sqrt{{{v}_{1}}{{v}_{2}}}\]
done
clear
B)
\[\left( \frac{{{v}_{1}}+{{v}_{2}}}{2} \right)\]
done
clear
C)
\[{{\left( \frac{1}{{{v}_{1}}}+\frac{1}{{{v}_{2}}} \right)}^{-1}}\]
done
clear
D)
\[2{{\left( \frac{1}{{{v}_{1}}}+\frac{1}{{{v}_{2}}} \right)}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 37) A particle of mass m moves in a circular path radius r under the action of a force\[\frac{m{{v}^{2}}}{r}\]. The work done during its motion over half of the circumference of the circular path will be
A)
\[\left( \frac{m{{v}^{2}}}{r} \right)\times 2\pi r\]
done
clear
B)
\[\left( \frac{m{{v}^{2}}}{r} \right)\times \pi r\]
done
clear
C)
\[\frac{(2\pi r)}{\left( \frac{m{{v}^{2}}}{r} \right)}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 38) Dimensions of self inductance are
A)
\[[ML{{T}^{-2}}{{A}^{-3}}]\]
done
clear
B)
\[[M{{L}^{-2}}{{T}^{-1}}{{A}^{-2}}]\]
done
clear
C)
\[[M{{L}^{-2}}{{T}^{-2}}{{A}^{-2}}]\]
done
clear
D)
\[[M{{L}^{2}}{{T}^{-2}}{{A}^{-1}}]\]
done
clear
View Answer play_arrow
question_answer 39) A car of mass m is moving with momentum p. If\[\mu \]be the coefficient of friction between the tyres and the road, what will be stopping distance due to friction alone?
A)
\[\frac{{{p}^{2}}}{2\mu g}\]
done
clear
B)
\[\frac{{{p}^{2}}}{2m\mu g}\]
done
clear
C)
\[\frac{{{p}^{2}}}{2{{m}^{2}}\mu g}\]
done
clear
D)
\[\frac{{{p}^{2}}}{2mg}\]
done
clear
View Answer play_arrow
question_answer 40) A neutron is moving with velocity u. It collides head on and elastically with an atom of mass number A. If the initial kinetic energy of the neutron be E, how much kinetic energy will be retained by the neutron after collision?
A)
\[{{\left( \frac{A}{A+1} \right)}^{2}}E\]
done
clear
B)
\[\frac{A}{{{(A+1)}^{2}}}E\]
done
clear
C)
\[{{\left( \frac{A-1}{A+1} \right)}^{2}}E\]
done
clear
D)
\[\frac{A-1}{{{(A+1)}^{2}}}E\]
done
clear
View Answer play_arrow
question_answer 41) If the momentum of a particle is increased by 20%, then its kinetic energy increases by
A)
44%
done
clear
B)
66%
done
clear
C)
80%
done
clear
D)
30%
done
clear
View Answer play_arrow
question_answer 42) Three point masses, each of mass M are placed at the comers of an equilateral triangle of side L. The moment of inertia of this system about an axis along one side of the triangle is
A)
\[\frac{1}{3}M{{L}^{2}}\]
done
clear
B)
\[\frac{3}{2}M{{L}^{2}}\]
done
clear
C)
\[\frac{3}{4}M{{L}^{2}}\]
done
clear
D)
\[M{{L}^{2}}\]
done
clear
View Answer play_arrow
question_answer 43) A thin circular ring of mass M and radius R is rotating about its axis with a constant angular velocity\[\omega \]. Two objects, each of mass m, are connected gently to the ring. The ring now rotates with an angular velocity
A)
\[\frac{\omega M}{M+m}\]
done
clear
B)
\[\frac{\omega (M-2M)}{(M+2m)}\]
done
clear
C)
\[\frac{\omega (M+2M)}{M}\]
done
clear
D)
\[\frac{\omega M}{M+2m}\]
done
clear
View Answer play_arrow
question_answer 44) A satellite of mass m is moving in a circular orbit of radius R above the surface of a planet of mass M and radius R. The amount of work done to shift the satellite to higher orbit of radius 2R is
A)
\[mgR\]
done
clear
B)
\[\frac{mgR}{6}\]
done
clear
C)
\[\frac{mMgR}{(M+m)}\]
done
clear
D)
\[\frac{mMgR}{6(M+m)}\]
done
clear
View Answer play_arrow
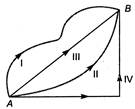
question_answer 45)
In a gravitational force field a particle is taken from A to B along different paths as shown in figure. Then
A)
work done along path I will be maximum
done
clear
B)
work done along path III will be maximum
done
clear
C)
work done along path IV will be maximum
done
clear
D)
work done along all the paths will be the same
done
clear
View Answer play_arrow
question_answer 46) A wire of length L and area of cross-section A is made of material of Young's modulus Y. If the wire is stretched by the amount\[x,\]the work done is
A)
\[\frac{YA{{x}^{2}}}{2L}\]
done
clear
B)
\[YA{{x}^{2}}L\]
done
clear
C)
\[\frac{YAx}{2L}\]
done
clear
D)
\[\frac{YA{{x}^{2}}}{L}\]
done
clear
View Answer play_arrow
question_answer 47) The potential energy of a molecule increases when it is brought to the surface from the interior of a liquid because
A)
at the free liquid surface gravitational potential energy is more
done
clear
B)
work has to be done to move a molecule to the surface against the repulsive
done
clear
C)
component of the inter molecular forces work has to be done to move a molecule to the surface against the attraction from other molecules
done
clear
D)
the temperature of the liquid surface is always more than that of the interior of the liquid
done
clear
View Answer play_arrow
question_answer 48) When a van der Waals' gas undergoes free expansion then its temperature
A)
decreases
done
clear
B)
increases
done
clear
C)
does not change
done
clear
D)
depends upon the nature of the gas
done
clear
View Answer play_arrow
question_answer 49) A cylinder of radius r and of thermal conductivity\[{{K}_{1}}\]is surrounded by a cylindrical shell of inner radius r and outer radius 2 r made of a material of thermal conductivity\[{{K}_{2}}\] The effective thermal conductivity of the system is
A)
\[\frac{1}{3}({{K}_{1}}+2{{K}_{2}})\]
done
clear
B)
\[\frac{1}{2}(2{{K}_{1}}+3{{K}_{2}})\]
done
clear
C)
\[\frac{1}{4}(3{{K}_{2}}+2{{K}_{1}})\]
done
clear
D)
\[\frac{1}{4}({{K}_{1}}+3{{K}_{2}})\]
done
clear
View Answer play_arrow
question_answer 50) The tungsten filament of an electric lamp has a surface area A and a power rating P. If the emissivity of the filament is\[\varepsilon \]and\[\sigma \]is Stefan's constant, the steady temperature of the tungsten will be
A)
\[T={{\left( \frac{P}{A\varepsilon \sigma } \right)}^{4}}\]
done
clear
B)
\[T=\left( \frac{P}{A\varepsilon \sigma } \right)\]
done
clear
C)
\[T={{\left( \frac{A\varepsilon \sigma }{P} \right)}^{\frac{1}{4}}}\]
done
clear
D)
\[T={{\left( \frac{P}{A\varepsilon \sigma } \right)}^{\frac{1}{4}}}\]
done
clear
View Answer play_arrow
question_answer 51) Glauber's salt is
A)
\[N{{a}_{2}}S{{O}_{4}}.10{{H}_{2}}O\]
done
clear
B)
\[N{{a}_{2}}{{S}_{2}}{{O}_{3}}.5{{H}_{2}}O\]
done
clear
C)
\[CuS{{O}_{4}}.5{{H}_{2}}O\]
done
clear
D)
\[N{{a}_{2}}{{B}_{4}}{{O}_{7}}.10{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 52) The most basic element is
A)
fluorine
done
clear
B)
iodine
done
clear
C)
chlorine
done
clear
D)
bromine
done
clear
View Answer play_arrow
question_answer 53) Ammonia is a Lewis base and it forms complexes with many cations. Which one of the following cations does not form a complex with ammonia?
A)
\[A{{g}^{+}}\]
done
clear
B)
\[C{{u}^{2+}}\]
done
clear
C)
\[C{{d}^{2+}}\]
done
clear
D)
\[P{{b}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 54) Which of the following substances consists of only one element?
A)
Marble
done
clear
B)
Sand
done
clear
C)
Diamond
done
clear
D)
Glass
done
clear
View Answer play_arrow
question_answer 55) Which one of the following cations gives a brick red flame by flame test?
A)
\[B{{a}^{2+}}\]
done
clear
B)
\[S{{r}^{2+}}\]
done
clear
C)
\[C{{a}^{2+}}\]
done
clear
D)
\[Z{{n}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 56) For which element of first transition series, the oxidation potential value\[(M\to {{M}^{2+}}+2{{e}^{-}})\]is lowest?
A)
\[Mn\]
done
clear
B)
\[Fe\]
done
clear
C)
\[Ni\]
done
clear
D)
\[Cu\]
done
clear
View Answer play_arrow
question_answer 57) A compound of zinc which is white in cold state and yellow in hot state, is
A)
\[ZnS\]
done
clear
B)
\[ZnO\]
done
clear
C)
\[ZnC{{l}_{2}}\]
done
clear
D)
\[ZnS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 58)
The isomer
A)
dextro isomer
done
clear
B)
laevo isomer
done
clear
C)
cis-isomer
done
clear
D)
trans-isomer
done
clear
View Answer play_arrow
question_answer 59) The compound which does not show paramagnetism, is
A)
\[[Cu{{(N{{H}_{3}})}_{4}}]C{{l}_{2}}\]
done
clear
B)
\[[Ag{{(N{{H}_{3}})}_{2}}]Cl\]
done
clear
C)
\[NO\]
done
clear
D)
\[N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 60) The equivalent weight of potassium permanganate\[(KMn{{O}_{4}})\]in neutral medium will be equal to
A)
molecular weight
done
clear
B)
\[\frac{molecular\text{ }weight}{2}\]
done
clear
C)
\[\frac{molecular\text{ }weight}{3}\]
done
clear
D)
\[\frac{molecular\text{ }weight}{5}\]
done
clear
View Answer play_arrow
question_answer 61) An element forms a solid oxide which when dissolved in water, forms an acidic solution. The element is
A)
neon
done
clear
B)
sodium
done
clear
C)
phosphorus
done
clear
D)
sulphur
done
clear
View Answer play_arrow
question_answer 62) What is the product obtained when\[MnS{{O}_{4}}\]in solution is boiled with\[Pb{{O}_{2}}\]and concentrated\[HN{{O}_{3}}\]?
A)
\[Mn{{O}_{2}}\]
done
clear
B)
\[HMn{{O}_{4}}\]
done
clear
C)
\[M{{n}_{3}}{{O}_{4}}\]
done
clear
D)
\[PbMn{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 63) Lead nitrate on heating gives lead oxide, nitrogen dioxide and oxygen. This reaction is known as
A)
combustion
done
clear
B)
combination
done
clear
C)
displacement
done
clear
D)
decomposition
done
clear
View Answer play_arrow
question_answer 64) The number of d-electrons in\[{{[Cr{{({{H}_{2}}O)}_{4}}]}^{3+}}\]is
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 65) Coordination number for copper (Cu) is
A)
1
done
clear
B)
6
done
clear
C)
8
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 66) Silver nitrate on heating gives
A)
\[AgO\]and\[N{{O}_{2}}\]
done
clear
B)
\[AgO,NO\]and\[{{O}_{2}}\]
done
clear
C)
\[Ag\]and\[N{{O}_{2}}\]
done
clear
D)
\[Ag,N{{O}_{2}}\]and\[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 67) Which one of the following is an example of a true peroxide?
A)
\[N{{O}_{2}}\]
done
clear
B)
\[Mn{{O}_{2}}\]
done
clear
C)
\[Ba{{O}_{2}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 68) The molarity of 98%\[{{H}_{2}}S{{O}_{4}}(d=1.8\text{ }g/mL)\]by weight, is
A)
6 M
done
clear
B)
18 M
done
clear
C)
10 M
done
clear
D)
4 M
done
clear
View Answer play_arrow
question_answer 69) 20 mL of\[10\text{ }N\text{ }HCl\]are mixed with 10 mL of 36 N\[{{H}_{2}}S{{O}_{4}}\]and the mixture is made 1 L. Normality of the mixture will be
A)
0.56 N
done
clear
B)
0.50 N
done
clear
C)
0.40 N
done
clear
D)
0.35 N
done
clear
View Answer play_arrow
question_answer 70) The energy of an electron in the 3rd orbit of an atom is\[-E\]. The energy of an electron in the first orbit will be
A)
\[-3E\]
done
clear
B)
\[-\frac{E}{3}\]
done
clear
C)
\[-\frac{E}{9}\]
done
clear
D)
\[-9E\]
done
clear
View Answer play_arrow
question_answer 71) Which emits\[\beta -\] pardcles?
A)
\[_{1}^{3}H\]
done
clear
B)
\[_{6}^{14}C\]
done
clear
C)
\[_{19}^{40}K\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 72) What is X in the nuclear reaction \[_{7}^{14}N+_{1}^{1}H\xrightarrow{{}}_{8}^{15}O+X?\]
A)
\[_{1}^{1}H\]
done
clear
B)
\[_{0}^{1}n\]
done
clear
C)
\[\gamma \]
done
clear
D)
\[_{-1}^{0}e\]
done
clear
View Answer play_arrow
question_answer 73) Which of the following is related with the colloidal solution?
A)
Tyndall effect
done
clear
B)
Fajan's rule
done
clear
C)
Le-Chateliefs principle
done
clear
D)
Aufbau principle
done
clear
View Answer play_arrow
question_answer 74) Who discovered that cathode rays are made up of electrons?
A)
William Crookes
done
clear
B)
G.J. Stoney
done
clear
C)
R. A. Millikan
done
clear
D)
J. J. Thomson
done
clear
View Answer play_arrow
question_answer 75) For the chemical reaction\[A\xrightarrow[{}]{{}}E,\], it is found that the rate of the reaction doubles when the concentration of A is increased four times. The order in terms of A for this reaction is
A)
two
done
clear
B)
one
done
clear
C)
zero
done
clear
D)
half
done
clear
View Answer play_arrow
question_answer 76) One mole of\[{{N}_{2}}{{O}_{4}}\]is heated in a flask with a volume of\[10\text{ }d{{m}^{3}}\]. At equilibrium 1.708 mole of\[N{{O}^{2}}\]and 0.146 mole of\[{{N}_{2}}{{O}_{4}}\]were found at\[134{}^\circ C\]. The equilibrium constant will be
A)
\[250\,mol\,d{{m}^{-3}}\]
done
clear
B)
\[300\,mol\,d{{m}^{-3}}\]
done
clear
C)
\[200\,mol\,d{{m}^{-3}}\]
done
clear
D)
\[230\,mol\,d{{m}^{-3}}\]
done
clear
E)
none of the above
done
clear
View Answer play_arrow
question_answer 77) Which one of the following is paramagnetic?
A)
\[{{O}_{2}}\]
done
clear
B)
\[{{N}_{2}}\]
done
clear
C)
\[He\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 78) The compound which is non-linear
A)
\[C{{O}_{2}}\]
done
clear
B)
\[C{{S}_{2}}\]
done
clear
C)
\[HgC{{l}_{2}}\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 79) The valency of the element having atomic number 9 is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 80) From the knowledge of the position of radium in the Periodic Table, which of the following statements would you expect to be false?
A)
\[RaS{{O}_{4}}\]is insoluble in water
done
clear
B)
\[RaS{{O}_{4}}\] is insoluble in\[HN{{O}_{3}}\]
done
clear
C)
\[RaS{{O}_{4}}\]is a white solid
done
clear
D)
\[RaS{{O}_{4}}\]is a colour less liquid
done
clear
View Answer play_arrow
question_answer 81) Hexa-2-ene-4-yne is
A)
\[C{{H}_{3}}C{{H}_{2}}C=CCH=C{{H}_{2}}\]
done
clear
B)
\[C{{H}_{3}}-C=CCH=CHC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CH=CHC=CH\]
done
clear
D)
\[C{{H}_{3}}C=CC{{H}_{2}}CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 82) The number of unpaired electrons in carbon atom is
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
question_answer 83) The end product of 4n series is
A)
\[_{82}^{208}Pb\]
done
clear
B)
\[_{82}^{207}Pb\]
done
clear
C)
\[_{82}^{209}Pb\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 84) Which statement is correct?
A)
Ethanol is more acidic than phenol
done
clear
B)
Phenol is more acidic than ethanol
done
clear
C)
Phenol is more acidic than benzoic acid
done
clear
D)
Acidity of phenol and benzoic acid is about equal
done
clear
View Answer play_arrow
question_answer 85) Which chloride is least reactive with the hydrolysis point of view?
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
C)
\[{{(C{{H}_{3}})}_{3}}CCl\]
done
clear
D)
\[C{{H}_{2}}=CH-Cl\]
done
clear
View Answer play_arrow
question_answer 86) The reaction of acetone with\[PC{{l}_{5}}\]gives
A)
\[C{{H}_{3}}COC{{H}_{2}}Cl\]
done
clear
B)
\[C{{H}_{3}}COCHC{{l}_{2}}\]
done
clear
C)
done
clear
D)
\[ClC{{H}_{2}}COC{{H}_{2}}Cl\]
done
clear
View Answer play_arrow
question_answer 87) Towards electrophilic reagents
A)
ethene is more reactive than ethyne
done
clear
B)
ethene is less reactive than ethyne
done
clear
C)
both have equal reactivity
done
clear
D)
the reactivity of both cannot be predicted
done
clear
View Answer play_arrow
question_answer 88) On heating a mixture of sodium benzoate and sodalime...... is formed.
A)
toluene
done
clear
B)
phenol
done
clear
C)
benzene
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 89) In the following reaction, \[2C{{H}_{2}}=C{{H}_{2}}+{{S}_{2}}C{{l}_{2}}\to Product\] The product is
A)
mustard gas
done
clear
B)
lewisite
done
clear
C)
polythene
done
clear
D)
Teflon
done
clear
View Answer play_arrow
question_answer 90) What is the initial product of the acidic hydrolysis of a cyanide?
A)
A primary amide
done
clear
B)
Anisocyanide
done
clear
C)
An isocyanate
done
clear
D)
A nitrile
done
clear
View Answer play_arrow
question_answer 91) In the preparation of nylon-6 from cyclohexanone oxime, use is made of a rearrangement reaction. This rearrangement reaction is called
A)
Wolff rearrangement
done
clear
B)
Amadori rearrangement
done
clear
C)
Curtius rearrangement
done
clear
D)
Beckmann rearrangement
done
clear
View Answer play_arrow
question_answer 92) In the reaction, \[C{{l}_{2}}+C{{H}_{4}}\xrightarrow[{}]{hv}C{{H}_{3}}Cl+HCl\] presence of a small amount of oxygen
A)
increases the rate of reaction for a brief period of time
done
clear
B)
decreases the rate of reaction for a brief period of time
done
clear
C)
does not affect the rate of reaction
done
clear
D)
completely stops the reaction
done
clear
View Answer play_arrow
question_answer 93) An example of a lipid is
A)
lard
done
clear
B)
keratin
done
clear
C)
glutathione
done
clear
D)
oxytocin
done
clear
View Answer play_arrow
question_answer 94) Which of the following reagents can be used for distinguishing the three classes of alcohols?
A)
Fenton's reagent
done
clear
B)
Lucas reagent
done
clear
C)
Schiff?s reagent
done
clear
D)
Tollen?s reagent
done
clear
View Answer play_arrow
question_answer 95) Natural rubber is
A)
a polymer of 1, 3-butadiene
done
clear
B)
a polymer of ethylene
done
clear
C)
a polymer of 2-methyl-l, 3-butadiene
done
clear
D)
a polymer of styrene
done
clear
View Answer play_arrow
question_answer 96) Test for an ester is
A)
Biurettest
done
clear
B)
Hydroxamic acid test
done
clear
C)
Mullicken test
done
clear
D)
Liebermann nitroso test
done
clear
View Answer play_arrow
question_answer 97) Number of resonating structures for Dewa?s benzene will be
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
question_answer 98) Which of the following reactions can be used for the synthesis of an alkene?
A)
Chugaev reaction
done
clear
B)
Dakin reaction
done
clear
C)
Reimer-Tiemann reaction
done
clear
D)
Wurtz-Fittig reaction
done
clear
View Answer play_arrow
question_answer 99) The reaction,\[{{R}_{2}}C=NN{{H}_{2}}\xrightarrow[{}]{{{C}_{2}}{{H}_{5}}ONa}{{R}_{2}}C{{H}_{2}}+{{N}_{2}}\]is called
A)
Clemmensen reduction
done
clear
B)
Hunsdiecker reaction
done
clear
C)
Tischenko reaction
done
clear
D)
Wolff-Kishner reduction
done
clear
View Answer play_arrow
question_answer 100) The monomer of cellulose is
A)
fructose
done
clear
B)
galactose
done
clear
C)
glucose
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 101) Which shows polymorphism?
A)
Physalia
done
clear
B)
Trypanosoma
done
clear
C)
Termite
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 102) Secondary radial symmetry is found in
A)
Cnidaria
done
clear
B)
Jelly fish
done
clear
C)
Echinodennata
done
clear
D)
Hemichordata
done
clear
View Answer play_arrow
question_answer 103) Basic unit of classification is
A)
genus
done
clear
B)
species
done
clear
C)
order
done
clear
D)
class
done
clear
View Answer play_arrow
question_answer 104) A Connecting link between Annelida and Mollusca is
A)
Peripatus
done
clear
B)
Lepidosiren
done
clear
C)
Neopilina
done
clear
D)
Protopterus
done
clear
View Answer play_arrow
question_answer 105) Select incorrect pair
A)
Porifera ? choanocytes
done
clear
B)
Coelenterata ? nematocysts
done
clear
C)
Annelida ? segmentation
done
clear
D)
Monera ? eukaryote
done
clear
View Answer play_arrow
question_answer 106) Bilateral symmetry, metameric segmentation, coelom and open circulatory are the features of
A)
Annelids
done
clear
B)
Arthropoda
done
clear
C)
Mollusca
done
clear
D)
Echinodermata
done
clear
View Answer play_arrow
question_answer 107) Ancestor of man who first stood erect was
A)
Australopithecus
done
clear
B)
Cro-magnon
done
clear
C)
Java-ape man
done
clear
D)
Peking man
done
clear
View Answer play_arrow
question_answer 108) Core zone, buffer zone and manipulation zone are found in
A)
national park
done
clear
B)
sanctuary
done
clear
C)
tiger reserve
done
clear
D)
biosphere reserve
done
clear
View Answer play_arrow
question_answer 109) Which insecticide is more hazardous to human health ?
A)
Rotenone
done
clear
B)
Pyrcthrum
done
clear
C)
DDT
done
clear
D)
Humulin
done
clear
View Answer play_arrow
question_answer 110) Universal donor is
A)
O Rh+
done
clear
B)
O Rh-
done
clear
C)
AB Rh+
done
clear
D)
AB Rh-
done
clear
View Answer play_arrow
question_answer 111) One of these is not concerned with wild-life conservation
A)
\[\text{IVF}\]
done
clear
B)
\[\text{IUCN}\]
done
clear
C)
\[\text{WWF}\]
done
clear
D)
\[\text{IBWL}\]
done
clear
View Answer play_arrow
question_answer 112) Largest tiger population is found in
A)
Sunderban national park
done
clear
B)
Corbett national park
done
clear
C)
Ranthambhor national park
done
clear
D)
Kanha national park
done
clear
View Answer play_arrow
question_answer 113) Genetic material found in Human Immunodeficiency Virus (HIV) is
A)
double stranded RNA
done
clear
B)
single stranded RNA
done
clear
C)
double stranded DNA
done
clear
D)
single stranded DNA
done
clear
View Answer play_arrow
question_answer 114) Gigantism and acromegaly are due to
A)
hypothyroidism
done
clear
B)
hype rthyroid ism
done
clear
C)
hypopituitarism
done
clear
D)
hyperpituitarism
done
clear
View Answer play_arrow
question_answer 115) If a child is of O blood group and his father is of B blood group, the genotype of father is
A)
\[{{\text{I}}^{\text{o}}}\,{{\text{I}}^{\text{o}}}\]
done
clear
B)
\[{{\text{I}}^{A}}\,{{\text{I}}^{B}}\]
done
clear
C)
\[{{\text{I}}^{o}}\,{{\text{I}}^{B}}\]
done
clear
D)
\[{{\text{I}}^{o}}\,{{\text{I}}^{A}}\]
done
clear
View Answer play_arrow
question_answer 116) Spennatogenesis is under the regulatory in fluence of
A)
ADH
done
clear
B)
FSH
done
clear
C)
LH
done
clear
D)
STH
done
clear
View Answer play_arrow
question_answer 117) Which of the following can be controlled by using biopesticides?
A)
Insects
done
clear
B)
Diseases
done
clear
C)
Weeds
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 118) Which hormone is secreted in a woman if pregnancy has occurred ?
A)
Estrogen
done
clear
B)
Progesterone
done
clear
C)
Luteinizing hormone
done
clear
D)
Chorionic gonadotropin
done
clear
View Answer play_arrow
question_answer 119) Product of biotechnology is
A)
transgenic crops (GM crops)
done
clear
B)
humulin
done
clear
C)
biofertilizer
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 120) Phase common in aerobic and anaerobic respiration is
A)
Krebs cycle
done
clear
B)
glycolysis
done
clear
C)
glycogenolysis
done
clear
D)
ETS
done
clear
View Answer play_arrow
question_answer 121) Oxyntic cells secrete
A)
\[\text{HCl}\]
done
clear
B)
trypsin
done
clear
C)
Na OH
done
clear
D)
pepsinogen
done
clear
View Answer play_arrow
question_answer 122) Menstruation is due to sudden
A)
reduction of FSH
done
clear
B)
increase of LH
done
clear
C)
reduction in estrogen and progesterone
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 123) Correctly matched set of phylum, class and example is
A)
Protozoa?Mastigophora?Entamoeba
done
clear
B)
Mollusca?Bivalvia?Pinctata
done
clear
C)
Arthropoda?Diplopoda?Swiopendra
done
clear
D)
Chordata?Cyclosromata?Phrynosoma
done
clear
View Answer play_arrow
question_answer 124) Urea synthesis occurs in
A)
kidney
done
clear
B)
liver
done
clear
C)
brain
done
clear
D)
muscles
done
clear
View Answer play_arrow
question_answer 125) Which is common to kidney and skeleton in mammals ?
A)
Cortex
done
clear
B)
Medulla
done
clear
C)
Pelvis
done
clear
D)
Radius
done
clear
View Answer play_arrow
question_answer 126) Which is regarded as urinary bladder of embryo?
A)
Amnion
done
clear
B)
Allantois
done
clear
C)
Chorion
done
clear
D)
Yolk sac
done
clear
View Answer play_arrow
question_answer 127) Deficiency of vitamin B12 causes
A)
cheilosis
done
clear
B)
thalassemia
done
clear
C)
beri-beri
done
clear
D)
pernicious anaemia
done
clear
View Answer play_arrow
question_answer 128) Blood is a kind of
A)
areolar tissue
done
clear
B)
connective tissue
done
clear
C)
fluid connective tissue
done
clear
D)
reticular connective tissue
done
clear
View Answer play_arrow
question_answer 129) Which of these is used to control human population?
A)
Estrogen + progesterone
done
clear
B)
\[\text{IUCD}\] and \[\text{MTP}\]
done
clear
C)
Tubectomy and vasectomy
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 130) Addiction to alcohol causes
A)
cirrhosis
done
clear
B)
epilepsy
done
clear
C)
neurosis
done
clear
D)
psychosis
done
clear
View Answer play_arrow
question_answer 131) The most primitive vertebrates are
A)
ostracoderms
done
clear
B)
cephalochordates
done
clear
C)
placoderms
done
clear
D)
cyclostomes
done
clear
View Answer play_arrow
question_answer 132) Change in the number of body parts is called
A)
continuous variation
done
clear
B)
discontinuous variation
done
clear
C)
meristic variation
done
clear
D)
substantive variation
done
clear
View Answer play_arrow
question_answer 133) Which has an additional \[\text{X}\] chromosome ?
A)
Turner's syndrome
done
clear
B)
Klinefeker's syndrome
done
clear
C)
Super female
done
clear
D)
Down's syndrome
done
clear
View Answer play_arrow
question_answer 134) Origin of life occurred in
A)
Precambrian
done
clear
B)
Coenozoic
done
clear
C)
Palaeozoic
done
clear
D)
Mesozoic
done
clear
View Answer play_arrow
question_answer 135) Branch of zoology dealing with the study of fishes is called
A)
Arthrology
done
clear
B)
Ichthyology
done
clear
C)
Saurology
done
clear
D)
Herpetology
done
clear
View Answer play_arrow
question_answer 136) Theory of continuity of germplasm was propounded by
A)
Mendel
done
clear
B)
Lamarck
done
clear
C)
Weismann
done
clear
D)
Haeckel
done
clear
View Answer play_arrow
question_answer 137) Who discovered oocysts m the stomach of female Anopheles ?
A)
Ronald Ross
done
clear
B)
Charles Lavern
done
clear
C)
Golgi
done
clear
D)
Lamble
done
clear
View Answer play_arrow
question_answer 138) Epimysium, perimysium and endomysium are found in
A)
nerve
done
clear
B)
blood vessel
done
clear
C)
striated muscle
done
clear
D)
uterus
done
clear
View Answer play_arrow
question_answer 139) Endothelium is made up of
A)
squamous cells
done
clear
B)
cuboidal cells
done
clear
C)
columnar cells
done
clear
D)
stratified epithelium
done
clear
View Answer play_arrow
question_answer 140) Bipolar neurons occur in
A)
vertebrate embryos
done
clear
B)
retina of eye
done
clear
C)
brain and spinal cord
done
clear
D)
skeletal muscles
done
clear
View Answer play_arrow
question_answer 141) Glisson's capsules are found, in which organ of mammals?
A)
Stomach
done
clear
B)
Kidney
done
clear
C)
Tesris
done
clear
D)
liver
done
clear
View Answer play_arrow
question_answer 142) Production of glucose from amino acids, fatty acids and glycerol is called
A)
glycogenesis
done
clear
B)
gluconeogenesis
done
clear
C)
glycogenolysis
done
clear
D)
glycolysis
done
clear
View Answer play_arrow
question_answer 143) Which is called Hamburger shift?
A)
Hydrogen shift
done
clear
B)
Bicarbonate shift
done
clear
C)
Chloride shift
done
clear
D)
Sodium shift
done
clear
View Answer play_arrow
question_answer 144) Papillary muscles are found in mammalian
A)
auricles
done
clear
B)
ventricles
done
clear
C)
pinna
done
clear
D)
eyes
done
clear
View Answer play_arrow
question_answer 145) Which foramen is paired in mammalian brain?
A)
Foramen of Luschka
done
clear
B)
Foramen of Magendie
done
clear
C)
Foramen of Monro
done
clear
D)
Inter-ventricular foramen
done
clear
View Answer play_arrow
question_answer 146) Corpus luteum produces
A)
progesterone
done
clear
B)
estrogen
done
clear
C)
luteotropin hormone
done
clear
D)
luteinizing hormone
done
clear
View Answer play_arrow
question_answer 147) Axis vertebra is identified by
A)
sigmoid notch
done
clear
B)
deltoid ridge
done
clear
C)
odontoid process
done
clear
D)
centrum
done
clear
View Answer play_arrow
question_answer 148) Which is thickened to form organ of Cora?
A)
Reissner's membrane
done
clear
B)
Basilar membrane
done
clear
C)
Tectorial membrane
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 149) Which stage of silkworm secretes silk?
A)
Adult
done
clear
B)
Larva
done
clear
C)
Cocoon
done
clear
D)
Pupa
done
clear
View Answer play_arrow
question_answer 150) Real product of apiculture is
A)
honey
done
clear
B)
bee wax
done
clear
C)
Both Ca) and
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 151) Who coined the term 'gene??
A)
Johanssen
done
clear
B)
Watson
done
clear
C)
Morgan
done
clear
D)
Williamson
done
clear
View Answer play_arrow
question_answer 152) Protein coat of a virus enclosing nucleic acid is called
A)
plasmid
done
clear
B)
capsid
done
clear
C)
vector
done
clear
D)
genome
done
clear
View Answer play_arrow
question_answer 153) Highest number of antibiotics are produced by
A)
Bacillus
done
clear
B)
Penicillium
done
clear
C)
Streptomyces
done
clear
D)
Cephalosporium
done
clear
View Answer play_arrow
question_answer 154) Which of the following is stained using carmine?
A)
Bacteria
done
clear
B)
Diatoms
done
clear
C)
Chromosomes
done
clear
D)
Viruses
done
clear
View Answer play_arrow
question_answer 155) A mature pollen grain of Pinus has
A)
2 cells
done
clear
B)
3 cells
done
clear
C)
4 cells
done
clear
D)
5 cells
done
clear
View Answer play_arrow
question_answer 156) Polyploidy can be induced by the application of
A)
auxin
done
clear
B)
kinetin
done
clear
C)
colchicine
done
clear
D)
ethylene
done
clear
View Answer play_arrow
question_answer 157) Quantasomes are present in
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
Golgi body
done
clear
D)
lysosome
done
clear
View Answer play_arrow
question_answer 158) In mitochondria, enzyme cytochrome oxidase is present in
A)
outer membrane
done
clear
B)
perimitochondrial space
done
clear
C)
inner membrane
done
clear
D)
matrix
done
clear
View Answer play_arrow
question_answer 159) Which of the following bio-engineered bacteria is utilized for cleaning of marine oil slicks?
A)
Escherichia coli
done
clear
B)
Pseudomonas syringae
done
clear
C)
Pseudomonas putida
done
clear
D)
Rhizoctonia solani
done
clear
View Answer play_arrow
question_answer 160) Green potatoes are toxic due to
A)
phytoalexins
done
clear
B)
solanin
done
clear
C)
triazine
done
clear
D)
hormones
done
clear
View Answer play_arrow
question_answer 161) Cells obtained from cancerous tumours are known as
A)
hybridomas
done
clear
B)
myelomas
done
clear
C)
lymphocytes
done
clear
D)
monoclonal cells
done
clear
View Answer play_arrow
question_answer 162) The plant of Triricum aestivum is
A)
haploid
done
clear
B)
diploid
done
clear
C)
tetraploid
done
clear
D)
hexaploid
done
clear
View Answer play_arrow
question_answer 163) Which of the following is a total root parasite?
A)
Cuscuca
done
clear
B)
Rafflesia
done
clear
C)
Santalum
done
clear
D)
Afonocrapa
done
clear
View Answer play_arrow
question_answer 164) Which of the following tissues consist of living cells?
A)
Vessels
done
clear
B)
Tracheids
done
clear
C)
Companion cell
done
clear
D)
Sclerenchyma
done
clear
View Answer play_arrow
question_answer 165) Which is a useful product of epidermal origin?
A)
Saffron
done
clear
B)
Cotton fibres
done
clear
C)
Clove
done
clear
D)
Jute
done
clear
View Answer play_arrow
question_answer 166) Fern spores are usually
A)
haploid
done
clear
B)
diploid
done
clear
C)
triploid
done
clear
D)
tetraploid
done
clear
View Answer play_arrow
question_answer 167) When pollen tube enters through micropyle, the process is called
A)
porogamy
done
clear
B)
chalazogamy
done
clear
C)
mesogamy
done
clear
D)
apogamy
done
clear
View Answer play_arrow
question_answer 168) Outer wall of pollen grain is made up of
A)
cellulose
done
clear
B)
sporopollenin
done
clear
C)
peclocellulose
done
clear
D)
lignin
done
clear
View Answer play_arrow
question_answer 169) Nucleotides are formed by
A)
purine, sugar and phosphate
done
clear
B)
purine, pyrimidine and phosphate
done
clear
C)
purine, pyrimidine, sugar and phosphate
done
clear
D)
pyrimidine, sugar and phosphate
done
clear
View Answer play_arrow
question_answer 170) DNA replication occurs in
A)
\[{{\text{G}}_{\text{1}}}\text{-}\]phase
done
clear
B)
S-phase
done
clear
C)
\[{{\text{G}}_{\text{2}}}\text{-}\]phase
done
clear
D)
M-phase
done
clear
View Answer play_arrow
question_answer 171) Which of the following plant cells is not surrounded by a cell wall?
A)
Root hair cell
done
clear
B)
Stem hair cell
done
clear
C)
Gamete cell
done
clear
D)
Bacterial cell
done
clear
View Answer play_arrow
question_answer 172) Which of the following cell organelles stores hydrolytic enzymes?
A)
Centriole
done
clear
B)
Lysosome
done
clear
C)
Chromoplast
done
clear
D)
Chloroplast
done
clear
View Answer play_arrow
question_answer 173) A monocarpic plant is one, which:
A)
has only one carpel
done
clear
B)
flowers once in a life-time
done
clear
C)
produces only one seed
done
clear
D)
produces only one fruit
done
clear
View Answer play_arrow
question_answer 174) AIDS virus contains
A)
RNA with protein
done
clear
B)
DNA with protein
done
clear
C)
DNA without protein
done
clear
D)
DNA only
done
clear
View Answer play_arrow
question_answer 175) Calyptra develops from
A)
venter wall of archegonium
done
clear
B)
outgrowth of gametophyte
done
clear
C)
neck wall of archegonium
done
clear
D)
paraphysis of the archegonial branch
done
clear
View Answer play_arrow
question_answer 176) Protonema is the stage in the life-cycle of
A)
Cycas
done
clear
B)
Funaria
done
clear
C)
Selaginella
done
clear
D)
Mucor
done
clear
View Answer play_arrow
question_answer 177) A fem differs from a moss in having
A)
swimming archegonia
done
clear
B)
swimming antherozoids
done
clear
C)
independent gametophytes
done
clear
D)
independent sporophytes
done
clear
View Answer play_arrow
question_answer 178) Female cone of pinus is a
A)
modified needles
done
clear
B)
modified long shoot
done
clear
C)
modified dwarf shoot
done
clear
D)
modified scale
done
clear
View Answer play_arrow
question_answer 179) Development of an embryo without fertilization is called as
A)
apomixis
done
clear
B)
polyembryony
done
clear
C)
parthenocarpy
done
clear
D)
parthenogenesis
done
clear
View Answer play_arrow
question_answer 180) Which of the following floras parts forms pericarp after fertilization?
A)
Nucellus
done
clear
B)
Outer integument
done
clear
C)
Ovary wall
done
clear
D)
Inner integument
done
clear
View Answer play_arrow
question_answer 181) Prothallus of the fem produces
A)
spores
done
clear
B)
gametes
done
clear
C)
Both and
done
clear
D)
cones
done
clear
View Answer play_arrow
question_answer 182) Which of the following cell organdies is associated with phocorespiration?
A)
Mitochondria
done
clear
B)
Peroxysome
done
clear
C)
Chloroplast
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 183) The thickness of unit membrane is
A)
20Å
done
clear
B)
35Å
done
clear
C)
55Å
done
clear
D)
75Å
done
clear
View Answer play_arrow
question_answer 184) Chromosomes are arranged along the equator during
A)
prophase
done
clear
B)
metaphase
done
clear
C)
anaphase
done
clear
D)
telophase
done
clear
View Answer play_arrow
question_answer 185) Width of the DNA molecule is
A)
15 Å
done
clear
B)
20 Å
done
clear
C)
25 Å
done
clear
D)
34 Å
done
clear
View Answer play_arrow
question_answer 186) In gymnosperms, the ovule is naked because
A)
ovary wall is absent
done
clear
B)
integuments are absent
done
clear
C)
perianth is absent
done
clear
D)
nucellus is absent
done
clear
View Answer play_arrow
question_answer 187) In Funaria capsule, dispersal of spores takes place through
A)
peristomial teeth
done
clear
B)
annulus
done
clear
C)
calyptra
done
clear
D)
operculum
done
clear
View Answer play_arrow
question_answer 188) Crossing over occurs during
A)
leptotene
done
clear
B)
diplotene
done
clear
C)
pachytene
done
clear
D)
zygotene
done
clear
View Answer play_arrow
question_answer 189) Genes are made up of
A)
histones
done
clear
B)
hydrocarbons
done
clear
C)
polynucleotides
done
clear
D)
lipoproteins
done
clear
View Answer play_arrow
question_answer 190) The site of photosynthesis in blue-green algae is
A)
chromatophores
done
clear
B)
mitochondria
done
clear
C)
chloroplast
done
clear
D)
root hair
done
clear
View Answer play_arrow
question_answer 191) Viral infection is usually absent in
A)
phloem cells
done
clear
B)
xylem cells
done
clear
C)
pith cells
done
clear
D)
apical meristem
done
clear
View Answer play_arrow
question_answer 192) Clamp connections are found in
A)
Phycomycetes
done
clear
B)
Ascomycetes
done
clear
C)
Basidiomyceies
done
clear
D)
Deuteromycetes
done
clear
View Answer play_arrow
question_answer 193) Which of the following characters is related with telophase?
A)
Formation of nuclear membrane
done
clear
B)
Formation of nucleolus
done
clear
C)
Elongation of chromosome
done
clear
D)
Formation of two daughter nuclei
done
clear
View Answer play_arrow
question_answer 194) In case of incomplete dominance, what will be the phenotypic ratio of F2 generation?
A)
3 : 1
done
clear
B)
1 : 2 : 1
done
clear
C)
1 :1: 1 :1
done
clear
D)
2 : 2
done
clear
View Answer play_arrow
question_answer 195) Which of the following does not contain DNA?
A)
Mitochondria
done
clear
B)
Chloroplast
done
clear
C)
Peroxysome
done
clear
D)
Nucleus
done
clear
View Answer play_arrow
question_answer 196) Genes exhibiting multiple effects are known as
A)
complementary genes
done
clear
B)
pleiotropic genes
done
clear
C)
cistrons
done
clear
D)
pseudogenes
done
clear
View Answer play_arrow
question_answer 197) Who coined the term 'cistron??
A)
Muller
done
clear
B)
Benzer
done
clear
C)
Khorana
done
clear
D)
Sutton
done
clear
View Answer play_arrow
question_answer 198) Which of the following is responsible for the origin of lysosome?
A)
Chloroplast
done
clear
B)
Mitochondria
done
clear
C)
Golgi body
done
clear
D)
Ribosome
done
clear
View Answer play_arrow
question_answer 199) In Selaginella, trabeculae are the modification of
A)
epidermal cells
done
clear
B)
cortical cells
done
clear
C)
endodermal cells
done
clear
D)
pericycle cells
done
clear
View Answer play_arrow
question_answer 200) Tonoplast is a membrane, which surrounds
A)
ribosome
done
clear
B)
mitochondria
done
clear
C)
vacuole
done
clear
D)
cytoplasm
done
clear
View Answer play_arrow
 Is
Is
