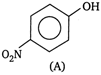
 and
and 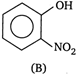
A) higher than that of A
B) lower than that of B
C) same as that of A
D) can be higher or lower depending upon the size of the vessel.
Correct Answer: A
Solution :
Due to intramolecular \[H-\]bonding in o-nitrophenol it exists as a monomer while due to intermolecular H-bonding in p-nitrophenol it exists as an associate molecule. As a result, vapour pressure of a o-nitrophenol is higher than that of p-nitrophenol.You need to login to perform this action.
You will be redirected in
3 sec
