question_answer 1) How many seconds are there in a light Fermi?
A)
\[{{10}^{-15}}s\]
done
clear
B)
\[3.0\times {{10}^{8}}s\]
done
clear
C)
\[3.33\times {{10}^{-24}}s\]
done
clear
D)
\[3.33\times {{10}^{-7}}s\]
done
clear
View Answer play_arrow
question_answer 2) A machine is delivering constant power to drive a body along a straight line. What is the relation between the distance travelled by the body against time?
A)
\[{{s}^{2}}\alpha {{t}^{3}}\]
done
clear
B)
\[{{s}^{2}}\alpha {{t}^{-3}}\]
done
clear
C)
\[{{s}^{2}}\alpha {{t}^{2}}\]
done
clear
D)
\[s\alpha {{t}^{3}}\]
done
clear
View Answer play_arrow
question_answer 3) The square of resultant of two equal forces is three times their product. Angle between the forces is
A)
\[\pi \]
done
clear
B)
\[\frac{\pi }{2}\]
done
clear
C)
\[\frac{\pi }{4}\]
done
clear
D)
\[\frac{\pi }{3}\]
done
clear
View Answer play_arrow
question_answer 4) An object placed on a ground is in stable equilibrium. If the object is given a slight push then initially the position of centre of gravity
A)
moves nearer to ground
done
clear
B)
rises higher above the ground
done
clear
C)
remains as such
done
clear
D)
may remain at same level
done
clear
View Answer play_arrow
question_answer 5) How much work must be done by a force on 50 kg body in order to accelerate it from rest to 20 m/s in 10 s ?
A)
\[{{10}^{3}}J\]
done
clear
B)
\[{{10}^{4}}J\]
done
clear
C)
\[2\times {{10}^{4}}J\]
done
clear
D)
\[4\times {{10}^{4}}J\]
done
clear
View Answer play_arrow
question_answer 6) Moment of inertia of circular loop of radius R about the axis of rotation parallel to horizontal diameter at a distance R/2 from it is
A)
\[M{{R}^{2}}\]
done
clear
B)
\[\frac{1}{2}M{{R}^{2}}\]
done
clear
C)
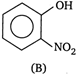
\[2M{{R}^{2}}\]
done
clear
D)
\[\frac{3}{4}M{{R}^{2}}\]
done
clear
View Answer play_arrow
question_answer 7) What will happen to the weight of the body at the South Pole, if the earth stops rotating about its polar axis?
A)
No change
done
clear
B)
Increases
done
clear
C)
Decreases but does not become zero
done
clear
D)
Reduces to zero
done
clear
View Answer play_arrow
question_answer 8) A beam of metal supported at the two ends is loaded at the centre. The depression at the centre is proportional to
A)
\[{{Y}^{2}}\]
done
clear
B)
Y
done
clear
C)
\[\frac{1}{Y}\]
done
clear
D)
\[\frac{1}{{{Y}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 9) A common hydrometer reads specific gravity of liquids. Compared to the 1.6 mark of the stem the mark 1.5 will be
A)
upwards
done
clear
B)
downwards
done
clear
C)
in the same place
done
clear
D)
may be upward or downward depending upon the hydrometer
done
clear
View Answer play_arrow
question_answer 10) A balloon contains 500\[{{m}^{3}}\] of Heat 27°C and atmospheric pressure. The volume of Heat -3°C and 0.5 atmospheric pressure will be
A)
700\[{{m}^{3}}\]
done
clear
B)
900\[{{m}^{3}}\]
done
clear
C)
1000\[{{m}^{3}}\]
done
clear
D)
500\[{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 11) Which of the following is different from others?
A)
Wavelength
done
clear
B)
Velocity
done
clear
C)
Frequency
done
clear
D)
Amplitude
done
clear
View Answer play_arrow
question_answer 12) Two pendulums have time periods T and 57/4. They starts SHM at the same time from the mean position. What will be the phase difference between them after the bigger pendulum completed one oscillation?
A)
\[45{}^\circ \]
done
clear
B)
\[90{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[30{}^\circ \]
done
clear
View Answer play_arrow
question_answer 13) A balloon is filled with hydrogen. For sound waves, this balloon behaves like
A)
a converging lens
done
clear
B)
a diverging lens
done
clear
C)
a concave mirror
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 14) Each of the two point charges are doubled and their distance is halved. Force of interaction becomes n times, where n is
A)
4
done
clear
B)
1
done
clear
C)
1/16
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 15) Two soap bubbles have radii in the ratio of 2:1. What is the ratio of excess pressures inside them?
A)
1 : 2
done
clear
B)
1 : 4
done
clear
C)
2 : 1
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
question_answer 16) The phenomenon of Brownian movement may be taken as evidence of
A)
kinetic theory of matter
done
clear
B)
EMT of radiation
done
clear
C)
corpuscular theory of light
done
clear
D)
photoelectric phenomenon
done
clear
View Answer play_arrow
question_answer 17) Two sound waves of slightly different frequencies propagating in the same direction produce beats due to
A)
interference
done
clear
B)
diffraction
done
clear
C)
reflection
done
clear
D)
refraction
done
clear
View Answer play_arrow
question_answer 18) An ice block floats in a liquid whose density is less than water. A part of block is outside the liquid. When whole of ice has melted, the liquid level will
A)
rise
done
clear
B)
go down
done
clear
C)
remain same
done
clear
D)
first rise then go down
done
clear
View Answer play_arrow
question_answer 19) Two bodies of different masses of 2 kg and 4 kg moving with velocities 2 m/s and 10 m/s towards each other due to mutual gravitational attraction. What is the velocity of their centre of mass?
A)
5 m/s
done
clear
B)
6 m/s
done
clear
C)
8 m/s
done
clear
D)
Zero
done
clear
View Answer play_arrow
question_answer 20) Given that the displacement of an oscillating particle is given by \[y=A\sin (Bx+Ct+D).\] The dimensional formula for (ABCD) is
A)
\[\left[ {{M}^{0}}{{L}^{-1}}{{T}^{0}} \right]\]
done
clear
B)
\[\left[ {{M}^{0}}{{L}^{0}}{{T}^{-1}} \right]\]
done
clear
C)
\[\left[ {{M}^{0}}{{L}^{-1}}{{T}^{-1}} \right]\]
done
clear
D)
\[\left[ {{M}^{0}}{{L}^{0}}{{T}^{0}} \right]\]
done
clear
View Answer play_arrow
question_answer 21) Two waves having intensities in the ratio of 9:1 produce interference. The ratio of maximum to minimum intensity is equal to
A)
10 : 8
done
clear
B)
9 : 1
done
clear
C)
4 : 1
done
clear
D)
2 : 1
done
clear
View Answer play_arrow
question_answer 22) Four wires each of same length, diameter and material are connected to each other to form a square. If the resistance of each wire is R, then equivalent resistance across the opposite comersis
A)
R
done
clear
B)
R/2
done
clear
C)
R/4
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 23) An electric motor runs on DC source of emf 200 V and draws a current of 10 A. If the efficiency be 40% then the resistance of armature is
A)
2 \[\Omega \]
done
clear
B)
8 \[\Omega \]
done
clear
C)
12 \[\Omega \]
done
clear
D)
16 \[\Omega \]
done
clear
View Answer play_arrow
question_answer 24) A capacitor having capacity of 2.0\[\mu F\] is charged to 200 V and then the plates of the capacitor are connected to a resistance wire. The heat produced in joule will be
A)
\[2\times {{10}^{-2}}\]
done
clear
B)
\[4\times {{10}^{-2}}\]
done
clear
C)
\[4\times {{10}^{4}}\]
done
clear
D)
\[4\times {{10}^{10}}\]
done
clear
View Answer play_arrow
question_answer 25) A voltmeter of range 2V and resistance 300\[\Omega \]cannot be converted into ammeter of range
A)
1 A
done
clear
B)
1 mA
done
clear
C)
100 mA
done
clear
D)
10 mA
done
clear
View Answer play_arrow
question_answer 26) If a magnet is suspended at angle 30° to the magnetic meridian, the dip needle makes angle of 45° with the horizontal. The real dip is
A)
\[{{\tan }^{-1}}(\sqrt{3/2})\]
done
clear
B)
\[{{\tan }^{-1}}(\sqrt{3})\]
done
clear
C)
\[{{\tan }^{-1}}(\sqrt{3/2})\]
done
clear
D)
\[{{\tan }^{-1}}(2/\sqrt{3})\]
done
clear
View Answer play_arrow
question_answer 27) Which quantity is increased in step-down transformer?
A)
Current
done
clear
B)
Voltage
done
clear
C)
Power
done
clear
D)
Frequency
done
clear
View Answer play_arrow
question_answer 28) The ratio of intensity at the centre of a bright fringe to the intensity at a point distant one fourth of the distance between two successive bright fringes will be
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 29) Which has more luminous efficiency?
A)
A 40 W bulb
done
clear
B)
A 40 W fluorescent tube
done
clear
C)
Both have same
done
clear
D)
cannot say
done
clear
View Answer play_arrow
question_answer 30) When a ray of light enters from one medium to another, then its velocity in second medium becomes double. The maximum value of angle of incidence, so that total internal reflection may not take place will be
A)
\[60{}^\circ \]
done
clear
B)
\[180{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[30{}^\circ \]
done
clear
View Answer play_arrow
question_answer 31) What should be the velocity of an electron so that its momentum becomes equal to that of a photon of wavelength \[5200\overset{\text{o}}{\mathop{\text{A}}}\,\]?
A)
700 m/s
done
clear
B)
1000 m/s
done
clear
C)
1400 m/s
done
clear
D)
2800 m/s
done
clear
View Answer play_arrow
question_answer 32) A radioactive element has half-life period of 600 years. After 3000 years, what amount will remain?
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{1}{16}\]
done
clear
C)
\[\frac{1}{8}\]
done
clear
D)
\[\frac{1}{32}\]
done
clear
View Answer play_arrow
question_answer 33) Beyond which frequency, the ionosphere bends any incident electromagnetic radiation but do not reflect it back towards the earth?
A)
50 MHz
done
clear
B)
40 MHz
done
clear
C)
30 MHz
done
clear
D)
20 MHz
done
clear
View Answer play_arrow
question_answer 34) In intrinsic semiconductor at room temperature number of electrons and holes are
A)
equal
done
clear
B)
zero
done
clear
C)
unequal
done
clear
D)
infinite
done
clear
View Answer play_arrow
question_answer 35) The unit of thermal conductance is
A)
\[W{{K}^{-1}}\]
done
clear
B)
\[J{{K}^{-1}}\]
done
clear
C)
WK
done
clear
D)
JK
done
clear
View Answer play_arrow
question_answer 36) The value of P so that the vectors \[2\mathbf{\hat{i}}-\mathbf{\hat{j}}+\mathbf{\hat{k}},\mathbf{\hat{i}}+2\mathbf{\hat{j}}-3\mathbf{\hat{k}}\] and \[3\mathbf{\hat{i}}+P\mathbf{\hat{j}}+5\mathbf{\hat{k}}\] are coplanar should be
A)
16
done
clear
B)
-4
done
clear
C)
4
done
clear
D)
-8
done
clear
View Answer play_arrow
question_answer 37) A capacitor of capacitance C has charge Q and stored .energy is W. If the charge is increased to 2Q, the stored energy will be
A)
\[\frac{W}{4}\]
done
clear
B)
\[\frac{W}{2}\]
done
clear
C)
2 W
done
clear
D)
4 W
done
clear
View Answer play_arrow
question_answer 38) Pure silicon at 300 K has equal electron ne and whole\[({{n}_{h}})\] concentration of\[1.5\times {{10}^{16}}\,\,{{m}^{-3}}\]. Doping by indium increases \[{{n}_{h}}\]to \[\,\,4.5\times {{10}^{22}}\,\,{{m}^{-3}}\]. The \[{{n}_{e}}\] in the doped silicon is
A)
\[9\times {{10}^{5}}\]
done
clear
B)
\[5\times {{10}^{9}}\]
done
clear
C)
\[2.25\times {{10}^{11}}\]
done
clear
D)
\[3\times {{10}^{19}}\]
done
clear
View Answer play_arrow
question_answer 39) A cylindrical conductor is placed near another positively charged conductor. The net charge acquired by the cylindrical conductor will be :
A)
positive only
done
clear
B)
negative only
done
clear
C)
zero
done
clear
D)
either positive or negative
done
clear
View Answer play_arrow
question_answer 40) If the unit of force is 1 kilo newton, the length is km and time 100 s, what will be the unit of mass?
A)
1,000 kg
done
clear
B)
1kg
done
clear
C)
10,000 kg
done
clear
D)
100 kg
done
clear
View Answer play_arrow
question_answer 41) The maximum tension which an in extensible ring of mass 0.1 kg/m can bear is 10 N. The maximum velocity in m/s with which it can be rotated is
A)
10
done
clear
B)
\[\sqrt{10}\]
done
clear
C)
20
done
clear
D)
15
done
clear
View Answer play_arrow
question_answer 42) If there were a reduction in gravitational effect, which of the following forces do you think would change in some respect?
A)
Magnetic force
done
clear
B)
Electrostatic force
done
clear
C)
Viscous force
done
clear
D)
Archimedes uplift
done
clear
View Answer play_arrow
question_answer 43) The breaking force for a wire of diameter D of a material is F. The breaking force for a wire of the same material of radius D is
A)
F
done
clear
B)
2F
done
clear
C)
\[\frac{F}{4}\]
done
clear
D)
4F
done
clear
View Answer play_arrow
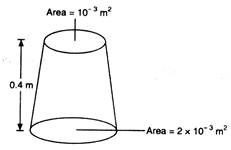
question_answer 44)
A uniformly tapering vessel is filled with a liquid of density \[900\text{ }kg/{{m}^{3}}.\] The force that acts on the base of the vessel due to the liquid is\[(g=10\text{ }m/{{s}^{2}})\]
A)
3.6 N
done
clear
B)
7.2 N
done
clear
C)
9.0 N
done
clear
D)
14.4 N
done
clear
View Answer play_arrow
question_answer 45) If pressure of a gas contained in a closed vessel is increased by 0.4% when heated by \[1{}^\circ C\], its initial temperature must be
A)
250 K
done
clear
B)
\[250{}^\circ C\]
done
clear
C)
2500 K
done
clear
D)
\[25{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 46) Lines of force due to earths horizontal magnetic field are
A)
parallel and straight
done
clear
B)
concentric circles
done
clear
C)
elliptical
done
clear
D)
curved lines
done
clear
View Answer play_arrow
question_answer 47) Two thermometers are constructed in the same way except that one has a spherical bulb and the other a cylindrical bulb, which one will respond quickly to temperature changes?
A)
Spherical bulb thermometer
done
clear
B)
Cylindrical bulb thermometer
done
clear
C)
Both equally
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 48) What is the fractional change in tension necessary in a sonometer of fixed length to produce a note one octave lower than before?
A)
\[\frac{1}{4}\]
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{2}{3}\]
done
clear
D)
\[\frac{3}{4}\]
done
clear
View Answer play_arrow
question_answer 49) If battery of 6 V is connected to the terminals of three metre long wire of uniform thickness and resistance of the order of 100 ohm, the difference of potential between two points separated by 50 cm in the wire will be
A)
1V
done
clear
B)
1.5 V
done
clear
C)
2 V
done
clear
D)
3 V
done
clear
View Answer play_arrow
question_answer 50) In an electromagnetic wave, the electric and magnetising fields are 100 V/m and 0.265 A/m. The maximum energy flow is
A)
26.5 W/\[{{m}^{2}}\]
done
clear
B)
36.5 W/\[{{m}^{2}}\]
done
clear
C)
46.7 W/\[{{m}^{2}}\]
done
clear
D)
765 W/\[{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 51) Ethyl acetate is obtained when methyl magnesium bromide reacts with
A)
ethyl formate
done
clear
B)
ethyl chloroformate
done
clear
C)
acetyl chloride
done
clear
D)
carbon dioxide
done
clear
View Answer play_arrow
question_answer 52) The most stable hydride is
A)
\[N{{H}_{3}}\]
done
clear
B)
\[P{{H}_{3}}\]
done
clear
C)
\[As{{H}_{3}}\]
done
clear
D)
\[Sb{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 53) The ratio of amounts of \[{{H}_{2}}S\]needed to precipitate all the metal ions from 100 mL of \[1M\,\,AgN{{O}_{3}}\]and 100 mL of 1M \[CuS{{O}_{4}}\] will be
A)
\[1:1\]
done
clear
B)
\[1:2\]
done
clear
C)
\[2:1\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 54) If the electronegativity difference between two atoms A and B is 2.0, then the percentage of co-valent character in the molecule is
A)
54%
done
clear
B)
46%
done
clear
C)
23%
done
clear
D)
72%
done
clear
View Answer play_arrow
question_answer 55) Which of the following reaction defines \[\Delta H_{f}^{o}\]?
A)
\[{{C}_{(diamond)}}+{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g)\]
done
clear
B)
\[\frac{1}{2}{{H}_{2}}(g)+\frac{1}{2}{{F}_{2}}(g)\xrightarrow{{}}HF(g)\]
done
clear
C)
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\xrightarrow{{}}2N{{H}_{2}}(g)\]
done
clear
D)
\[CO(g)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g)\]
done
clear
View Answer play_arrow
question_answer 56) Formaldehyde polymerizes to form glucose according to the reaction\[6HCHO\rightleftharpoons {{C}_{6}}{{H}_{12}}{{O}_{6}}\] The theoretically computed equilibrium constant for this reaction is found to be \[6\times {{10}^{22}}\] If 1 M solution of glucose dissociates according to the above equilibrium, the concentration of formaldehyde in the solution will be
A)
\[1.6\times {{10}^{-2}}M\]
done
clear
B)
\[1.6\times {{10}^{-4}}M\]
done
clear
C)
\[1.6\times {{10}^{-6}}M\]
done
clear
D)
\[1.6\times {{10}^{-8}}M\]
done
clear
View Answer play_arrow
question_answer 57) The electronic configuration of a dispositive ion \[{{M}^{2+}}\] is 2, 8, 14 and its mass number is 56. The number of neutrons present is
A)
32
done
clear
B)
42
done
clear
C)
30
done
clear
D)
34
done
clear
View Answer play_arrow
question_answer 58) If X is the total number of collisions which a gas molecule register with others per unit time under particular conditions, then the collision frequency of the gas containing N molecules per unit volume is
A)
X/N
done
clear
B)
NX
done
clear
C)
2 NX
done
clear
D)
NX/2
done
clear
View Answer play_arrow
question_answer 59) A hypothetical reaction \[{{A}_{2}}+{{B}_{2}}\to 2AB\] follows the mechanism as given below, \[{{A}_{2}}\rightleftharpoons A+A(fast)\] \[A+{{B}_{2}}\xrightarrow{{}}AB+B(slow)\] \[A+B\xrightarrow{{}}AB(fast)\] The order of the overall reaction is
A)
\[2\]
done
clear
B)
\[1\]
done
clear
C)
\[1\frac{1}{2}\]
done
clear
D)
\[0\]
done
clear
View Answer play_arrow
question_answer 60) The mass of helium atom of mass number 4 is 4.0026 amu, while that of the neutron and proton are 1.0087 and 1.0078 respectively on the same scale. Hence, the nuclear binding energy per nucleon in the helium atom is nearly
A)
\[5MeV\]
done
clear
B)
\[7MeV\]
done
clear
C)
\[10MeV\]
done
clear
D)
\[14MeV\]
done
clear
View Answer play_arrow
question_answer 61) Which of the following statements is correct? Dielectric constant of
A)
increases with dilution
done
clear
B)
decreases with dilution
done
clear
C)
is unaffected on dilution
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 62) For the square planar complex [M] (where, M = central metal and a, b, c and d are mono dentate ligands), the number of possible geometrical isomers are
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 63) Potash alum dissolves in water to give a/an
A)
acidic solution of \[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
alkaline solution
done
clear
C)
acidic solution of \[HCl\]
done
clear
D)
neutral solution
done
clear
View Answer play_arrow
question_answer 64) The discovery of which of the following group of elements gave death blow to the Newlands law of octaves?
A)
Inert gases
done
clear
B)
Alkaline earths
done
clear
C)
Rare earths
done
clear
D)
Actinides
done
clear
View Answer play_arrow
question_answer 65) Vant Hoff factor more than unity indicates that the solute in solution has
A)
dissociated
done
clear
B)
associated
done
clear
C)
Both (a) and (b)
done
clear
D)
cannot say anything
done
clear
View Answer play_arrow
question_answer 66) How many number of atoms are there in a cube based unit cell having one atom on each corner and two atoms on each body diagonal of cube?
A)
8
done
clear
B)
6
done
clear
C)
4
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 67) Bleeding due to a cut can be stopped by applying ferric chloride solution in the laboratory. This is due to
A)
co-agulation of negatively charged blood particles by \[F{{e}^{3+}}\] ions
done
clear
B)
co-agulation of positively charged blood particles by \[C{{l}^{-}}\] ions
done
clear
C)
reaction taking place between ferric ions and the haemoglobin forming a complex
done
clear
D)
common element, iron, in both \[FeC{{l}_{3}}\] and haemoglobin.
done
clear
View Answer play_arrow
question_answer 68) Which one of the following solutions will have highest conductivity?
A)
\[0.1M\,C{{H}_{3}}COOH\]
done
clear
B)
\[0.1M\,NaCl\]
done
clear
C)
\[0.1M\,KN{{O}_{3}}\]
done
clear
D)
\[0.1M\,HCl\]
done
clear
View Answer play_arrow
question_answer 69) One of the following metals forms a volatile compound and this property is taken advantage for its extraction. This metal is
A)
iron
done
clear
B)
nickel
done
clear
C)
cobalt
done
clear
D)
tungsten
done
clear
View Answer play_arrow
question_answer 70) If \[N{{a}^{+}}ion\] is larger than \[M{{g}^{2+}}\] ion and \[{{S}^{2-}}\]ion is larger than CF ion, which of the following will be stable soluble in water?
A)
Sodium chloride
done
clear
B)
Sodium sulphide
done
clear
C)
Magnesium chloride
done
clear
D)
Magnesium sulphide
done
clear
View Answer play_arrow
question_answer 71) Impurities of Cu and Ag from gold are removed by
A)
boiling impure gold with dil. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
boiling impure gold with cone. \[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
electrolytically
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 72) Which of the following salt would give \[S{{O}_{2}}\] with hot and dil. \[{{H}_{2}}S{{O}_{4}}\] and also decolourises \[B{{r}_{2}}\] water?
A)
\[N{{a}_{2}}S{{O}_{3}}\]
done
clear
B)
\[NaHS{{O}_{4}}\]
done
clear
C)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
D)
\[N{{a}_{2}}S\]
done
clear
View Answer play_arrow
question_answer 73) If two compounds have the same empirical formula but different molecular formulae, they must have
A)
different percentage composition
done
clear
B)
different molecular weights
done
clear
C)
same viscocity
done
clear
D)
same vapour density
done
clear
View Answer play_arrow
question_answer 74) Among the following which one has weakest carbon-halogen bond?
A)
Benzyl bromide
done
clear
B)
Bromobenzene
done
clear
C)
Vinyl bromide
done
clear
D)
Benzyl chloride
done
clear
View Answer play_arrow
question_answer 75) Petrochemicals can be used to prepare
A)
synthetic fibres
done
clear
B)
pesticides
done
clear
C)
plastics
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 76) tert-butyl methyl ether on heating with anhydrous HI in ether gives
A)
\[C{{H}_{3}}OH+{{(C{{H}_{3}})}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}I+{{(C{{H}_{3}})}_{3}}COH\]
done
clear
C)
\[C{{H}_{3}}I+{{(C{{H}_{3}})}_{3}}Cl\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 77) The correctly reported answer of the addition of 4.523, 2.3 and 6.24 will have significant figures
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 78) What happens if \[CC{{l}_{4}}\] is treated with \[AgN{{O}_{3}}\]?
A)
A white ppt. of \[AgCl\] will form
done
clear
B)
\[N{{O}_{2}}\] will be evolved
done
clear
C)
\[CC{{l}_{4}}\] will dissolve in \[AgN{{O}_{3}}\]
done
clear
D)
Nothing will happen
done
clear
View Answer play_arrow
question_answer 79) \[^{23}Na\] is more stable isotope of Na. Find out the process by which \[_{11}^{24}Na\] can undergo radioactive decay
A)
\[{{\beta }^{-}}\] emission
done
clear
B)
\[\alpha \] -emission
done
clear
C)
\[{{\beta }^{+}}\] emission
done
clear
D)
K electron capture
done
clear
View Answer play_arrow
question_answer 80) The heat of combustion of solid benzoic acid at constant volume is \[-321.30kJ\] at \[{{27}^{o}}C\]. The heat of combustion at constant pressure is
A)
\[-321.30-300R\]
done
clear
B)
\[-321.30+300R\]
done
clear
C)
\[-321.30-150R\]
done
clear
D)
\[-321.30-900R\]
done
clear
View Answer play_arrow
question_answer 81) In which of the following compounds -OH group is least reactive?
A)
done
clear
B)
done
clear
C)
done
clear
D)
All are equally reactive.
done
clear
View Answer play_arrow
question_answer 82) lodoform is obtained when ethanol is heated with
A)
\[KI\]and aq \[KOH\]
done
clear
B)
\[{{I}_{2}}\] and aq \[KOH\]
done
clear
C)
\[{{I}_{2}}\]/ aq \[KI\]
done
clear
D)
\[HI\] and \[HI{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 83) The total number of acylic isomers including the stereoisomers (geometrical and optical), with the molecular formula \[{{C}_{4}}{{H}_{7}}Cl\] is
A)
12
done
clear
B)
11
done
clear
C)
10
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 84) The alkyl halides that can be made by free radical halogenation of alkanes are
A)
RCl, and RBr but not RF or RI
done
clear
B)
RF. RCl and RBr but not RI
done
clear
C)
RF, RCl, RBr, RI
done
clear
D)
RF, RCl and RI but RBr
done
clear
View Answer play_arrow
question_answer 85) Silica is a/an
A)
acidic flux only
done
clear
B)
gangue only
done
clear
C)
basic flux only
done
clear
D)
both gangue and acidic flux
done
clear
View Answer play_arrow
question_answer 86) The nodes present in 3p-orbitals are
A)
one spherical, one planar
done
clear
B)
two spherical
done
clear
C)
two planar
done
clear
D)
one planar
done
clear
View Answer play_arrow
question_answer 87) The number of a-and p-particles emitted in nuclear reaction \[_{90}T{{h}^{228}}{{\xrightarrow{{}}}_{83}}B{{i}^{212}}\] are respectively
A)
4, 1
done
clear
B)
3, 7
done
clear
C)
8, 1
done
clear
D)
4, 7
done
clear
View Answer play_arrow
question_answer 88) Two bottles A and B contains 1 M and 1 m aqueous solution of sulphuric acid respectively
A)
A is more concentrated than B
done
clear
B)
B is more concentrated than A
done
clear
C)
concentration of A is equal to concentration of B
done
clear
D)
it is not possible to compare the concentrations
done
clear
View Answer play_arrow
question_answer 89) A salt on treatment with dil. \[HCl\] gives a pungent smelling gas and a yellow precipitate. The sak gives green flame test and a yellow precipitate with potassium chromate the salt is
A)
\[NiS{{O}_{4}}\]
done
clear
B)
\[Ba{{S}_{2}}{{O}_{3}}\]
done
clear
C)
\[Pb{{S}_{2}}{{O}_{3}}\]
done
clear
D)
\[CuS{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 90) Which of the oxide of manganese is 1 amphoteric?
A)
\[Mn{{O}_{2}}\]
done
clear
B)
\[M{{n}_{2}}{{O}_{3}}\]
done
clear
C)
\[M{{n}_{2}}{{O}_{7}}\]
done
clear
D)
\[MnO\]
done
clear
View Answer play_arrow
question_answer 91) Which of the following alkenes is most reactive , towards cationic polymerization?
A)
\[C{{H}_{2}}=CHC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{2}}=CH-C{{H}_{2}}OH\]
done
clear
C)
\[C{{H}_{3}}-CO-C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{2}}=CH-O-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 92) An organic compound, \[{{C}_{3}}{{H}_{6}}O\] does not give a precipitate with 2, 4-dinitrophenyl hydrazine reagent and does not react with metallic sodium, It could be
A)
\[C{{H}_{3}}-C{{H}_{2}}-CHO\]
done
clear
B)
\[C{{H}_{2}}=CH-C{{H}_{2}}OH\]
done
clear
C)
\[C{{H}_{3}}-CO-C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{2}}=CH-O-C{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 93) Oxidation of 1-butene with hot \[KMn{{O}_{4}}\] solution produces
A)
\[C{{H}_{3}}C{{H}_{2}}COOH+HCOOH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COOH+C{{O}_{2}}\]
done
clear
C)
\[C{{H}_{3}}COOH+C{{O}_{2}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}}C=O+C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 94) A mixture of 1-chlorobutane and 2-chloro- butane when treated with alcoholic \[KOH\] gives
A)
1-butene
done
clear
B)
2-butene
done
clear
C)
isobutylene
done
clear
D)
mixture of 1-butene + 2-butene
done
clear
View Answer play_arrow
question_answer 95)
Out of the two compounds shown below, the vapour pressure of B at a particular temperature is expected to be
A)
higher than that of A
done
clear
B)
lower than that of B
done
clear
C)
same as that of A
done
clear
D)
can be higher or lower depending upon the size of the vessel.
done
clear
View Answer play_arrow
question_answer 96) Roasted tin stone ore after washing with water is known as
A)
block tin
done
clear
B)
white tin
done
clear
C)
black tin
done
clear
D)
granulated tin
done
clear
View Answer play_arrow
question_answer 97) Which of the following has strongest hydrogen bonding ?
A)
Ethylamine
done
clear
B)
Ammonia
done
clear
C)
Ethyl alcohol
done
clear
D)
Diethyl ether
done
clear
View Answer play_arrow
question_answer 98) Consider the following statements The race law for the acid catalysed hydrolysis of an ester being given as \[Rate=k[{{H}^{+}}][ester]=k[ester]\]. If the acid concentration is doubled at constant ester concentration 1. The second order rate constant, k is doubled. 2. The pseudo first order rate constant, k is doubled. 3. The rate of the reaction is doubled Which of the above statements are correct?
A)
1 and 2
done
clear
B)
2 and 3
done
clear
C)
1 and 3
done
clear
D)
1, 2 and 3
done
clear
View Answer play_arrow
question_answer 99) A fibrous mineral which can withstand red hot flames without any damage is
A)
talc
done
clear
B)
glass wool
done
clear
C)
soap stone
done
clear
D)
asbestos
done
clear
View Answer play_arrow
question_answer 100) When \[{{o}^{-}}\] or p-phenolsulphonic acid is treated with bromine water, the product formed is
A)
2, 4-dibromophenol
done
clear
B)
2, 4, 6-tribromophenol
done
clear
C)
3-bromophenol boric acid
done
clear
D)
3, 5-dibromophenol
done
clear
View Answer play_arrow
question_answer 101) The book Genera plantarum was written by
A)
Bessey
done
clear
B)
Hutchinson
done
clear
C)
Engler and Pranti
done
clear
D)
Bentham and Hooker
done
clear
View Answer play_arrow
question_answer 102) Which of the following is not a true fish?
A)
Dogfish
done
clear
B)
Devilfish
done
clear
C)
Catfish
done
clear
D)
Sawfish
done
clear
View Answer play_arrow
question_answer 103) During mitosis, the number of chromosomes get
A)
change
done
clear
B)
no change
done
clear
C)
may be change if cell is mature
done
clear
D)
may be change if cell is immature
done
clear
View Answer play_arrow
question_answer 104) During transcription, the DNA site at which RNA polymerase binds is called:
A)
promoter
done
clear
B)
regulator
done
clear
C)
receptor
done
clear
D)
enhancer
done
clear
View Answer play_arrow
question_answer 105) Bacteria do not possess
A)
capsule
done
clear
B)
ribosome
done
clear
C)
mitochondria
done
clear
D)
plasma membrane
done
clear
View Answer play_arrow
question_answer 106) Thermal cycler is used in this reaction
A)
radioactivity
done
clear
B)
enzyme catalysed
done
clear
C)
chemical reactions
done
clear
D)
PCR
done
clear
View Answer play_arrow
question_answer 107) Sporozoite, infectious stage of Plasmodium parasite contains
A)
two nucleus and a vacuole
done
clear
B)
one nucleus and several vacuoles
done
clear
C)
vacuoles and chloroplasts
done
clear
D)
a nucleus
done
clear
View Answer play_arrow
question_answer 108) The shape of chromosome is determined by
A)
centrosome
done
clear
B)
centromere
done
clear
C)
chromomere
done
clear
D)
telomere
done
clear
View Answer play_arrow
question_answer 109) Heteroneries stage is
A)
transformation of sexual individual into asexual
done
clear
B)
sexually matured with two regions atoke and epitoke
done
clear
C)
sexually immatured with two regions etoke
done
clear
D)
and epitoke
done
clear
View Answer play_arrow
question_answer 110) All of the above In Funaria, calypu-a is derived from
A)
capsule
done
clear
B)
columella
done
clear
C)
antheridium
done
clear
D)
archegonium
done
clear
View Answer play_arrow
question_answer 111) Biologically most resistant plant material is
A)
lignin
done
clear
B)
curin
done
clear
C)
suberin
done
clear
D)
sporopollenin
done
clear
View Answer play_arrow
question_answer 112) Which of the following techniques other than microscopy is used for the study of cell?
A)
Plasmolysis
done
clear
B)
Maceration
done
clear
C)
Chromatography
done
clear
D)
Auto-radiography
done
clear
View Answer play_arrow
question_answer 113) Earthworms have no special sense organs. Sdll they are sensitive to
A)
touch and sound
done
clear
B)
light and sound
done
clear
C)
touch, taste and sound
done
clear
D)
touch, taste and light
done
clear
View Answer play_arrow
question_answer 114) What will be the gametic chromosome number in a cell, if somatic cells have 40 chromosomes?
A)
10
done
clear
B)
20
done
clear
C)
30
done
clear
D)
40
done
clear
View Answer play_arrow
question_answer 115) The ATP production in photosynthesis is called
A)
phototropism
done
clear
B)
phosphorylation
done
clear
C)
photooxidation
done
clear
D)
photophosphorylation
done
clear
View Answer play_arrow
question_answer 116) Duramen of heart wood is
A)
region of pericycle
done
clear
B)
another name of cambium
done
clear
C)
inner region of secondary wood
done
clear
D)
outer region of secondary wood
done
clear
View Answer play_arrow
question_answer 117) The term heterosis was first used by
A)
Shull
done
clear
B)
NE Borlaug
done
clear
C)
MS Swaminachan
done
clear
D)
R Mishra
done
clear
View Answer play_arrow
question_answer 118) The larva of Bombyx mori is
A)
cocoon
done
clear
B)
nymph
done
clear
C)
caterpillar
done
clear
D)
trochophore
done
clear
View Answer play_arrow
question_answer 119) Gemmule formation in sponges is helpful in
A)
sexual reproduction
done
clear
B)
asexual reproduction
done
clear
C)
only dissemination
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 120) Organs not similar in structure and origin but perform the same function
A)
homologous structures
done
clear
B)
analogous structures
done
clear
C)
adaptive structures
done
clear
D)
variable structures
done
clear
View Answer play_arrow
question_answer 121) In Dryopteris, the opening mechanism of sporangium is effectively operated by
A)
stalk
done
clear
B)
annulus
done
clear
C)
stomium
done
clear
D)
peristome
done
clear
View Answer play_arrow
question_answer 122) Tunica corpus theory is used for
A)
shoot apex
done
clear
B)
root apex
done
clear
C)
leaf apex
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 123) The part of earth and atmosphere supporting life is
A)
biota
done
clear
B)
biome
done
clear
C)
ecotone
done
clear
D)
biosphere
done
clear
View Answer play_arrow
question_answer 124) Lenticels and hydathodes are small pores with which of the following common attributes?
A)
They allow exchange of gases
done
clear
B)
Their opening and closing is not regulated
done
clear
C)
They always remain closed
done
clear
D)
They are found on the same organ of plants
done
clear
View Answer play_arrow
question_answer 125) Chloragogen cells of earthworms are analogous to vertebrate
A)
lungs
done
clear
B)
liver
done
clear
C)
gut
done
clear
D)
kidney
done
clear
View Answer play_arrow
question_answer 126) Change in one base in mRNA leading to termination of polypeptide is known as which type of mutations ?
A)
Sense
done
clear
B)
None-sense
done
clear
C)
Gibberish
done
clear
D)
Framshift
done
clear
View Answer play_arrow
question_answer 127) Taenia solium derives its energy from the breakdown of
A)
nucleic acids
done
clear
B)
amino acids
done
clear
C)
glycogen
done
clear
D)
glycerol
done
clear
View Answer play_arrow
question_answer 128) Which of the following hormone is produced during leaf fall ?
A)
ABA
done
clear
B)
Cytokinin
done
clear
C)
Florigen
done
clear
D)
Vemalin
done
clear
View Answer play_arrow
question_answer 129) Which fish selectively feed on larva of mosquito?
A)
Gambusia
done
clear
B)
Rohu
done
clear
C)
Glorias
done
clear
D)
Exocoetus
done
clear
View Answer play_arrow
question_answer 130) Lomentum is a term used to describe a kind of
A)
fruit
done
clear
B)
seed
done
clear
C)
inflorescence
done
clear
D)
outgrowth from seed
done
clear
View Answer play_arrow
question_answer 131) House-fly possesses
A)
two pairs of wings
done
clear
B)
one pair of wings
done
clear
C)
three pairs of wings
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 132) Sunken stomata are found in
A)
Nerium
done
clear
B)
Hydrilla
done
clear
C)
mango
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 133) Which fish has an electric organ?
A)
Torpedo
done
clear
B)
Rhinoceros
done
clear
C)
Scoiiodon
done
clear
D)
Cat fish
done
clear
View Answer play_arrow
question_answer 134) Three most abundant elements in protoplasm are
A)
carbon, sodium and hydrogen
done
clear
B)
carbon, hydrogen and \[{{O}_{2}}\]
done
clear
C)
nitrogen, carbon and \[{{O}_{2}}\]
done
clear
D)
carbon, phosphorus and oxygen
done
clear
View Answer play_arrow
question_answer 135) Genetically engineered bacteria is used for the production of
A)
thyroxin
done
clear
B)
human insulin
done
clear
C)
epinephrine
done
clear
D)
cortisol
done
clear
View Answer play_arrow
question_answer 136) Inulin is a
A)
lipid
done
clear
B)
protein
done
clear
C)
human insulin
done
clear
D)
polysaccharide
done
clear
View Answer play_arrow
question_answer 137) Jumping genes in maize were discovered by
A)
Hugo de Vries
done
clear
B)
TH Morgan
done
clear
C)
Barbara McClintock
done
clear
D)
Mendel
done
clear
View Answer play_arrow
question_answer 138) Which of the following is not a pentose sugar?
A)
arabinose
done
clear
B)
xylose
done
clear
C)
xylulose
done
clear
D)
mannose
done
clear
View Answer play_arrow
question_answer 139) Nucleolus is the site for the synthesis of
A)
DNA
done
clear
B)
m RNA
done
clear
C)
t RNA
done
clear
D)
ribosomes
done
clear
View Answer play_arrow
question_answer 140) Which of the following statement is not true?
A)
Animal cells never contain chloroplast unlike many plant cells
done
clear
B)
Animal cells contain ultrascopic chloroplast while plant ceils contain microscopic chloroplast
done
clear
C)
Plant cells have cellulosic cell wall while animal cells do not
done
clear
D)
Plant cells have central vacuoles while animal-cells do not
done
clear
View Answer play_arrow
question_answer 141) Which of the following factor controls the human population density ?
A)
Industry
done
clear
B)
Climate
done
clear
C)
Communication
done
clear
D)
Natural resources
done
clear
View Answer play_arrow
question_answer 142) Chromosomal aberration is due to
A)
aneuploidy
done
clear
B)
polyploidy
done
clear
C)
physical effects
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 143) What used to described as Nisei granules in a nerve cell are now identified as
A)
cell metabolites
done
clear
B)
fat granules
done
clear
C)
ribosomes
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
question_answer 144) Ageing of an animal body is associated with deterioration in its
A)
connective tissue
done
clear
B)
glandular tissue
done
clear
C)
epithelial tissue
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 145) Photochemical smog always contains
A)
\[{{O}_{3}}\]
done
clear
B)
\[C{{H}_{4}}\]
done
clear
C)
\[CO\]
done
clear
D)
\[P{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 146) The posterior end of Ancylostoma has
A)
anus
done
clear
B)
curved tail
done
clear
C)
bursa
done
clear
D)
caudal spine
done
clear
View Answer play_arrow
question_answer 147) Quantasomes are found in
A)
mitochondria
done
clear
B)
chloroplast
done
clear
C)
lysosome
done
clear
D)
endoplasmic reticulum
done
clear
View Answer play_arrow
question_answer 148) In Ptinus, male cone bears a large number of
A)
ligules
done
clear
B)
anthers
done
clear
C)
microsporophylls
done
clear
D)
megasporophyus
done
clear
View Answer play_arrow
question_answer 149) Hydroponics is a
A)
airless culture
done
clear
B)
waterless culture
done
clear
C)
soilless culture
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 150) Lysosomes are formed by
A)
endoptasmic reticulum
done
clear
B)
mitocliondrion
done
clear
C)
Golgi membrane
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 151) Nissls granules are characteristic of
A)
muscle tissue
done
clear
B)
connective tissue
done
clear
C)
nerve tissue
done
clear
D)
epithelial tissue
done
clear
View Answer play_arrow
question_answer 152) Gastric vacuoles are analogous to the
A)
peroxisomes
done
clear
B)
alimentary canal of higher animals
done
clear
C)
mouth of higher animals
done
clear
D)
liver of higher animals
done
clear
View Answer play_arrow
question_answer 153) Genetic information is carried by long chain macromolecules made up of
A)
amino acids
done
clear
B)
nucleotides
done
clear
C)
chromosomes
done
clear
D)
enzymes
done
clear
View Answer play_arrow
question_answer 154) Fertilization of ovum in human being occur in
A)
fallopian tube
done
clear
B)
cervix
done
clear
C)
fundus
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 155) Which one are correcrely marched?
A)
Vitamin-E-tocopherol
done
clear
B)
Vitamin-D-riboflavin
done
clear
C)
Vitamin-B-calciferol
done
clear
D)
Vitamin-A-thiamine
done
clear
View Answer play_arrow
question_answer 156) Believers in spontaneous generation theory assumed that
A)
organisms arose only from other similar organisms
done
clear
B)
organisms could arise only from air
done
clear
C)
organisms arose-from non-living material
done
clear
D)
organisms always arise from air
done
clear
View Answer play_arrow
question_answer 157) Half-life period of \[{{C}^{14}}\]is
A)
500 years
done
clear
B)
5000 years
done
clear
C)
50 years
done
clear
D)
\[5\times {{10}^{4}}\]years
done
clear
View Answer play_arrow
question_answer 158) Maximum number of plasmids discovered so far
A)
50 kilo base
done
clear
B)
500 kilo base
done
clear
C)
5000 kilo base
done
clear
D)
5 kilo base
done
clear
View Answer play_arrow
question_answer 159) The lyric enzymes released by sperm is
A)
acrosome
done
clear
B)
ligase
done
clear
C)
androgamone
done
clear
D)
hyaluronidase
done
clear
View Answer play_arrow
question_answer 160) Transfer of genes from one gene pool to another is called
A)
speciation
done
clear
B)
genetic drift
done
clear
C)
gene flow
done
clear
D)
mutation
done
clear
View Answer play_arrow
question_answer 161) A material which arrests cell division, is obtained from
A)
Crocus
done
clear
B)
Colchicum
done
clear
C)
Dalbergia
done
clear
D)
Chrysanthemum
done
clear
View Answer play_arrow
question_answer 162) Chromosomes in a bacterial cell can be 1-3 in number and
A)
are always circular
done
clear
B)
are always linear
done
clear
C)
can be either circular or linear, but never both within the same cell
done
clear
D)
can be circular as well as linear within the same cell
done
clear
View Answer play_arrow
question_answer 163) Example of an aggregate fruit is
A)
Pyrus malus
done
clear
B)
Ananas sativus
done
clear
C)
Annona squamosal
done
clear
D)
Artocarpits integrifolia
done
clear
View Answer play_arrow
question_answer 164) Identify the edible freshwater teleoscs
A)
sharks
done
clear
B)
rays and skates
done
clear
C)
Hifsa hilsa
done
clear
D)
Catia catia
done
clear
View Answer play_arrow
question_answer 165) The blood cell which shows phagocytosis is
A)
monocyte
done
clear
B)
barsophil
done
clear
C)
eosinophil
done
clear
D)
platelets
done
clear
View Answer play_arrow
question_answer 166) In angiosperms, which of the following part is triploid?
A)
Zygote
done
clear
B)
Endosperm
done
clear
C)
Archegonia
done
clear
D)
Coralloid root
done
clear
View Answer play_arrow
question_answer 167) Mitochondria are often seen aggregated around
A)
food vacuole
done
clear
B)
contractile vacuole
done
clear
C)
water vacuole
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 168) Addisons disease results from
A)
hypertrophy of gonad
done
clear
B)
hyposecretion of adrenal cortex
done
clear
C)
hyperactivity of leydig cells
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 169) Main difference between active and passive transport across cell membrane is that
A)
active transport occurs more rapidly than passive
done
clear
B)
passive transport is non-selective
done
clear
C)
passive transport is confined to anions and cations
done
clear
D)
passive transport requires a concentration gradient across the membrane, whereas active transport requires metabolic energy
done
clear
View Answer play_arrow
question_answer 170) Which statement is correct for muscle- contraction?
A)
Length of H zone decreases
done
clear
B)
Length of A band decreases
done
clear
C)
Length of I band remain constant
done
clear
D)
Length of two Z line increases
done
clear
View Answer play_arrow
question_answer 171) Aleurone layer helps in
A)
growth
done
clear
B)
nutrition
done
clear
C)
protection
done
clear
D)
water absorption
done
clear
View Answer play_arrow
question_answer 172) Internal cork disease in apple is due to the deficiency of
A)
iron
done
clear
B)
copper
done
clear
C)
boron
done
clear
D)
phosphorus
done
clear
View Answer play_arrow
question_answer 173) When an animal turns sideways to the Sun to get the maximum amount of Sun on its flanks, this is an example of:
A)
motivation
done
clear
B)
habitation
done
clear
C)
posture
done
clear
D)
orientation
done
clear
View Answer play_arrow
question_answer 174) The main components of plasma membrane are
A)
lipids and proteins
done
clear
B)
lipids, proteins and carbohydrates
done
clear
C)
lipids only
done
clear
D)
proteins only
done
clear
View Answer play_arrow
question_answer 175) Pollination by insects is called
A)
zoophily
done
clear
B)
chiropterophily
done
clear
C)
anemophily
done
clear
D)
entomophily
done
clear
View Answer play_arrow
question_answer 176) The genetic material in fX174 is
A)
single stranded DNA
done
clear
B)
single stranded RNA
done
clear
C)
double stranded DNA
done
clear
D)
double stranded RNA
done
clear
View Answer play_arrow
question_answer 177) The pH of stomach ranges between
A)
7.1 to 8.2
done
clear
B)
7.6 to 8.6
done
clear
C)
1.5 to 2.0
done
clear
D)
6.8 to 7.2
done
clear
View Answer play_arrow
question_answer 178) Removal of ring wood of tissue outside the vascular cambium from the tree trunk kills it because
A)
water cannot move up
done
clear
B)
food does not travel down and roots become starved
done
clear
C)
shoot become starved
done
clear
D)
annual ring are not produced
done
clear
View Answer play_arrow
question_answer 179) The high amount of E. coil in water is an indicator of
A)
hardness of water
done
clear
B)
industrial pollution
done
clear
C)
sewage pollution
done
clear
D)
presence of chlorine in water
done
clear
View Answer play_arrow
question_answer 180) Which of the following connects glycolysis to Krebs cycle ?
A)
Acetyl Co-A
done
clear
B)
Ribozyme
done
clear
C)
Cyrochrome oxidase
done
clear
D)
N-acetyl glucosamine
done
clear
View Answer play_arrow
question_answer 181) Which of the following theory explain structure of protoplasm?
A)
Surface tension theory
done
clear
B)
Colloidal theory
done
clear
C)
Sol-gel theory
done
clear
D)
Viscosity theory
done
clear
View Answer play_arrow
question_answer 182) An insect without metamorphosis is
A)
silver fish
done
clear
B)
cray fish
done
clear
C)
bedbug
done
clear
D)
pediculus
done
clear
View Answer play_arrow
question_answer 183) Lichens show
A)
mutualism
done
clear
B)
commensalism
done
clear
C)
parasitism
done
clear
D)
saprophytism
done
clear
View Answer play_arrow
question_answer 184) In which of the following animal anal tail is found?
A)
Earthworm
done
clear
B)
Lower invertebrates
done
clear
C)
Scorpion
done
clear
D)
Snake
done
clear
View Answer play_arrow
question_answer 185) Which of the following statements is true?
A)
All enzymes are proteins
done
clear
B)
All proteins are enzymes
done
clear
C)
All enzymes are not proteins
done
clear
D)
All enzymes and hormones are proteins
done
clear
View Answer play_arrow
question_answer 186) This is not the cell of areolar tissue
A)
macrophages
done
clear
B)
Schwann cell
done
clear
C)
plasma cell
done
clear
D)
adipose cell
done
clear
View Answer play_arrow
question_answer 187) Anaerobic respiration after glycolysis is called
A)
restoration
done
clear
B)
fermentation
done
clear
C)
multiplication
done
clear
D)
fragmentation
done
clear
View Answer play_arrow
question_answer 188) Portuguese man of war is
A)
Obelia
done
clear
B)
Pennatula
done
clear
C)
Coral
done
clear
D)
Physalia
done
clear
View Answer play_arrow
question_answer 189) In photosynthesis, photolysis of water is useful for
A)
reduction of NADP
done
clear
B)
oxidation of NADP
done
clear
C)
oxidation of FAD
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 190) Liquid food drinking is
A)
imbibition
done
clear
B)
pinocytosis
done
clear
C)
phagocytosis
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 191) Terminating codons are also called
A)
initiating codons
done
clear
B)
stop signals
done
clear
C)
central dogma
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 192) A pome fruit is said to be false because
A)
the pericarp is inconspicuous
done
clear
B)
the endocarp is cartilaginous
done
clear
C)
fruit is derived from superior ovary
done
clear
D)
fruit is present in fleshy edible thalamus
done
clear
View Answer play_arrow
question_answer 193) In photosynthesis, the light energy is utilized in
A)
convening ATP into ADP
done
clear
B)
converting \[C{{H}_{3}}\] into \[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
converting ADP into ATP
done
clear
D)
converting \[C{{O}_{2}}\] into carbohydrate
done
clear
View Answer play_arrow
question_answer 194) Receptors of sensation produced when a person eats red chillies are located on which part of tongue?
A)
Tip
done
clear
B)
Base
done
clear
C)
Side
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 195) In which of the following haemocyanin pigment is found?
A)
Annelida
done
clear
B)
Echinodennata
done
clear
C)
Insects
done
clear
D)
Lower invertebrates
done
clear
View Answer play_arrow
question_answer 196) Photosystem-II occurs in
A)
grana
done
clear
B)
stroma
done
clear
C)
entire chloroplast
done
clear
D)
chloroplast membrane
done
clear
View Answer play_arrow
question_answer 197) When a cluster of genes show linkage behavior they
A)
do not show a chromosome map
done
clear
B)
show recombination during meiosis
done
clear
C)
do not show independent assortment
done
clear
D)
induce cell division
done
clear
View Answer play_arrow
question_answer 198) What is the function of centrosome?
A)
Cell division
done
clear
B)
Cell plate formation
done
clear
C)
Cell differentiation
done
clear
D)
Cell wall formation
done
clear
View Answer play_arrow
question_answer 199) Keratinization of the skin is prevented by
A)
vitamin-A
done
clear
B)
vitamin-B
done
clear
C)
vitamin-C
done
clear
D)
vitamin-D
done
clear
View Answer play_arrow
question_answer 200) Christmas disease is another name for
A)
haemophilia-B
done
clear
B)
hepatitis-B
done
clear
C)
Downs syndrome
done
clear
D)
sleeping sickness
done
clear
View Answer play_arrow
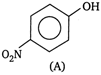 and
and