question_answer 1) If M is the mass of the earth and R its radius, the ratio of the gravitational acceleration and the gravitational constant is:
A)
\[\frac{{{R}^{2}}}{M}\]
done
clear
B)
\[\frac{M}{{{R}^{2}}}\]
done
clear
C)
\[M{{R}^{2}}\]
done
clear
D)
\[\frac{M}{R}\]
done
clear
View Answer play_arrow
question_answer 2) A student unable to answer a question on Newtons laws of motion attempts to pull himself up by tugging on his hair. He will not succeed:
A)
as the force exerted is small
done
clear
B)
the frictional force while gripping, is small
done
clear
C)
Newtons law of inertia is not applicable to living beings
done
clear
D)
as the force applied is internal to the system
done
clear
View Answer play_arrow
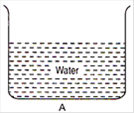
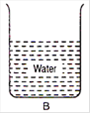
question_answer 3)
From the adjacent figure, the correct observation is:
A)
the pressure on the bottom of tank A is greater than at the bottom of B
done
clear
B)
the pressure on the bottom of the tank A is smaller than at the bottom of B
done
clear
C)
the pressure depends on the shape of the container
done
clear
D)
the pressure on the bottom of A and B is the same
done
clear
View Answer play_arrow
question_answer 4) Which one of the following is not a unit of Youngs modulus?
A)
\[N{{m}^{-1}}\]
done
clear
B)
\[N{{m}^{-2}}\]
done
clear
C)
dyne \[c{{m}^{-2}}\]
done
clear
D)
mega pascal
done
clear
View Answer play_arrow
question_answer 5) A piece of blue glass heated to a high temperature and a piece of red glass at room temperature, are taken inside a dimly lit room, then:
A)
the blue piece will look blue and red will look as usual
done
clear
B)
red looks brighter red and blue looks ordinary blue
done
clear
C)
blue shines like brighter red compared to the red piece
done
clear
D)
both the pieces will look equally red
done
clear
View Answer play_arrow
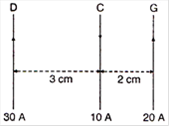
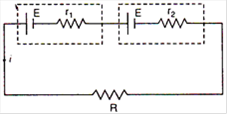
question_answer 6)
Three long, straight parallel wires, carrying current, are arranged as shown in figure. The force experienced by a 25 cm length of wire C is:
A)
\[{{10}^{-3}}N\]
done
clear
B)
\[2.5\times {{10}^{-3}}N\]
done
clear
C)
zero
done
clear
D)
\[1.5\times {{10}^{-3}}N\]
done
clear
View Answer play_arrow
question_answer 7) A 5.0 amp current is setup in an external circuit by a 6.0 volt storage battery for 6.0 minutes. The chemical energy of the battery is reduced by:
A)
\[1.08\times {{10}^{4}}J\]
done
clear
B)
\[1.08\times {{10}^{4}}J\]
done
clear
C)
\[1.8\times {{10}^{4}}J\]
done
clear
D)
\[1.8\times {{10}^{4}}J\]
done
clear
View Answer play_arrow
question_answer 8) The current in a simple series circuit is 5.0 amp. When an additional resistance of 2.0 ohms is inserted, the current drops to 4.0 amp. The original resistance of the circuit in ohms was:
A)
1.25
done
clear
B)
8
done
clear
C)
10
done
clear
D)
20
done
clear
View Answer play_arrow
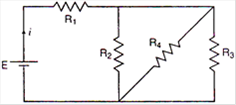
question_answer 9)
In the circuit given \[E=6.0\]volt, \[{{R}_{1}}=100\,\Omega \]\[{{R}_{2}}={{R}_{3}}=50\,\,\Omega ,{{R}_{4}}=75\,\,\Omega \]. The equivalent resistance of the circuit, in ohms, is:
A)
11.875
done
clear
B)
26.31
done
clear
C)
118.75
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 10) Two resistances are connected in two gaps of a metre bridge. The balance point is 20 cm from the zero end. A resistance of 15 ohms is connected in series with the smaller of the two. The null point shifts to 40 cm. The value of the smaller resistance in ohms is:
A)
3
done
clear
B)
6
done
clear
C)
9
done
clear
D)
12
done
clear
View Answer play_arrow
question_answer 11)
If the potential difference across the internal resistance \[{{r}_{1}}\] is equal to the emf E of die battery, then:
A)
\[R={{r}_{1}}+{{r}_{2}}\]
done
clear
B)
\[R=\frac{{{r}_{1}}}{{{r}_{2}}}\]
done
clear
C)
\[R={{r}_{1}}-{{r}_{2}}\]
done
clear
D)
\[R=\frac{{{r}_{2}}}{{{r}_{1}}}\]
done
clear
View Answer play_arrow
question_answer 12) By using only two resistance coils-singly, in series or ill parallel one should be able to obtain resistances of 3, 4, 12 and 16 ohms. The separate resistances of the coil are:
A)
3 and 4
done
clear
B)
4 and 12
done
clear
C)
12 and 16
done
clear
D)
16 and 3
done
clear
View Answer play_arrow
question_answer 13) The electrons in the beam of a television tube move horizontally from south to north. The vertical component of the earths magnetic field points down. The electron is deflected towards:
A)
west
done
clear
B)
no deflection
done
clear
C)
east
done
clear
D)
north to south
done
clear
View Answer play_arrow
question_answer 14) A tangent galvanometer has a reduction factor of 1 A and it is placed with the plane of its coil perpendicular to the magnetic meridian. The deflection produced when a current of 1 A is passed through it is:
A)
\[{{60}^{o}}\]
done
clear
B)
\[{{45}^{o}}\]
done
clear
C)
\[{{30}^{o}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
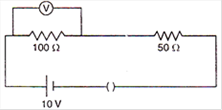
question_answer 15)
In the given circuit, the voltmeter records 5 volts. The resistance of the voltmeter in ohms is:
A)
200
done
clear
B)
100
done
clear
C)
10
done
clear
D)
50
done
clear
View Answer play_arrow
question_answer 16) The wavelength of the radiation emitted by a body depends upon:
A)
the nature of the surface
done
clear
B)
the area of the surface
done
clear
C)
the temperature of the surface
done
clear
D)
all of the above factors
done
clear
View Answer play_arrow
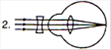
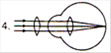
question_answer 17) Which mirror is to be used to obtain a parallel beam of light from a small lamp?
A)
Plane mirror
done
clear
B)
Convex mirror
done
clear
C)
Concave mirror
done
clear
D)
Any one of the above
done
clear
View Answer play_arrow
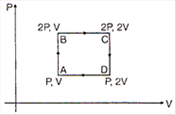
question_answer 18)
An ideal monoatomic gas is taken around the cycle ABCDA as shown in the PV diagram. The work done during the cycle is given by:
A)
\[\frac{1}{2}PV\]
done
clear
B)
PV
done
clear
C)
2 PV
done
clear
D)
4 PV
done
clear
View Answer play_arrow
question_answer 19) Which of the following is a wrong statement?
A)
\[D=1\text{ }/f\] where f is the focal length and D is called the refractive power of a lens.
done
clear
B)
Power is expressed in a diopter when f is in metres
done
clear
C)
Power is expressed in diopter and does not depend on the system of unit used to measure f
done
clear
D)
D is positive for convergent lens and negative for divergent lens.
done
clear
View Answer play_arrow
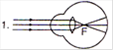
question_answer 20)
Identify the wrong description of the above figures:
A)
1 represents far-sightedness
done
clear
B)
2 correction for short sightedness
done
clear
C)
3 represents far-sightedness
done
clear
D)
4 correction for far-sightedness
done
clear
View Answer play_arrow
question_answer 21) An electric field of 1500 V/m and a magnetic field of 0.40 weber/\[metr{{e}^{2}}\] act on a moving electron. The minimum uniform speed along a straight line the electron could have is:
A)
\[1.6\times {{10}^{15}}m/s\]
done
clear
B)
\[1.6\times {{10}^{15}}m/s\]
done
clear
C)
\[3.75\times {{10}^{3}}m/s\]
done
clear
D)
\[3.75\times {{10}^{2}}m/s\]
done
clear
View Answer play_arrow
question_answer 22) In an ammeter 10% of main current is passing through the galvanometer. If the resistance of the galvanometer is G, then the shunt resistance, in ohms is:
A)
9 G
done
clear
B)
\[\frac{G}{9}\]
done
clear
C)
90 G
done
clear
D)
\[\frac{G}{90}\]
done
clear
View Answer play_arrow
question_answer 23) Among the following properties describing diamagnetism identify the property that is wrongly stated:
A)
Diamagnetic material do not have permanent magnetic moment
done
clear
B)
Ddiamagnetism is explained in terms of electromagnetic induction
done
clear
C)
Diamagnetic materials have a small positive susceptibility
done
clear
D)
The magnetic moment of individual electrons neutralize each other
done
clear
View Answer play_arrow
question_answer 24) The induction coil works on the principle of:
A)
self - induction
done
clear
B)
mutual induction
done
clear
C)
Amperes rule
done
clear
D)
Flemings right hand rule
done
clear
View Answer play_arrow
question_answer 25) The square root of the product of inductance and capacitance has the dimension of:
A)
length
done
clear
B)
mass
done
clear
C)
time
done
clear
D)
no dimension
done
clear
View Answer play_arrow
A)
\[1<2<3\]
done
clear
B)
\[2<3<1\]
done
clear
C)
\[3<2<1\]
done
clear
D)
\[3<1<2\]
done
clear
View Answer play_arrow
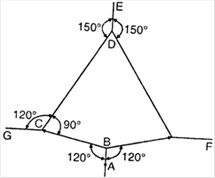
question_answer 27)
The adjacent figure is the part of a horizontally stretched net. Section AB is stretched with a force of 10 N. The tensions in the sections BC and BF are:
A)
10 N, 11 N
done
clear
B)
10 N, 6 N
done
clear
C)
10 N, 10 N
done
clear
D)
cant be calculated due to insufficient data
done
clear
View Answer play_arrow
question_answer 28) Out of the following four dimensional quantities, which one qualifies to be called a dimensional constant?
A)
Acceleration due to gravity
done
clear
B)
Surface tension of water
done
clear
C)
Weight of a standard kilogram mass
done
clear
D)
The velocity of light hi vacuum
done
clear
View Answer play_arrow
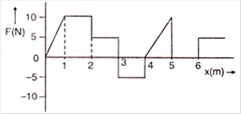
question_answer 29)
The relationship between the force F and position x of a body is as shown in figure. The work done in displacing the body from \[x=1\]m to \[x=5\,m\] will be:
A)
30 J
done
clear
B)
15 J
done
clear
C)
25 J
done
clear
D)
20 J
done
clear
View Answer play_arrow
question_answer 30) From the top of a tower of two stones, whose masses are in the ratio \[1:2\] are thrown on straight up with an initial speed k and the second straight down with the same speed it. Then neglecting air resistance:
A)
the heavier stone hits the ground with a higher speed
done
clear
B)
the lighter stone hits the ground with a higher speed
done
clear
C)
both the stones will have the same speed when they hit the ground
done
clear
D)
the speed cant be determined with the given data
done
clear
View Answer play_arrow
question_answer 31) Infrared radiation was discovered in 1800 by:
A)
William Wollaston
done
clear
B)
William Herschel
done
clear
C)
Wilhelm Roentgen
done
clear
D)
Thomas Young
done
clear
View Answer play_arrow
question_answer 32) A particle on the trough of a wave at any instant will come to the mean position after a time (T = time period)
A)
T/2
done
clear
B)
T/4
done
clear
C)
T
done
clear
D)
2 T
done
clear
View Answer play_arrow
question_answer 33) The disc of a siren containing 60 holes rotates at a constant speed of 360 rpm. The emitted sound is in unison with a tuning fork of frequency:
A)
10 Hz
done
clear
B)
360 Hz
done
clear
C)
216 Hz
done
clear
D)
60 Hz
done
clear
View Answer play_arrow
question_answer 34) The ratio of velocity of sound in hydrogen and oxygen at STP is:
A)
\[16:1\]
done
clear
B)
\[8:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
question_answer 35) In an experiment with sonometer a tuning fork of frequency 256 Hz resonates with a length of 25 cm and another tuning fork resonates with a length of 16 cm. Tension of the string remaining constant the frequency of the second tuning fork is:
A)
163.84 Hz
done
clear
B)
400 Hz
done
clear
C)
320 Hz
done
clear
D)
204.8 Hz
done
clear
View Answer play_arrow
question_answer 36) The apparent frequency of a note is 200 Hz, when a listener is moving with a velocity of \[40\text{ }m{{s}^{-1}}\] towards a stationary source. When he moves away from the same source with the same speed, the apparent frequency of the same note is 160 Hz. The velocity of sound in air in m/s is:
A)
340
done
clear
B)
330
done
clear
C)
360
done
clear
D)
320
done
clear
View Answer play_arrow
question_answer 37) The wave theory of light, in its original form, was first postulated by:
A)
Isaac Newton
done
clear
B)
Christian Huygens
done
clear
C)
Thomas Young
done
clear
D)
Augustin Jean Fresnel
done
clear
View Answer play_arrow
question_answer 38) If a liquid does not wet glass, its angle of contact is:
A)
zero
done
clear
B)
acute
done
clear
C)
obtuse
done
clear
D)
right angle
done
clear
View Answer play_arrow
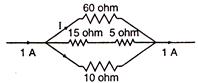
question_answer 39)
The magnitude of J in ampere is:
A)
0.1
done
clear
B)
0.3
done
clear
C)
0.6
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 40) Electron of mass m and charge q is travelling with a speed v along a circular path of radius r at right angles to a uniform magnetic field of intensity B. If the speed of the electron is doubled and the magnetic field is halved the resulting path would have a radius:
A)
2 r
done
clear
B)
4 r
done
clear
C)
r/4
done
clear
D)
r/2
done
clear
View Answer play_arrow
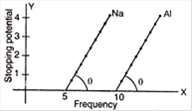
question_answer 41)
From the figure describing photoelectric effect we may infer correctly that:
A)
Na and Al both have the same threshold frequency
done
clear
B)
maximum kinetic energy for both the metals depend linearly on the frequency
done
clear
C)
the stopping potentials are different for Na and Al for the same change m frequency
done
clear
D)
Al is a better photo sensitive material than Na.
done
clear
View Answer play_arrow
question_answer 42) Two coherent light beams of intensity \[I\]and \[4I\] are superposed. The maximum and minimum possible intensities in the resulting beam are:
A)
\[9I\] and \[I\]
done
clear
B)
\[9I\] and \[3I\]
done
clear
C)
\[5I\] and \[I\]
done
clear
D)
\[5I\] and \[3I\]
done
clear
View Answer play_arrow
question_answer 43) The electron in a hydrogen atom makes a transition from \[n={{n}_{1}}\] to \[n={{n}_{2}}\] state. The time period of the electron in the initial state \[({{n}_{1}})\] is eight times that in the final state \[({{n}_{2}})\]. The possible values of \[{{n}_{1}}\] and \[{{n}_{2}}\]are:
A)
\[{{n}_{1}}=8,{{n}_{2}}=1\]
done
clear
B)
\[{{n}_{1}}=4,{{n}_{2}}=2\]
done
clear
C)
\[{{n}_{1}}=2,{{n}_{2}}=4\]
done
clear
D)
\[{{n}_{1}}=1,{{n}_{2}}=8\]
done
clear
View Answer play_arrow
question_answer 44) If the forward voltage in a diode is increased, the width of the depletion region:
A)
increases
done
clear
B)
decreases
done
clear
C)
fluctuates
done
clear
D)
no change
done
clear
View Answer play_arrow
question_answer 45) Two nucleons are at a separation of one Fermi. Protons have a charge of\[+1.6\times {{10}^{-19}}C\]. The net nuclear force between them is \[{{F}_{1}}\], if both are neutrons, \[{{F}_{2}}\]if both are protons and \[{{F}_{3}}\] if one is proton and the other is neutron. Then:
A)
\[{{F}_{1}}={{F}_{2}}>{{F}_{3}}\]
done
clear
B)
\[{{F}_{1}}={{F}_{2}}={{F}_{3}}\]
done
clear
C)
\[{{F}_{1}}<{{F}_{2}}<{{F}_{3}}\]
done
clear
D)
\[{{F}_{1}}>{{F}_{2}}>{{F}_{3}}\]
done
clear
View Answer play_arrow
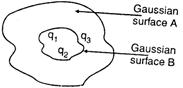
question_answer 46)
The electric flux for Gaussian surface A that enclose the charged particles in free space is (given \[{{q}_{1}}=14nC,{{q}_{2}}=78.85nC,{{q}_{3}}=-56nC\])
A)
\[{{10}^{3}}N{{m}^{2}}{{C}^{-1}}\]
done
clear
B)
\[{{10}^{3}}C{{N}^{-1}}{{m}^{-2}}\]
done
clear
C)
\[6.32\times {{10}^{3}}N{{m}^{2}}{{C}^{-1}}\]
done
clear
D)
\[6.32\times {{10}^{3}}C{{N}^{-1}}{{m}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 47) Four metal conductors having different shapes (a) a sphere (b) cylinder (c) pear (d) lightning conductor are mounted on insulating stands and charged. The one which is best suited to retain the charges for a longer time is:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 48) The potential to which a conductor is raised, depends on:
A)
the amount of charge
done
clear
B)
geometry and size of the conductor
done
clear
C)
both (a) and (b)
done
clear
D)
only on (a)
done
clear
View Answer play_arrow
question_answer 49) The work done in carrying a charge q once round a circle of radius r with a charge Q at die centre is:
A)
\[\frac{qQ}{4\pi {{\varepsilon }_{0}}r}\]
done
clear
B)
\[\frac{qQ}{4\pi {{\varepsilon }_{0}}^{2}{{r}^{2}}}\]
done
clear
C)
\[\frac{qQ}{4\pi {{\varepsilon }_{0}}{{r}^{2}}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 50) An air filled parallel plate condenser has a capacity of 2 pF. The separation of the plates is doubled and the interspace between the plates is filled with wax. If the capacity is increased to 6 pF, the dielectric constant of wax is:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 51) The energy that should be added to an electron to reduce its de-Broglie wavelength from 1 nm to 0.5 rim is:
A)
four times the initial energy
done
clear
B)
equal to the initial energy
done
clear
C)
twice the initial energy
done
clear
D)
thrice the initial energy
done
clear
View Answer play_arrow
question_answer 52) Mean life of a radioactive sample is 100 seconds. Then its half-life (in minutes) is:
A)
0.693
done
clear
B)
1
done
clear
C)
\[{{10}^{-4}}\]
done
clear
D)
1.155
done
clear
View Answer play_arrow
question_answer 53) Consider two nuclei of the same radioactive nuclide. One of the nuclei was created in a supernova explosion 5 billion years ago. The other was created in a nuclear reactor 5 minutes ago. The probability of decay during the next time is:
A)
different for each nuclei
done
clear
B)
nuclei created in explosion decays first
done
clear
C)
nuclei created in the reactor decays first
done
clear
D)
independent of the time of creation
done
clear
View Answer play_arrow
question_answer 54) Bohrs atom model assumes:
A)
the nucleus is of infinite mass and is at rest
done
clear
B)
electrons in a quantized orbit will not radiate energy
done
clear
C)
mass of electron remains constant
done
clear
D)
all the above conditions
done
clear
View Answer play_arrow
question_answer 55) Identify the property which is not characteristic for a semiconductor?
A)
At a very low temperatures, it behaves like an insulator
done
clear
B)
At higher temperatures two types of charge carriers will cause conductivity
done
clear
C)
The charge carriers are electrons and holes in the valence band at higher temperatures
done
clear
D)
The semiconductor is electrically neutral.
done
clear
View Answer play_arrow
question_answer 56) Identify the wrong statement in the following. Coulombs law correctly described the electric force that:
A)
binds the electrons of an atom to its nucleus
done
clear
B)
binds the protons and neutrons in the nucleus of an atom
done
clear
C)
binds atoms together to form molecules
done
clear
D)
binds atoms and molecules to form solids
done
clear
View Answer play_arrow
question_answer 57) A single slit of width d is illuminated by violet light of wavelength 400 nm and the width of the diffraction pattern is measured as y. When half of the slit width is covered and illuminated by yellow light of wavelength 600 nm, the width of the diffraction pattern is:
A)
the pattern vanishes and the width is zero
done
clear
B)
\[y/3\]
done
clear
C)
3 y
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 58) At Kavalur in India, the astronomers using a telescope whose objective had a diameter of one metre started using telescope of diameter 2.54 m. This resulted in:
A)
the increase m the resolving power by 2.54 times for the same \[\lambda \]
done
clear
B)
the increase in the limiting angle by 2.54 times for the same \[\lambda \]
done
clear
C)
decrease in the resolving power
done
clear
D)
no effect on the limiting angle
done
clear
View Answer play_arrow
question_answer 59) When unpolarised light beam is incident from air onto glass \[(n=1.5)\] at the polarizing angle:
A)
reflected beam is polarised 100 percent
done
clear
B)
reflected and refracted beams are partially polarized
done
clear
C)
the reason for (a) is that almost all the light is reflected
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 60) Select the right option in the following:
A)
Christian Huygens, a contemporary of Newton established the wave theory of light by assuming that light waves were transverse.
done
clear
B)
Maxwell provided the theoretical evidence that light is transverse wave
done
clear
C)
Thomas Young experimentally proved the wave behaviour of light and Huygens assumption
done
clear
D)
All the statements given above, correctly answers the question what is light?
done
clear
View Answer play_arrow
question_answer 61) 15 moles of \[{{H}_{2}}\] and 5.2 moles of \[{{I}_{2}}\] are mixed and allowed to attain equilibrium at \[{{500}^{o}}C\]. At equilibrium, the concentration of HI is found to be 10 moles. The equilibrium constant for the formation of HI is :
A)
50
done
clear
B)
15
done
clear
C)
100
done
clear
D)
25
done
clear
View Answer play_arrow
question_answer 62) If, in the reaction \[{{N}_{2}}{{O}_{4}}2N{{O}_{2}},\,\,x\] is that part of \[{{N}_{2}}{{O}_{4}}\] which dissociates, then the number of molecules at equilibrium will be :
A)
1
done
clear
B)
3
done
clear
C)
\[(1+x)\]
done
clear
D)
\[{{(1+x)}^{2}}\]
done
clear
View Answer play_arrow
question_answer 63) Which of these does not influence the rate of reaction?
A)
Nature of the reactants
done
clear
B)
Concentration of the reactants
done
clear
C)
Temperature of the reaction
done
clear
D)
Molecularity of die reaction
done
clear
View Answer play_arrow
question_answer 64) For the reaction \[A+B\to C\], it is found that doubling the concentration of A increases the rate by 4 times, and doubling the concentration of B doubles the reaction rate. What is the overall order of the reaction?
A)
4
done
clear
B)
3/2
done
clear
C)
3
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 65) The rate at which a substance reacts depends on its :
A)
atomic weight
done
clear
B)
atomic number
done
clear
C)
molecular weight
done
clear
D)
active mass
done
clear
View Answer play_arrow
question_answer 66) A compound A has a molecular formula\[{{C}_{2}}C{{l}_{3}}OH\]. It reduces Fehlings solution and on oxidation, gives a monocarboxylic acid B.A can be obtained by the action of chlorine on ethyl alcohol. A is :
A)
chloroform
done
clear
B)
chloral
done
clear
C)
methyl chloride
done
clear
D)
monochloroacetic acid
done
clear
View Answer play_arrow
question_answer 67) Which of the following haloalkanes is most reactive?
A)
1-chloropropane
done
clear
B)
1-bromopropane
done
clear
C)
2-chloropropane
done
clear
D)
2-bromopropane
done
clear
View Answer play_arrow
question_answer 68) The reaction in which phenol differs from alcohol is :
A)
it undergoes esterification with carboxylic acid
done
clear
B)
it reacts with ammonia
done
clear
C)
it forms yellow crystals of iodoform
done
clear
D)
it liberates \[{{H}_{2}}\] with Na metal.
done
clear
View Answer play_arrow
question_answer 69) An organic compound A containing C, H and O has a pleasant odour with boiling point of\[{{78}^{o}}C\]. On boiling A with concentrated \[{{H}_{2}}S{{O}_{4}}\], a colourless gas is produced which decolourises bromine water and alkaline\[KMn{{O}_{4}}\]. The organic liquid A is :
A)
\[{{C}_{2}}{{H}_{5}}Cl\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}COOC{{H}_{3}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 70) Which of the following is an amphoteric acid?
A)
Glycine
done
clear
B)
Salicylic acid
done
clear
C)
Benzoic acid
done
clear
D)
Citric acid
done
clear
View Answer play_arrow
question_answer 71) Gold is extracted by hydrometallurgical process, based on its property :
A)
of being electropositive
done
clear
B)
of being less reactive
done
clear
C)
to form complexes which are water soluble
done
clear
D)
to form salts which are water soluble
done
clear
View Answer play_arrow
question_answer 72) In blast furnace, iron oxide is reduced by:
A)
hot blast of air
done
clear
B)
carbon monoxide
done
clear
C)
carbon
done
clear
D)
silica
done
clear
View Answer play_arrow
question_answer 73) Which of the following pairs of elements cannot form an alloy?
A)
Zn, Cu
done
clear
B)
Fe, Hg
done
clear
C)
Fe, C
done
clear
D)
Hg, Na
done
clear
View Answer play_arrow
question_answer 74) Which compound is zero valent metal complex?
A)
\[[Cu{{(N{{H}_{3}})}_{4}}]S{{O}_{4}}\]
done
clear
B)
\[[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]
done
clear
C)
\[[Ni{{(CO)}_{4}}]\]
done
clear
D)
\[{{K}_{3}}[Fe{{(CN)}_{6}}]\]
done
clear
View Answer play_arrow
question_answer 75) Alum is a water purifier because it :
A)
coagulates the impurities
done
clear
B)
softens hard water
done
clear
C)
gives taste
done
clear
D)
destroys the pathogenic bacteria
done
clear
View Answer play_arrow
question_answer 76) For the reaction\[{{N}_{2(g)}}+{{O}_{2(g)}}2N{{O}_{(g)}}\], the value of \[{{K}_{c}}\] at \[{{800}^{o}}C\] is 0.1. When the equilibrium concentration of both die reactants is 0.5 mol, what is the value of \[{{K}_{p}}\] at the same temperature?
A)
0.5
done
clear
B)
0.1
done
clear
C)
0.01
done
clear
D)
0.025
done
clear
View Answer play_arrow
question_answer 77) The extent of adsorption of a gas on a solid depends on :
A)
nature of the gas
done
clear
B)
pressure of the gas
done
clear
C)
temperature of the gas
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 78) A emulsifier is a substance which :
A)
stabilises the emulsion
done
clear
B)
homogenises the emulsion
done
clear
C)
coagulates the emulsion
done
clear
D)
accelerates the dispersion of liquid in liquid
done
clear
View Answer play_arrow
question_answer 79) Which of the following types of metals form the most efficient catalysts?
A)
Alkali metals
done
clear
B)
Alkaline earth metals
done
clear
C)
Transition metals
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 80) The species among the following, which can act as an acid and a base is :
A)
\[HSO_{4}^{-}\]
done
clear
B)
\[SO_{4}^{2-}\]
done
clear
C)
\[{{H}_{3}}{{O}^{+}}\]
done
clear
D)
\[C{{l}^{-}}\]
done
clear
View Answer play_arrow
question_answer 81) Benzyl alcohol and sodium benzoate is obtained by the action of sodium hydroxide on benzaldehyde. This reaction is known as :
A)
Perkins reaction
done
clear
B)
Cannizaros reaction
done
clear
C)
Sandmeyers reaction
done
clear
D)
Claisen condensation
done
clear
View Answer play_arrow
question_answer 82) Ethyl chloride on heating with \[AgCN\] forms a compound X. The functional isomer of X is :
A)
\[{{C}_{2}}{{H}_{5}}NC\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}CN\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 83) A compound, containing only carbon, hydrogen and oxygen, has a molecular weight of 44. On complete oxidation it is converted into a compound of molecular weight 60. The original compound is :
A)
an aldehyde
done
clear
B)
an acid
done
clear
C)
ail alcohol
done
clear
D)
an ether
done
clear
View Answer play_arrow
question_answer 84) Grignard reagent adds to :
A)
\[<C=O\]
done
clear
B)
\[C=N\]
done
clear
C)
\[<C=S\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 85) Which of the following biomolecules contain non-transition metal ion?
A)
Vitamin \[{{B}_{12}}\]
done
clear
B)
Chlorophyll
done
clear
C)
Haemoglobin
done
clear
D)
Insulin
done
clear
View Answer play_arrow
question_answer 86) A mixture of two moles of carbon monoxide and one mole of oxygen, in a closed vessel is ignited to convert the carbon monoxide to carbon dioxide. If \[\Delta H\] is the enthalpy change and \[\Delta E\] is the change in internal energy, then:
A)
\[\Delta H>\Delta E\]
done
clear
B)
\[\Delta H<\Delta E\]
done
clear
C)
\[\Delta H=\Delta E\]
done
clear
D)
the relationship depends on the capacity of the vessel
done
clear
View Answer play_arrow
question_answer 87) The cooling in refrigerator is due to :
A)
reaction of the refrigerator gas
done
clear
B)
expansion of ice
done
clear
C)
the expansion of the gas in die refrigerator
done
clear
D)
the work of die compressor
done
clear
View Answer play_arrow
question_answer 88) For a system in equilibrium, \[\Delta G=0\], under conditions of constant .........
A)
temperature and pressure
done
clear
B)
temperature arid volume
done
clear
C)
pressure and volume
done
clear
D)
energy and volume
done
clear
View Answer play_arrow
question_answer 89) Molar heat of vaporisation of a liquid is\[6\,kJ\,mo{{l}^{-1}}\]. If die entropy change is \[16\,J\,mo{{l}^{-1}}{{K}^{-1}}\], the boiling point of the liquid is:
A)
\[{{375}^{o}}C\]
done
clear
B)
375K
done
clear
C)
273 K
done
clear
D)
\[{{102}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 90) The temperature of the system decreases in an :
A)
adiabatic compression
done
clear
B)
isothermal compression
done
clear
C)
isothermal expansion
done
clear
D)
adiabatic expansion
done
clear
View Answer play_arrow
question_answer 91) A buffer solution has equal volumes of 0.2 M \[N{{H}_{4}}OH\] and 0.02 M \[N{{H}_{4}}Cl\]. The \[p{{K}_{b}}\], of the base is 5. The pH is :
A)
10
done
clear
B)
9
done
clear
C)
4
done
clear
D)
7
done
clear
View Answer play_arrow
question_answer 92) The hydrogen electrode is dipped in a solution of pH 3 at \[{{25}^{o}}C\]. The potential would be (the value of 2.303 RT/F is 0.059 V) :
A)
0.177 V
done
clear
B)
0.087 V
done
clear
C)
0.059V
done
clear
D)
-0.177V
done
clear
View Answer play_arrow
question_answer 93) 20 mL of 0.5 N \[HCl\] and 35 mL of 0.1 N \[NaOH\]are mixed. The resulting solution will :
A)
be neutral
done
clear
B)
be basic
done
clear
C)
turn phenolphthalein solution pink
done
clear
D)
turn methyl orange red
done
clear
View Answer play_arrow
question_answer 94) Corrosion of iron is essentially an electrochemical phenomenon where the cell reactions are :
A)
Fe is oxidised to \[F{{e}^{2+}}\]and dissolved oxygen in water is reduced to \[O{{H}^{-}}\]
done
clear
B)
Fe is oxidised to \[F{{e}^{2+}}\] and \[{{H}_{2}}O\] is reduced to \[O_{2}^{2-}\]
done
clear
C)
Fe is oxidised to \[F{{e}^{2+}}\] and \[{{H}_{2}}O\] is reduced to \[O_{2}^{-}\]
done
clear
D)
Fe is oxidised to \[F{{e}^{2+}}\] and \[{{H}_{2}}O\] is reduced to \[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 95) The standard electrode potential is measured by :
A)
electrometer
done
clear
B)
voltmeter
done
clear
C)
pyrometer
done
clear
D)
galvanometer
done
clear
View Answer play_arrow
question_answer 96) A precipitate of \[AgCl\] is formed when equal volumes of the following are mixed : [\[{{K}_{sp}}\] for\[AgCl={{10}^{-10}}\]]
A)
\[{{10}^{-4}}M\,AgN{{O}_{3}}\] and \[{{10}^{-7}}M\,HCl\]
done
clear
B)
\[{{10}^{-5}}M\,AgN{{O}_{3}}\] and \[{{10}^{-6}}M\,HCl\]
done
clear
C)
\[{{10}^{-5}}M\,AgN{{O}_{3}}\] and \[{{10}^{-4}}M\,HCl\]
done
clear
D)
\[{{10}^{-6}}M\,AgN{{O}_{3}}\] and \[{{10}^{-6}}M\,HCl\]
done
clear
View Answer play_arrow
question_answer 97) Which one of the following defects in the crystals lowers its density?
A)
Frenkel defect
done
clear
B)
Schottky defect
done
clear
C)
F-centres
done
clear
D)
Interstitial defect
done
clear
View Answer play_arrow
question_answer 98) A radioactive isotope has a half-life of 10 days. If today 125 mg is left over, what was its original weight 40 days earlier?
A)
2 g
done
clear
B)
600 mg
done
clear
C)
1 g
done
clear
D)
1.5 g
done
clear
View Answer play_arrow
question_answer 99) Which of the following cannot be accelerated?
A)
\[\alpha \]-particle
done
clear
B)
\[\beta \]-particle
done
clear
C)
Protons
done
clear
D)
Neutrons
done
clear
View Answer play_arrow
question_answer 100) In which of the following nuclear reaction neutrons is emitted?
A)
\[_{13}^{27}Al+_{2}^{4}He\to _{15}^{30}P\]
done
clear
B)
\[_{6}^{27}C+_{1}^{1}H\to _{7}^{13}N\]
done
clear
C)
\[_{15}^{30}P\to _{14}^{30}Si\]
done
clear
D)
\[_{96}^{241}Am+_{2}^{4}He\to _{97}^{245}Bk\]
done
clear
View Answer play_arrow
question_answer 101) Molarity of \[0.2\,N\,{{H}_{2}}S{{O}_{4}}\] is :
A)
0.2
done
clear
B)
0.4
done
clear
C)
0.6
done
clear
D)
0.1
done
clear
View Answer play_arrow
question_answer 102) In the equation of state of an ideal gas\[PV=nRT\], the value of the universal gas constant would depend only on :
A)
the nature of the gas
done
clear
B)
the pressure of the gas
done
clear
C)
the units of the measurement
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 103) A commercial sample of hydrogen peroxide is labelled as 10 volume. Its percentage strength is nearly :
A)
1%
done
clear
B)
3%
done
clear
C)
10%
done
clear
D)
90%
done
clear
View Answer play_arrow
question_answer 104) Activated charcoal is used to remove colouring matter from pure substances. It works by :
A)
oxidation
done
clear
B)
reduction
done
clear
C)
bleaching
done
clear
D)
adsorption
done
clear
View Answer play_arrow
question_answer 105) When plants and animals decay, the organic nitrogen is converted into inorganic nitrogen. The inorganic nitrogen is in the form of:
A)
ammonia
done
clear
B)
elements of nitrogen
done
clear
C)
nitrates
done
clear
D)
nitrides
done
clear
View Answer play_arrow
question_answer 106) Three dimensional molecules with cross links are formed in the case of a :
A)
thermoplastic
done
clear
B)
thermosetting plastic
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 107) Sucrose molecule is made up of :
A)
a gluco pyranose and a fructo pyranose
done
clear
B)
a gluco pyranose and a fructo furanose
done
clear
C)
a gluco furanose and a fructo pyranose
done
clear
D)
a gluco furanose and a fructo furanose
done
clear
View Answer play_arrow
question_answer 108) Water insoluble component of starch is :
A)
amylopectin
done
clear
B)
amylose
done
clear
C)
cellulose
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 109) An example for a saturated fatty acid, present in nature is :
A)
oleic acid
done
clear
B)
linoleic acid
done
clear
C)
linolenic acid
done
clear
D)
palmitic acid
done
clear
View Answer play_arrow
question_answer 110) A nanopeptide contains......peptide linkages :
A)
10
done
clear
B)
8
done
clear
C)
9
done
clear
D)
18
done
clear
View Answer play_arrow
question_answer 111) A gas decolourised by \[KMn{{O}_{4}}\] solution but gives no precipitate with ammoniacal cuprous chloride is :
A)
ethane
done
clear
B)
methane
done
clear
C)
ethane
done
clear
D)
acetylene
done
clear
View Answer play_arrow
question_answer 112) \[{{H}_{3}}C-\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,=CH-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,H-C{{H}_{3}}\]
A)
2-chloro-4-methyl-2pentene
done
clear
B)
4-chloro-2-methyl-3-pentene
done
clear
C)
4-methyl-2-chloro-2-pentene
done
clear
D)
2-chloro-4,4-dimethyl-2-butene
done
clear
View Answer play_arrow
question_answer 113) Amongst the following the compound that can most readily get sulphonated is :
A)
benzene
done
clear
B)
toluene
done
clear
C)
nitrobenzene
done
clear
D)
chlorobenzene
done
clear
View Answer play_arrow
question_answer 114) Household gaseous fuel (LPG) mainly con tarns:
A)
\[C{{H}_{4}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{4}}{{H}_{10}}\]
done
clear
View Answer play_arrow
question_answer 115) Use of chlorofluoro carbons is not encouraged because :
A)
they are harmful to the eyes of people that use it
done
clear
B)
they damage the refrigerators and air conditioners
done
clear
C)
they eat away the ozone in the atmosphere
done
clear
D)
they destroy the oxygen layer
done
clear
View Answer play_arrow
question_answer 116) An example of a sulphur containing amino acid is :
A)
lysine
done
clear
B)
serine
done
clear
C)
cysteine
done
clear
D)
tyrosine
done
clear
View Answer play_arrow
question_answer 117) Which of the following is not present in a nucleotide?
A)
Cytosine
done
clear
B)
Guanine
done
clear
C)
Adenine
done
clear
D)
Tyrosine
done
clear
View Answer play_arrow
question_answer 118) Antiseptic chloroxylenol is :
A)
4-chloro-3,5-dimethyl phenol
done
clear
B)
3-chloro-4,5-dimethyl phenol
done
clear
C)
4-chloro-2,5-dimethyl phenol
done
clear
D)
5-chloro-3,4-dimethyl phenol
done
clear
View Answer play_arrow
question_answer 119) An atom of an element A has three electrons in its outermost orbit and that of B has six electrons in its outermost orbit. The formula of the compound between these two will be :
A)
\[{{A}_{3}}{{B}_{6}}\]
done
clear
B)
\[{{A}_{2}}{{B}_{3}}\]
done
clear
C)
\[{{A}_{3}}{{B}_{2}}\]
done
clear
D)
\[{{A}_{2}}B\]
done
clear
View Answer play_arrow
question_answer 120) Among \[N{{a}^{+}},\,Na,\,Mg\] and \[M{{g}^{2+}}\], the largest particle is :
A)
\[M{{g}^{2+}}\]
done
clear
B)
Mg
done
clear
C)
Na
done
clear
D)
\[N{{a}^{+}}\]
done
clear
View Answer play_arrow
question_answer 121) When \[{{2}^{301}}\] is divided by 5, the least positive remainder is :
A)
4
done
clear
B)
8
done
clear
C)
2
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 122) The contrapositive of If two triangles are identical, then these are similar is :
A)
If two triangles are not similar, then these are not identical
done
clear
B)
If two triangles are not identical, then these are not similar
done
clear
C)
If two triangles are not identical, then these are similar
done
clear
D)
If two. triangles are not similar, then these are identical
done
clear
View Answer play_arrow
question_answer 123) The contrapositive of the inverse of \[p\to \,\sim \,\,q\]is :
A)
\[\sim q\to p\]
done
clear
B)
\[p\to q\]
done
clear
C)
\[\sim q\to \sim p\]
done
clear
D)
\[\sim p\to \sim q\]
done
clear
View Answer play_arrow
question_answer 124) The converse of the contrapositive of \[p\to q\] is :
A)
\[\sim p\to q\]
done
clear
B)
\[p\to \sim q\]
done
clear
C)
\[\sim p\to \sim q\]
done
clear
D)
\[\sim q\to p\]
done
clear
View Answer play_arrow
question_answer 125) If \[\omega \] is a complex cube-root of unity, then\[\left| \begin{matrix} 1 & \omega & {{\omega }^{2}} \\ \omega & {{\omega }^{2}} & 1 \\ {{\omega }^{2}} & 1 & \omega \\ \end{matrix} \right|\] is equal to :
A)
-1
done
clear
B)
1
done
clear
C)
0
done
clear
D)
\[\omega \]
done
clear
View Answer play_arrow
question_answer 126) The ends of the latus rectum of the conic\[{{x}^{2}}+10x-16y+25=0\] are :
A)
(3,-4), (13, 4)
done
clear
B)
(-3,-4), (13,-4)
done
clear
C)
(3, 4), (-13, 4)
done
clear
D)
(5,-8), (-5, 8)
done
clear
View Answer play_arrow
question_answer 127) The equation to die hyperbola having its eccentricity 2 and the distance between its foci is 8, is :
A)
\[\frac{{{x}^{2}}}{12}-\frac{{{y}^{2}}}{4}=1\]
done
clear
B)
\[\frac{{{x}^{2}}}{4}-\frac{{{y}^{2}}}{12}=1\]
done
clear
C)
\[\frac{{{x}^{2}}}{8}-\frac{{{y}^{2}}}{2}=1\]
done
clear
D)
\[\frac{{{x}^{2}}}{16}-\frac{{{y}^{2}}}{9}=1\]
done
clear
View Answer play_arrow
question_answer 128) The solution of \[{{\sin }^{-1}}x-{{\sin }^{-1}}2x=\pm \frac{\pi }{3}\] is :
A)
\[\pm \frac{1}{3}\]
done
clear
B)
\[\pm \frac{1}{3}\]
done
clear
C)
\[\pm \frac{\sqrt{3}}{2}\]
done
clear
D)
\[\pm \frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 129) In a \[\Delta ABC\] if the sides are \[a=3,b=5\] and\[c=4\], then \[\sin \frac{B}{2}+\cos \frac{B}{2}\] is equal to :
A)
\[\sqrt{2}\]
done
clear
B)
\[\frac{\sqrt{3}+1}{2}\]
done
clear
C)
\[\frac{\sqrt{3}-1}{2}\]
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 130) The value of\[\cos \left( {{270}^{o}}+\theta \right)\cos \left( {{90}^{o}}-\theta \right)-\sin \left( {{270}^{o}}-\theta \right)\cos \theta \]is :
A)
0
done
clear
B)
- 1
done
clear
C)
1/2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 131) The two circles \[{{x}^{2}}+{{y}^{2}}-2x+22y+5=0\] and \[{{x}^{2}}+{{y}^{2}}+14x+6y+k=0\] intersect orthogonally provided k is equal to :
A)
47
done
clear
B)
-47
done
clear
C)
49
done
clear
D)
-49
done
clear
View Answer play_arrow
question_answer 132) The radius of the circle \[{{x}^{2}}+{{y}^{2}}+4x+6y+13=0\] is ;
A)
\[\sqrt{26}\]
done
clear
B)
\[\sqrt{13}\]
done
clear
C)
\[\sqrt{23}\]
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 133) The centre of the circle \[x=2+3\cos \theta ,\]\[y=3\text{ }\sin \theta -1\] is :
A)
(3, 3)
done
clear
B)
(2,-1)
done
clear
C)
(-2,1)
done
clear
D)
(-1,2)
done
clear
View Answer play_arrow
question_answer 134) The sum of the focal distances of any point on the conic \[\frac{{{x}^{2}}}{25}+\frac{{{y}^{2}}}{16}=1\] is :
A)
10
done
clear
B)
9
done
clear
C)
41
done
clear
D)
18
done
clear
View Answer play_arrow
question_answer 135) The eccentricity of the hyperbola \[\frac{{{x}^{2}}}{16}-\frac{{{y}^{2}}}{25}=1\] is:
A)
\[\frac{3}{4}\]
done
clear
B)
\[\frac{3}{5}\]
done
clear
C)
\[\frac{\sqrt{41}}{4}\]
done
clear
D)
\[\frac{\sqrt{41}}{5}\]
done
clear
View Answer play_arrow
question_answer 136) The solutions of me equation \[\left| \begin{matrix} x & 2 & -1 \\ 2 & 5 & x \\ -1 & 2 & x \\ \end{matrix} \right|=0\] are:
A)
3,-1
done
clear
B)
-3, 1
done
clear
C)
3, 1
done
clear
D)
-3,-1
done
clear
View Answer play_arrow
question_answer 137) If \[A=\left[ \begin{matrix} 3 & 5 \\ 2 & 0 \\ \end{matrix} \right]\] and \[B=\left[ \begin{matrix} 1 & 17 \\ 0 & -10 \\ \end{matrix} \right]\], then \[|AB|\] is equal to :
A)
80
done
clear
B)
100
done
clear
C)
- 110
done
clear
D)
92
done
clear
View Answer play_arrow
question_answer 138) The inverse of the matrix \[\left[ \begin{matrix} 5 & -2 \\ 3 & 1 \\ \end{matrix} \right]\] is :
A)
\[\frac{1}{11}\left[ \begin{matrix} 1 & 2 \\ -3 & 5 \\ \end{matrix} \right]\]
done
clear
B)
\[\left[ \begin{matrix} 1 & 2 \\ -3 & 5 \\ \end{matrix} \right]\]
done
clear
C)
\[\frac{1}{13}\left[ \begin{matrix} -2 & 5 \\ 1 & 3 \\ \end{matrix} \right]\]
done
clear
D)
\[\left[ \begin{matrix} 1 & 3 \\ -2 & 5 \\ \end{matrix} \right]\]
done
clear
View Answer play_arrow
question_answer 139) The projection of the vector \[2\hat{i}+\hat{j}-3\hat{k}\] on the vector \[\hat{i}-2\hat{j}+\hat{k}\] is :
A)
\[-\frac{3}{\sqrt{14}}\]
done
clear
B)
\[\frac{3}{\sqrt{14}}\]
done
clear
C)
\[-\frac{\sqrt{3}}{2}\]
done
clear
D)
\[\frac{3}{\sqrt{2}}\]
done
clear
View Answer play_arrow
question_answer 140) A unit vector perpendicular to the plane containing the vectors \[\hat{i}-\hat{j}+\hat{k}\] and \[-\hat{i}+\hat{j}+\hat{k}\] is :
A)
\[\pm \frac{\hat{j}-\hat{j}}{\sqrt{2}}\]
done
clear
B)
\[\pm \frac{\hat{j}+\hat{k}}{\sqrt{2}}\]
done
clear
C)
\[\pm \frac{\hat{j}-\hat{k}}{\sqrt{2}}\]
done
clear
D)
\[\pm \frac{\hat{i}+\hat{j}}{\sqrt{2}}\]
done
clear
View Answer play_arrow
question_answer 141) If \[12\,{{\cot }^{2}}\theta -31\cos ec\theta +32=0\], then the value of \[\sin \theta \] is
A)
\[\frac{3}{5}\]or 1
done
clear
B)
\[\frac{2}{3}\] or \[-\frac{2}{3}\]
done
clear
C)
\[\frac{4}{5}\] or \[\frac{3}{4}\]
done
clear
D)
\[\pm \frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 142) The circus-radius of the triangle whose sides are 13, 12 and 5, is :
A)
15
done
clear
B)
13/2
done
clear
C)
15/2
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 143) If \[{{\tan }^{-1}}x+{{\tan }^{-1}}y=\frac{\pi }{4}\], then :
A)
\[x+y+xy=1\]
done
clear
B)
\[x+y-xy=1\]
done
clear
C)
\[x+y+xy+1=0\]
done
clear
D)
\[~x+y-xy+1=0\]
done
clear
View Answer play_arrow
question_answer 144) The general solution of \[\sin x-\cos x=\sqrt{2}\] for any integer n is :
A)
\[n\pi \]
done
clear
B)
\[2n\pi +\frac{3\pi }{4}\]
done
clear
C)
\[2n\pi \]
done
clear
D)
\[(2n\pi +1)\]
done
clear
View Answer play_arrow
question_answer 145) The amplitude of \[\frac{1+i\sqrt{3}}{\sqrt{3}+i}\] is :
A)
\[\frac{\pi }{3}\]
done
clear
B)
\[\frac{\pi }{4}\]
done
clear
C)
\[\frac{2\pi }{3}\]
done
clear
D)
\[\frac{\pi }{6}\]
done
clear
View Answer play_arrow
question_answer 146) The sum to infinity of the progression\[9-3+\frac{1}{3}+.........\] is :
A)
9
done
clear
B)
9/2
done
clear
C)
27/4
done
clear
D)
15/2
done
clear
View Answer play_arrow
question_answer 147) If\[{{\,}^{n}}{{C}_{12}}={{\,}^{n}}{{C}_{6}}\], then \[^{n}{{C}_{2}}=\]
A)
72
done
clear
B)
153
done
clear
C)
306
done
clear
D)
2556
done
clear
View Answer play_arrow
question_answer 148) The middle term in the expansion of \[{{\left( x-\frac{1}{x} \right)}^{18}}\]
A)
\[^{18}{{C}_{9}}\]
done
clear
B)
\[\,{{-}^{18}}{{C}_{9}}\]
done
clear
C)
\[^{18}{{C}_{10}}\]
done
clear
D)
\[{{-}^{18}}{{C}_{10}}\]
done
clear
View Answer play_arrow
question_answer 149) If \[\alpha ,\,\beta ,\,\,\gamma \] are the roots of the equation\[2{{x}^{3}}-3{{x}^{2}}+6x+1=0\], then \[{{\alpha }^{2}}+{{\beta }^{2}}+{{\gamma }^{2}}\] is equal to :
A)
\[-\frac{15}{4}\]
done
clear
B)
\[\frac{15}{4}\]
done
clear
C)
\[\frac{9}{4}\]
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 150) The digit in the units place in the number \[{{7}^{289}}\] is :
A)
9
done
clear
B)
7
done
clear
C)
1
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 151) If \[\hat{a},\,\,\hat{b}\] and \[\hat{c}\] are mutually perpendicular unit vectors, then \[|\hat{a}+\hat{b}+\hat{c}\,|\] is equal to :
A)
3
done
clear
B)
\[\sqrt{3}\]
done
clear
C)
\[(\sqrt{{{a}^{2}}+{{b}^{2}}+{{c}^{3}})}/3\]
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 152) The identity element ill die group\[M=\left\{ \left[ \begin{matrix} x & x \\ x & x \\ \end{matrix} \right]\,\,\left| x\in R\,;\,x\ne 0 \right. \right\}\,\]with respect to matrix multiplication is :
A)
\[\left[ \begin{matrix} 1 & 1 \\ 1 & 1 \\ \end{matrix} \right]\]
done
clear
B)
\[\frac{1}{2}\left[ \begin{matrix} 1 & 1 \\ 1 & 1 \\ \end{matrix} \right]\]
done
clear
C)
\[\left[ \begin{matrix} 1 & 0 \\ 0 & 1 \\ \end{matrix} \right]\]
done
clear
D)
\[\left[ \begin{matrix} 0 & 1 \\ 1 & 0 \\ \end{matrix} \right]\]
done
clear
View Answer play_arrow
question_answer 153) In the group \[G=\{1,3,7,9\}\] under multiplication modulo 10, the inverse of 3 is :
A)
1
done
clear
B)
3
done
clear
C)
7
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 154) In the group (0,1, 2, 4, 5} under addition modulo 6 a subgroup is :
A)
{ 0, 2, 5}
done
clear
B)
( 1, 4, 5}
done
clear
C)
( 0, 1, 3)
done
clear
D)
( 0..2, 4}
done
clear
View Answer play_arrow
question_answer 155) In the group \[({{Q}^{+}},*)\] of positive rational numbers w.r.t. the binary operation * defined by \[a*b=\frac{ab}{3}\,\,\forall \,\,a,b\in {{Q}^{+}}\] the solution of the equation \[5*x={{4}^{-1}}\] in \[{{Q}^{+}}\] is :
A)
\[\frac{27}{20}\]
done
clear
B)
\[\frac{20}{27}\]
done
clear
C)
\[\frac{1}{20}\]
done
clear
D)
20
done
clear
View Answer play_arrow
question_answer 156) (0, -1) and (0, 3) are two opposite vertices of a square. The other two vertices are:
A)
(0,1),(0,-3)
done
clear
B)
(3,-1),(0,0)
done
clear
C)
(2,1), (-2,1)
done
clear
D)
(2, 2), (1,1)
done
clear
View Answer play_arrow
question_answer 157) The equation to the line bisecting the join of (3, - 4) and (5, 2) and having its intercepts on the ^-axis and the y-axis in the ratio \[2:1\] is :
A)
\[x+y-3=0\]
done
clear
B)
\[2x-y=9\]
done
clear
C)
\[x+2y=2\]
done
clear
D)
\[2x+y=7\]
done
clear
View Answer play_arrow
question_answer 158) The distance between the pair of parallel lines \[{{x}^{2}}+2xy+{{y}^{2}}-8ax-8ay-9{{a}^{2}}=0\] is :
A)
\[2\sqrt{5}\,a\]
done
clear
B)
\[\sqrt{10}\,a\]
done
clear
C)
10 a
done
clear
D)
\[5\sqrt{2}\,a\]
done
clear
View Answer play_arrow
question_answer 159) The equation to the circle with centre (2,1) and touching the line \[3x+4y=5\] is :
A)
\[{{x}^{2}}+{{y}^{2}}-4y-2y+5=0\]
done
clear
B)
\[{{x}^{2}}+{{y}^{2}}-4x-2y-5=0\]
done
clear
C)
\[{{x}^{2}}+{{y}^{2}}-4x-2y+4=0\]
done
clear
D)
\[{{x}^{2}}+{{y}^{2}}-4x-2y-4=0\]
done
clear
View Answer play_arrow
question_answer 160) The condition for a line \[y=2x+c\] to touch the circle \[{{x}^{2}}+{{y}^{2}}=16\] is :
A)
\[c=10\]
done
clear
B)
\[{{c}^{2}}=80\]
done
clear
C)
\[c=12\]
done
clear
D)
\[{{c}^{2}}=64\]
done
clear
View Answer play_arrow
question_answer 161) \[\int{\frac{\sin \,\,(2x)}{1+{{\cos }^{2}}x}d\,x=}\]
A)
\[-\frac{1}{2}\log \,(1+{{\cos }^{2}}x)+c\]
done
clear
B)
\[2\,\,\log \,(1+{{\cos }^{2}}x)+c\]
done
clear
C)
\[\frac{1}{2}\,\,\log \,(1+{{\cos }^{2}}2x)+c\]
done
clear
D)
\[c-\,\log \,\,(1+{{\cos }^{2}}x)\]
done
clear
View Answer play_arrow
question_answer 162) \[\int{\frac{{{e}^{x}}(1+\sin x)}{1+\cos x}dx=}\]
A)
\[{{e}^{x}}\tan \left( \frac{x}{2} \right)+c\]
done
clear
B)
\[{{e}^{x}}\tan x+c\]
done
clear
C)
\[{{e}^{x}}\left( \frac{1+\sin x}{1-\cos x} \right)+c\]
done
clear
D)
\[c-{{e}^{x}}\cot \left( \frac{x}{2} \right)\]
done
clear
View Answer play_arrow
question_answer 163) \[\int{\frac{1+\tan x}{{{e}^{-x}}\cos x}d\,x}=\]
A)
\[{{e}^{-x}}\tan x+c\]
done
clear
B)
\[{{e}^{-x}}\sec x+c\]
done
clear
C)
\[{{e}^{x}}\sec x+c\]
done
clear
D)
\[{{e}^{x}}\tan x+c\]
done
clear
View Answer play_arrow
question_answer 164) \[\int\limits_{\pi /4}^{\pi /2}{\cos e{{c}^{2}}x\,dx=}\]
A)
-1
done
clear
B)
1
done
clear
C)
0
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
question_answer 165) \[\int\limits_{0}^{\pi /4}{\log \,(1+\tan x)d\,x=}\]
A)
\[\frac{\pi }{8}{{\log }_{e}}2\]
done
clear
B)
\[\frac{\pi }{4}{{\log }_{2}}e\]
done
clear
C)
\[\frac{\pi }{4}{{\log }_{e}}2\]
done
clear
D)
\[\frac{\pi }{4}{{\log }_{e}}\left( \frac{1}{2} \right)\]
done
clear
View Answer play_arrow
question_answer 166) The modulus and amplitude of \[\frac{1+2i}{1-{{(1-i)}^{2}}}\], are :
A)
\[\sqrt{2}\] and \[\frac{\pi }{6}\]
done
clear
B)
1 and 0
done
clear
C)
1 and \[\frac{\pi }{3}\]
done
clear
D)
1 and \[\frac{\pi }{4}\]
done
clear
View Answer play_arrow
question_answer 167) The real part of \[\frac{1}{1+\cos \theta +i\sin \theta }\] is :
A)
\[-\frac{1}{2}\]
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\sqrt{2}\]
done
clear
D)
\[\frac{1}{\sqrt{2}}\]
done
clear
View Answer play_arrow
question_answer 168) \[\underset{x\to 0}{\mathop{\lim }}\,\frac{\tan x-\sin x}{{{x}^{3}}}\] is equal to :
A)
\[\frac{1}{2}\]
done
clear
B)
\[-\frac{1}{2}\]
done
clear
C)
0
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 169) If \[y=\frac{{{e}^{x}}+{{e}^{-x}}}{{{e}^{x}}-{{e}^{-x}}}\], then \[\frac{dy}{dx}\] is equal to :
A)
\[\sec \,{{h}^{2}}x\]
done
clear
B)
\[\cos ec\,{{h}^{2}}x\]
done
clear
C)
\[\sec \,\,{{h}^{2}}x\]
done
clear
D)
\[-\cos ec\,{{h}^{2}}x\]
done
clear
View Answer play_arrow
question_answer 170) If \[f(x)=\left\{ \begin{matrix} \frac{\sin \,5x}{{{x}^{2}}+2x}, & x\ne 0 \\ k+\frac{1}{2}, & x=0 \\ \end{matrix} \right.\] is continuous at\[x=0\], then the value of k is :
A)
1
done
clear
B)
-2
done
clear
C)
2
done
clear
D)
½
done
clear
View Answer play_arrow
question_answer 171) The area bounded by the parabola \[{{y}^{2}}=4\text{ }ax\] and the line \[x=a\] and \[x=4\,a\] is :
A)
\[\frac{35\,{{a}^{2}}}{3}\]
done
clear
B)
\[\frac{4\,{{a}^{2}}}{3}\]
done
clear
C)
\[\frac{7\,{{a}^{2}}}{3}\]
done
clear
D)
\[\frac{56\,{{a}^{2}}}{3}\]
done
clear
View Answer play_arrow
question_answer 172) A population p (t) of 1000 bacteria introduced into nutrient medium grows according to the relation \[p\,(t)=1000+\frac{1000\,t}{100+{{t}^{2}}}\]. The maximum size of this bacterial population is :
A)
1100
done
clear
B)
1250
done
clear
C)
1050
done
clear
D)
5250
done
clear
View Answer play_arrow
question_answer 173) The differential equation representing a family of circles touching the y-axis at the origin is :
A)
\[{{x}^{2}}+{{y}^{2}}-2xy\frac{dy}{dx}=0\]
done
clear
B)
\[{{x}^{2}}+{{y}^{2}}+2xy\frac{dy}{dx}=0\]
done
clear
C)
\[{{x}^{2}}-{{y}^{2}}-2xy\frac{dy}{dx}=0\]
done
clear
D)
\[{{x}^{2}}-{{y}^{2}}+2xy\frac{dy}{dx}=0\]
done
clear
View Answer play_arrow
question_answer 174) The area of the region bounded by tile curve \[9{{x}^{2}}+4{{y}^{2}}-36=0\] is :
A)
\[9\pi \]
done
clear
B)
\[4\pi \]
done
clear
C)
\[36\pi \]
done
clear
D)
\[6\pi \]
done
clear
View Answer play_arrow
question_answer 175) The general solution of the differential equation \[(2x-y+1)\,dx+(2y-x+1)\,\,dy=0\] is:
A)
\[{{x}^{2}}+{{y}^{2}}+xy-x+y=c\]
done
clear
B)
\[{{x}^{2}}+{{y}^{2}}-xy+x+y=c\]
done
clear
C)
\[{{x}^{2}}-{{y}^{2}}+2xy-x+y=c\]
done
clear
D)
\[{{x}^{2}}-{{y}^{2}}-2xy+x-y=c\]
done
clear
View Answer play_arrow
question_answer 176) If \[y={{\tan }^{-1}}\frac{\sqrt{1+{{x}^{2}}}-\sqrt{1-{{x}^{2}}}}{\sqrt{1+{{x}^{2}}}+\sqrt{1-{{x}^{2}}}}\], then \[\frac{dy}{dx}\] is equal to :
A)
\[\frac{{{x}^{2}}}{\sqrt{1-{{x}^{4}}}}\]
done
clear
B)
\[\frac{{{x}^{2}}}{\sqrt{1+{{x}^{4}}}}\]
done
clear
C)
\[\frac{{{x}^{2}}}{\sqrt{1+{{x}^{4}}}}\]
done
clear
D)
\[\frac{x}{\sqrt{1-{{x}^{4}}}}\]
done
clear
View Answer play_arrow
question_answer 177) If \[x=\sin \,t,\,y=\cos \,pt\], then :
A)
\[(1-{{x}^{2}}){{y}_{2}}+x{{y}_{1}}+{{p}^{2}}y=0\]
done
clear
B)
\[(1-{{x}^{2}}){{y}_{2}}+x{{y}_{1}}-{{p}^{2}}y=0\]
done
clear
C)
\[(1+{{x}^{2}}){{y}_{2}}-x{{y}_{1}}+{{p}^{2}}y=0\]
done
clear
D)
\[(1-{{x}^{2}}){{y}_{2}}-x{{y}_{1}}+{{p}^{2}}y=0\]
done
clear
View Answer play_arrow
question_answer 178) If ST and SN are the lengths of the sub tangent and the subnormal at the point \[\theta =\frac{\pi }{2}\] on the curve \[x=a\,(\theta +\sin \theta )\], \[y=a\,(1-\cos \theta ),\,a\ne 1\], then :
A)
\[ST=SN\]
done
clear
B)
\[Sr=2SN\]
done
clear
C)
\[S{{T}^{2}}=a\,S{{N}^{3}}\]
done
clear
D)
\[S{{T}^{3}}=a\,SN\]
done
clear
View Answer play_arrow
question_answer 179) If \[\theta \] is the acute angle of intersection at a real point of intersection of the circle \[x+y=5\]and the parabola \[y=4x\], then \[\tan \theta \] is equal to :
A)
1
done
clear
B)
\[\sqrt{3}\]
done
clear
C)
3
done
clear
D)
\[\frac{1}{\sqrt{3}}\]
done
clear
View Answer play_arrow
question_answer 180) A spherical balloon is being inflated at the rate of 35 cc/min. The rate of increase of the surface area of the balloon when its diameter is 14 cm, is :
A)
7 sq.cm/min
done
clear
B)
10 sq.cm/min
done
clear
C)
17.5 sq.cm/min
done
clear
D)
28 sq.cm/min
done
clear
View Answer play_arrow