question_answer 1) Two simple harmonic motions A and B are given respectively by the following equations \[{{y}_{1}}=a\,\sin \,\left( \omega t+\frac{\pi }{6} \right),\] \[{{y}_{2}}=a\,\sin \,\left( \omega t+\frac{3\pi }{6} \right)\] The phase difference between the waves is
A)
\[\frac{\pi }{2}\]
done
clear
B)
\[\frac{\pi }{6}\]
done
clear
C)
\[\frac{\pi }{3}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 2) A stretched string of length \[l\] fixed at both ends can sustain stationary waves of wavelength \[\lambda \] given by
A)
\[\lambda =\frac{{{n}^{2}}}{2l}\]
done
clear
B)
\[\lambda =\frac{{{l}^{2}}}{2n}\]
done
clear
C)
\[\lambda =\frac{2l}{n}\]
done
clear
D)
\[\lambda =2\ln \] (Here, n is a integer)
done
clear
View Answer play_arrow
question_answer 3) An observer moves towards a stationary source of sound with a velocity that is one-tenth the velocity of sound. The apparent change in frequency is
A)
zero
done
clear
B)
5%
done
clear
C)
10%
done
clear
D)
0.1%
done
clear
View Answer play_arrow
question_answer 4) Light of wavelength \[7200\,\overset{\text{o}}{\mathop{\text{A}}}\,\] in air has a wavelength in glass \[(\mu =1.5)\] equal to
A)
\[7200\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[4800\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[10800\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[6000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 5) When a thin convex lens is put in contact with a thin concave lens of the same focal length \[(f),\] the resultant combination has a focal length equal to
A)
\[\frac{f}{2}\]
done
clear
B)
\[2f\]
done
clear
C)
0
done
clear
D)
\[\infty \]
done
clear
View Answer play_arrow
question_answer 6) A cylindrical lens is required to correct
A)
myopia
done
clear
B)
presbyopia
done
clear
C)
hypermetropia
done
clear
D)
astigmatism
done
clear
View Answer play_arrow
question_answer 7) Which of the following phenomenona is not common to sound and light waves ?
A)
Interference
done
clear
B)
Diffraction
done
clear
C)
Polarisation
done
clear
D)
Reflection
done
clear
View Answer play_arrow
question_answer 8) The nearer point of hypermetropic eye is 40 cm. The lens to be used for its correction should have the power
A)
+ 1.5 D
done
clear
B)
-1.5 D
done
clear
C)
+2.5D
done
clear
D)
+0.5D
done
clear
View Answer play_arrow
question_answer 9) The work of Sir C.V. Raman for which he was awarded Nobel Prize was concerned with
A)
nuclear energy
done
clear
B)
scattering of light
done
clear
C)
laser
done
clear
D)
transistors
done
clear
View Answer play_arrow
question_answer 10) Young's double slit experiment is based on
A)
Newton's corpuscular theory of light
done
clear
B)
Huygen's wave theory
done
clear
C)
Quantum theory
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 11) One kilowatt hour equals
A)
\[36\times {{10}^{s}}J\]
done
clear
B)
\[34\times {{10}^{s}}J\]
done
clear
C)
\[3\times {{10}^{5}}J\]
done
clear
D)
\[6\times {{10}^{s}}J\]
done
clear
View Answer play_arrow
question_answer 12) Velocity of electromagnetic waves in vacuum is given by
A)
\[\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{\sqrt{{{\mu }_{0}}}}{{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{\sqrt{{{\varepsilon }_{0}}}}{{{\mu }_{0}}}\]
done
clear
D)
\[\frac{1}{\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}}\]
done
clear
View Answer play_arrow
question_answer 13) Two spheres A and B of radii a and b respectively are at the same potential. The ratio of the surface charge densities of A to B is
A)
\[\frac{a}{b}\]
done
clear
B)
\[\frac{b}{a}\]
done
clear
C)
\[\frac{{{a}^{2}}}{{{b}^{2}}}\]
done
clear
D)
\[\frac{{{b}^{2}}}{{{a}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 14) A hollow metal sphere of radius 5 cm is charged such that the potential on its surface is 10 V. The potential at a distance of 2 cm from the centre of sphere is
A)
zero
done
clear
B)
10 V
done
clear
C)
4V
done
clear
D)
5V
done
clear
View Answer play_arrow
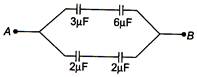
question_answer 15)
The equivalent capacitance between points A and B in the given figure, is
A)
\[\frac{36}{13\mu F}\]
done
clear
B)
\[2\mu F\]
done
clear
C)
\[1\mu F\]
done
clear
D)
\[3\mu F\]
done
clear
View Answer play_arrow
question_answer 16) An electric bulb is rated 220 V and 100 W. Power consumed by it when operated on 110 V is
A)
50 W
done
clear
B)
75 W
done
clear
C)
90 W
done
clear
D)
25 W
done
clear
View Answer play_arrow
question_answer 17) The temperature coefficient of resistance of a wire is \[0.00125\text{ }per\text{ }{}^\circ C\]. At 300 K temperature its resistance is \[1\Omega .\] The resistance of the wire will be \[2\Omega \] at
A)
1154 K
done
clear
B)
1100 K
done
clear
C)
1400 K
done
clear
D)
1127 K
done
clear
View Answer play_arrow
question_answer 18) How much heat is produced by 1500 W heater in 7 min ?
A)
1.5 kcal
done
clear
B)
15 kcal
done
clear
C)
150 kcal
done
clear
D)
1500 kcal
done
clear
View Answer play_arrow
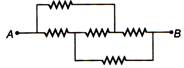
question_answer 19)
Five equal resistors, each equal to r are connected as shown in the figure, then the equivalent resistance between points A and B
A)
r
done
clear
B)
5r
done
clear
C)
\[\frac{r}{5}\]
done
clear
D)
\[\frac{2r}{3}\]
done
clear
View Answer play_arrow
question_answer 20) A solenoid of 1.5 m length and 4.0 cm diameter possesses 10 turns/cm. A current of 5 A is flowing through it. The magnetic induction a axis inside the solenoid is
A)
\[\text{2}\,\text{ }\!\!\pi\!\!\text{ }\,\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-3}}}\,\text{T}\]
done
clear
B)
\[\text{2}\,\text{ }\!\!\pi\!\!\text{ }\,\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-5}}}\,\text{T}\]
done
clear
C)
\[\text{2}\,\text{ }\!\!\pi\!\!\text{ }\,\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-3}}}\,\text{T}\]
done
clear
D)
\[\text{2}\,\text{ }\!\!\pi\!\!\text{ }\,\text{ }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-5}}}\,\text{T}\]
done
clear
View Answer play_arrow
question_answer 21) The unit of intensity of magnetisation (\[I\]) is
A)
A/m
done
clear
B)
A-m2
done
clear
C)
A-m
done
clear
D)
W/m
done
clear
View Answer play_arrow
question_answer 22) The resistance of an ideal voltmeter is
A)
low
done
clear
B)
high
done
clear
C)
infinite
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 23) The inductive time constant in an electrical circuit is
A)
\[LR\]
done
clear
B)
\[\frac{L}{R}\]
done
clear
C)
\[\sqrt{\frac{L}{R}}\]
done
clear
D)
\[\frac{R}{L}\]
done
clear
View Answer play_arrow
question_answer 24) The relation between triode constants is
A)
\[\mu \times {{r}_{P}}\times {{g}_{m}}=1\]
done
clear
B)
\[\mu \frac{{{r}_{P}}}{{{g}_{m}}}\]
done
clear
C)
\[\mu \frac{{{g}_{m}}}{{{r}_{P}}}\]
done
clear
D)
\[\mu ={{r}_{P}}\times {{g}_{m}}\]
done
clear
View Answer play_arrow
question_answer 25) An n-type semiconductor is formed
A)
when germanium crystal is doped with an impurity containing 3 valence electrons
done
clear
B)
when germanium crystal is doped with an impurity containing 5 valence electrons
done
clear
C)
from pure germanium
done
clear
D)
from pure silicon
done
clear
View Answer play_arrow
question_answer 26) In a common base configuration; (transistor circuit) \[{{I}_{E}}=1\]mA, \[{{I}_{c}}=\] 0.95mA. The value of base current is
A)
1.95mA
done
clear
B)
0.05mA
done
clear
C)
1.05mA
done
clear
D)
0.95mA
done
clear
View Answer play_arrow
question_answer 27) The work function for aluminium surface is 4.2eV. The cut-off wavelength for the photoelectric effect is
A)
\[2955\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[4200\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[2000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 28) X-rays are being produced in a tube operating at 105V The velocity of X-rays produced is
A)
\[3\times {{10}^{8}}m/s\]
done
clear
B)
\[2.8\times {{10}^{8}}m/s\]
done
clear
C)
\[3.1\times {{10}^{8}}m/s\]
done
clear
D)
\[3\times {{10}^{10}}m/s\]
done
clear
View Answer play_arrow
question_answer 29) If the potential energy of the electron in the hydrogen atom is \[\frac{-K{{e}^{2}}}{r},\]its Kinetic energy is
A)
\[\frac{-K{{e}^{2}}}{2r}\]
done
clear
B)
\[\frac{-K{{e}^{2}}}{r}\]
done
clear
C)
\[\frac{K{{e}^{2}}}{2r}\]
done
clear
D)
\[\frac{K{{e}^{2}}}{r}\]
done
clear
View Answer play_arrow
question_answer 30) The decay constant of radium is \[4.28\times {{10}^{-4}}\] per \[yr\]Its half-life is approximately
A)
\[1250\,yr\]
done
clear
B)
\[1620\,yr\]
done
clear
C)
\[200\,yr\]
done
clear
D)
\[60\,yr\]
done
clear
View Answer play_arrow
question_answer 31) Dimensions of the three physical quantities in which En up are the same ?
A)
Work, energy, force
done
clear
B)
Velocity, momentum, impulse
done
clear
C)
Potential energy, kinetic energy, momentum
done
clear
D)
Pressure, stress, coefficient of elasticity
done
clear
View Answer play_arrow
question_answer 32) One nanometre is equal to
A)
\[\text{1}{{\text{0}}^{\text{9}}}\text{mm}\]
done
clear
B)
\[\text{1}{{\text{0}}^{\text{-6}}}\text{cm}\]
done
clear
C)
\[\text{1}{{\text{0}}^{\text{-7}}}\text{cm}\]
done
clear
D)
\[\text{1}{{\text{0}}^{\text{-9}}}\text{cm}\]
done
clear
View Answer play_arrow
question_answer 33)
A life is going up. The variation in the speed of the lift is as given in the graph
A)
3.6m
done
clear
B)
28.8m
done
clear
C)
36.0m
done
clear
D)
40.0m
done
clear
View Answer play_arrow
question_answer 34)
34. A2 kg mass is suspended using two strings AB CD as shown in figure
A)
part AB of the string breaks
done
clear
B)
part CD of the string breaks
done
clear
C)
no part of the string breaks
done
clear
D)
both the strings will simultaneously break
done
clear
View Answer play_arrow
question_answer 35)
A body of mass 2 kg slides down a curved track which is quadrant of a circle of radius 1m (figure)
A)
4.43 m/s
done
clear
B)
2 m/s
done
clear
C)
0.5 m/s
done
clear
D)
19.6 m/s
done
clear
View Answer play_arrow
question_answer 36) A lift moving up comes to rest with a retardation. The weight of the man in it
A)
increases
done
clear
B)
decreases
done
clear
C)
remains constant
done
clear
D)
either increases or decreases
done
clear
View Answer play_arrow
question_answer 37) A car of mass m is taking a circular turn of radius r on a frictional level road with a speed v. In order that the car does not skid (\[\mu =\] coefficient of friction)
A)
\[\frac{m{{v}^{2}}}{r}\ge \mu mg\]
done
clear
B)
\[\frac{m{{v}^{2}}}{r}\le \mu mg\]
done
clear
C)
\[\frac{m{{v}^{2}}}{r}=\mu mg\]
done
clear
D)
\[\frac{v}{r}=\mu g\]
done
clear
View Answer play_arrow
question_answer 38) Moment of inertia of a rod of length\[l\] about an axis passing through its edge is (mass of the rod M)
A)
\[\frac{M{{l}^{2}}}{12}\]
done
clear
B)
\[\frac{M{{l}^{2}}}{3}\]
done
clear
C)
\[\frac{M{{l}^{2}}}{2}\]
done
clear
D)
\[\frac{M{{l}^{2}}}{4}\]
done
clear
View Answer play_arrow
question_answer 39) Two bodies of masses \[{{m}_{1}}\] and \[{{m}_{2}}\] have equal momentum. Their kinetic energies \[{{E}_{1}}\] and \[{{E}_{2}}\] are in the ratio
A)
\[\sqrt{{{m}_{1}}:}\sqrt{{{m}_{2}}}\]
done
clear
B)
\[{{m}_{1}}:{{m}_{2}}\]
done
clear
C)
\[{{m}_{2}}:{{m}_{1}}\]
done
clear
D)
\[m_{1}^{2}:m_{2}^{2}\]
done
clear
View Answer play_arrow
question_answer 40) Time period of revolution of a satellite around a planet of radius R is T. The period of revolution around another planet whose radius is 3R is
A)
T
done
clear
B)
3T
done
clear
C)
9T
done
clear
D)
\[3\sqrt{3}T\]
done
clear
View Answer play_arrow
question_answer 41) The total energy of the body executing simple harmonic motion (SHM) is E. Then the kinetic energy when the displacement is half of the amplitude is
A)
\[\frac{E}{2}\]
done
clear
B)
\[\frac{E}{4}\]
done
clear
C)
\[\frac{3E}{4}\]
done
clear
D)
\[\frac{\sqrt{3E}}{4}\]
done
clear
View Answer play_arrow
question_answer 42) An ice-berg is floating in sea water. What percentage of volume of the ice-berg is above the water ? (Given : relative density of sea water 1.2 and relative density or ice = 0.9)
A)
20%
done
clear
B)
25%
done
clear
C)
33.3%
done
clear
D)
50%
done
clear
View Answer play_arrow
question_answer 43) A cylindrical vessel is filled with a liquid of density d upto a height h, if the vessel is at rest, then the pressure at the bottom is
A)
zero
done
clear
B)
\[\frac{h}{2}dg\]
done
clear
C)
\[hdg\]
done
clear
D)
\[2hdg\]
done
clear
View Answer play_arrow
question_answer 44) 44. A thick copper rope of density \[1.5\times {{10}^{3}}kg/{{m}^{3}}\]and young's modulus \[5\times {{10}^{6}}N/{{m}^{2}},\text{ }8m\] in length is hung from the ceiling of a room. The increase in its length due to its own weight is
A)
\[\text{9}\text{.6 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-5}}}\text{m}\]
done
clear
B)
\[\text{19}\text{.6 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-7}}}\text{m}\]
done
clear
C)
\[\text{19}\text{.2 }\!\!\times\!\!\text{ 1}{{\text{0}}^{\text{-7}}}\text{m}\]
done
clear
D)
\[\text{9}\text{.6}\,\text{m}\]
done
clear
View Answer play_arrow
question_answer 45) The excess of pressure due to surface tension in a spherical liquid drop of radius R is proportional To
A)
\[R\]
done
clear
B)
\[{{R}^{3}}\]
done
clear
C)
\[{{R}^{-2}}\]
done
clear
D)
\[{{R}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 46) If one mole of a monoatomic gas \[\left( y=\frac{5}{3} \right)\] is mixed with one mole of a diatomic gas \[\left( y=\frac{7}{3} \right),\] the value of y for the mixture is
A)
1.40
done
clear
B)
1.50
done
clear
C)
1.53
done
clear
D)
3.07
done
clear
View Answer play_arrow
question_answer 47) Oxygen boils at \[-183{}^\circ C\].This temperature is approximately in fahrenheit is
A)
\[-329{}^\circ F\]
done
clear
B)
\[-261{}^\circ F\]
done
clear
C)
\[-215{}^\circ F\]
done
clear
D)
\[-297{}^\circ F\]
done
clear
View Answer play_arrow
question_answer 48) 420 J of energy supplied to 10 g of water will raise its temperature by nearly
A)
\[1{}^\circ C\]
done
clear
B)
\[4.2{}^\circ C\]
done
clear
C)
\[10{}^\circ C\]
done
clear
D)
\[42{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 49) Using which of the following instruments, the temperature of the sun can be determined ?
A)
Platinum thermometer
done
clear
B)
Gas thermometer
done
clear
C)
Pyrometer
done
clear
D)
Vapour pressure thermometer
done
clear
View Answer play_arrow
question_answer 50) An ideal gas undergoes an adiabatic change in volume (V) with pressure (p). Then
A)
\[{{p}^{\gamma }}V\]= constant
done
clear
B)
\[{{p}^{\gamma }}V\]= constant
done
clear
C)
\[{{(pV)}^{\gamma }}\] = constant
done
clear
D)
\[pV\]= constant
done
clear
View Answer play_arrow
question_answer 51) The most electronegative element in periodic table is
A)
cesium
done
clear
B)
chlorine
done
clear
C)
fluorine
done
clear
D)
barium
done
clear
View Answer play_arrow
question_answer 52) A neutral atom can accept electron to form an anion. The process involves
A)
loss of energy
done
clear
B)
gain of energy
done
clear
C)
no change in energy
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 53) The outermost electronic configuration of the most electronegative element is
A)
\[n{{s}^{2}}n{{p}^{3}}\]
done
clear
B)
\[n{{s}^{2}}n{{p}^{4}}\]
done
clear
C)
\[n{{s}^{2}}n{{p}^{5}}\]
done
clear
D)
\[n{{s}^{2}}n{{p}^{6}}\]
done
clear
View Answer play_arrow
question_answer 54) The total number of electrons that take part in forming bond in \[{{O}_{2}}\] is
A)
\[2\]
done
clear
B)
\[4\]
done
clear
C)
\[6\]
done
clear
D)
\[8\]
done
clear
View Answer play_arrow
question_answer 55) Methanol is soluble in water due to
A)
covalent bond nature
done
clear
B)
hydrogen bonding
done
clear
C)
ionic bond nature
done
clear
D)
its poisonous nature
done
clear
View Answer play_arrow
question_answer 56) The number of bonding and anti-bonding respectively in \[CO\] molecule are
A)
\[8,2\]
done
clear
B)
\[2,8\]
done
clear
C)
\[4,2\]
done
clear
D)
\[2,4\]
done
clear
View Answer play_arrow
question_answer 57) The bond order in \[He_{2}^{+}\] ion is
A)
\[0.5\]
done
clear
B)
\[1.0\]
done
clear
C)
\[1.5\]
done
clear
D)
\[2.0\]
done
clear
View Answer play_arrow
question_answer 58) Food irradiation is used to
A)
precook food
done
clear
B)
kill micro-organism
done
clear
C)
kill insects
done
clear
D)
increase nutrient value
done
clear
View Answer play_arrow
question_answer 59) The name catalyst was given by
A)
J.J. Thomson
done
clear
B)
J.J Thomson
done
clear
C)
Berzelius
done
clear
D)
Rutherford
done
clear
View Answer play_arrow
question_answer 60) Vinegar is
A)
\[HCHO\]
done
clear
B)
\[H\,COOH\]
done
clear
C)
\[C{{H}_{3}}CHO\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 61) In which solvent are alkenes most soluble?
A)
water
done
clear
B)
ethyl alcohol
done
clear
C)
carbon tetrachloride
done
clear
D)
ammonia
done
clear
View Answer play_arrow
question_answer 62) Which of the following compounds will show geometrical isomerism?
A)
Propene
done
clear
B)
2-butene
done
clear
C)
Propyne
done
clear
D)
2-butyne
done
clear
View Answer play_arrow
question_answer 63) How many \[\sigma \](sigma) bonds are there in \[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]?
A)
\[3\]
done
clear
B)
\[6\]
done
clear
C)
\[9\]
done
clear
D)
\[12\]
done
clear
View Answer play_arrow
question_answer 64) When acetylene is passed through hot iron tube at \[{{400}^{o}}C,\]it gives
A)
benzene
done
clear
B)
toluene
done
clear
C)
o-xylene
done
clear
D)
methylene
done
clear
View Answer play_arrow
question_answer 65) Which of the following hydrocarbons has acidic hydrogen?s?
A)
1-butane
done
clear
B)
1-butyne
done
clear
C)
2-butene
done
clear
D)
2-butyne
done
clear
View Answer play_arrow
question_answer 66) Which of the following has the highest boiling point?
A)
Diethyl ether
done
clear
B)
n-butyraldehyde
done
clear
C)
n-butyl alcohol
done
clear
D)
n-propyl chloride
done
clear
View Answer play_arrow
question_answer 67) Rectified spirit is
A)
100% ethanol
done
clear
B)
90% ethanol
done
clear
C)
100% methanol
done
clear
D)
95% ethanol
done
clear
View Answer play_arrow
question_answer 68) Which one of the following reagents will react readily with both aldehydes and ketones?
A)
Grignard reagent
done
clear
B)
Fehling?s reagent
done
clear
C)
Tollen?s reagent
done
clear
D)
Schiff?s reagent
done
clear
View Answer play_arrow
question_answer 69) Which of the following compounds will give an iodoform test?
A)
Benzoic acid
done
clear
B)
Ethanol
done
clear
C)
Benzyl chloride
done
clear
D)
M' ethanol
done
clear
View Answer play_arrow
question_answer 70) Acetone undergoes reduction with hydrazine in the presence of \[NaOH\]to form propane. This reaction is known as
A)
Clemmensen reduction
done
clear
B)
Wolff-Kishner reduction
done
clear
C)
Rosenmund reduction
done
clear
D)
Reformat sky reaction
done
clear
View Answer play_arrow
question_answer 71) Which of the following will give acetic acid on acid hydrolysis?
A)
Ethyl acetate
done
clear
B)
Acetone
done
clear
C)
Methyl propionate
done
clear
D)
Lactic acid
done
clear
View Answer play_arrow
question_answer 72) Methylamine reacts with nitrous acid to form
A)
\[C{{H}_{3}}C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}N{{O}_{2}}\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 73) Which of the following compounds does not react with acetyl chloride?
A)
Methylamine
done
clear
B)
Dimethylamine
done
clear
C)
Aniline
done
clear
D)
Trimethylamine
done
clear
View Answer play_arrow
question_answer 74) Which of the following compounds undergoes nitration most readily?
A)
Benzene
done
clear
B)
Toluene
done
clear
C)
Benzoic acid
done
clear
D)
Nitrobenzene
done
clear
View Answer play_arrow
question_answer 75) Which element is used to vulcanise rubber?
A)
\[P\]
done
clear
B)
\[Br\]
done
clear
C)
\[N\]
done
clear
D)
\[S\]
done
clear
View Answer play_arrow
question_answer 76) Epsom salt is
A)
\[MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
B)
\[CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
C)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
D)
\[BaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 77) Oxygen exhibits positive oxidation state in
A)
\[CO\]
done
clear
B)
\[{{F}_{2}}O\]
done
clear
C)
\[NO\]
done
clear
D)
\[{{N}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 78) The poison for platinum catalyst is
A)
sulphur
done
clear
B)
arsenic
done
clear
C)
selenium
done
clear
D)
vanadium
done
clear
View Answer play_arrow
question_answer 79) Chlorine gas is dried over
A)
quicklime
done
clear
B)
caustic soda
done
clear
C)
caustic potash stick
done
clear
D)
cone. sulphuric acid
done
clear
View Answer play_arrow
question_answer 80) The noble gas which is radioactive is
A)
argon
done
clear
B)
helium
done
clear
C)
radon
done
clear
D)
xenon
done
clear
View Answer play_arrow
question_answer 81) The total number of Bravais lattices in crystal is
A)
\[7\]
done
clear
B)
\[32\]
done
clear
C)
\[230\]
done
clear
D)
\[14\]
done
clear
View Answer play_arrow
question_answer 82) If the de-Broglie wavelength of the fourth Bohr's orbit of hydrogen atom is \[4\overset{\text{o}}{\mathop{\text{A}}}\,\] the circumference of the orbit is
A)
\[4\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[4nm\]
done
clear
C)
\[16\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[16\text{ }nm\]
done
clear
View Answer play_arrow
question_answer 83) The s and p characters in bond formed by central atom are equal in
A)
\[C{{H}_{4}}\]
done
clear
B)
\[CH\equiv CH\]
done
clear
C)
\[CH_{3}^{+}\]
done
clear
D)
\[CH_{3}^{-}\]
done
clear
View Answer play_arrow
question_answer 84) The maximum bond strength is in
A)
\[{{O}_{2}}\]
done
clear
B)
\[O_{2}^{+}\]
done
clear
C)
\[O_{2}^{-}\]
done
clear
D)
\[O_{2}^{2-}\]
done
clear
View Answer play_arrow
question_answer 85) The ratio of the radii of the nucleic He and } H is
A)
\[4\]
done
clear
B)
\[2\]
done
clear
C)
\[3\]
done
clear
D)
\[1.6\]
done
clear
View Answer play_arrow
question_answer 86) Radiocarbon dating was developed by
A)
M. Curie
done
clear
B)
M. Calvin
done
clear
C)
W. F. Libby
done
clear
D)
H. Becquerel
done
clear
View Answer play_arrow
question_answer 87) The pH of \[0.1\text{ }M\]aqueous solution of \[N{{H}_{4}}OH\]is \[({{K}_{b}}=1.0\times {{10}^{-5}})\]
A)
\[3\]
done
clear
B)
\[10.5\]
done
clear
C)
\[11\]
done
clear
D)
\[7.5\]
done
clear
View Answer play_arrow
question_answer 88) The work differential, dW, is
A)
a state function
done
clear
B)
an inexact differential
done
clear
C)
exact differential
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 89) For a real gas\[\left( \frac{\partial U}{\partial V} \right)\]is
A)
zero
done
clear
B)
positive
done
clear
C)
negative
done
clear
D)
infinity
done
clear
View Answer play_arrow
question_answer 90) \[10\text{ }moles\]of an ideal gas expands into a vacuum. The work done is
A)
infinity
done
clear
B)
\[10\text{ }J\]
done
clear
C)
zero
done
clear
D)
\[5\text{ }J\]
done
clear
View Answer play_arrow
question_answer 91) In Arrhenius equation for activation energy, \[k=A{{e}^{-{{E}_{a}}/Rt}},\] A represents the following [a] Pre exponential factor [b] Frequency factor [c] Arrhenius factor [d] Collision factor and frequency The correct answer is
A)
\[1\]and \[3\]
done
clear
B)
\[1\] and \[2\]
done
clear
C)
\[3\] and \[4\]
done
clear
D)
\[-2\] and \[3\]
done
clear
View Answer play_arrow
question_answer 92) Which is correct for an endothermic reaction?
A)
\[\Delta E\] is negative
done
clear
B)
\[\Delta H\] is negative
done
clear
C)
\[\Delta H\] is positive
done
clear
D)
\[\Delta H\] is equal to zero
done
clear
View Answer play_arrow
question_answer 93) In the lead-acid battery during charging, the cathode reaction is
A)
reduction of \[P{{b}^{2+}}\] to \[Pb\]
done
clear
B)
formation of \[PbS{{O}_{4}}\]
done
clear
C)
deposition of \[Pb\] at the anode
done
clear
D)
formation of \[Pb{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 94) For an adiabaric process
A)
T = constant
done
clear
B)
Q = 0
done
clear
C)
Q = constant
done
clear
D)
W = 0
done
clear
View Answer play_arrow
question_answer 95) A real solution is that which
A)
obeys Raoult's law
done
clear
B)
obeys Henry's law
done
clear
C)
does not obey Raoult's law
done
clear
D)
obeys Ohm's law
done
clear
View Answer play_arrow
question_answer 96) The continuous rapid zig-zag movement executed by a colloidal particle in the dispersion medium is called
A)
Tyndall effect
done
clear
B)
Brownian movement
done
clear
C)
electrophoresis
done
clear
D)
peptization
done
clear
View Answer play_arrow
question_answer 97) The enthalpy change in the reaction \[2CO+{{O}_{2}}\xrightarrow{{}}2C{{O}_{2}}\]is termed as
A)
enthalpy of reaction
done
clear
B)
enthalpy of fusion
done
clear
C)
enthalpy of combustion
done
clear
D)
enthalpy of formation
done
clear
View Answer play_arrow
question_answer 98) Which of the following can change the value of equilibrium constant for a reaction
A)
changing the concentration of the reactant
done
clear
B)
removing the products as they are formed
done
clear
C)
changing the solvent
done
clear
D)
adding a catalyst
done
clear
View Answer play_arrow
question_answer 99) The vapour pressure of a dilute aqueous solution of glucose is \[740\text{ }mm\]of mercury at\[373\text{ }K\]. The mole fraction of the solute is
A)
\[\frac{1}{20}\]
done
clear
B)
\[\frac{1}{38}\]
done
clear
C)
\[\frac{1}{76}\]
done
clear
D)
\[\frac{1}{740}\]
done
clear
View Answer play_arrow
question_answer 100) How many grams of nickel can be electroplated from a solution of nickel chloride by 4F of electricity?
A)
\[29.3\]
done
clear
B)
\[58.7\]
done
clear
C)
\[117.4\]
done
clear
D)
\[176.1\]
done
clear
View Answer play_arrow
question_answer 101) Which is the unit of DNA?
A)
Chromatin
done
clear
B)
Euchromatm
done
clear
C)
Chromosomes
done
clear
D)
Chromatids
done
clear
View Answer play_arrow
question_answer 102) Which is the property of bile juice?
A)
Not have any enzymes
done
clear
B)
It has crypsin
done
clear
C)
It has enterogastrone
done
clear
D)
It has trypofnomide
done
clear
View Answer play_arrow
question_answer 103) In humans, which side of cerebral hemisphere recognises the faces of friends and family members?
A)
Left cerebral hemisphere
done
clear
B)
Right cerebral hemisphere
done
clear
C)
Both [a] and [b]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 104) The extra embryonic membranes of mammalian embryo are derived from
A)
inner cell mass
done
clear
B)
trophoblast
done
clear
C)
formative cells
done
clear
D)
follicle cells
done
clear
View Answer play_arrow
question_answer 105) Red tide is caused by
A)
Protozoans
done
clear
B)
corals
done
clear
C)
dinoflagellates
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 106) Find the out odd one
A)
Guinea worm
done
clear
B)
Hookworm
done
clear
C)
Ringworm
done
clear
D)
Tapeworm
done
clear
View Answer play_arrow
question_answer 107) The major constituent of vertebrate bone is
A)
calcium phosphate
done
clear
B)
calcium carbonate
done
clear
C)
sodium chloride
done
clear
D)
potassium hydroxide
done
clear
View Answer play_arrow
question_answer 108) Which of the following animal belongs to phylum-Mollusca ?
A)
Globe fish
done
clear
B)
Star fish
done
clear
C)
Silver fish
done
clear
D)
Cattle fish
done
clear
View Answer play_arrow
question_answer 109) Honey that has a high concentration of sugar, does not decay because
A)
it contains natural antioxidant that prevent bacterial attack
done
clear
B)
bacteria cannot survive in an active state in a solution of high osmotic strength, as water is drawn out from honey
done
clear
C)
in honey bacteria are totally deprived of oxygen
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 110) Which of the following is iron storing protein?
A)
Alpha globulin
done
clear
B)
Gamma globulin
done
clear
C)
Ferritin
done
clear
D)
Albumins
done
clear
View Answer play_arrow
question_answer 111) Corpus striatum is found in
A)
paracoel
done
clear
B)
diocoel
done
clear
C)
metacoel
done
clear
D)
cerebral hemisphere
done
clear
View Answer play_arrow
question_answer 112) A rabbit runs very fast but after some time feel tired, because
A)
formation of lactic acid in muscles
done
clear
B)
formation of succinic acid in muscles
done
clear
C)
loss of energy
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 113) Body cells infected with virus produce a protein Called
A)
interferon
done
clear
B)
properdin
done
clear
C)
Gamma globulin
done
clear
D)
hybridoma
done
clear
View Answer play_arrow
question_answer 114) The cytoplasmic segement of striated muscle fibre is termed
A)
metamere
done
clear
B)
neuromere
done
clear
C)
sarcoplasm
done
clear
D)
sarcomere
done
clear
View Answer play_arrow
question_answer 115) Capacitation of sperm in mammals involves
A)
sperm size change
done
clear
B)
acrosome reaction
done
clear
C)
nuclear chromatin change
done
clear
D)
mitochondrial alignment
done
clear
View Answer play_arrow
question_answer 116) Which one of the following is a pandemic disease ?
A)
Amoebic dysentery
done
clear
B)
Hepatitis
done
clear
C)
Filariasis
done
clear
D)
Influenza
done
clear
View Answer play_arrow
question_answer 117) Products of adenine and guanine metabolism are excreted by humans as
A)
ammonia
done
clear
B)
urea
done
clear
C)
uric acid
done
clear
D)
allantoin
done
clear
View Answer play_arrow
question_answer 118) Peripatus is a connecting link of
A)
Reptiles and Mammals
done
clear
B)
Flat worms and Annelida
done
clear
C)
Mollusca and Arthropoda
done
clear
D)
Annelida and Arthropoda
done
clear
View Answer play_arrow
question_answer 119) Which of the following is a hormonal disease ?
A)
Cold
done
clear
B)
Goitre
done
clear
C)
Tuberculosis
done
clear
D)
Leprosy
done
clear
View Answer play_arrow
question_answer 120) One gene-one enzyme hypothesis is proposed by
A)
Watson and Crick
done
clear
B)
Beadle and Tatum
done
clear
C)
Singer and Nicolson
done
clear
D)
Schleiden and Schwan
done
clear
View Answer play_arrow
question_answer 121) The skin colour of the humans are regulated by
A)
multiple alleles
done
clear
B)
lethal genes
done
clear
C)
polygenes
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 122) The thickening of wall of artery is called
A)
aneurysm
done
clear
B)
arteriosclerosis
done
clear
C)
arthritis
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
question_answer 123) Which blood group has both 'a' and V antibodies ?
A)
0
done
clear
B)
AB
done
clear
C)
A
done
clear
D)
B
done
clear
View Answer play_arrow
question_answer 124) Passive food ingestion in Amoeba is known as
A)
invagination
done
clear
B)
import
done
clear
C)
drcumfluence
done
clear
D)
circumvallation
done
clear
View Answer play_arrow
question_answer 125) Palaeontology is the study of
A)
birds
done
clear
B)
bones
done
clear
C)
primates
done
clear
D)
fossils
done
clear
View Answer play_arrow
question_answer 126) The thread-like structures that begin to radiate from each centromere by the end of prophase are
A)
kinetochore microtubules
done
clear
B)
polar microtubules
done
clear
C)
aster microtubule
done
clear
D)
spindle microtubules
done
clear
View Answer play_arrow
question_answer 127) In which vitamin the cobalt metal is present ?
A)
\[{{B}_{1}}\]
done
clear
B)
\[{{B}_{2}}\]
done
clear
C)
\[{{B}_{6}}\]
done
clear
D)
\[{{B}_{12}}\]
done
clear
View Answer play_arrow
question_answer 128) Which of the following is the least poisonous nitrogenous waste ?
A)
Ammonia
done
clear
B)
Urea
done
clear
C)
Uric acid
done
clear
D)
D-Ammonia and urea
done
clear
View Answer play_arrow
question_answer 129) Chloride shift is essential for the transportation of
A)
\[C{{O}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[C{{O}_{2}}\] and \[{{O}_{2}}\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 130) The sensory neurons of a vertebrate retina are
A)
rods and cones
done
clear
B)
ganglion cells
done
clear
C)
amacrine cells
done
clear
D)
bipolar cells
done
clear
View Answer play_arrow
question_answer 131) At which stage does cytokinesis begin ?
A)
Anaphase
done
clear
B)
Prophase
done
clear
C)
Metaphase
done
clear
D)
Interphase
done
clear
View Answer play_arrow
question_answer 132) Each restriction enzymes cleaves a molecule only at
A)
methyl groups
done
clear
B)
the ends of genes
done
clear
C)
particular nucleotide sequence
done
clear
D)
the time of DNA replication
done
clear
View Answer play_arrow
question_answer 133) The bio concentration of DDT in the body fat of Indian people is
A)
13-31 ppm
done
clear
B)
18-36 ppm
done
clear
C)
5-10 ppm
done
clear
D)
2-5 ppm
done
clear
View Answer play_arrow
question_answer 134) In oogenesis, when a diploid cell in the ovary undergoes meiosis, how many ova results ?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 135) Natural selection acts on an organism's
A)
dominant alleles
done
clear
B)
recessive alleles
done
clear
C)
phenorype
done
clear
D)
genotype
done
clear
View Answer play_arrow
question_answer 136) An allergic reaction is initiated by antibodies of the
A)
\[\text{IgG}\]group
done
clear
B)
\[\text{Ig}\,\text{M}\]
done
clear
C)
\[\text{Ig}\,\text{A}\]
done
clear
D)
\[\text{Ig}\,\text{E}\]
done
clear
View Answer play_arrow
question_answer 137) The virus that causes Acquired Immune Deficiency Syndrome (AIDS) parasitizes
A)
B-cells
done
clear
B)
cytotoxic T-cells
done
clear
C)
helper T-cells
done
clear
D)
the membrane of lymph nodes
done
clear
View Answer play_arrow
question_answer 138) The geographic limits within which a population exists is its
A)
niche
done
clear
B)
habitat
done
clear
C)
range
done
clear
D)
territory
done
clear
View Answer play_arrow
question_answer 139) Which of the following taxonomic categories contains organisms least similar to one another?
A)
Class
done
clear
B)
Genus
done
clear
C)
Family
done
clear
D)
Species
done
clear
View Answer play_arrow
question_answer 140) Which of the following is a type of intercellular Junction between animals cells ?
A)
Middle lamella
done
clear
B)
Plasmodesma
done
clear
C)
Desmosome
done
clear
D)
Glycocalyx
done
clear
View Answer play_arrow
question_answer 141) Human sperm cells move by means of a
A)
dlium
done
clear
B)
flagellum
done
clear
C)
basal body
done
clear
D)
hair
done
clear
View Answer play_arrow
question_answer 142) Which of the following processes makes direct use of oxygen ?
A)
Glycolysis
done
clear
B)
Fermentation
done
clear
C)
Krebs' cycle
done
clear
D)
Electron transport
done
clear
View Answer play_arrow
question_answer 143) Genes that are inactive for long periods of time tend to be bonded to
A)
each other
done
clear
B)
methyl groups
done
clear
C)
actin and myosin
done
clear
D)
the nucleolus
done
clear
View Answer play_arrow
question_answer 144) M. A virus that can reproduce without killing its host is called a
A)
lyric virus
done
clear
B)
retroactive virus
done
clear
C)
temperate virus
done
clear
D)
virion
done
clear
View Answer play_arrow
question_answer 145) An organism with two different alleles is
A)
homozygous for that trait
done
clear
B)
homologous for the allele
done
clear
C)
heterozygous for that trait
done
clear
D)
heterologous for the alleles
done
clear
View Answer play_arrow
question_answer 146) The gerotypic expression seen in a person of blood group AB is called
A)
dominant-recessive
done
clear
B)
codominance
done
clear
C)
incomplete dominance
done
clear
D)
corecession
done
clear
View Answer play_arrow
question_answer 147) The genetic basis of haemophilia was revealed by studies of
A)
DNA sequences
done
clear
B)
chromosome in gametes
done
clear
C)
family pedigree
done
clear
D)
prenatal gene products
done
clear
View Answer play_arrow
question_answer 148) Diseases caused by pleiotropic genes are
A)
syndromes
done
clear
B)
reversible by diet therapy
done
clear
C)
reversible by gene therapy
done
clear
D)
extremely rare
done
clear
View Answer play_arrow
question_answer 149) Tobacoo smoke contains
A)
\[C{{O}_{2}}\] hydrocarbons and tar
done
clear
B)
\[C{{O}_{2}}\]and tar
done
clear
C)
\[C{{O}_{2}}\], polycyclic aromatic hydrocarbons and tar
done
clear
D)
\[C{{O}_{2}},CO\] and tar
done
clear
View Answer play_arrow
question_answer 150) Alcohol depresses nervous system and acts as
A)
sedative agents
done
clear
B)
analgesic agent
done
clear
C)
anesthetic agents
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 151) Merozygore is formed in
A)
Yeast
done
clear
B)
Spirogyra
done
clear
C)
Escherichia coli
done
clear
D)
Neurospora
done
clear
View Answer play_arrow
question_answer 152) The fermented food of Mucor is
A)
tempe kedel
done
clear
B)
tofu
done
clear
C)
tempe kedel
done
clear
D)
sufu
done
clear
View Answer play_arrow
question_answer 153) The theory that all the cells arise from pre-existing cells (Omnis cellula e cellula) was proposed by
A)
Schwann (1839)
done
clear
B)
Brown (1831)
done
clear
C)
Virchow (1855)
done
clear
D)
Schleiden (1938)
done
clear
View Answer play_arrow
question_answer 154) C He most studied prokaryote is
A)
Klebsiella pneumonia
done
clear
B)
Pseudomonas sp
done
clear
C)
Escherichia coli
done
clear
D)
Badus thuringiensis
done
clear
View Answer play_arrow
question_answer 155) Which bacterium has two chromosomes in nucleoid?
A)
Artho bacterium sp
done
clear
B)
Vibrio cholera
done
clear
C)
Xanthomonas citri
done
clear
D)
Closmdium
done
clear
View Answer play_arrow
question_answer 156) Which of the following ribosomal RNA is most extensively used to establish phylogenetic relationship among eukaryotes?
A)
16S
done
clear
B)
18S
done
clear
C)
5S
done
clear
D)
34S
done
clear
View Answer play_arrow
question_answer 157) Annealing' is a phenomenon related to
A)
denaturation of DNA
done
clear
B)
renamration of DNA
done
clear
C)
melting point of DNA (Tm)
done
clear
D)
separation of G from C
done
clear
View Answer play_arrow
question_answer 158) One of the following plasmid produces lacteriocin' which is lethal to other strains of same genus
A)
\[Ti\]
done
clear
B)
\[Cpl\text{ }E\]
done
clear
C)
\[{{\text{R}}_{\text{100}}}\]
done
clear
D)
\[pB{{r}^{322}}\]
done
clear
View Answer play_arrow
question_answer 159) Lysosomes are not found in
A)
Leucocytes
done
clear
B)
Amoeba
done
clear
C)
Hela cells
done
clear
D)
Mammalian erythrocytes
done
clear
View Answer play_arrow
question_answer 160) Chromoplast contains pigment other than.
A)
chlorophyll
done
clear
B)
anthocyanin
done
clear
C)
\[\text{ }\!\!\beta\!\!\text{ -}\]carotene
done
clear
D)
xanthophylls
done
clear
View Answer play_arrow
question_answer 161) A dihybrid cross between pea plants with "yellow round' and 'green wrinkled', seed in \[{{F}_{2}}\] generation genotype yy Rr yy rR means
A)
heterozygous yellow wrinkled
done
clear
B)
heterozygous yellow round
done
clear
C)
heterozygous green round
done
clear
D)
heterozygous green wrinkled
done
clear
View Answer play_arrow
question_answer 162) In male human, how many linkage groups occur in their chromosome ?
A)
22
done
clear
B)
24
done
clear
C)
23
done
clear
D)
46
done
clear
View Answer play_arrow
question_answer 163) During crossing over, synaptonemal complex in between two paired chromosome is formed in which stage of meiosis?
A)
Diplotene
done
clear
B)
Diakinesis
done
clear
C)
Pachytene
done
clear
D)
Leptotene
done
clear
View Answer play_arrow
question_answer 164) The enzyme which act to hydrolyse or breakdown a polynucleotide chain into its component nucleotide is
A)
gyrases
done
clear
B)
polymerases
done
clear
C)
nucleases
done
clear
D)
primases
done
clear
View Answer play_arrow
question_answer 165) In most organisms the 'initiation codon' is
A)
UAG
done
clear
B)
AUG
done
clear
C)
GUA
done
clear
D)
GAU
done
clear
View Answer play_arrow
question_answer 166) In E. coli, the average life of mRNA is about
A)
2h
done
clear
B)
2 min
done
clear
C)
2 s
done
clear
D)
indefinite period
done
clear
View Answer play_arrow
question_answer 167) Antibiotic which inhibits cell wall synthesis in bacteria
A)
chloramphenicol
done
clear
B)
erythromycin
done
clear
C)
penicillin
done
clear
D)
tetracycline
done
clear
View Answer play_arrow
question_answer 168) Cell wall of xylem is rich in
A)
starch
done
clear
B)
lignin
done
clear
C)
lipid
done
clear
D)
protein
done
clear
View Answer play_arrow
question_answer 169) Anatomically jute fibres are
A)
xylem fibres
done
clear
B)
phloem fibres
done
clear
C)
sclerenchyma fibres
done
clear
D)
cortical fibres
done
clear
View Answer play_arrow
question_answer 170) The first sporophyte arising from pollen grain was made by Guha and Maheshwari (1966) in
A)
Hyacinthus
done
clear
B)
Datura
done
clear
C)
Drosera
done
clear
D)
Glycine max
done
clear
View Answer play_arrow
question_answer 171) The haustorial behaviour where entire embryo sac grow beyond the ovular tissue occur in
A)
Santalum
done
clear
B)
Zea mays
done
clear
C)
Argemone
done
clear
D)
Datura
done
clear
View Answer play_arrow
question_answer 172) The chief source of nutrition of embryo inside the ovule is
A)
endosperm
done
clear
B)
ovular tissue
done
clear
C)
embryo sac
done
clear
D)
haustoria
done
clear
View Answer play_arrow
question_answer 173) Cereal proteins are deficient in one of the following amino acid
A)
methionine
done
clear
B)
threonine
done
clear
C)
lysine
done
clear
D)
glutamic acid
done
clear
View Answer play_arrow
question_answer 174) 'Elicitors' are molecule which stimulate production of
A)
primary metabolites
done
clear
B)
secondary metabolites
done
clear
C)
tertiary metabolites
done
clear
D)
all metabolites
done
clear
View Answer play_arrow
question_answer 175) Malolactic fermentation during wine production is performed by
A)
Lactobacillus
done
clear
B)
Leuconostoc
done
clear
C)
Saccharomyces
done
clear
D)
As pergillus
done
clear
View Answer play_arrow
question_answer 176) 'Downstream process' in fermentation technology means
A)
secretion of metabolites in stationary phase
done
clear
B)
actual recovery of useful product
done
clear
C)
removal of waste after fermentation
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 177) Cephalosporum acremonium a fungus which produces cephalosporin is
A)
marine fungus
done
clear
B)
fresh water fungus
done
clear
C)
soil fungus
done
clear
D)
endophyte
done
clear
View Answer play_arrow
question_answer 178) Eukaryotes lack
A)
membrane bound nucleus
done
clear
B)
complex membranous internal organ
done
clear
C)
gas vesicle
done
clear
D)
mRNA in trons
done
clear
View Answer play_arrow
question_answer 179) T series bacteriophage possess complex tail structure is
A)
\[{{T}_{1}}\]
done
clear
B)
\[{{T}_{2}}\]
done
clear
C)
\[{{T}_{3}}\]
done
clear
D)
\[{{T}_{4}}\]
done
clear
View Answer play_arrow
question_answer 180) Mycoplasmas differs from other bacteria in their
A)
DNA
done
clear
B)
RNA
done
clear
C)
cell wall
done
clear
D)
nutrition
done
clear
View Answer play_arrow
question_answer 181) Cyanobacteria differs from other groups of bacteria in their
A)
DNA
done
clear
B)
cell wall
done
clear
C)
nutrition
done
clear
D)
reproduction
done
clear
View Answer play_arrow
question_answer 182) Nostoc does not form symbiotic association with
A)
Cycos
done
clear
B)
Anthoceros
done
clear
C)
Pinus
done
clear
D)
Gunnera
done
clear
View Answer play_arrow
question_answer 183) Penicillin is produced commercially from
A)
Penidllium notatum
done
clear
B)
P. chrysogenum
done
clear
C)
P. citrinum
done
clear
D)
P. rubrum
done
clear
View Answer play_arrow
question_answer 184) Which of the following statement is true?
A)
Pure water has minimum water potential
done
clear
B)
Greater the water molecule in a system lesser the kinetic energy
done
clear
C)
Water always move from lower water potential to higher one
done
clear
D)
The water potential of pure water is zero.
done
clear
View Answer play_arrow
question_answer 185) Alga commonly known as "water silk' or 'pond silk' is
A)
Zygnema
done
clear
B)
Spirogyra
done
clear
C)
Cladophora
done
clear
D)
Oedogonium
done
clear
View Answer play_arrow
question_answer 186) In Funaria stomata are found in
A)
capsule
done
clear
B)
foot
done
clear
C)
seta
done
clear
D)
thallus
done
clear
View Answer play_arrow
question_answer 187) Trabeculae in Selaginella is
A)
modified pericycle
done
clear
B)
elongated endodermis
done
clear
C)
elongated cortical cell
done
clear
D)
modified xylem vessel
done
clear
View Answer play_arrow
question_answer 188) Rhizobia form nodule in Glycine max (soybean) is
A)
Azorhizobium
done
clear
B)
Bradyrhizobium
done
clear
C)
Sinorhizobium
done
clear
D)
Mesorhizobium
done
clear
View Answer play_arrow
question_answer 189) The scientific name of 'Haldi' is
A)
Curcuma longa
done
clear
B)
C. aromatic
done
clear
C)
C. zedoaria
done
clear
D)
C. leucorhiza
done
clear
View Answer play_arrow
question_answer 190) The most primitive family of dicot is
A)
Rosaceae
done
clear
B)
Magnoliaceae
done
clear
C)
Utricaceae
done
clear
D)
Cactaceae
done
clear
View Answer play_arrow
question_answer 191) The present level (2009-10) of \[C{{O}_{2}}\] (ppm) in atmosphere is about
A)
346
done
clear
B)
385
done
clear
C)
365
done
clear
D)
355
done
clear
View Answer play_arrow
question_answer 192) My corrhizal association of which plant enables it to utilize amino acid as N-source?
A)
Strawberry
done
clear
B)
Cranberry
done
clear
C)
Orchids
done
clear
D)
Pinus
done
clear
View Answer play_arrow
question_answer 193) Which part of the root is most active in absorption of phosphate?
A)
Root cap
done
clear
B)
Meristematic zone
done
clear
C)
Root hairs
done
clear
D)
Root apex
done
clear
View Answer play_arrow
question_answer 194) The main part of entry of \[C{{O}_{3}}\] into leaf is
A)
cuticle
done
clear
B)
epidermis
done
clear
C)
stomata
done
clear
D)
pore
done
clear
View Answer play_arrow
question_answer 195) Oxygen liberated during photosynthesis comes from
A)
carbon dioxide
done
clear
B)
water
done
clear
C)
chlorophyll
done
clear
D)
phosphoglyceric acid
done
clear
View Answer play_arrow
question_answer 196) In anaerobic respiration ATP produced (mol.) is
A)
24
done
clear
B)
12
done
clear
C)
2
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 197) Which is an example of 'hydrolases'?
A)
Catalase
done
clear
B)
Trypsin
done
clear
C)
Oxidases
done
clear
D)
Transaminases
done
clear
View Answer play_arrow
question_answer 198) Which one of the following is 100 times more reactive than CFC in destroying ozone ?
A)
\[\text{IFC}\]
done
clear
B)
\[\text{Br}\,\text{FC}\]
done
clear
C)
\[\text{UV-B}\]
done
clear
D)
\[\text{UV-C}\]
done
clear
View Answer play_arrow
question_answer 199) Which of the following process helps in nutrient conservation ?
A)
Meneralization
done
clear
B)
Leaching
done
clear
C)
Immobilization
done
clear
D)
Nitrification
done
clear
View Answer play_arrow
question_answer 200) Engine fitted in two wheeler perform compel combustion of fuel is
A)
two stroke
done
clear
B)
three stroke
done
clear
C)
four stroke
done
clear
D)
one stroke
done
clear
View Answer play_arrow
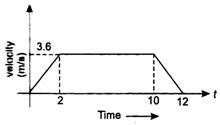 What is the height to which the lift takes the passengers ?
What is the height to which the lift takes the passengers ?
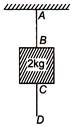 A sudden jerk is given to the end D of the string, then
A sudden jerk is given to the end D of the string, then
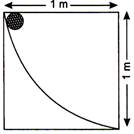 All the surfaces are frictionless. If the body starts from rest, its speed at the bottom of the track is
All the surfaces are frictionless. If the body starts from rest, its speed at the bottom of the track is
