A) \[2CuS{{O}_{4}}+4KI\to C{{u}_{2}}{{I}_{2}}+2{{K}_{2}}S{{O}_{4}}+{{I}_{2}}\]
B) \[S{{O}_{2}}+{{H}_{2}}O\to {{H}_{2}}S{{O}_{3}}\]
C) \[N{{a}_{2}}S{{O}_{4}}+BaC{{l}_{2}}\to BaS{{O}_{4}}+2NaCl\]
D) \[CuS{{O}_{4}}+4N{{H}_{3}}\to [Cu{{(N{{H}_{3}})}_{4}}]S{{O}_{4}}\]
E) \[{{C}_{12}}{{H}_{22}}{{O}_{11}}+{{H}_{2}}O\to {{C}_{6}}{{H}_{12}}{{O}_{6}}+{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
Correct Answer: A
Solution :
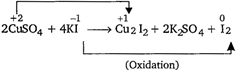 In redox reaction, oxidation and reduction occurs simultaneously in a reaction.
In redox reaction, oxidation and reduction occurs simultaneously in a reaction.
You need to login to perform this action.
You will be redirected in
3 sec
