question_answer 1) A ray of light is incident at an angle of, incidence \[60{}^\circ \] from air to liquid. What is the 1 ratio of width from air to liquid if angle of refraction is\[45{}^\circ \]?
A)
\[\frac{\sqrt{2}}{3}\]
done
clear
B)
\[\sqrt{\frac{3}{2}}\]
done
clear
C)
\[\sqrt{\frac{2}{3}}\]
done
clear
D)
\[\frac{1}{\sqrt{2}}\]
done
clear
E)
\[\frac{\sqrt{3}}{2}\]
done
clear
View Answer play_arrow
question_answer 2) The nature of path of an electron when its moves in transverse electric field is
A)
circle
done
clear
B)
ellipse
done
clear
C)
parabola
done
clear
D)
hyperbola
done
clear
E)
straight line
done
clear
View Answer play_arrow
question_answer 3) In photoelectric emission, one photon is capable of emitting
A)
one electron
done
clear
B)
two electrons
done
clear
C)
more electrons
done
clear
D)
Both (a) and (b)
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 4) Identify the incorrect relation.
A)
\[\alpha =\frac{\beta }{1-\beta }\]
done
clear
B)
\[\beta =\frac{\alpha }{1-\alpha }\]
done
clear
C)
\[\alpha =\frac{\beta }{1+\beta }\]
done
clear
D)
\[1-\alpha =\frac{1}{1+\beta }\]
done
clear
E)
\[\beta =\alpha (1+\beta )\]
done
clear
View Answer play_arrow
question_answer 5) The depletion layer in p - n junction region is caused by
A)
drift of electrons
done
clear
B)
migration of impurity ions
done
clear
C)
drift of holes
done
clear
D)
diffusion of charge carriers
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 6) Dimensional formula for entropy is identical to that of
A)
universal gas constant
done
clear
B)
specific heat
done
clear
C)
Boltzmann constant
done
clear
D)
gravitational potential
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 7) If the velocity of projection is increased by 1% (other things remaining constant), the horizontal range will increase by
A)
1 %
done
clear
B)
2 %
done
clear
C)
4 %
done
clear
D)
8 %
done
clear
E)
10 %
done
clear
View Answer play_arrow
question_answer 8) A mass m is placed on an inclined plane. If the mass is in equilibrium, the maximum inclination of the plane with the horizontal would be: (where \[\mu \] is the coefficient of friction between the mass and surface)
A)
\[{{\tan }^{-1}}\left( \mu \right)\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{\mu }{2} \right)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \frac{\mu }{m} \right)\]
done
clear
D)
\[{{\cos }^{-1}}\left( \mu \right)\]
done
clear
E)
\[{{\tan }^{-1}}\left( \frac{m}{\mu } \right)\]
done
clear
View Answer play_arrow
question_answer 9) Light of two different frequencies whose photons have energies 1 eV and 2.5 eV successively illuminate a metal of work function 0.5 eV. The ratio of the maximum speeds of the emitted electrons will be
A)
1 : 5
done
clear
B)
1 : 4
done
clear
C)
1 : 2
done
clear
D)
1 : 1
done
clear
E)
2 : 1
done
clear
View Answer play_arrow
question_answer 10) The half - life of a radioactive element depends upon
A)
temperature
done
clear
B)
pressure
done
clear
C)
nature of element
done
clear
D)
amount of radioactive element
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 11) The gravitational field in a region is given by\[I=\left( 4\widehat{i}+\widehat{j} \right)\]N/kg. The work done by this field is zero when a particle is moved along the line
A)
\[~x+y=6\]
done
clear
B)
\[x+4y=6\]
done
clear
C)
\[y+4x=6\]
done
clear
D)
\[x-y=6\]
done
clear
E)
\[x+y=8\]
done
clear
View Answer play_arrow
question_answer 12) A satellite with kinetic energy E revolving round the earth in a circular orbit. The minimum additional KE required for it to escape into outer space is
A)
\[\sqrt{2}E\]
done
clear
B)
2 E
done
clear
C)
\[E/\sqrt{2}\]
done
clear
D)
E
done
clear
E)
\[\frac{E}{2}\]
done
clear
View Answer play_arrow
question_answer 13) The magnitude of the force developed by raising the temperature from \[0{}^\circ C\] to \[100{}^\circ C\] of the iron bar 1.0 m long and \[1c{{m}^{2}}\]cross-section, when it is held so that it is not permitted to expand or bend is \[\left( \alpha ={{10}^{5}}{{/}^{0}}C,Y={{10}^{11}}N/{{m}^{2}} \right)\]
A)
\[{{10}^{3}}N\]
done
clear
B)
\[{{10}^{4}}N\]
done
clear
C)
\[{{10}^{5}}N\]
done
clear
D)
\[{{10}^{9}}N\]
done
clear
E)
\[{{10}^{2}}N\]
done
clear
View Answer play_arrow
question_answer 14) The alpha and beta particles cause ionization because of
A)
photoelectric emission
done
clear
B)
compton collision
done
clear
C)
pair production
done
clear
D)
the electrostatic force
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 15) Which one of the following is the weakest kind of bonding in solids?
A)
Ionic
done
clear
B)
Metallic
done
clear
C)
vander Waals
done
clear
D)
Covalent
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 16) A wave travelling along a stretched string is represented by y= 3 cos\[\pi \](100 t - x). Its wavelength is
A)
3 cm
done
clear
B)
100 cm
done
clear
C)
2 cm
done
clear
D)
5 cm
done
clear
E)
8 cm
done
clear
View Answer play_arrow
question_answer 17) A disc of mass 10 g is kept horizontally in air by firing bullets of mass 5 g each at the rate of 10 per second. If the bullets rebound with the same the same speed, what is the velocity with which the bullets are fired?
A)
49 cm/s
done
clear
B)
98 cm/s
done
clear
C)
147 cm/s
done
clear
D)
196 cm/s
done
clear
E)
208 cm/s
done
clear
View Answer play_arrow
question_answer 18) Frequency of the series limit of Balmer series of hydrogen atom in term of Rydberg constant R and velocity of light c is
A)
\[\frac{4}{Rc}\]
done
clear
B)
4 Rc
done
clear
C)
\[\frac{Rc}{4}\]
done
clear
D)
\[Rc\]
done
clear
E)
\[\frac{Rc}{2}\]
done
clear
View Answer play_arrow
question_answer 19) The inductive reactance \[{{X}_{L}}\] of induction is
A)
always constant in AC circuit
done
clear
B)
directly proportional to frequency of AC
done
clear
C)
inversely proportional to frequency of AC
done
clear
D)
directly proportional to time period J
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 20) A 100 mH coil carries 1 A current. Energy stored in its magnetic field is
A)
0.1 J
done
clear
B)
0.05 J
done
clear
C)
0.5 J
done
clear
D)
1 J
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 21) Liquids and gases never show
A)
diamagnetic properties
done
clear
B)
paramagnetic properties
done
clear
C)
ferromagnetic properties
done
clear
D)
Both (a) and (b)
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 22) The deflection of tangent galvanometer is governed
A)
by a horse shoe magnet
done
clear
B)
by current in a circular coil
done
clear
C)
by current in a rectangular coil placed between the pole pieces of a magnet
done
clear
D)
by pole pieces of an external bar magnet
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 23) Two small magnets each of magnetic moment \[10\text{ }A{{m}^{2}}\]are placed in end on position 0.1 m apart from their centres. The force acting between them is
A)
\[0.6\times {{10}^{7}}N\]
done
clear
B)
\[0.06\times {{10}^{-7}}N\]
done
clear
C)
\[0.6\text{ }N\]
done
clear
D)
\[0.06\text{ }N\]
done
clear
E)
\[0.06\times {{10}^{-10}}N\]
done
clear
View Answer play_arrow
question_answer 24) A coil of cross-sectional area \[400\text{ }c{{m}^{2}}\]having 30 turns is making 180 rev/min in a magnetic field of 1 T. The peak value of the induced emf is
A)
0.4 V
done
clear
B)
0.6 V
done
clear
C)
226 V
done
clear
D)
2.26 V
done
clear
E)
22.6 V
done
clear
View Answer play_arrow
question_answer 25) For a thermocouple, the temperature of cold junction\[({{T}_{c}}),\]the neutral temperature \[({{T}_{n}})\] and the temperature of inversion \[({{T}_{i}})\] are \[0{}^\circ C,\] \[285{}^\circ C,\] \[570{}^\circ C\] respectively. If the temperature of cold junction \[({{T}_{c}})\] is raised to \[10{}^\circ C,\] then
A)
\[{{T}_{n}}=275{}^\circ C\] and \[{{T}_{i}}=570{}^\circ C\]
done
clear
B)
\[{{T}_{n}}=275{}^\circ C\] and \[{{T}_{i}}=560{}^\circ C\]
done
clear
C)
\[{{T}_{n}}=285{}^\circ C\] and \[{{T}_{i}}=560{}^\circ C\]
done
clear
D)
\[{{T}_{n}}=295{}^\circ C\] and \[{{T}_{i}}=580{}^\circ C\]
done
clear
E)
\[{{T}_{n}}=560{}^\circ C\] and \[{{T}_{i}}=285{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 26) The wires A and B are of same material and same length but their radii are in the ratio 1 : 2. They are stretched by the same force. Strain produced in two wires is
A)
2 : 1
done
clear
B)
1 : 2
done
clear
C)
1 : 4
done
clear
D)
4 : 1
done
clear
E)
1 : 8
done
clear
View Answer play_arrow
question_answer 27) Heat is transmitted from higher to lower temperature due to the molecular collision in
A)
conduction
done
clear
B)
radiation
done
clear
C)
convection
done
clear
D)
convection and radiation
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 28) A wire of length 10 cm is placed horizontal on the surface of water and is gently pulled up with a force of \[1.8\times {{10}^{-2}}N\]to keep the wire in equilibrium. The surface tension of water will be
A)
0.99 N/m
done
clear
B)
0.09 N/m
done
clear
C)
0.59 m
done
clear
D)
59 N/m
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 29) A solid sphere rolls down without slipping from rest on a 30° incline. Its linear acceleration is
A)
\[\frac{5g}{7}\]
done
clear
B)
\[\frac{5g}{14}\]
done
clear
C)
\[\frac{2g}{3}\]
done
clear
D)
\[\frac{g}{3}\]
done
clear
E)
\[\frac{5g}{17}\]
done
clear
View Answer play_arrow
question_answer 30) The time period of simple pendulum in a satellite is
A)
4 s
done
clear
B)
zero
done
clear
C)
cannot be calculated
done
clear
D)
infinite
done
clear
E)
\[\frac{3}{2}\]s
done
clear
View Answer play_arrow
question_answer 31) An electric bulb has power rating of 60 W, 220 V. If it is connected to a source of rms voltage 110 V, the power consumed is
A)
125 W
done
clear
B)
15 W
done
clear
C)
30 W
done
clear
D)
60 W
done
clear
E)
40 W
done
clear
View Answer play_arrow
question_answer 32) In an AC circuit, E = 220 sin 100\[\pi t\]. If the impedance is 110\[\Omega \] and phase angle\[\phi =60{}^\circ ,\] the power consumption is
A)
440 W
done
clear
B)
220 W
done
clear
C)
30 W
done
clear
D)
55 W
done
clear
E)
1000 W
done
clear
View Answer play_arrow
question_answer 33) The Lyman series of hydrogen lies in the region
A)
microwave
done
clear
B)
infrared
done
clear
C)
visible
done
clear
D)
ultraviolet
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 34) A radioactive element disintegrates S - h and its 1/16th part remain undisimegrated. Half life of the element will be
A)
0.5 h
done
clear
B)
1 h
done
clear
C)
8 h
done
clear
D)
4 h
done
clear
E)
2 h
done
clear
View Answer play_arrow
question_answer 35) Ozone layer blocks the radiation of wavelength
A)
more than \[3\times {{10}^{-7}}m\]
done
clear
B)
less than \[3\times {{10}^{-7}}m\]
done
clear
C)
equal to \[3\times {{10}^{-7}}m\]
done
clear
D)
equal to \[{{10}^{-7}}m\]
done
clear
E)
more than \[{{10}^{6}}m\]
done
clear
View Answer play_arrow
question_answer 36) The resistance of discharge tube is
A)
non-ohmic
done
clear
B)
zero
done
clear
C)
ohmic
done
clear
D)
one
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 37) A bulb has specifications of 1 kW and 250 V, the resistance of bulb is
A)
625\[\Omega \]
done
clear
B)
0.25\[\Omega \]
done
clear
C)
6.25\[\Omega \]
done
clear
D)
62.5\[\Omega \]
done
clear
E)
2.5\[\Omega \]
done
clear
View Answer play_arrow
question_answer 38) The internal energy of the working substance in any cyclic process
A)
increases
done
clear
B)
decreases
done
clear
C)
remain constant
done
clear
D)
becomes zero
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 39) Out of the following electromagnetic radiations which has the shortest wavelength?
A)
Radio waves
done
clear
B)
Infrared
done
clear
C)
Ultraviolet
done
clear
D)
Visible light
done
clear
E)
X-rays
done
clear
View Answer play_arrow
question_answer 40) The modulus of elasticity is dimensionally equivalent to
A)
strain
done
clear
B)
force
done
clear
C)
stress
done
clear
D)
coefficient of viscosity
done
clear
E)
Work
done
clear
View Answer play_arrow
question_answer 41) A tiger chases a deer 30 m ahead of it and gains 3 m in 5 s after the chase began. The distance gained by the tiger in 10 s is
A)
6 m
done
clear
B)
12 m
done
clear
C)
18 m
done
clear
D)
20 m
done
clear
E)
16 m
done
clear
View Answer play_arrow
question_answer 42) The speed of projection of a projectile is increased by 10% without changing the angle of projection. The percentage increase in the range will be
A)
10%
done
clear
B)
20%
done
clear
C)
15%
done
clear
D)
5%
done
clear
E)
25%
done
clear
View Answer play_arrow
question_answer 43) A sphere is suspended by a thread of length 1. The minimum horizontal velocity which has to be imparted to the sphere for it to reach the height of suspension is
A)
\[2\sqrt{gR}\]
done
clear
B)
\[\sqrt{2gl}\]
done
clear
C)
2gl
done
clear
D)
gl
done
clear
E)
\[\sqrt{gl/2}\]
done
clear
View Answer play_arrow
question_answer 44) A vehicle of mass 120 kg is moving with a uniform velocity of 108 km/h. The force required to step the vehicle in 10 s is
A)
90 N
done
clear
B)
180 N
done
clear
C)
360 N
done
clear
D)
720 N
done
clear
E)
810 N
done
clear
View Answer play_arrow
question_answer 45) The change in potential energy when a body of mass m is raised to a height nR from the centre of earth (R = radius of earth)
A)
\[mgR\frac{\left( n-1 \right)}{n}\]
done
clear
B)
\[nmgR\]
done
clear
C)
\[mgR\left( \frac{{{n}^{2}}}{{{n}^{2}}+1} \right)\]
done
clear
D)
\[mgR\left( \frac{n}{n+1} \right)\]
done
clear
E)
mgR
done
clear
View Answer play_arrow
question_answer 46) A particle of mass 1 kg is moving in SHM with an amplitude 0.02 m and a frequency of 60 Hz. The maximum force in newton acting on the particle is
A)
188\[\pi \]2
done
clear
B)
144\[\pi \]2
done
clear
C)
288 \[\pi \]2
done
clear
D)
12\[{{\pi }^{2}}\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 47) The equation of a spherical progressive wave is
A)
y = \[\alpha \]sin \[\omega \]t
done
clear
B)
y = \[\alpha \] sin (\[\omega \]t - kr)
done
clear
C)
y = \[\frac{a}{\sqrt{r}}\]sin(\[\omega \]t - kr)
done
clear
D)
y = \[\frac{a}{r}\] sin(\[\omega \]t - fr)
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 48) The moment of inertia of a rod (length I mass m) about an axis perpendicular to the length of the rod and passing through a J point equidistant from its mid-point and one end, is
A)
\[\frac{m{{l}^{2}}}{12}\]
done
clear
B)
\[\frac{7}{48}m{{l}^{2}}\]
done
clear
C)
\[\frac{13}{48}m{{l}^{2}}\]
done
clear
D)
\[\frac{19}{48}m{{l}^{2}}\]
done
clear
E)
\[\frac{14}{48}m{{l}^{2}}\]
done
clear
View Answer play_arrow
question_answer 49) At constant volume, temperature is increased, then
A)
collision on walls will be less
done
clear
B)
number of collisions per unit time will increase
done
clear
C)
collisions will be in straight lines
done
clear
D)
collisions will not change
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 50) A rain drop of radius 0.3 mm has a terminal velocity 1 m/s in the air. The viscosity of air is\[18\times {{10}^{-5}}P.\] The viscous force on it is
A)
\[101.73\times {{10}^{-4}}\]dyne
done
clear
B)
\[101.73\times {{10}^{-5}}\]dyne
done
clear
C)
\[16.95\times {{10}^{-5}}\]dyne
done
clear
D)
\[16.95\times {{10}^{-4}}\]dyne
done
clear
E)
\[1.6\times {{10}^{-4}}\]dyne
done
clear
View Answer play_arrow
question_answer 51) Magnetic field intensity due to a dipole varies as \[{{d}^{n}};\] \[d=\]distance of observation point from dipole, where n is equal to
A)
2
done
clear
B)
-2
done
clear
C)
3
done
clear
D)
-3
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 52) If the photon of energy 12.1 eV is incident on hydrogen gas, the gas will emit per second
A)
few lines in Balmer and Lymen series
done
clear
B)
all the lines in Balmer series
done
clear
C)
few lines in Balmer series
done
clear
D)
Balmer series only
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 53) A body is moving in circular motion of constant radius, then
A)
the net acceleration of the body may be towards the centre of the circle
done
clear
B)
the net acceleration of the body may not be towards the centre of the circle
done
clear
C)
the velocity of the body must change
done
clear
D)
All of the above
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 54) For a constant hydraulic stress on an object, the fractional change in the objects volume \[\left( \frac{\Delta V}{V} \right)\]and its bulk modulus are related as
A)
\[\frac{\Delta V}{V}\propto B\]
done
clear
B)
\[\frac{\Delta V}{V}\propto \frac{1}{B}\]
done
clear
C)
\[\frac{\Delta V}{V}\propto {{B}^{2}}\]
done
clear
D)
\[\frac{\Delta V}{V}\propto {{B}^{-2}}\]
done
clear
E)
\[\frac{\Delta V}{V}\propto {{B}^{3/2}}\]
done
clear
View Answer play_arrow
question_answer 55) A person cannot see the object beyond 100 cm. The power of a lens to correct his vision will be
A)
+2 D
done
clear
B)
-1 D
done
clear
C)
+5 D
done
clear
D)
0.5 D
done
clear
E)
+6 D
done
clear
View Answer play_arrow
question_answer 56) The moment of inertia of a body about a given axis is 1.2 kg\[{{m}^{2}}\]. Initially the body is at rest. In order to produce a rotational kinetic energy of 1500 J, an angular acceleration of 25 \[rad\text{/}{{s}^{2}}\]must be applied about that axis for a duration of
A)
4 s
done
clear
B)
2 s
done
clear
C)
8 s
done
clear
D)
10 s
done
clear
E)
3 s
done
clear
View Answer play_arrow
question_answer 57) The following equation represents induced transmutation\[_{4}B{{e}^{9}}{{+}_{2}}H{{e}^{4}}{{\xrightarrow[{}]{{}}}_{6}}{{C}^{12}}+X\]In this equation, X represents
A)
one negative \[\beta \text{-}\]particle
done
clear
B)
\[\alpha \]-particle
done
clear
C)
a positron
done
clear
D)
a neutron
done
clear
E)
\[\gamma -\]particle
done
clear
View Answer play_arrow
question_answer 58) Cyclotron is a device which is used to
A)
measure the charge
done
clear
B)
measure the voltage
done
clear
C)
accelerate protons
done
clear
D)
accelerate electrons
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 59) If boiling point of water is 95 F, what will be the reduction at Celsius scale?
A)
\[7{}^\circ C\]
done
clear
B)
\[65{}^\circ C\]
done
clear
C)
\[63{}^\circ C\]
done
clear
D)
\[35{}^\circ C\]
done
clear
E)
\[70{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 60) The refractive indices of violet and red light are 1.54 and 1.52 respectively. If the angle of prism is \[10{}^\circ ,\] the angular dispersion (in degree) is
A)
0.02
done
clear
B)
0.20
done
clear
C)
3.06
done
clear
D)
30.6
done
clear
E)
306
done
clear
View Answer play_arrow
question_answer 61) Which of the following reacts fastest with a mixture of anhydrous \[ZnC{{l}_{2}}\] and cone. \[HCl\]?
A)
Trimethyl carbinol
done
clear
B)
Ethanol
done
clear
C)
Propanol
done
clear
D)
Methanol
done
clear
E)
Iso-propanol
done
clear
View Answer play_arrow
question_answer 62) The reagent with which both acetaldehyde and acetophenone reacts easily are
A)
Fehlings solution
done
clear
B)
Schiffs reagent
done
clear
C)
Tollens reagent
done
clear
D)
sodium bisulphite
done
clear
E)
2, 4-dinitrophenylhydrazine
done
clear
View Answer play_arrow
question_answer 63) The main component of glass which gives heat resistance to laboratory glassware is
A)
\[PbO\]
done
clear
B)
\[MgO\]
done
clear
C)
\[{{B}_{2}}{{O}_{3}}\]
done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
E)
\[{{P}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 64) Thomas slag is referred to as
A)
calcium silicate
done
clear
B)
calcium phosphate
done
clear
C)
barium phosphate
done
clear
D)
strontium silicate
done
clear
E)
barium silicate
done
clear
View Answer play_arrow
question_answer 65) Each \[B-H-B\] bridge in \[{{B}_{2}}{{H}_{6}}\] is formed by the sharing of
A)
2 electrons
done
clear
B)
4 electrons
done
clear
C)
1 electron
done
clear
D)
3 electrons
done
clear
E)
8 electrons
done
clear
View Answer play_arrow
question_answer 66) The one electron species having ionization energy of 54.4 eV is
A)
\[H\]
done
clear
B)
\[H{{e}^{+}}\]
done
clear
C)
\[{{B}^{4+}}\]
done
clear
D)
\[L{{i}^{2+}}\]
done
clear
E)
\[B{{e}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 67) Density of a crystal remains unchanged as a result of
A)
ionic defect
done
clear
B)
Schottky defect
done
clear
C)
Frenkel defect
done
clear
D)
crystal defect
done
clear
E)
point defect
done
clear
View Answer play_arrow
question_answer 68) The mass of 11.2 L of ammonia gas at STP is
A)
8.5 g
done
clear
B)
85 g
done
clear
C)
17 g
done
clear
D)
1.7 g
done
clear
E)
4.25 g
done
clear
View Answer play_arrow
question_answer 69) Identify the correct statement from below, concerning the structure of\[C{{H}_{2}}=C=C{{H}_{2}}\].
A)
The molecule is planar
done
clear
B)
One of the three carbon atoms is in \[s{{p}^{3}}\]hybridized state
done
clear
C)
The molecule is non-planar with the two\[-C{{H}_{2}}\] groups being in planes perpendicular to each other
done
clear
D)
All the carbon atoms are sp-hybridized
done
clear
E)
The molecule is bent with the\[-C-C-C-\]angle being 120 degrees
done
clear
View Answer play_arrow
question_answer 70) The enthalpy of a monoatomic gas at T Kelvin is
A)
\[\frac{7}{2}RT\]
done
clear
B)
\[\frac{3}{2}RT\]
done
clear
C)
\[\frac{1}{2}RT\]
done
clear
D)
\[\frac{1}{2}m{{v}^{2}}\]
done
clear
E)
\[\frac{5}{2}RT\]
done
clear
View Answer play_arrow
question_answer 71) The dissociation constant of acetic acid \[{{K}_{a}}\]is\[1.74\times {{10}^{-5}}\] at 298 K. The pH of a solution of 0.1 M acetic acid is
A)
2.88
done
clear
B)
3.6
done
clear
C)
4.0
done
clear
D)
1.0
done
clear
E)
2.0
done
clear
View Answer play_arrow
question_answer 72) In the given reaction,\[2X(g)+Y(g)\rightleftharpoons 2Z(g)+80\,kcal.\] Which combination of pressure and temperature will give the highest yield of Z at equilibrium?
A)
1000 atm and \[200{}^\circ C\]
done
clear
B)
500 atm and \[500{}^\circ C\]
done
clear
C)
1000 atm and \[100{}^\circ C\]
done
clear
D)
500 atm and \[100{}^\circ C\]
done
clear
E)
1000 atm and \[500{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 73) \[E_{C{{u}^{\,2+}}/Cu}^{\text{o}}=0.34\,V,\]\[E_{Z{{n}^{\,2+}}/Zn}^{\text{o}}=-\,0.76\,V.\] A Daniell cell contains 0.1 M \[ZnS{{O}_{4}}\]solution and 0.01 M \[CuS{{O}_{4}}\]solution at its electrodes. EMF of the cell is
A)
1.10 V
done
clear
B)
1.04 V
done
clear
C)
1.16 V
done
clear
D)
1.07 V
done
clear
E)
1.00 V
done
clear
View Answer play_arrow
question_answer 74) A radioactive isotope has a half-life of 8 days. If today 125 mg is left over, what was its original weight 32 days earlier?
A)
6 g
done
clear
B)
5 g
done
clear
C)
4 g
done
clear
D)
2 g
done
clear
E)
1 g
done
clear
View Answer play_arrow
question_answer 75) On addition of 1 mL solution of 10% \[NaCl\]to 10 mL gold solution in the presence of 0.025 g of starch, the coagulation is prevented because starch has the following gold number
A)
25
done
clear
B)
0.025
done
clear
C)
0.25
done
clear
D)
2.5
done
clear
E)
0.0025
done
clear
View Answer play_arrow
question_answer 76) IUPAC name of acraldehyde is
A)
but-3-en-1-al
done
clear
B)
propenyl aldehyde
done
clear
C)
but-2-ene-1-al
done
clear
D)
propanal
done
clear
E)
prop-2-ene-1-al
done
clear
View Answer play_arrow
question_answer 77) A molecule of urea can show
A)
chain isomerism
done
clear
B)
position isomerism
done
clear
C)
geometrical isomerism
done
clear
D)
tautomerism
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 78) The presence of\[A{{g}^{+}}\]ion increases the solubility of alkenes due to the formation of
A)
\[d\pi \text{-}d\sigma \] bonding
done
clear
B)
\[p\sigma \text{-}p\pi \] bonding
done
clear
C)
\[p\pi \text{-}d\pi \] bonding
done
clear
D)
\[p\pi \text{-}p\pi \]bonding
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 79) Glycerine contains
A)
\[1{}^\circ \]carbon
done
clear
B)
\[2{}^\circ \]carbon
done
clear
C)
\[3{}^\circ \]carbon
done
clear
D)
both \[1{}^\circ \] and \[2{}^\circ \]carbon
done
clear
E)
both \[2{}^\circ \]and \[3{}^\circ \]carbon
done
clear
View Answer play_arrow
question_answer 80) \[\alpha \] and \[\beta \text{-}\]glucose differ in the orientation of -OH group around
A)
\[{{C}_{1}}\]
done
clear
B)
\[{{C}_{2}}\]
done
clear
C)
\[{{C}_{3}}\]
done
clear
D)
\[{{C}_{4}}\]
done
clear
E)
\[{{C}_{5}}\]
done
clear
View Answer play_arrow
question_answer 81) Which one of the following has the highest molar conductivity?
A)
Diamminedichloroplatinum (II)
done
clear
B)
Tetramminedichlorocobalt (III) chloride
done
clear
C)
Potassium hexacyanoferrate (II)
done
clear
D)
Hexaaquochromium (III) bromide
done
clear
E)
Pentacarbonyl iron (0).
done
clear
View Answer play_arrow
question_answer 82) The first law of thermodynamic is expressed as
A)
\[Q-W=\Delta E\]
done
clear
B)
\[\Delta E=Q-p\Delta V\]
done
clear
C)
\[Q=\Delta E-W\]
done
clear
D)
\[W=Q+\Delta E\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
question_answer 83) The Markownikoffs rule is the best applicable to the reaction between
A)
\[{{C}_{2}}{{H}_{4}}+HCl\]
done
clear
B)
\[{{C}_{3}}{{H}_{6}}+B{{r}_{2}}\]
done
clear
C)
\[{{C}_{3}}{{H}_{6}}+HBr\]
done
clear
D)
\[{{C}_{3}}{{H}_{8}}+C{{l}_{2}}\]
done
clear
E)
\[{{C}_{2}}{{H}_{4}}+{{I}_{2}}\]
done
clear
View Answer play_arrow
question_answer 84) Phenol can be distinguished from ethanol by the following reagents except
A)
sodium
done
clear
B)
\[NaOH\text{/}{{I}_{2}}\]
done
clear
C)
neutral \[FeC{{l}_{3}}\]
done
clear
D)
\[B{{r}_{2}}\text{/}{{H}_{2}}O\]
done
clear
E)
phthalic anhydride/cone. \[{{H}_{2}}S{{O}_{4}}\] and \[NaOH\]
done
clear
View Answer play_arrow
question_answer 85) The enol form of acetone after treatment with \[{{D}_{2}}O,\]gives
A)
\[{{H}_{3}}C-\underset{OD}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,=C{{H}_{2}}\]
done
clear
B)
\[{{H}_{3}}C-\underset{O}{\mathop{\underset{\mathbf{||}}{\mathop{C}}\,}}\,-C{{D}_{3}}\]
done
clear
C)
\[{{H}_{2}}C=\underset{OH}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,-C{{H}_{2}}D\]
done
clear
D)
\[{{H}_{2}}C=\underset{OH}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,-CH{{D}_{2}}\]
done
clear
E)
\[{{D}_{2}}C=\underset{OD}{\mathop{\underset{\mathbf{|}}{\mathop{C}}\,}}\,-C{{D}_{3}}\]
done
clear
View Answer play_arrow
question_answer 86) An alkene on reductive ozonolysis gives 2-molecules of \[C{{H}_{2}}{{(CHO)}_{2}}.\] The alkene is
A)
2, 4-hexadiene
done
clear
B)
1, 3-cyclohexadiene
done
clear
C)
1, 4-cyclohexadiene
done
clear
D)
1-methyl-1, 3-cyclopentadiene
done
clear
E)
1, 2-dimethylcyclopropene
done
clear
View Answer play_arrow
question_answer 87) Identify the compound\[Z\]in this reaction sequence. \[C{{H}_{3}}C{{H}_{2}}COOH\xrightarrow[\Delta ]{N{{H}_{3}}}X\xrightarrow{B{{r}_{2}}+KOH}\]\[Y\xrightarrow{HN{{O}_{2}}}Z\]
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
E)
\[C{{H}_{3}}N{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 88)
The following homogeneous gaseous reactions were experimentally found to be second order overall. I. \[2NO\xrightarrow{{}}{{N}_{2}}+{{O}_{2}}\] II. \[3{{O}_{2}}\xrightarrow{{}}2{{O}_{3}}\] III. \[{{N}_{2}}{{O}_{3}}\xrightarrow{{}}NO+N{{O}_{2}}\] IV. \[{{H}_{2}}+{{I}_{2}}\xrightarrow{{}}2HI\]
Which of these are most likely to be elementary reactions that occur in one step?
A)
III only
done
clear
B)
I and III
done
clear
C)
I and IV
done
clear
D)
III and IV
done
clear
E)
I, II and III
done
clear
View Answer play_arrow
question_answer 89) On a humid day in summer, the mole fraction of gaseous \[{{H}_{2}}O\](water vapour) in the air at \[25{}^\circ C\] can be as high as 0.0287. Assuming a total pressure of 0.977 arm, what is the partial pressure of dry air?
A)
94.9 atm
done
clear
B)
0.949 atm
done
clear
C)
949 atm
done
clear
D)
0.648 atm
done
clear
E)
1.248 atm
done
clear
View Answer play_arrow
question_answer 90) For which of the following sparingly soluble salt, the solubility (s) and solubility product (Kgp) are related by the expression\[s={{({{K}_{sp}}\text{/}4)}^{1/3}}\]?
A)
\[BaS{{O}_{4}}\]
done
clear
B)
\[C{{a}_{3}}{{(P{{O}_{4}})}_{2}}\]
done
clear
C)
\[H{{g}_{2}}C{{l}_{2}}\]
done
clear
D)
\[A{{g}_{3}}P{{O}_{4}}\]
done
clear
E)
\[CuS\]
done
clear
View Answer play_arrow
question_answer 91) At certain temperature, a 5.12% solution of cane sugar is isotonic with a 0.9% solution of an unknown solute. The molar mass of solute is
A)
60
done
clear
B)
46.17
done
clear
C)
120
done
clear
D)
90
done
clear
E)
92.34
done
clear
View Answer play_arrow
question_answer 92) Which of the following is true in respect of adsorption?
A)
\[\Delta G<0;\Delta \,S>0;\,\Delta \,H<0\]
done
clear
B)
\[\Delta G<0;\Delta \,S<0;\,\Delta \,H<0\]
done
clear
C)
\[\Delta G>0;\Delta \,S>0;\,\Delta \,H<0\]
done
clear
D)
\[\Delta G<0;\Delta \,S<0;\,\Delta \,H>0\]
done
clear
E)
\[\Delta G>0;\Delta \,S>0;\,\Delta \,H>0\]
done
clear
View Answer play_arrow
question_answer 93) The amine which will not liberate nitrogen on reaction with nitrous acid is
A)
trimethyl amine
done
clear
B)
ethyl amine
done
clear
C)
sec-butyl amine
done
clear
D)
\[t\text{-}\]butyl amine
done
clear
E)
iso-propyl amine
done
clear
View Answer play_arrow
question_answer 94) One mole of acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]on reaction with excess KI will liberate ?... mole(s) of \[{{I}_{2}}\].
A)
6
done
clear
B)
1
done
clear
C)
7
done
clear
D)
2
done
clear
E)
3
done
clear
View Answer play_arrow
question_answer 95) Which of the following exists as Zwitter ion?
A)
p-aminophenol
done
clear
B)
Salicylic acid
done
clear
C)
Sulphanilic acid
done
clear
D)
Ethanolamine
done
clear
E)
p-aminoacetophenone
done
clear
View Answer play_arrow
question_answer 96)
Consider the following halogen containing compounds I. \[CHC{{l}_{3}}\] II. \[CC{{l}_{4}}\] III. \[C{{H}_{2}}C{{l}_{2}}\] IV. \[C{{H}_{3}}Cl\] V.
The compounds with a net zero dipole moment are
A)
II and V only
done
clear
B)
III only
done
clear
C)
III and IV only
done
clear
D)
I and IV only
done
clear
E)
II only
done
clear
View Answer play_arrow
question_answer 97) Which of the following is bacteriostatic?
A)
Penicillin
done
clear
B)
Erythromycin
done
clear
C)
Amino glycodine
done
clear
D)
Ofloxacin
done
clear
E)
Bithional
done
clear
View Answer play_arrow
question_answer 98) Which one of the following set of quantum numbers is not possible for electron in the ground state of an atom with atomic number 19?
A)
\[n=2,\,l=0,\,m=0\]
done
clear
B)
\[n=2,\,l=1,\,m=0\]
done
clear
C)
\[n=3,\,l=1,\,m=-1\]
done
clear
D)
\[n=3,\,l=2,\,m=+\,2\]
done
clear
E)
\[n=4,\,l=0,\,m=0\]
done
clear
View Answer play_arrow
question_answer 99) The sequence that correctly describes the relative bond strength pertaining to oxygen molecule and its cation or anions is
A)
\[O_{2}^{2-}>O_{2}^{-}>{{O}_{2}}>O_{2}^{+}\]
done
clear
B)
\[{{O}_{2}}>O_{2}^{+}>O_{2}^{-}>O_{2}^{2-}\]
done
clear
C)
\[O_{2}^{+}>{{O}_{2}}>O_{2}^{2-}>O_{2}^{-}\]
done
clear
D)
\[O_{2}^{+}>{{O}_{2}}>O_{2}^{-}>O_{2}^{2-}\]
done
clear
E)
\[{{O}_{2}}>O_{2}^{-}>O_{2}^{2-}>O_{2}^{+}\]
done
clear
View Answer play_arrow
question_answer 100) The hybrid rocket propellant consists of
A)
acrylic rubber and liquid nitrogen tetraoxide
done
clear
B)
polyurethane and ammonium perchlorate
done
clear
C)
nitroglycerine and nitrocellulose
done
clear
D)
liquid hydrogen and liquid oxygen
done
clear
E)
hydrogen peroxide
done
clear
View Answer play_arrow
question_answer 101) Boric acid is used in carom boards for smooth gliding of pawns because
A)
\[{{H}_{3}}B{{O}_{3}}\] molecules are loosely chemically bonded and hence soft
done
clear
B)
its low density makes it fluffy
done
clear
C)
it can be powdered to a very small grain size
done
clear
D)
it is chemically inert with the plywood
done
clear
E)
H-bonding in \[{{H}_{3}}B{{O}_{3}}\] gives it a layered structure
done
clear
View Answer play_arrow
question_answer 102) The pair of \[[Co(S{{O}_{4}}){{(N{{H}_{3}})}_{5}}]\,Cl\]and\[[CoCl(N{{H}_{3}}){{(N{{H}_{3}})}_{5}}]\,S{{O}_{4}}\] constitutes
A)
optical isomers
done
clear
B)
linkage isomers
done
clear
C)
coordination isomers
done
clear
D)
hydrate isomers
done
clear
E)
ionisation isomers
done
clear
View Answer play_arrow
question_answer 103) Which one of the following pairs of elements is called chemical twins because of their very similar chemical properties?
A)
Mn and W
done
clear
B)
Mo and Tc
done
clear
C)
Fe and Re
done
clear
D)
Hf and Zr
done
clear
E)
Fe and Co
done
clear
View Answer play_arrow
question_answer 104) The IUPAC name of \[{{[Co{{(N{{H}_{3}})}_{5}}ONO]}^{2+}}\]ion is
A)
pentammine nitrito, cobalt (IV) ion
done
clear
B)
pentammine nitrito cobalt (III) ion
done
clear
C)
pentammine nitro cobalt (III) ion
done
clear
D)
pentammine nitro cobalt (IV) ion
done
clear
E)
None of the above
done
clear
View Answer play_arrow
question_answer 105) Hydration of different ions in aqueous solution is an example of
A)
ion-induced dipole interaction
done
clear
B)
dipole-dipole interaction
done
clear
C)
dipole-induced dipole interaction
done
clear
D)
attractive dispersion forces between atoms
done
clear
E)
ion-dipole interaction
done
clear
View Answer play_arrow
question_answer 106) The vapour pressure of two liquids X and Y are 80 and 60 Torr respectively. The total vapour pressure of the ideal solution obtained by mixing 3 moles of X and 2 moles of Y would be
A)
68 Torr
done
clear
B)
140 Torr
done
clear
C)
48 Torr
done
clear
D)
72 Torr
done
clear
E)
54 Torr
done
clear
View Answer play_arrow
question_answer 107) The elements present in the core of earth are collectively known as
A)
lithophiles
done
clear
B)
nucleophiles
done
clear
C)
chalcophiles
done
clear
D)
siderophiles
done
clear
E)
atmophiles
done
clear
View Answer play_arrow
question_answer 108) Concentrated sulphuric acid can be reduced by
A)
\[NaCl\]
done
clear
B)
\[NaF\]
done
clear
C)
\[NaOH\]
done
clear
D)
\[NaN{{O}_{3}}\]
done
clear
E)
\[NaBr\]
done
clear
View Answer play_arrow
question_answer 109) Consider the ions : \[{{K}^{+}},\,{{S}^{2-}},C{{l}^{-}}\] and \[C{{a}^{2+}}.\] The radii of these ionic species follow the order
A)
\[C{{a}^{2+}}>{{K}^{+}}>C{{l}^{-}}>{{S}^{2-}}\]
done
clear
B)
\[C{{l}^{-}}>{{S}^{2-}}>{{K}^{+}}>C{{a}^{2+}}\]
done
clear
C)
\[C{{a}^{2+}}>C{{l}^{-}}>{{K}^{+}}>{{S}^{2-}}\]
done
clear
D)
\[{{K}^{+}}>{{S}^{2-}}>C{{l}^{-}}>C{{a}^{2+}}\]
done
clear
E)
\[{{S}^{2-}}>C{{l}^{-}}>{{K}^{+}}>C{{a}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 110) A compound in which a metal ion\[{{M}^{x+}}(Z=25)\] has a spin only magnetic moment of \[\sqrt{24}\] BM. The number of unpaired electrons in the compound and the oxidation state of the metal ion are respectively
A)
4 and 2
done
clear
B)
5 and 3
done
clear
C)
3 and 2
done
clear
D)
4 and 3
done
clear
E)
3 and 1
done
clear
View Answer play_arrow
question_answer 111) To an aqueous solution containing anions, a few drops of acidified \[KMn{{O}_{4}}\] are added. Which one of the following anions, if present will not decolourise the \[KMn{{O}_{4}}\] solution?
A)
\[{{I}^{-}}\]
done
clear
B)
\[CO_{3}^{2-}\]
done
clear
C)
\[{{S}^{2-}}\]
done
clear
D)
\[NO_{2}^{-}\]
done
clear
E)
\[C{{l}^{-}}\]
done
clear
View Answer play_arrow
question_answer 112) The ion that is more effective for the coagulation of \[A{{s}_{2}}{{S}_{3}}\] sol is
A)
\[B{{a}^{2+}}\]
done
clear
B)
\[N{{a}^{+}}\]
done
clear
C)
\[PO_{4}^{3-}\]
done
clear
D)
\[SO_{4}^{2-}\]
done
clear
E)
\[A{{l}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 113) Lassaignes test for the detection of nitrogen fails in
A)
\[{{H}_{2}}N-CO-NHN{{H}_{2}}\cdot HCl\]
done
clear
B)
\[N{{H}_{2}}-N{{H}_{2}}\cdot HCl\]
done
clear
C)
\[N{{H}_{2}}-CO-N{{H}_{2}}\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}-NH-N{{H}_{2}}\cdot HCl\]
done
clear
E)
\[{{C}_{6}}{{H}_{5}}CON{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 114) The alkyl halide that undergoes \[{{S}_{N}}1\] reaction more readily is
A)
ethyl bromide
done
clear
B)
iso-propyl bromide
done
clear
C)
vinyl bromide
done
clear
D)
n-propyl bromide
done
clear
E)
t-butyl bromide
done
clear
View Answer play_arrow
question_answer 115)
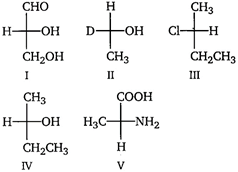
Select .R-isomers from the following
A)
I and III
done
clear
B)
II, IV and V
done
clear
C)
I, II and III
done
clear
D)
II and III
done
clear
E)
I, III and V
done
clear
View Answer play_arrow
question_answer 116) Which of the following is a better reducing I agent for the following reduction? \[RCOOH\xrightarrow{{}}RC{{H}_{2}}OH\]
A)
\[SnC{{l}_{2}}/HCl\]
done
clear
B)
\[NaB{{H}_{4}}/ether\]
done
clear
C)
\[{{H}_{2}}/Pd\]
done
clear
D)
\[{{N}_{2}}{{H}_{4}}/{{C}_{2}}{{H}_{5}}ONa\]
done
clear
E)
\[{{B}_{2}}{{H}_{6}}/{{H}_{3}}{{O}^{+}}\]
done
clear
View Answer play_arrow
question_answer 117) The ore that is concentrated by froth floatation process is
A)
zincite
done
clear
B)
cinnabar
done
clear
C)
bauxite
done
clear
D)
malachite
done
clear
E)
corundum
done
clear
View Answer play_arrow
question_answer 118) The oxide of an element whose electronic configuration is \[1{{s}^{2}},2{{s}^{2}},2{{p}^{6}},3{{s}^{1}}\]is
A)
neutral
done
clear
B)
amphoteric
done
clear
C)
basic
done
clear
D)
acidic
done
clear
E)
data insufficient
done
clear
View Answer play_arrow
question_answer 119) Among the following, the compound that contains ionic, covalent and coordinate linkage is
A)
\[N{{H}_{3}}\]
done
clear
B)
\[N{{H}_{4}}Cl\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[CaO\]
done
clear
E)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
View Answer play_arrow
question_answer 120) Which of the following is fully fluorinated polymer?
A)
PVC
done
clear
B)
Thiokol
done
clear
C)
Teflon
done
clear
D)
Neoprene
done
clear
E)
Saran
done
clear
View Answer play_arrow
question_answer 121) Plants restricted to certain localities are referred to as
A)
exotic
done
clear
B)
endemic
done
clear
C)
introduced
done
clear
D)
natural
done
clear
View Answer play_arrow
question_answer 122) Some bacteria are not easily killed by antibiotics or heat treatment because of their
A)
capsule
done
clear
B)
chitinous cell wall
done
clear
C)
mucopeptides in cell wall
done
clear
D)
mesosomes
done
clear
View Answer play_arrow
question_answer 123) Which type of ribosome is found in Nostoc cells?
A)
70 S
done
clear
B)
80 S
done
clear
C)
55 S
done
clear
D)
30 S
done
clear
View Answer play_arrow
question_answer 124) Haploid cells can be obtained from
A)
leaf
done
clear
B)
stem
done
clear
C)
seed
done
clear
D)
anther
done
clear
View Answer play_arrow
question_answer 125) Bromelain is an enzyme extracted from
A)
yeast
done
clear
B)
Ficus
done
clear
C)
pineapple
done
clear
D)
papaya
done
clear
View Answer play_arrow
question_answer 126) Genes not located within the nucleus are almost always found in
A)
cytosol
done
clear
B)
ribosome
done
clear
C)
cytoskeleton
done
clear
D)
cell membrane
done
clear
View Answer play_arrow
question_answer 127) Spine formation is found in
A)
Opuntia
done
clear
B)
Nepenthes
done
clear
C)
Asparagus
done
clear
D)
Drosera
done
clear
View Answer play_arrow
question_answer 128) Which one reproduces vegetatively?
A)
Allium cepa
done
clear
B)
Allium sativum
done
clear
C)
Pisum sativum
done
clear
D)
Zea mays
done
clear
View Answer play_arrow
question_answer 129) Gall flowers of figs are
A)
staminate
done
clear
B)
pistillate
done
clear
C)
neutar
done
clear
D)
naked
done
clear
View Answer play_arrow
question_answer 130) Quiescent centre possesses
A)
actively dividing cells
done
clear
B)
meristematic cells
done
clear
C)
passive cell
done
clear
D)
storage cells
done
clear
View Answer play_arrow
question_answer 131) Stomata are evenly distributed on upper and lower surfaces of leaf in
A)
potato
done
clear
B)
mulberry
done
clear
C)
Vallisneria
done
clear
D)
Eichhornia
done
clear
View Answer play_arrow
question_answer 132) Cork cells are dead because they do not possess
A)
cellulosic cell wall
done
clear
B)
permeable cell wall
done
clear
C)
protoplast
done
clear
D)
meristematic activities
done
clear
View Answer play_arrow
question_answer 133) Transfusion tissue is meant for translocation of
A)
organic solutes
done
clear
B)
sap
done
clear
C)
food materials
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 134) An alcohol- dipped cell kept in hypertonic sucrose solution will
A)
burst
done
clear
B)
plasmolysed
done
clear
C)
remain unchanged
done
clear
D)
become turgid
done
clear
View Answer play_arrow
question_answer 135) The plant ash is an indication of
A)
mineral salts absorbed by the plant
done
clear
B)
organic matter of the plant
done
clear
C)
both mineral salts and organic matter
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 136) The smallest known plant having the same pigments as grasses and trees, belongs to
A)
Chlorophyceae
done
clear
B)
Schizomycetes
done
clear
C)
Bryophyta
done
clear
D)
Angiosperms
done
clear
View Answer play_arrow
question_answer 137) Synthesis of ATP in mitochondria requires
A)
NADP
done
clear
B)
FMN
done
clear
C)
oxygen
done
clear
D)
pyruvic acid
done
clear
View Answer play_arrow
question_answer 138) Polygonum type 7- celled embryo sac closely resembles to
A)
Allium type
done
clear
B)
Drusa type
done
clear
C)
Adoxa type
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 139) Fruit formation is directly associated with stimulus of
A)
pollination
done
clear
B)
fertilization
done
clear
C)
endospore formation
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 140) Hormone related to phototropism is
A)
IAA
done
clear
B)
\[G{{A}_{3}}\]
done
clear
C)
kinetin
done
clear
D)
2, 4-D
done
clear
View Answer play_arrow
question_answer 141) In mangrove forest, there occur
A)
rich biodiversity
done
clear
B)
very severe interspecific competition
done
clear
C)
very severe intraspecific competition
done
clear
D)
no competition
done
clear
View Answer play_arrow
question_answer 142) Secondary growth is not seen in
A)
succulents
done
clear
B)
xerophytes
done
clear
C)
hydrophytes
done
clear
D)
mangroves
done
clear
View Answer play_arrow
question_answer 143) Important Value Index (IVI) is determined using quantitative data of
A)
relative density
done
clear
B)
relative frequency
done
clear
C)
relative dominance
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 144) Maximum radiant energy is trapped by
A)
algae
done
clear
B)
herbs
done
clear
C)
shrubs
done
clear
D)
trees
done
clear
View Answer play_arrow
question_answer 145) Which of the weed is most harmful?
A)
Azolla
done
clear
B)
Parthenium
done
clear
C)
Salvinia
done
clear
D)
Salicomia
done
clear
View Answer play_arrow
question_answer 146) Early blight disease is related to
A)
sugarcane
done
clear
B)
potato
done
clear
C)
onion
done
clear
D)
rose
done
clear
View Answer play_arrow
question_answer 147) Bioenergy is always
A)
ecofriendly
done
clear
B)
cheap
done
clear
C)
non-polluting
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 148) Axenic culture means
A)
cell culture
done
clear
B)
cell culture free from microorganisms
done
clear
C)
cell culture free from plants
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 149) Nif genes are fixing genes for
A)
nitrogen
done
clear
B)
oxygen
done
clear
C)
carbon
done
clear
D)
nitrate
done
clear
View Answer play_arrow
question_answer 150) A cell having genetically different DNA strands is called
A)
callus
done
clear
B)
cybrid
done
clear
C)
chimera
done
clear
D)
plasmid
done
clear
View Answer play_arrow
question_answer 151) Glissons capsule is related to
A)
intestine
done
clear
B)
liver
done
clear
C)
lung
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 152) Crypts of Lieberkuhn have
A)
a-cells
done
clear
B)
p-cells
done
clear
C)
paneth cells
done
clear
D)
Kupffers cells
done
clear
View Answer play_arrow
question_answer 153) Circulation is closed type in
A)
frog
done
clear
B)
human
done
clear
C)
monkey
done
clear
D)
earthworm
done
clear
View Answer play_arrow
question_answer 154) Frogs and toads belong to the order
A)
Caudata
done
clear
B)
Apoda
done
clear
C)
Anura
done
clear
D)
Gymnophiona
done
clear
View Answer play_arrow
question_answer 155) The Vibrissae of a rat is arranged in
A)
2 groups
done
clear
B)
3 groups
done
clear
C)
4 groups
done
clear
D)
5 groups
done
clear
View Answer play_arrow
question_answer 156) Kwashiorkor is a deficiency disease of
A)
protein
done
clear
B)
fat
done
clear
C)
carbohydrate
done
clear
D)
minerals
done
clear
View Answer play_arrow
question_answer 157) Which refers to biocatalysts?
A)
Erepsin, amylase, rennin
done
clear
B)
Rhodopsin, pepsin, steapsin
done
clear
C)
Myosin, oxytocin, adrenaline
done
clear
D)
Glucose, amino acids, fatty acids
done
clear
View Answer play_arrow
question_answer 158) Which of them possess non-elastic lungs which elastic air sacs connected to them?
A)
Birds
done
clear
B)
Reptiles
done
clear
C)
Mammals
done
clear
D)
Amphibians
done
clear
View Answer play_arrow
question_answer 159) The yellow color of urine is due to
A)
urea
done
clear
B)
bilirubin
done
clear
C)
uric acid
done
clear
D)
urochrome
done
clear
View Answer play_arrow
question_answer 160) Retinal cells involved in color vision are
A)
cones
done
clear
B)
cornea
done
clear
C)
neurons
done
clear
D)
neuroglial cells
done
clear
View Answer play_arrow
question_answer 161) Pituitary lies in the sella turcica of
A)
nasal
done
clear
B)
vomer
done
clear
C)
ethmoid
done
clear
D)
sphenoid
done
clear
View Answer play_arrow
question_answer 162) Glucagon characteristically increases all the following except
A)
ketogenesis in the liver
done
clear
B)
glycogenolysis in muscle
done
clear
C)
lipolysis in adipose tissue
done
clear
D)
urea synthesis in liver
done
clear
View Answer play_arrow
question_answer 163) Membrane attack complex (Mac) is formed by
A)
B-lymphocytes
done
clear
B)
macrophages
done
clear
C)
T-lymphocytes
done
clear
D)
complements
done
clear
View Answer play_arrow
question_answer 164) Which one is not related to hallucinogens?
A)
LSD
done
clear
B)
Marijuana
done
clear
C)
Psilocybin
done
clear
D)
Heroin
done
clear
View Answer play_arrow
question_answer 165) Chromatoid bodies in Entamoeba histolytica are found in
A)
cyst
done
clear
B)
minuta
done
clear
C)
metacyst
done
clear
D)
trophozoite
done
clear
View Answer play_arrow
question_answer 166) Animals which have well-marked digestive cavity are put under
A)
Metazoa
done
clear
B)
Bryozoa
done
clear
C)
Parazoa
done
clear
D)
Enterozoa
done
clear
View Answer play_arrow
question_answer 167) Blood of earthworm is red because its haemoglobin is
A)
reduced
done
clear
B)
oxidized
done
clear
C)
intracellular
done
clear
D)
intercellular
done
clear
View Answer play_arrow
question_answer 168) Larva of housefly lacks
A)
eyes
done
clear
B)
abdomen
done
clear
C)
fins
done
clear
D)
spiracles
done
clear
View Answer play_arrow
question_answer 169) Which of the following stain is obtained from the female scale insect Coccus cacti?
A)
Carmine
done
clear
B)
Orcein
done
clear
C)
Haematoxytin
done
clear
D)
Crystal violet
done
clear
View Answer play_arrow
question_answer 170) Creatinine is formed metabolically from
A)
arginine
done
clear
B)
histidine
done
clear
C)
tryptophan
done
clear
D)
phenylalanine
done
clear
View Answer play_arrow
question_answer 171) Example of a phosphoprotein is
A)
mucin
done
clear
B)
casein
done
clear
C)
ferritin
done
clear
D)
haemoglobin
done
clear
View Answer play_arrow
question_answer 172) Which of the following diseases is related to colour blindness?
A)
Night blindness
done
clear
B)
Haemophilia
done
clear
C)
Cataract
done
clear
D)
Cancer
done
clear
View Answer play_arrow
question_answer 173) The category of molecules produced by Milter-Urey experiment was
A)
organic polymers
done
clear
B)
inorganic polymers
done
clear
C)
organic monomers
done
clear
D)
inorganic monomers
done
clear
View Answer play_arrow
question_answer 174) Which of them are not homologous?
A)
Insect legs
done
clear
B)
Insect mouth parts
done
clear
C)
(c ) Vertebrate forelimbs
done
clear
D)
Birds and insect wings
done
clear
View Answer play_arrow
question_answer 175) An example of vestigial organ is
A)
hair of bear
done
clear
B)
ear of cow
done
clear
C)
tusk of elephant
done
clear
D)
nictitating membrane of man
done
clear
View Answer play_arrow
question_answer 176) The closest relative of modem man is considered to be
A)
monkey
done
clear
B)
chimpanzee
done
clear
C)
ape
done
clear
D)
gorilla
done
clear
View Answer play_arrow
question_answer 177) Why pea plants were more suitable than dogs for Mendels experiment?
A)
Dogs have many genetic traits
done
clear
B)
Pea plants can be self fertilized
done
clear
C)
No pedigree records of dogs
done
clear
D)
Pea plants favour cross fertilization
done
clear
View Answer play_arrow
question_answer 178) Super position image formation takes place in cockroach during
A)
dim light
done
clear
B)
diffused light
done
clear
C)
bright light
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 179) Scales are absent in
A)
Catia
done
clear
B)
Wallago
done
clear
C)
Cirrhinus
done
clear
D)
Scoliodon
done
clear
View Answer play_arrow
question_answer 180) The mammals evolved from the reptile in the
A)
Cretaceous
done
clear
B)
Triassic
done
clear
C)
Devonian
done
clear
D)
Carboniferous
done
clear
View Answer play_arrow
question_answer 181) The ICC Executive Board has declared that 14 - team will be eligible for the 2015 World Cup, to be held in Australia and New Zealand. What number has been decided for 2019 World Cup?
A)
10
done
clear
B)
12
done
clear
C)
14
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 182) In which of following States the Sick Newborn Care Units CSNCUs) are being set up in all district hospital with a view to minimizing the infant and child mortality rate?
A)
Gujarat
done
clear
B)
Punjab
done
clear
C)
Bihar
done
clear
D)
Haryana
done
clear
View Answer play_arrow
question_answer 183) Which famous temple in Kerala sparked widespread interest when a treasure trove amounting to $ 22 was unearthed during a stock-taking exercise?
A)
Sri Guruvayr Temple
done
clear
B)
Sri Padmanabhaswamy Temple
done
clear
C)
Sril Lakshman Temple
done
clear
D)
Puthukulangara Sree Bhadrakli Temple
done
clear
View Answer play_arrow
question_answer 184) Which of these is the capital of the newly constituted country South Sudan?
A)
Abei
done
clear
B)
Juba
done
clear
C)
Jongli
done
clear
D)
Bahr al Ghazal
done
clear
View Answer play_arrow
question_answer 185) The 17th SAARC Summit will be held in the Republic of Maldives from Nov 10-11, 2011. What will be the theme of this summit?
A)
Investing for Future
done
clear
B)
Building Cross-Border Confidence
done
clear
C)
Respect for Elders and Love for Children
done
clear
D)
Building Bridges for Peace, Friendship and Security
done
clear
View Answer play_arrow
question_answer 186) The first UID number was issued on Sept 29, 2010, in a village of Nandurbar district of Maharashtra. Since then, one crore Indians have been issued their Unique Identification Numbers. The government targets of issuing 60 crore Aadhaar numbers by which year?
A)
2013
done
clear
B)
2014
done
clear
C)
2015
done
clear
D)
2016
done
clear
View Answer play_arrow
question_answer 187) Which of the following biosphere reserves (NBR) is going to be showcased for the award instituted by the UNESCO to mark the 40th year of its theme Man and Biosphere?
A)
Sunderban
done
clear
B)
Nilgiris
done
clear
C)
Manas
done
clear
D)
Nokrek
done
clear
View Answer play_arrow
question_answer 188) The World Chess Championship Title match in 2012 will be held in Chennai in April-May. The title clash will be between defending champion Viswanathan Anand of India and Boris Gelfand, who belongs to
A)
Spain
done
clear
B)
Belarus
done
clear
C)
Israel
done
clear
D)
Russia
done
clear
View Answer play_arrow
question_answer 189) Basant Nayak, who passed away recently, was a film-maker of which among the following film industry?
A)
Tamil
done
clear
B)
Telugu
done
clear
C)
Oriya
done
clear
D)
Bhojpuri
done
clear
View Answer play_arrow
question_answer 190) Which day is observed as the international day against drug abuse and illicit trafficking throughout the world?
A)
June 23
done
clear
B)
June 24
done
clear
C)
June 25
done
clear
D)
June 26
done
clear
View Answer play_arrow
question_answer 191) Which of the following rivers is also called Ganga of the South?
A)
Krishna
done
clear
B)
Godavari
done
clear
C)
Cauveri
done
clear
D)
Tapti
done
clear
View Answer play_arrow
question_answer 192) Maximum crude petroleum in India is produced in
A)
Asom
done
clear
B)
Gujarat
done
clear
C)
Off- shor Bombay High
done
clear
D)
Coastal Tamil Nadu
done
clear
View Answer play_arrow
question_answer 193) Lines which join the places receiving equal amount of rainfall are called
A)
Contours
done
clear
B)
Isohyets
done
clear
C)
Isotherms
done
clear
D)
Isobars
done
clear
View Answer play_arrow
question_answer 194) Which among the following rivers makes an estuary?
A)
Cauveri
done
clear
B)
Krishna
done
clear
C)
Narmada
done
clear
D)
Ganga
done
clear
View Answer play_arrow
question_answer 195) Which State of India has the largest area under forests?
A)
Arunachal Pradesh
done
clear
B)
Madhya Pradesh
done
clear
C)
Maharashtra
done
clear
D)
Nagaland
done
clear
View Answer play_arrow
question_answer 196) The highest peak of India is
A)
Everest
done
clear
B)
K2
done
clear
C)
Pokalde
done
clear
D)
Nanda Devi
done
clear
View Answer play_arrow
question_answer 197) Oil reserves are found mainly in following rocks
A)
Metamorphic
done
clear
B)
Sedimentary
done
clear
C)
Igneous
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 198) Which State in India is famous for sane wood?
A)
Maharashtra
done
clear
B)
Karnataka
done
clear
C)
Kerala
done
clear
D)
Andhra Pradesh
done
clear
View Answer play_arrow
question_answer 199) Indian Standard Time is calculated on the basis of that longitude, which is near
A)
Allahabad
done
clear
B)
Lucknow
done
clear
C)
Kanpur
done
clear
D)
Varanasi
done
clear
View Answer play_arrow
question_answer 200) The shape of the earth is oblong spheroid due to
A)
attraction of moon
done
clear
B)
internal structure
done
clear
C)
revolution around the sun
done
clear
D)
rotation about its axis
done
clear
View Answer play_arrow
question_answer 201) During which geological age did dinosaurs roam on the earth?
A)
Jurrasic
done
clear
B)
Tommotian
done
clear
C)
Cambrian
done
clear
D)
Precambrian
done
clear
View Answer play_arrow
question_answer 202) The Southernmost point of mainland India is
A)
Chennai
done
clear
B)
Kanyakumari
done
clear
C)
Thiruvananthpuram
done
clear
D)
Mangalore
done
clear
View Answer play_arrow
question_answer 203) Which of the following scales is used to measure earthquake intensity?
A)
Mohrs scale
done
clear
B)
Richter scale
done
clear
C)
Beaufort scale
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 204) Teen- Bigha Corridor links
A)
Bangladesh and India
done
clear
B)
India and Nepal
done
clear
C)
Bangladesh and Nepal
done
clear
D)
India and Pakistan
done
clear
View Answer play_arrow
question_answer 205) A solar eclipse occurs when
A)
the earth comes in between the sun and the moon
done
clear
B)
the sun comes in between the moon and the earth
done
clear
C)
the moon, the sun and the earth are in a straight line
done
clear
D)
the moon comes in between the sun and the earth
done
clear
View Answer play_arrow
question_answer 206) The Panama Canal connects
A)
the Atlantic ocean and the mediterranean sea
done
clear
B)
the Atlantic ocean and the Indian ocean
done
clear
C)
the Pacific and the Atlantic oceans
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 207) On which river is the highest waterfall in India located?
A)
Krishna
done
clear
B)
Godavari
done
clear
C)
Sharavati
done
clear
D)
Tapti
done
clear
View Answer play_arrow
question_answer 208) The country with highest population density in the world is
A)
England
done
clear
B)
Nauru
done
clear
C)
Japan
done
clear
D)
India
done
clear
View Answer play_arrow
question_answer 209) The Chipko Movement was about
A)
forest conservation
done
clear
B)
wildlife preservation
done
clear
C)
scientific agriculture
done
clear
D)
deforestation
done
clear
View Answer play_arrow
question_answer 210) Which is the longest National Highway in India?
A)
NH 1
done
clear
B)
NH 24
done
clear
C)
NH 7
done
clear
D)
NH 6
done
clear
View Answer play_arrow
question_answer 211) The city known as the Pittsburg of India is
A)
Jamshedpur
done
clear
B)
Tatanagar
done
clear
C)
Hardwar
done
clear
D)
Bhilai
done
clear
View Answer play_arrow
question_answer 212) Which of the following is the best type of coal?
A)
Anthracite
done
clear
B)
Lignite
done
clear
C)
Peat
done
clear
D)
Limonite
done
clear
View Answer play_arrow
question_answer 213) Indias largest lake Chilka is situated in
A)
Odisha
done
clear
B)
Maharashtra
done
clear
C)
West Bengal
done
clear
D)
Karnataka
done
clear
View Answer play_arrow
question_answer 214) A storm is indicated if atmospheric pressure
A)
falls suddenly
done
clear
B)
rises gradually
done
clear
C)
rises suddenly
done
clear
D)
falls gradually
done
clear
View Answer play_arrow
question_answer 215) Who is credited with the design of Taj Mahal?
A)
Ustad lsa
done
clear
B)
Mir Ali
done
clear
C)
Syed Hussain
done
clear
D)
Abdus Samad
done
clear
View Answer play_arrow
question_answer 216) The Qutub Minar at Delhi was built to commemorate honour
A)
Qutubuddin Aibak
done
clear
B)
Iltutmish
done
clear
C)
Qutubuddin Bakhriyar Kaki
done
clear
D)
Alauddin Khalji
done
clear
View Answer play_arrow
question_answer 217) Tripitakas are the sacred texts of which religion?
A)
Jainism
done
clear
B)
Buddhism
done
clear
C)
Pashupatas
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 218) Which leader started the now hugely popular Ganapati Festival in Maharashtra?
A)
Dadabhai Nauroji
done
clear
B)
MG Ranade
done
clear
C)
Bal Gangadhar Tilak
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 219) Bahadur Shah Zafar, the last Mughal, was exiled to
A)
Maldives
done
clear
B)
Kala Pani
done
clear
C)
Rangoon
done
clear
D)
London
done
clear
View Answer play_arrow
question_answer 220) The Mughal Emperor who again imposed Jaziya on Hindus was
A)
Aurangzeb
done
clear
B)
Akbar
done
clear
C)
Shah Jahan
done
clear
D)
Humayun
done
clear
View Answer play_arrow
question_answer 221) The Bengal Partition was announced in
A)
1911
done
clear
B)
1901
done
clear
C)
1905
done
clear
D)
1916
done
clear
View Answer play_arrow
question_answer 222) Who was called the Nightingale of India?
A)
Pandita Ramabai
done
clear
B)
Kalpana Dutt
done
clear
C)
Sucheta Kriplani
done
clear
D)
Sarojini Naidu
done
clear
View Answer play_arrow
question_answer 223) Where did in India Mahatma Gandhi first try his techniques of Satyagraha?
A)
Poona
done
clear
B)
Champaran
done
clear
C)
Delhi
done
clear
D)
Sabarmati
done
clear
View Answer play_arrow
question_answer 224) Lilavati an ancient work on Mathematics, was written by
A)
Aryabhatta
done
clear
B)
Varahmihira
done
clear
C)
Bhaskaracharya
done
clear
D)
Banabhatta
done
clear
View Answer play_arrow
question_answer 225) Which of the following was a famous physician?
A)
Charvak
done
clear
B)
Kamban
done
clear
C)
Charak
done
clear
D)
Bhaskar
done
clear
View Answer play_arrow
question_answer 226) Which Gupta ruler has been depicted on his coins as playing veena?
A)
Skandagupta
done
clear
B)
Chandragupta
done
clear
C)
Samudragupta
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 227) In which year was the battle of Plassy fought?
A)
1761
done
clear
B)
1757
done
clear
C)
1857
done
clear
D)
1843
done
clear
View Answer play_arrow
question_answer 228) Alberuni has given the detailed account of his visit to India in
A)
Kitab-ul-Hind
done
clear
B)
Tarikh-i-Yamini
done
clear
C)
Futuh-us-Alamgiri
done
clear
D)
Hamzanamah
done
clear
View Answer play_arrow
question_answer 229) Who was the British Prime Minister at the time of independence of India?
A)
McDonald
done
clear
B)
Atlee
done
clear
C)
Churchill
done
clear
D)
Cripps
done
clear
View Answer play_arrow
question_answer 230) The ancient name of Bengal was
A)
Gaud
done
clear
B)
Sopara
done
clear
C)
Kamarupa
done
clear
D)
Panchala
done
clear
View Answer play_arrow
question_answer 231) Name the founder of the Widow Remarriage Association in the 19th century.
A)
Ishwar Chandra Vidyasagar
done
clear
B)
Vishnu Shastri Pandit
done
clear
C)
Raja Ram Mohan Roy
done
clear
D)
Gopal Hari Deshmukh
done
clear
View Answer play_arrow
question_answer 232) Which Mughal Emperor got constructed the Peacock throne?
A)
Jahangir
done
clear
B)
Aurangzeb
done
clear
C)
Shahjahan
done
clear
D)
Akbar
done
clear
View Answer play_arrow
question_answer 233) Who was the only Indian Governor-General of India?
A)
Jawahar Lal Nehru
done
clear
B)
Narendra Kriplani
done
clear
C)
Rajendra Prasad
done
clear
D)
C Rajagopalachari
done
clear
View Answer play_arrow
question_answer 234) In which language was the Sangam literature composed?
A)
Sanskrit
done
clear
B)
Aramaic
done
clear
C)
Prakrit
done
clear
D)
Tamil
done
clear
View Answer play_arrow
question_answer 235) Which of the following objects has been discovered at all the Harappan sites?
A)
Seals
done
clear
B)
Ploughshare
done
clear
C)
Gold ornaments
done
clear
D)
Spindles
done
clear
View Answer play_arrow
question_answer 236) The revenue system of Mughal Empire was revamped by
A)
Raja Birbal
done
clear
B)
Raja Man Singh
done
clear
C)
Todarmal
done
clear
D)
Bairam Khan
done
clear
View Answer play_arrow
question_answer 237) At which session of the Congress it split due to the differences between the Extremisa and the Moderates?
A)
1903
done
clear
B)
1904
done
clear
C)
1906
done
clear
D)
1907
done
clear
View Answer play_arrow
question_answer 238) Who among the following was called The Grand Old Man of India?
A)
Mahatma Gandhi
done
clear
B)
Dadabhai Nauroji
done
clear
C)
AO Hume
done
clear
D)
Badruddin Tyabji
done
clear
View Answer play_arrow
question_answer 239) Which Mughal ruler got executed Teg Bahadur, the 9th Sikh Guru?
A)
Jahangir
done
clear
B)
Aurangzeb
done
clear
C)
Bahadur Shah I
done
clear
D)
Shahjahan
done
clear
View Answer play_arrow
question_answer 240) Vrithviraj Raso was written by
A)
Bilhan
done
clear
B)
AmirKhusro
done
clear
C)
Chand Bardai
done
clear
D)
Kalhana
done
clear
View Answer play_arrow
question_answer 241) Gandhiji withdrew the Non-Cooperation Movement due to
A)
agreement with Lord Irwin
done
clear
B)
Jalianwala Bagh violence
done
clear
C)
Chauri Chaura incident
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 242) Mahatma Gandhi was the President of die Congress in which session?
A)
Bombay, 1942
done
clear
B)
Belgaum, 1924
done
clear
C)
Calcutta, 1920
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 243) Which of the following bodies is not a constitutional body?
A)
Planning Commission
done
clear
B)
Election Commission
done
clear
C)
Finance Commission
done
clear
D)
Union Public Service Commission
done
clear
View Answer play_arrow
question_answer 244) The Zero hour is set aside for
A)
asking questions
done
clear
B)
rest break between two meetings of the House
done
clear
C)
discussing budget proposals
done
clear
D)
miscellaneous business
done
clear
View Answer play_arrow
question_answer 245) The Pro-tern Speaker
A)
swears-in members and hold charge till a regular Speaker is elected
done
clear
B)
officiates as Speaker when the Speaker is unlikely to be elected
done
clear
C)
conducts the proceedings of Houses in absence of the Speaker and the Deputy Speaker
done
clear
D)
checks if the election certificates of the members are in order
done
clear
View Answer play_arrow
question_answer 246) Which among the following is referred to as the First Citizen of India?
A)
The Prime Minister
done
clear
B)
The Chief Justice of India
done
clear
C)
The Lok Sabha Speaker
done
clear
D)
The President
done
clear
View Answer play_arrow
question_answer 247) The ninth schedule of Indian Constitution deals with
A)
anti- defection legislation
done
clear
B)
Panchayati Raj
done
clear
C)
land reforms
done
clear
D)
distribution of powers between the Union and States
done
clear
View Answer play_arrow
question_answer 248) Which of the following UTs sends most MPs to the Lok Sabha?
A)
Chhattisgarh
done
clear
B)
Delhi
done
clear
C)
Pondicherry
done
clear
D)
Andaman
done
clear
View Answer play_arrow
question_answer 249) Indian Constitution heavily borrowed from
A)
the erstwhile USSR Constitution
done
clear
B)
the British Constitution
done
clear
C)
the Govt of India Act, 1935
done
clear
D)
the US Constitution
done
clear
View Answer play_arrow
question_answer 250) Who officiates as the President when offices of both the President and the Vice-President are vacant?
A)
The Lok Sabha Speaker
done
clear
B)
The Rajya Sabha Chairman
done
clear
C)
The Chief Justice
done
clear
D)
The Home Minster
done
clear
View Answer play_arrow
question_answer 251) Who was the first woman Governor of a State?
A)
Indira Gandhi
done
clear
B)
Sarojini Naidu
done
clear
C)
Vijaya Lakshmi Pandit
done
clear
D)
Lakshmi Sehgal
done
clear
View Answer play_arrow
question_answer 252) The Constitution states that India shall be
A)
a Union of States
done
clear
B)
Unitary State
done
clear
C)
a Federation
done
clear
D)
a Confederation
done
clear
View Answer play_arrow
question_answer 253) The number of Fundamental Duties mentioned in the Constitution is
A)
12
done
clear
B)
10
done
clear
C)
11
done
clear
D)
13
done
clear
View Answer play_arrow
question_answer 254) The maximum permissible time-gap between two consecutive sessions of Parliament is
A)
3 months
done
clear
B)
2 months
done
clear
C)
6 months
done
clear
D)
4 months
done
clear
View Answer play_arrow
question_answer 255) Right to education to all children of the age-group of 6 to 14 years is
A)
a Directive Principle of State Policy
done
clear
B)
a Fundamental Right
done
clear
C)
a Statutory Right
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 256) A joint sitting of the two Houses of Parliament is summoned by the
A)
Prime Minister
done
clear
B)
President
done
clear
C)
Members of Lok Sabha
done
clear
D)
Members of Rajya Sabha
done
clear
View Answer play_arrow
question_answer 257) Who among the following is called the First Law Officer of India?
A)
Attorney-General
done
clear
B)
Law Minister
done
clear
C)
Chief Justice
done
clear
D)
Law Secretary
done
clear
View Answer play_arrow
question_answer 258) The President can impose central rule on a State under
A)
Article 356
done
clear
B)
Article 256
done
clear
C)
Article 370
done
clear
D)
Article 377
done
clear
View Answer play_arrow
question_answer 259) How many members can the President nominate to the Rajya Sabha?
A)
12 members
done
clear
B)
18 members
done
clear
C)
6 members
done
clear
D)
15 members
done
clear
View Answer play_arrow
question_answer 260) World Bank is also known as
A)
International Bank for Research and Development
done
clear
B)
International Bank for Rehabilitation and Development
done
clear
C)
International Bank for Refinance and Development
done
clear
D)
International Bank for Reconstruction and Development
done
clear
View Answer play_arrow
question_answer 261) The headquarters of the Asian Development Bank is at
A)
Manila
done
clear
B)
Singapore
done
clear
C)
Delhi
done
clear
D)
Bangkok
done
clear
View Answer play_arrow
question_answer 262) CAPART is engaged primarily in
A)
e-governance
done
clear
B)
share market
done
clear
C)
rural development
done
clear
D)
pollution control
done
clear
View Answer play_arrow
question_answer 263) The difference between the GNP and the NNP is equal to the
A)
consumer expenditure on durable goods
done
clear
B)
indirect tax revenue
done
clear
C)
direct tax revenue
done
clear
D)
capital depreciation
done
clear
View Answer play_arrow
question_answer 264) Bank rate is the rate at which the RBI provides loans to
A)
Public Sector Undertakings
done
clear
B)
Private Corporate Sector
done
clear
C)
Scheduled Commercial Banks
done
clear
D)
Non-Banking Financial institutions
done
clear
View Answer play_arrow
question_answer 265) Which sector of Indias economy employs the maximum number of people?
A)
Primary sector
done
clear
B)
Secondary sector
done
clear
C)
Tertiary sector
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 266) What does AGMARK signify?
A)
A quality guarantee stamp for agricultural produce
done
clear
B)
Regulated agricultural produce markets
done
clear
C)
Farmers cooperatives
done
clear
D)
A cooperative for egg production
done
clear
View Answer play_arrow
question_answer 267) The term Green Revolution indicates higher production through
A)
enhanced agricultural productivity
done
clear
B)
planting more trees
done
clear
C)
creation of grasslands
done
clear
D)
creation of garden in urban areas
done
clear
View Answer play_arrow
question_answer 268) Wall Street is
A)
the financial centre of USA
done
clear
B)
the newspaper centre of Britain
done
clear
C)
the great wall of China
done
clear
D)
a renowned street in France
done
clear
View Answer play_arrow
question_answer 269) GDP is defined as the value of all
A)
final value of goods and saw produced in an economy in a year
done
clear
B)
goods and services produced in, economy in a year
done
clear
C)
final goods produced in an economy in year
done
clear
D)
goods produced in an economy in a year
done
clear
View Answer play_arrow
question_answer 270) ATM stands for
A)
Automatic Tally Machine
done
clear
B)
Automotive Teller Machine
done
clear
C)
Automatic Teller Machine
done
clear
D)
Automated Tally Mechanism
done
clear
View Answer play_arrow
question_answer 271) Which States of India benefited most from Green Revolution?
A)
Punjab, Haryana and Western Uttar Pradesh
done
clear
B)
Rajasthan, Gujarat and Maharashtra
done
clear
C)
Bihar, Uttar Pradesh and Asom
done
clear
D)
Tamil Nadu, Andhra Pradesh and Karnataka
done
clear
View Answer play_arrow
question_answer 272) Who is the brand ambassador of Thums Up?
A)
Salman Khan
done
clear
B)
Amir Khan
done
clear
C)
Akshay Kumar
done
clear
D)
Shahrukh Khan
done
clear
View Answer play_arrow
question_answer 273) When was the General Agreement on Tariffs and Trade (GATT) absorbed into the World Trade Organization (WTO)?
A)
1991
done
clear
B)
2000
done
clear
C)
1995
done
clear
D)
2005
done
clear
View Answer play_arrow
question_answer 274) The First Five Year Plan adopted
A)
Harrod Domar model
done
clear
B)
Mahalanobis model
done
clear
C)
Lewis Fei model
done
clear
D)
Stalin model
done
clear
View Answer play_arrow
question_answer 275) The Mid-day Meal Scheme was launched by the Union Ministry of
A)
Home Affairs
done
clear
B)
Social Welfare
done
clear
C)
HRD
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 276) Public Distribution System was launched to
A)
prevent hoarding and black-marketing
done
clear
B)
provide food security to the poor
done
clear
C)
prevent overcharging by traders
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 277) Which year is known as the Year of Great Divide with respect to population growth in India?
A)
1947
done
clear
B)
1921
done
clear
C)
1911
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 278) Which type of unemployment can occur even in a situation of full employment?
A)
Structural unemployment
done
clear
B)
Functional unemployment
done
clear
C)
Cyclical unemployment
done
clear
D)
Disguised unemployment
done
clear
View Answer play_arrow
question_answer 279) The number of companies covered under the BSE Sensex is
A)
20
done
clear
B)
25
done
clear
C)
30
done
clear
D)
50
done
clear
View Answer play_arrow
question_answer 280) Nuclear fission is caused by the impact of
A)
proton
done
clear
B)
electron
done
clear
C)
neutron
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 281) Which of the following has the least penetrating power?
A)
Alpha particles
done
clear
B)
Beta panicles
done
clear
C)
Gamma rays
done
clear
D)
All have the same penetrating power
done
clear
View Answer play_arrow
question_answer 282) Which of the following are the discoveries of Einstein?
A)
Radioactivity and photoelectric effect
done
clear
B)
Radioactivity and theory of relativity
done
clear
C)
Theory of relativity and photoelectric effect
done
clear
D)
X-rays and photoelectric effect
done
clear
View Answer play_arrow
question_answer 283) Cryogenic engines find applications in
A)
rocket technology
done
clear
B)
frost-free refrigerators
done
clear
C)
submarine propulsion
done
clear
D)
researches in superconductivity
done
clear
View Answer play_arrow
question_answer 284) Optic fibres are mainly use for which of the following?
A)
Communication
done
clear
B)
Weaving
done
clear
C)
Musical instruments
done
clear
D)
Food industry
done
clear
View Answer play_arrow
question_answer 285) Which of the following is used as a coolant in nuclear reactors?
A)
Heavy water
done
clear
B)
Cadmium
done
clear
C)
Liquid sodium
done
clear
D)
Graphite
done
clear
View Answer play_arrow
question_answer 286) Which of the following is the lightest metal?
A)
Mercury
done
clear
B)
Silver
done
clear
C)
Lithium
done
clear
D)
Lead
done
clear
View Answer play_arrow
question_answer 287) An alloy used in making heating elements for electric heating devices is
A)
solder
done
clear
B)
alloy steel
done
clear
C)
nichrome
done
clear
D)
German silver
done
clear
View Answer play_arrow
question_answer 288) Carbon, diamond and graphite are together called
A)
isomers
done
clear
B)
allotropes
done
clear
C)
isomorphs
done
clear
D)
isotopes
done
clear
View Answer play_arrow
question_answer 289) Which of the following is used in beauty parlours for hair setting?
A)
Chlorine
done
clear
B)
Sulphur
done
clear
C)
Phosphorus
done
clear
D)
Silicon
done
clear
View Answer play_arrow
question_answer 290) Balloons are filled with
A)
helium
done
clear
B)
oxygen
done
clear
C)
nitrogen
done
clear
D)
argon
done
clear
View Answer play_arrow
question_answer 291) Which of the following compounds are responsible for acid rain? 1. Nitrogen dioxide 2. Silicon dioxide 3. Sulphur dioxide 4. Carbon monoxide Select the correct answer from the codes given below
A)
1 and 2
done
clear
B)
1 and 3
done
clear
C)
2 and 4
done
clear
D)
3 and 4
done
clear
View Answer play_arrow
question_answer 292) Washing soda is the common name for
A)
calcium carbonate
done
clear
B)
calcium bicarbonate
done
clear
C)
sodium carbonate
done
clear
D)
sodium bicarbonate
done
clear
View Answer play_arrow
question_answer 293) A person who lives exclusively on milk, egg and bread is likey to become a victim of
A)
rickets
done
clear
B)
scurvy
done
clear
C)
xerophthalmia
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 294) During sleep a mans blood pressure
A)
increases
done
clear
B)
decreases
done
clear
C)
remains constant
done
clear
D)
fluctuates
done
clear
View Answer play_arrow
question_answer 295) Ready source of energy available for athletes is
A)
carbohydrates
done
clear
B)
fats
done
clear
C)
proteins
done
clear
D)
vitamins
done
clear
View Answer play_arrow
question_answer 296) Alzheimers disease in human beings is characterized by the degeneration of
A)
kideny cells
done
clear
B)
nerve cells
done
clear
C)
brain cells
done
clear
D)
liver cells
done
clear
View Answer play_arrow
question_answer 297) The pH of human blood is between
A)
6.5-7
done
clear
B)
7.5-8
done
clear
C)
8-9
done
clear
D)
4.5-5
done
clear
View Answer play_arrow
question_answer 298) Flower colors are due to
A)
chlorophyll
done
clear
B)
melanin
done
clear
C)
phytochromes
done
clear
D)
anthocyanin
done
clear
View Answer play_arrow
question_answer 299) Which vitamin is provided by sunlight to die body?
A)
Vitamin-A
done
clear
B)
Vitamin-B
done
clear
C)
Vitamin-C
done
clear
D)
Vitamin-D
done
clear
View Answer play_arrow
question_answer 300) Human body is
A)
heart
done
clear
B)
liver
done
clear
C)
kidney
done
clear
D)
brain
done
clear
View Answer play_arrow