question_answer 1) Hygens wave theory of light cannot explain
A)
photoelectric effect
done
clear
B)
polarisation
done
clear
C)
interference
done
clear
D)
diffraction
done
clear
View Answer play_arrow
question_answer 2) A truck is moving eastwards with a velocity of 15 m/s. In 10 s, the velocity changes to 15 m/s northwards. Its average acceleration during this time will be
A)
\[3\sqrt{2}\]m/s northwards
done
clear
B)
\[\sqrt{2}\]m/s northwards
done
clear
C)
\[3\text{/}\sqrt{2}\]m/s northwards
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 3) A body sliding on a smooth inclined plane requires 4 s to reach the bottom starting from rest at the top. Then the time taken to travel one eight the distance starting rest at the top, will be
A)
16 s
done
clear
B)
4 s
done
clear
C)
\[\sqrt{2}\,s\]
done
clear
D)
0.5 s
done
clear
View Answer play_arrow
question_answer 4) Energy of simple harmonic motion depends on
A)
\[\frac{1}{{{\omega }^{2}}}\]
done
clear
B)
\[\omega \]
done
clear
C)
\[\frac{1}{{{a}^{2}}}\]
done
clear
D)
\[{{a}^{2}}\]
done
clear
View Answer play_arrow
question_answer 5) If the potential of a capacitor having capacity \[6\mu F\]is increased from 10 V to 20 V then increase in its energy is
A)
\[12\times {{10}^{-6}}J\]
done
clear
B)
\[9\times {{10}^{-4}}J\]
done
clear
C)
\[4.5\times {{10}^{-6}}J\]
done
clear
D)
\[2.25\times {{10}^{-6}}J\]
done
clear
View Answer play_arrow
question_answer 6) A bullet of mass 0.1 kg is fired with a speed of 100 m/s. The mass of gun being 50 kg. Then the velocity of recoil become
A)
0.05 m/s
done
clear
B)
0.5 m/s
done
clear
C)
0.1 m/s
done
clear
D)
0.2 m/s
done
clear
View Answer play_arrow
question_answer 7) The electrical conductivity of earths atmosphere increases with altitudes of
A)
reflection of atmosphere
done
clear
B)
accelerator protons
done
clear
C)
measure the voltage
done
clear
D)
measure the charge
done
clear
View Answer play_arrow
question_answer 8) In an AC circuit, the potential difference across an inductance and resistance connected in series are 16 V and 20 V respectively. Total potential difference across the circuit will be
A)
10.0V
done
clear
B)
20.6V
done
clear
C)
25.6V
done
clear
D)
30.6V
done
clear
View Answer play_arrow
question_answer 9) If a wire of resistance R is stretched to triple its length, then the new resistance is
A)
\[9R\]
done
clear
B)
\[3R\]
done
clear
C)
\[R/2\]
done
clear
D)
\[R\]
done
clear
View Answer play_arrow
question_answer 10) Atomic number of nucleus is Z, while its mass number is M, what will be number of neutrons in nucleus?
A)
M + Z
done
clear
B)
M - Z
done
clear
C)
M
done
clear
D)
Z
done
clear
View Answer play_arrow
question_answer 11) The nuclear energy produced in nuclear reactor is used to run
A)
AC motor
done
clear
B)
induction generator
done
clear
C)
electric generator
done
clear
D)
DC motor
done
clear
View Answer play_arrow
question_answer 12) If the phase difference between two points separated by 0.8 in wave of frequency 120 Hz, is 0.5\[\pi \]then the velocity of the wave is will be
A)
768 Hz
done
clear
B)
695 Hz
done
clear
C)
710 Hz
done
clear
D)
384 Hz
done
clear
View Answer play_arrow
question_answer 13) A 12 pF capacitor is connected to a 50 V. The electrostatic energy is stored in the capacitor will be
A)
\[2.5\times {{10}^{-3}}J\]
done
clear
B)
\[3.5\times {{10}^{-12}}J\]
done
clear
C)
\[0.5\times {{10}^{-2}}J\]
done
clear
D)
\[1.5\times {{10}^{-8}}J\]
done
clear
View Answer play_arrow
question_answer 14) Cyclotron is a device which used to
A)
measuring the charge
done
clear
B)
measure of the voltage
done
clear
C)
acceleration protons
done
clear
D)
acceleration of electrons
done
clear
View Answer play_arrow
question_answer 15) The dimensions of potential difference are
A)
\[[M{{L}^{2}}{{T}^{2}}{{Q}^{-1}}]\]
done
clear
B)
\[[M{{L}^{2}}{{T}^{2}}{{Q}^{2}}]\]
done
clear
C)
\[[L{{T}^{2}}Q]\]
done
clear
D)
\[[{{M}^{2}}L{{T}^{2}}{{Q}^{-1}}]\]
done
clear
View Answer play_arrow
question_answer 16) Light takes \[{{t}_{1}}\]sec to travel a distance x cm in vacuum and the same light takes \[{{t}_{2}}\]sec to travel 10 x cm in the medium, critical angle for the corresponding medium is
A)
\[{{\sin }^{-1}}\left( \frac{10{{t}_{1}}}{{{t}_{2}}} \right)\]
done
clear
B)
\[{{\sin }^{-1}}\left( \frac{{{t}_{1}}}{10\,{{t}_{2}}} \right)\]
done
clear
C)
\[{{\sin }^{-1}}\left( \frac{{{t}_{2}}}{10\,{{t}_{1}}} \right)\]
done
clear
D)
\[{{\sin }^{-1}}\left( \frac{10\,{{t}_{2}}}{{{t}_{1}}} \right)\]
done
clear
View Answer play_arrow
question_answer 17) If the critical angle for total internal reflection from a medium to vacuum is \[30{}^\circ ,\] then the velocity of light in the medium will be
A)
\[3\times {{10}^{8}}m\text{/}s\]
done
clear
B)
\[2\times {{10}^{8}}m\text{/}s\]
done
clear
C)
\[1.5\times {{10}^{8}}m\text{/}s\]
done
clear
D)
\[6\times {{10}^{8}}m\text{/}s\]
done
clear
View Answer play_arrow
question_answer 18) Which one of the following discovered cyclotron?
A)
Maxwell
done
clear
B)
Lawrence
done
clear
C)
Levels
done
clear
D)
Bohr
done
clear
View Answer play_arrow
question_answer 19) A gas initially at \[18{}^\circ C\]is compressed adiabatically to one eight of its original volume. Then the temperature of the gas after compression is
A)
\[395.4{}^\circ C\]
done
clear
B)
\[144{}^\circ C\]
done
clear
C)
\[887.4{}^\circ C\]
done
clear
D)
\[18{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 20) A stone is dropped into a lake from a tower of 500 m height. The sound of splash will be heard after
A)
11.5 s
done
clear
B)
21 s
done
clear
C)
3.75 s
done
clear
D)
10 s
done
clear
View Answer play_arrow
question_answer 21) An aeroplane having a wing space of 35 m files due to north with the speed of 90 m/s given B is \[4\times {{10}^{-5}}T.\] The potential difference between the tips of the wings will be
A)
0.013 V
done
clear
B)
1.26 V
done
clear
C)
12.6 V
done
clear
D)
0.126 V
done
clear
View Answer play_arrow
question_answer 22) A radioactive element has half-life of 3.6 days. In what time will it be left \[1/{{32}^{\text{nd}}}\]undecayed?
A)
4 days
done
clear
B)
12 days
done
clear
C)
18 days
done
clear
D)
24 days
done
clear
View Answer play_arrow
question_answer 23) The wavelength associated with an electron accelerated through a potential difference of 100 V is of the order of
A)
\[1.2\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[10.5\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[100\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 24) The thermodynamic coordinates of a jar A filled with a gas are p, V and T and another j ar B filled with another gas 2p, V/4 and 2T, where the symbols have their usual meanings. The ratio of the number of molecules of jar A to those of jar B is
A)
4 : 1
done
clear
B)
2 : 2
done
clear
C)
1 : 2
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 25) When the kinetic energy of a body executing SHM is I/3 of the potential energy. The displacement of the body is x per cent of the amplitude, where x is
A)
33
done
clear
B)
87
done
clear
C)
67
done
clear
D)
50
done
clear
View Answer play_arrow
question_answer 26) In the case of horse is pulling a cart, force that causes the horse to move forward is the force that
A)
the horse exerts on the ground
done
clear
B)
the horse exerts on the cart
done
clear
C)
the ground exerts on the horse
done
clear
D)
the cart exerts on the horse
done
clear
View Answer play_arrow
question_answer 27) If the pressure of a gas contained in a vessel is increased by 0.4%. When heated through \[1{}^\circ C,\] the initial temperature had been
A)
2500 K
done
clear
B)
250 K
done
clear
C)
\[250{}^\circ C\]
done
clear
D)
\[25{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 28) The capacitance of a spherical conductor with radius 1 m is
A)
\[9\times {{10}^{-9}}F\]
done
clear
B)
\[1\,\mu F\]
done
clear
C)
\[2.5\times {{10}^{-10}}F\]
done
clear
D)
\[1\times {{10}^{-6}}F\]
done
clear
View Answer play_arrow
question_answer 29) Which of the following have higher specific charge?
A)
Positron
done
clear
B)
Proton
done
clear
C)
He
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 30) For an encloses maintained at 1000 K, the maximum radiation occurs at wavelength \[{{\lambda }_{m}}\]. If the temperature is raised to 2000 K, the peak will shift to
A)
\[\frac{{{\lambda }_{m}}}{2}\]
done
clear
B)
\[2{{\lambda }_{m}}\]
done
clear
C)
\[{{2}^{4}}{{\lambda }_{m}}\]
done
clear
D)
\[{{2}^{-4}}{{\lambda }_{m}}\]
done
clear
View Answer play_arrow
question_answer 31) A coil of area 5\[c{{m}^{2}}\] and of 20 turns is placed in uniform magnetic field of \[{{10}^{3}}\]T. The normal to the plane of the coil makes an angle of \[60{}^\circ \] with the magnetic field. The flux in Maxwell through the coil is
A)
\[5\times {{10}^{5}}\]
done
clear
B)
\[5\times {{10}^{4}}\]
done
clear
C)
\[2\times {{10}^{4}}\]
done
clear
D)
\[5\times {{10}^{3}}\]
done
clear
View Answer play_arrow
question_answer 32) An electron jumps from the 4th orbits to the 2nd orbit of hydrogen atom. Given the Rydbergs constant\[R={{10}^{5}}c{{m}^{-1}},\]the frequency in Hz of the emitted radiation will be
A)
\[\frac{3}{6}\times {{10}^{5}}\]
done
clear
B)
\[\frac{16}{3}\times {{10}^{15}}\]
done
clear
C)
\[\frac{9}{16}\times {{10}^{15}}\]
done
clear
D)
\[\frac{3}{4}\times {{10}^{15}}\]
done
clear
View Answer play_arrow
question_answer 33) The Keplers second law states that the straight line joining the planet to the sun sweeps out equal areas in equal times. The statement is equivalent to saying that
A)
total acceleration is zero
done
clear
B)
transverse acceleration is zero
done
clear
C)
longitudinal acceleration is zero
done
clear
D)
radial acceleration is zero
done
clear
View Answer play_arrow
question_answer 34) A diatomic gas is heated at constant pressure. What fraction of the heat energy is used to increases the internal energy?
A)
\[\frac{3}{5}\]
done
clear
B)
\[\frac{3}{7}\]
done
clear
C)
\[\frac{5}{7}\]
done
clear
D)
\[\frac{5}{9}\]
done
clear
View Answer play_arrow
question_answer 35) In interference pattern the energy is
A)
created at the maximum
done
clear
B)
destroyed at the minimum
done
clear
C)
conserved but redistributed
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 36) A lens behaves as a converging lens in air and diverging lens in water. The refractive index of the material of the lens is
A)
equal to that of water
done
clear
B)
less than that of water
done
clear
C)
greater than that of water
done
clear
D)
Nothing can be predicted
done
clear
View Answer play_arrow
question_answer 37) The electron emitted in beta radiation originates from
A)
inner orbits of atoms
done
clear
B)
free electron existing in nucleus
done
clear
C)
decay of neutran in the nucleus
done
clear
D)
photon escaping from the nucleus
done
clear
View Answer play_arrow
question_answer 38) If elements with principal quantum number n > 4 were not allowed in nature than the number of possible elements would be
A)
32
done
clear
B)
60
done
clear
C)
10
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 39) The square of resultant of two equal forces is three times their product. Angle between the forces is
A)
\[\pi \]
done
clear
B)
\[\pi /2\]
done
clear
C)
\[\pi /4\]
done
clear
D)
\[\pi /3\]
done
clear
View Answer play_arrow
question_answer 40) Which of the following is different from others?
A)
Wavelength
done
clear
B)
Velocity
done
clear
C)
Frequency
done
clear
D)
Amplitude
done
clear
View Answer play_arrow
question_answer 41) A capacitor of capacitance C has charge Q and stored energy is W. If the charge is increased to 2Q, the stored energy will be
A)
\[\frac{W}{4}\]
done
clear
B)
\[\frac{W}{2}\]
done
clear
C)
\[2\,W\]
done
clear
D)
\[4\,W\]
done
clear
View Answer play_arrow
question_answer 42) A cylindrical conductor is placed near another positively charged conductor the net charge acquired by the cylindrical conductor will be
A)
positive only
done
clear
B)
negative only
done
clear
C)
zero
done
clear
D)
Either positive or negative
done
clear
View Answer play_arrow
question_answer 43) A point moves such that its displacement as function of time is given by \[{{x}^{2}}={{t}^{2}}+1.\] Its acceleration at time\[t\]is
A)
\[\frac{1}{{{x}^{4}}}\]
done
clear
B)
\[-\frac{t}{{{x}^{2}}}\]
done
clear
C)
\[\frac{1}{x}-\frac{{{t}^{2}}}{{{x}^{3}}}\]
done
clear
D)
\[\frac{1}{x}-\frac{t}{{{x}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 44) If the speed of light wave \[\frac{2}{3}\] of its present value, the energy released in a given atomic explosion will be decreased by a fraction
A)
\[\frac{2}{3}\]
done
clear
B)
\[\frac{4}{9}\]
done
clear
C)
\[\frac{5}{9}\]
done
clear
D)
\[\frac{2}{9}\]
done
clear
View Answer play_arrow
question_answer 45) Alcohol is more volatile than water, because
A)
its boiling point is lower than water
done
clear
B)
it is an organic liquid
done
clear
C)
its freezing point is lower than water
done
clear
D)
its vapour pressure is 2.5 times greater than water
done
clear
View Answer play_arrow
question_answer 46) A body has same temperature as that of the surrounding, then
A)
it radiates same heat as it absorbs
done
clear
B)
it absorbs more, radiates less heat
done
clear
C)
If radiates more, absorbs less heat
done
clear
D)
it never radiates heat
done
clear
View Answer play_arrow
question_answer 47) In a transistor configuration\[\beta \]parameter is
A)
\[{{I}_{b}}/{{I}_{c}}\]
done
clear
B)
\[{{I}_{c}}/{{I}_{b}}\]
done
clear
C)
\[{{I}_{c}}/{{I}_{c}}\]
done
clear
D)
\[{{I}_{b}}/{{I}_{b}}\]
done
clear
View Answer play_arrow
question_answer 48) The ratio of forward biased to reverse biased resistance for p-n junction diode is
A)
\[{{10}^{-1}}:1\]
done
clear
B)
\[{{10}^{-2}}:1\]
done
clear
C)
\[{{10}^{-3}}:1\]
done
clear
D)
\[{{10}^{-4}}:1\]
done
clear
View Answer play_arrow
question_answer 49) A water film is made between two 10 cm long straight wire and at a distance of 0.5 cm. If distance between the wire is increased by 1 mm. Then work done will be
A)
\[9.22\times {{10}^{-6}}J\]
done
clear
B)
\[1.44\times {{10}^{-5}}J\]
done
clear
C)
\[2.88\times {{10}^{-5}}J\]
done
clear
D)
\[5.76\times {{10}^{-5}}J\]
done
clear
View Answer play_arrow
question_answer 50) A capacitor of 20\[\mu F\] capacity charged upto 500 V is connected in parallel with another capacitor of 10 aF which is charged upto 200 V. Their common potential is
A)
500V
done
clear
B)
400V
done
clear
C)
300V
done
clear
D)
200V
done
clear
View Answer play_arrow
question_answer 51) What will happen when a 40 W, 220 V lamp and 100 W, 220 V lamp are connected in series across 40 V supply?
A)
100 W lamp will fuse
done
clear
B)
40 W lamp will fuse
done
clear
C)
Both lamps will fuse
done
clear
D)
Neither lamp will fuse
done
clear
View Answer play_arrow
question_answer 52) The displacement of a particle of mass 3 g executing simple harmonic motion is given by Y = 3 sin\[(0.2\,t)\]in SI units. The kinetic energy of the particle at a points which is at a distance equal to \[\frac{1}{3}\] of its amplitude from its mean position is
A)
\[12\times {{10}^{3}}J\]
done
clear
B)
\[25\times {{10}^{-3}}J\]
done
clear
C)
\[0.48\times {{10}^{-3}}J\]
done
clear
D)
\[0.24\times {{10}^{-3}}J\]
done
clear
View Answer play_arrow
question_answer 53) The radius of gyration of a rod of length L and mass At about an axis perpendicular to its length and passing through a point at a distance L/3 from one if its ends is
A)
\[\frac{\sqrt{7}}{6}L\]
done
clear
B)
\[\frac{{{L}^{2}}}{9}\]
done
clear
C)
\[\frac{L}{3}\]
done
clear
D)
\[\frac{\sqrt{5}}{3}L\]
done
clear
View Answer play_arrow
question_answer 54) The apparent weight of a person inside a lift is \[{{w}_{1}}\]when lift moves up with a certain acceleration and is \[{{w}_{2}}\]when lift moves down with same acceleration. The weight of the person when lift moves up with constant speed is
A)
\[\frac{{{w}_{1}}+{{w}_{2}}}{2}\]
done
clear
B)
\[\frac{{{w}_{1}}-{{w}_{2}}}{2}\]
done
clear
C)
\[2\,{{w}_{1}}\]
done
clear
D)
\[2\,{{w}_{2}}\]
done
clear
View Answer play_arrow
question_answer 55) The magnitude of magnetic induction for a current carrying toroid of uniform cross-section is
A)
uniform over the whole cross-section
done
clear
B)
maximum onthe outer edge
done
clear
C)
maximum on the outer edge.
done
clear
D)
maximum at the centre of cross-section
done
clear
View Answer play_arrow
question_answer 56) Isogonic lines are those for which
A)
declination is the same at all places on the line
done
clear
B)
angle of dip is the same at all place on the line
done
clear
C)
the value of horizontal component of earths magnetic field is the same
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 57) Each atom of an iron bar (5 cm x 1 cm x 1 cm) has a magnetic moment \[1.8\times {{10}^{-23}}A{{m}^{-2}}\]that the density of iron is \[7.18\times {{10}^{3}}\,kg/{{m}^{3}},\], atomic weight is 56 and Avogadro number is \[6.02\times {{10}^{23}}.\] The magnetic .moment of bar is the state of magnetic saturation will be
A)
4,75 A/m
done
clear
B)
5.74 A/m
done
clear
C)
7.54 A/m
done
clear
D)
75.4 A/m
done
clear
View Answer play_arrow
question_answer 58) A current of 1A is flowing on the sides of an equilateral triangle of sides \[4.5\times {{10}^{-2}}m.\] The magnetic field at the centroid of the triangle is
A)
\[2\times {{10}^{-5}}T\]
done
clear
B)
\[4\times {{10}^{-5}}T\]
done
clear
C)
\[8\times {{10}^{-5}}T\]
done
clear
D)
\[1.2\times {{10}^{-5}}T\]
done
clear
View Answer play_arrow
question_answer 59) If the equation of transverse wave is\[y=5\,\sin 2\pi \left[ \frac{t}{0.04}-\frac{x}{40} \right],\] where distance is in cm and time in second and then the wavelength of the wave is
A)
60 cm
done
clear
B)
40 cm
done
clear
C)
35cm
done
clear
D)
25cm
done
clear
View Answer play_arrow
question_answer 60) A 700 pF capacitor is charged by a 50 V battery. The electrostatic energy stored by it is
A)
\[17.0\times {{10}^{-8}}J\]
done
clear
B)
\[13.5\times {{10}^{-8}}J\]
done
clear
C)
\[9.5\times {{10}^{-9}}J\]
done
clear
D)
\[8.7\times {{10}^{-7}}J\]
done
clear
View Answer play_arrow
question_answer 61) When benzene sulphonic acid and p-nitrophenol are treated with \[NaHC{{O}_{3}}\] the gases released respectively are
A)
\[S{{O}_{2}},\,C{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}},\,C{{O}_{2}}\]
done
clear
C)
\[S{{O}_{2}},\,NO\]
done
clear
D)
\[S{{O}_{2}},\,N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 62) Amongst the following antihistamines, which are antacids? I. Ranitidine II, Cimetidine III. Terfenadine IV. Brompheniramine
A)
I and II
done
clear
B)
III only
done
clear
C)
I and III
done
clear
D)
III and IV
done
clear
View Answer play_arrow
question_answer 63) Which of the following polymers cannot have vinylic monomer units?
A)
Acrilan
done
clear
B)
Polystyrene
done
clear
C)
Nylon
done
clear
D)
Teflon
done
clear
View Answer play_arrow
question_answer 64)
Optical rotations of some compounds along with their structures are given below. Which of them have D- configuration?
A)
I, II, III
done
clear
B)
II, III
done
clear
C)
l, ll
done
clear
D)
lll
done
clear
View Answer play_arrow
question_answer 65) Due to the presence of ambidentate ligands coordination compounds show isomerism. Palladium complexes of the type \[[Pd{{({{C}_{6}}{{H}_{5}})}_{2}}{{(SCN)}_{2}}]\]and\[[Pd({{C}_{6}}{{H}_{5}})\,{{(NCS)}_{2}}]\]are
A)
linkage isomerism
done
clear
B)
coordination isomerism
done
clear
C)
ionisation isomerism
done
clear
D)
geometrical isomerism
done
clear
View Answer play_arrow
question_answer 66) Amongst the following, the most stable complex is
A)
\[{{[Fe{{({{H}_{2}}O)}_{6}}]}^{3+}}\]
done
clear
B)
\[{{[Fe{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
C)
\[{{[Fe{{({{C}_{2}}{{O}_{4}})}_{3}}]}^{3-}}\]
done
clear
D)
\[{{[FeC{{l}_{6}}]}^{3-}}\]
done
clear
View Answer play_arrow
question_answer 67) In a 0.010M solution of oxalic acid, \[{{H}_{2}}{{C}_{2}}{{O}_{4}},\] \[{{K}_{1}}=5.9\times {{10}^{-2}},\] \[{{K}_{{{a}_{2}}}}=6.4\times {{10}^{-5}},\]the species present in the lowest concentration is
A)
\[{{H}_{2}}{{C}_{2}}{{O}_{4}}\]
done
clear
B)
\[{{H}_{3}}{{O}^{+}}\]
done
clear
C)
\[HC{{r}_{2}}O_{4}^{-}\]
done
clear
D)
\[CrO_{4}^{2-}\]
done
clear
View Answer play_arrow
question_answer 68) \[{{K}_{a}}\] for HF is \[3.5\times {{10}^{-7}}\]calculate \[{{K}_{b}}\] for the fluoride ion.
A)
\[3.5\times {{10}^{-4}}\]
done
clear
B)
\[1.0\times {{10}^{-7}}\]
done
clear
C)
\[2.85\times {{10}^{-11}}\]
done
clear
D)
\[1.0\times {{10}^{-14}}\]
done
clear
View Answer play_arrow
question_answer 69) A face centred cubic lattice is made up of two types of atoms A and B, in which A occupies the comer positions and B occupies the face centres. If atom along an axis joining the diagonally opposite comers on a face are removed, the empirical formula of the remaining solid would be
A)
\[{{A}_{2}}{{B}_{5}}\]
done
clear
B)
\[{{A}_{3}}{{B}_{7}}\]
done
clear
C)
\[{{A}_{3}}{{B}_{10}}\]
done
clear
D)
\[{{A}_{7}}{{B}_{24}}\]
done
clear
View Answer play_arrow
question_answer 70) The normal oxidation potential of zinc referred to the standard hydrogen electrode is 0.76V and that of copper is 0.34V at \[25{}^\circ C,\]when excess of zinc is added to solution of copper sulphate, zinc displaces copper till equilibrium is reached. What is the ratio of concentration to\[Z{{n}^{2+}}\]to\[C{{u}^{2+}}\]ions at equilibrium?
A)
\[1.69\times {{10}^{37}}:1\]
done
clear
B)
\[1.243\times {{10}^{22}}:1\]
done
clear
C)
\[1:1679\times {{10}^{37}}\]
done
clear
D)
\[1:1.24243\times {{10}^{22}}\]
done
clear
View Answer play_arrow
question_answer 71)
For the following metals A, B, C, D react with each other (N.R. = no reaction). \[A+{{B}^{+}}\to N.\,R\] \[;\,\,B+{{C}^{+}}\to N.\,R\] \[B+{{D}^{+}}\to {{B}^{+}}+D\] \[;\,\,C+{{D}^{+}}\to {{C}^{+}}+D\] \[A+C\to {{C}^{+}}+A\] \[;\,\,D+{{A}^{+}}\to {{D}^{+}}+A\]
What is the order of the metals in increasing reducing strength?
A)
\[B<D<A<C\]
done
clear
B)
\[C<B<D<A\]
done
clear
C)
\[A<D<B<C\]
done
clear
D)
\[D<C<A<B\]
done
clear
View Answer play_arrow
question_answer 72) Suppose that the electron spin quantum number could have three possible values which of the following atomic numbers would not correspond to a noble gas?
A)
3
done
clear
B)
15
done
clear
C)
27
done
clear
D)
42
done
clear
View Answer play_arrow
question_answer 73) Calculate the number of revolutions per second made around the nucleus by an electron in the third Bohrs orbit of a hydrogen atom.
A)
\[2.43\times {{10}^{14}}\]
done
clear
B)
\[2.43\times {{10}^{15}}\]
done
clear
C)
\[2.43\times {{10}^{16}}\]
done
clear
D)
\[2.43\times {{10}^{17}}\]
done
clear
View Answer play_arrow
question_answer 74) Which of the following regarding decay of \[A{{l}^{25}}\]is not true?
A)
It may decay by position emission
done
clear
B)
It may decay by a neutron emission
done
clear
C)
It may decay by electron capture
done
clear
D)
The product nucleus is Mg
done
clear
View Answer play_arrow
question_answer 75) 2.0 g of benzoic acid dissolved in 25.0g of benzene shows a depression in freezing point equal to 1.62 K. Molal depression constant, \[{{K}_{f}}\] of benzene is 4.9 K kg \[mo{{l}^{-1}}\], what is the percentage association of the acid if it forms dimer in the solution?
A)
78.2%
done
clear
B)
82.6%
done
clear
C)
89.7%
done
clear
D)
99.2%
done
clear
View Answer play_arrow
question_answer 76) A gas X at 1 atm is bubbled through a solution containing a mixture of 1\[M\,{{Y}^{-}}\] and 1 \[M\,{{Z}^{-}}\]ions at\[25{}^\circ C\]. If the reduction potential of \[Z>Y>X,\] then
A)
\[Y\] will oxidise \[Z\] but not \[Z\]
done
clear
B)
\[Y\] will oxidise both \[X\] and \[Z\]
done
clear
C)
\[Y\] will oxidise \[Z\] but
done
clear
D)
\[Y\] will reduce both \[X\] and \[Z\]
done
clear
View Answer play_arrow
question_answer 77) Conductivity of 0.00241 M acetic acid solution is \[7.896\times {{10}^{-5}}Sc{{m}^{-1}},\]what would be its dissociation constant?
A)
\[1.85\times {{10}^{-5}}\]
done
clear
B)
\[32.76\times {{10}^{-5}}\]
done
clear
C)
\[1.85\times {{10}^{-7}}\]
done
clear
D)
\[32.76\times {{10}^{-7}}\]
done
clear
View Answer play_arrow
question_answer 78) The rate of a gaseous reaction is given by the expression k[A][B]. If the volume of the reaction vessel is suddenly reduced to \[\frac{1}{4}\]th of the initial volume, the reaction rate relating to original rate will be
A)
\[\frac{1}{10}\]
done
clear
B)
\[\frac{1}{8}\]
done
clear
C)
\[8\]
done
clear
D)
\[16\]
done
clear
View Answer play_arrow
question_answer 79) Consider the reaction :\[C{{l}_{2}}(aq)+{{H}_{2}}S(aq)\xrightarrow{{}}\]\[S(s)+2{{H}^{+}}(aq)+2C{{l}^{-}}(aq)\] The rate equation for this reaction is , rate \[=k[C{{l}_{2}}]\,[{{H}_{2}}S]\] Which of these mechanisms is consistent with this rate equation? A. \[C{{l}_{2}}+{{H}_{2}}S\to {{H}^{+}}+C{{l}^{-}}+C{{l}^{+}}+H{{S}^{-}}\](slow) \[C{{l}^{+}}+H{{S}^{-}}\to {{H}^{+}}+C{{l}^{-}}+S\](fast) B. \[{{H}_{2}}S\rightleftharpoons {{H}^{+}}+H{{S}^{-}}\] (fast equilibrium); \[C{{l}_{2}}+H{{S}^{-}}\to 2C{{l}^{-}}+{{H}^{+}}+S\](slow)
A)
Neither A nor B
done
clear
B)
A only
done
clear
C)
B only
done
clear
D)
Both A and B
done
clear
View Answer play_arrow
question_answer 80)
Which of the following phenomenon occurs when a chalk stick is dipped in the ink? I. absorption of solvent II. adsorption of coloured substance III. absorption and adsorption both of solvent IV. adsorption of solvent
A)
I and II
done
clear
B)
II and III
done
clear
C)
III only
done
clear
D)
IV only
done
clear
View Answer play_arrow
question_answer 81) \[Pb{{O}_{2}}+Pb\xrightarrow{{}}2PbO\,;\,{{\Delta }_{r}}G{}^\circ <0\]\[Sn{{O}_{2}}+Sn\xrightarrow{{}}2SnO;\,{{\Delta }_{r}}G{}^\circ >0\] Which oxidation states are more characteristic for lead and tin?
A)
For lead +4, for tin +2
done
clear
B)
for lead +2 , for tin +4
done
clear
C)
For lead +2, for tin +2
done
clear
D)
For lead +4, for tin +4
done
clear
View Answer play_arrow
question_answer 82) The structure of \[I{{F}_{7}}\]is
A)
trigonal bipyramid
done
clear
B)
octahedral
done
clear
C)
pentagonal bipyramid
done
clear
D)
square pyramid
done
clear
View Answer play_arrow
question_answer 83) The correct order of increasing bond angles in the following species is
A)
\[ClO_{2}^{-}<C{{l}_{2}}O<Cl{{O}_{2}}\]
done
clear
B)
\[C{{l}_{2}}O<Cl{{O}_{2}}<ClO_{2}^{-}\]
done
clear
C)
\[Cl{{O}_{2}}<C{{l}_{2}}O<ClO_{2}^{-}\]
done
clear
D)
\[Cl{{O}_{2}}<ClO_{2}^{-}<ClO\]
done
clear
View Answer play_arrow
question_answer 84) In case of lanthanides, which of the following statements is not correct?
A)
All the members exhibit +3 oxidation state
done
clear
B)
Because of similar properties, the separation of lanthanoids is not easy
done
clear
C)
Availability of 4f electrons results in the formation of compounds in +4 state for all the members of the series
done
clear
D)
There is a gradual decrease in the radii of the members with increasing atomic number in the series
done
clear
View Answer play_arrow
question_answer 85) The acidic, basic or amphoteric nature of\[M{{n}_{2}}{{O}_{7}},\]\[{{V}_{2}}{{O}_{5}}\] and\[CrO\]are respectively
A)
acidic, amphoteric, basic
done
clear
B)
acidic, acidic, basic
done
clear
C)
basic, amphoteric acidic
done
clear
D)
acidic, basic, basic
done
clear
View Answer play_arrow
question_answer 86)
The correct order of increasing reactivity of C - X bond towards nucleophile in the following compounds is
A)
\[I<II<IV<III\]
done
clear
B)
\[III<II<IV<I\]
done
clear
C)
\[I<II<III<IV\]
done
clear
D)
\[III<IV<II<I\]
done
clear
View Answer play_arrow
question_answer 87) Alkyl halides undergoing nucleophilic bimolecular substitution involve
A)
formation of carbocation
done
clear
B)
racemic mixture
done
clear
C)
inversion of configuration
done
clear
D)
retention of configuration
done
clear
View Answer play_arrow
question_answer 88)
Arrange these compounds in order of increasing\[{{S}_{N}}1\]reaction rate
A)
\[IV<II<III<I\]
done
clear
B)
\[I<II<III<IV\]
done
clear
C)
\[IV<I<III<II\]
done
clear
D)
\[IV<I<II<III\]
done
clear
View Answer play_arrow
question_answer 89) \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-Br+C{{H}_{3}}C{{H}_{2}}OH\xrightarrow{C{{H}_{3}}C{{H}_{2}}OH}A\,(major)\]\[\xrightarrow[C{{H}_{3}}C{{H}_{2}}OH]{C{{H}_{3}}C{{H}_{2}}ONa}\,B\,(major)\] Possible products can be\[\begin{align} & C{{H}_{3}}\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{CO}}}\,}}}\,C{{H}_{2}}C{{H}_{3}}\,\,\,\,C{{H}_{3}}-\underset{C{{H}_{2}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\, \\ & \,\,\,\,\,\,\,\,\,\,\,\begin{matrix} I & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,II \\ \end{matrix} \\ \end{align}\]A and 5 can be respectively
A)
I, II
done
clear
B)
II, I
done
clear
C)
II in both cases
done
clear
D)
I in both cases
done
clear
View Answer play_arrow
question_answer 90)
Consider the following reaction, (I) Dehydration (II) Carbon skeleton migration (III) \[{{E}_{2}}\]mechanism (IV) Most stable carbocation is formed.
A)
I, III
done
clear
B)
I, II, III
done
clear
C)
I, II, IV
done
clear
D)
I, III, IV
done
clear
View Answer play_arrow
question_answer 91) Scheme I; Mg / Ether / \[{{D}_{2}}O\] Scheme II; CaO / NaOD (soda lime) Which is used to replace Br by D in bromocyclohexane?
A)
I
done
clear
B)
II
done
clear
C)
Both I and II
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 92)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 93) Identify a reagent from the following list which can be easily distinguish between 1-butane and 2-butyne.
A)
Bromine, \[CC{{l}_{4}}\]
done
clear
B)
Hg Lindlars catalyst
done
clear
C)
Dilute \[{{H}_{2}}S{{O}_{4}},\,HgS{{O}_{4}}\]
done
clear
D)
Ammoniacal \[C{{u}_{2}}C{{l}_{2}}\]solution
done
clear
View Answer play_arrow
question_answer 94)
A)
\[C{{H}_{3}}\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\]
done
clear
C)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-OH\]
done
clear
D)
No reaction
done
clear
View Answer play_arrow
question_answer 95) \[C{{H}_{2}}=CH\underset{OH}{\mathop{\underset{|}{\mathop{CH}}\,}}\,C{{H}_{2}}C{{H}_{2}}OH\xrightarrow[{}]{Mn{{O}_{2}}}A,\,A\,\,\text{is}\]
A)
\[C{{H}_{2}}=CHCOC{{H}_{2}}C{{H}_{2}}OH\]
done
clear
B)
\[C{{H}_{2}}=CH-\underset{OH}{\mathop{\underset{|}{\mathop{CH}}\,}}\,C{{H}_{2}}CHO\]
done
clear
C)
\[C{{H}_{2}}=CHCOC{{H}_{2}}CHO\]
done
clear
D)
\[C{{H}_{2}}=CHCOC{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
question_answer 96) \[C{{H}_{3}}\overset{O}{\mathop{\overset{|\,|}{\mathop{C}}\,}}\,C{{H}_{2}}C\equiv CH\xrightarrow{HOOH}\xrightarrow{NaN{{H}_{2}}/C{{H}_{3}}I}\]\[\xrightarrow{{{H}^{+}}}A,\,\,A\,\,\text{is}\]
A)
\[C{{H}_{3}}COC{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{2}}C{{H}_{2}}COC{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{2}}C\equiv CC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{2}}HC\equiv CHC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 97) How will you convert butan-2-one to propanoic acid?
A)
Tollens reagent
done
clear
B)
Fehlings solution
done
clear
C)
\[NaOH/{{I}_{2}}/{{H}^{+}}\]
done
clear
D)
\[NaOH/NaI/{{H}^{+}}\]
done
clear
View Answer play_arrow
question_answer 98) The smallest ketone and its next homologue are reacted with \[N{{H}_{2}}OH\]to form oxime
A)
two different oximes are formed
done
clear
B)
three different oximes are formed
done
clear
C)
two oximes are optically active
done
clear
D)
all oximes are optically active
done
clear
View Answer play_arrow
question_answer 99) Which of the following has the highest value of energy gap?
A)
Aluminium
done
clear
B)
Silver
done
clear
C)
Germanium
done
clear
D)
Diamond
done
clear
View Answer play_arrow
question_answer 100) \[x\]grams of water is mixed in 69 g ethanol. Mole fraction of ethanol in the resultant solution is 0.6. What is the value of\[x\]in grams?
A)
54
done
clear
B)
36
done
clear
C)
18
done
clear
D)
9
done
clear
View Answer play_arrow
question_answer 101) Blood cells will remain as such in
A)
hypertonic solution
done
clear
B)
hypotonic solution
done
clear
C)
isotonic solution
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 102) Number of atoms of He in 100 u of He (atomic weight of He = 4u) are
A)
25
done
clear
B)
50
done
clear
C)
100
done
clear
D)
\[100\times 6\times {{10}^{-23}}\]
done
clear
View Answer play_arrow
question_answer 103) 4.5 g of a metallic chloride contains 35.5 g of chlorine, the equivalent weight of the metal is
A)
19.5
done
clear
B)
35.5
done
clear
C)
39
done
clear
D)
78.0
done
clear
View Answer play_arrow
question_answer 104) Benzene diazoinum chloride on reaction with phenol in weakly basic medium gives
A)
diphenyl ether
done
clear
B)
p-hydroxy azo benzene
done
clear
C)
chloro benzene
done
clear
D)
benzene
done
clear
View Answer play_arrow
question_answer 105) \[CHC{{l}_{3}}+{{C}_{6}}{{H}_{5}}N{{H}_{2}}+3NaOH\xrightarrow[{}]{{}}A+3B+3C\]In the above reaction, the product A is
A)
chlorobenzene
done
clear
B)
phenyl isocyanide
done
clear
C)
phenyl cyanide
done
clear
D)
phenyl chloride
done
clear
View Answer play_arrow
question_answer 106) Complete hydrolysis of cellulose gives
A)
D- fructose
done
clear
B)
D- ribose
done
clear
C)
D- glucose
done
clear
D)
L- glucose
done
clear
View Answer play_arrow
question_answer 107) Vitamin \[{{B}_{6}}\]is known as
A)
pyridoxin
done
clear
B)
thiamine
done
clear
C)
tocopherol
done
clear
D)
riboflavin
done
clear
View Answer play_arrow
question_answer 108)
The drug
A)
vasodilator
done
clear
B)
antacid
done
clear
C)
analgesics
done
clear
D)
antiseptic
done
clear
View Answer play_arrow
question_answer 109) \[{{H}_{2}}O\]is dipolar, whereas \[Be{{F}_{2}}\]is not. It is because
A)
the electronegativity of F is greater than that of O
done
clear
B)
\[{{H}_{2}}O\]involves hydrogen bonding whereas \[Be{{F}_{2}}\]is a discrete molecule
done
clear
C)
\[{{H}_{2}}O\]is linear and \[Be{{F}_{2}}\] is angular
done
clear
D)
\[{{H}_{2}}O\]is angular and \[Be{{F}_{2}}\]is linear
done
clear
View Answer play_arrow
question_answer 110) Among the following the paramagnetic compound is
A)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
B)
\[{{O}_{3}}\]
done
clear
C)
\[{{N}_{2}}O\]
done
clear
D)
\[K{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 111) Hydration of different ions in aqueous solution is an example of
A)
ion induced dipole interaction
done
clear
B)
dipole-dipole interaction
done
clear
C)
dipole-induced dipole interaction
done
clear
D)
ion-dipole interaction
done
clear
View Answer play_arrow
question_answer 112) Generally, IE, increases along a period But there are some exceptions. One which is not an exception is
A)
N and O
done
clear
B)
Na and Mg
done
clear
C)
Mg and AI
done
clear
D)
Be and B
done
clear
View Answer play_arrow
question_answer 113) The electron affinity values (in kJ\[mo{{l}^{-1}}\]) of three halogens X, Y, Z are respectively -349, -333 and -325. Then X, Y and Z respectively are
A)
\[{{F}_{2}},\,C{{l}_{2}}\]and\[B{{r}_{2}}\]
done
clear
B)
\[C{{l}_{2}},\,{{F}_{2}}\]and\[B{{r}_{2}}\]
done
clear
C)
\[C{{l}_{2}},\,B{{r}_{2}}\]and\[{{F}_{2}}\]
done
clear
D)
\[B{{r}_{2}},\,C{{l}_{2}}\]and\[{{F}_{2}}\]
done
clear
View Answer play_arrow
question_answer 114) According to the kinetic theory of gases, in an ideal gas, between two successive collisions a gas molecule travels
A)
in a circular path
done
clear
B)
in a wavy path
done
clear
C)
in a straight line path
done
clear
D)
with an accelerated velocity
done
clear
View Answer play_arrow
question_answer 115) Root mean square speed of\[{{N}_{2}}(g)\]in air is \[515\,m{{s}^{-1}}.\] What is the average speed of He gas in the same air?
A)
\[536.8\,m{{s}^{-1}}\]
done
clear
B)
\[839.2\,m{{s}^{-1}}\]
done
clear
C)
\[1245.5\,m{{s}^{-1}}\]
done
clear
D)
\[1255.3\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 116) Photochemical dissociation produces a normal oxygen atom and a oxygen atom 2.5 eV more energetic than normal one. Also the average bond energy of \[{{O}_{2}}\] into normal oxygen is 498 kJ\[mo{{l}^{-1}}\]. Determine the longest wavelength required for photochemical decomposition of\[{{O}_{2}}\].
A)
126 nm
done
clear
B)
175 nm
done
clear
C)
190 nm
done
clear
D)
205 nm
done
clear
View Answer play_arrow
question_answer 117) If \[l=3,\]what can be said about\[n?\]
A)
n must be less than 3
done
clear
B)
n must be equal to 3
done
clear
C)
n must be greater than 3
done
clear
D)
n must be equal to 4
done
clear
View Answer play_arrow
question_answer 118) \[{{H}_{2}}(g)+2NO\,(g)\rightleftharpoons {{N}_{2}}O(g)+{{H}_{2}}O\,(g)\,;\]\[{{K}_{c}}=1.4\times {{10}^{5}}\]at \[25{}^\circ C\] If the reaction mixture contains 0.05 M\[{{H}_{2}}(g),\]\[0.02\,M\,NO\,(g),\]\[5.4\,M\,{{N}_{2}}O\,(g)\] and \[8.7\,M{{H}_{2}}O\,(g)\] at some particular moment in time then we accurately predict that
A)
the reaction is very close to equilibrium
done
clear
B)
the raction is very far to equilibrium
done
clear
C)
the reaction is at equilibrium
done
clear
D)
both the\[{{H}_{2}}(g)\]and\[NO(g)\]concentration must increase significantly to reach equilibrium
done
clear
View Answer play_arrow
question_answer 119) A cone. aq. solution of \[{{H}_{2}}S{{O}_{4}}\] is 86% by mass and has a density of 1.78 g \[m{{L}^{-1}}\]. 50 mL of this solution is diluted to 1 L with water. What is the \[{{H}^{+}}\]ion concentration of the dilute solution in mol\[{{L}^{-1}}\]?
A)
0.15
done
clear
B)
0.51
done
clear
C)
0.78
done
clear
D)
1.56
done
clear
View Answer play_arrow
question_answer 120) A certain process releases 64.0 kJ of heat which is transferred to the surroundings at a constant pressure and a constant temperature of 300 K. For this process\[\Delta {{S}_{surr}}\]is
A)
\[64.0\text{ }KJ\]
done
clear
B)
\[-\,64.0\text{ }KJ\]
done
clear
C)
\[-\,213\,J{{K}^{-1}}\]
done
clear
D)
\[213\,J{{K}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 121) Iodine is obtained from the members of
A)
green algae
done
clear
B)
brown algae
done
clear
C)
red algae
done
clear
D)
blue-green algae
done
clear
View Answer play_arrow
question_answer 122) If large quantities of domestic sewage are continuously emptied into a small stream, it leads to
A)
algal bloom
done
clear
B)
eutrophication
done
clear
C)
increase in temperature
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 123) Which of the following planets is called twin of the earth
A)
Mars
done
clear
B)
Pluto
done
clear
C)
Venus
done
clear
D)
Mercury
done
clear
View Answer play_arrow
question_answer 124) In which one of following would you except to find glyoxysomes?
A)
Endosperm of wheat
done
clear
B)
Endosperm of castor
done
clear
C)
Palisade cells in leaf
done
clear
D)
Root hair
done
clear
View Answer play_arrow
question_answer 125) Vaccines prepeared through recombinant DNA technology are called
A)
First generation vaccines
done
clear
B)
2nd generation vaccine
done
clear
C)
3rd generation vaccine
done
clear
D)
None of above
done
clear
View Answer play_arrow
question_answer 126) Continued exposure of vinyl chloride may cause cances of the
A)
Liver
done
clear
B)
WBCs
done
clear
C)
Lymphoid tissue
done
clear
D)
Spleen
done
clear
View Answer play_arrow
question_answer 127) Miracidium larva occurs in the life history of
A)
roundworm
done
clear
B)
liver fluke
done
clear
C)
earthworm
done
clear
D)
tapeworm
done
clear
View Answer play_arrow
question_answer 128) Male Ascaris is differentiable from female Ascaris in
A)
presence of post-anal papillae
done
clear
B)
presence of pre-anal papillae
done
clear
C)
presence of penial setae
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 129) Penetrant, valvent and glutinant are type of
A)
nematocyst of Hydra
done
clear
B)
tentacles of Hydra
done
clear
C)
zooids of Obelia
done
clear
D)
tentacles of Obelia
done
clear
View Answer play_arrow
question_answer 130) Which of the following enzyme is used in PCR (Polymerase Chain Reaction)?
A)
Taq polymerase
done
clear
B)
Vent polymerase
done
clear
C)
Both (a) and (b)
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 131) A woman with straight hair marries with a man with curly hair and who is known to be heterozygous for the trait. What is the chance that their first child will have curly hair?
A)
No change
done
clear
B)
One in two
done
clear
C)
It is certain
done
clear
D)
One in four
done
clear
View Answer play_arrow
question_answer 132) Which of the following are required in minimum amount by human?
A)
Iron, iodine, carbon, \[Mn,\,Cu,\,{{O}_{2}}\]
done
clear
B)
Iron, iodine,\[Mn,\,\,Cu,\,\,Zn,\]fluorine
done
clear
C)
Iron, iodine, \[Mn,\,\,Zn,\]hydrogen
done
clear
D)
\[N,\,\,{{O}_{2}},\,\,Zn,\]fluorine
done
clear
View Answer play_arrow
question_answer 133) Which of the following nerve of man is both sensory and motor?
A)
Olfactory
done
clear
B)
Trigeminal
done
clear
C)
Optic
done
clear
D)
Auditory
done
clear
View Answer play_arrow
question_answer 134) Highest number of antibiotics are produced by
A)
Bacillus
done
clear
B)
Penicillium.
done
clear
C)
Streptomyces
done
clear
D)
Grise of ulvin
done
clear
View Answer play_arrow
question_answer 135) Nurse tissue technique is applied in
A)
pollen culture
done
clear
B)
embryo culture
done
clear
C)
ovule culture
done
clear
D)
ovary culture
done
clear
View Answer play_arrow
question_answer 136) The reflectivity percentage of incident light on earth is meteorologically called as
A)
Albedo
done
clear
B)
Tornado
done
clear
C)
Refraction
done
clear
D)
Reradiation
done
clear
View Answer play_arrow
question_answer 137) Eusthenopteron connects
A)
reptiles and birds
done
clear
B)
birds and mammals
done
clear
C)
fishes and amphibians
done
clear
D)
amphibians and reptiles
done
clear
View Answer play_arrow
question_answer 138)
The numbers of stomata and epidermal cells in 1 \[m{{m}^{2}}\] leaf area of lower epidermis of leaves of X, Y and Z plants are given below. Arrange the plants in decreasing order of their stomatal index. Cell Number of Stomata No. of epidermal Cells X 30 150 Y 60 240 Z 90 400
The correct answer is
A)
X Y Z
done
clear
B)
Y Z X
done
clear
C)
Z Y X
done
clear
D)
Y X Z
done
clear
View Answer play_arrow
question_answer 139) Which one of the following components of ecosystem comes from outside?
A)
Energy
done
clear
B)
Oxygen
done
clear
C)
Insects
done
clear
D)
Temperature
done
clear
View Answer play_arrow
question_answer 140) The genome of Caenorhabiditis elgans consist of
A)
3 billion bp (base pair) and 30000 genes
done
clear
B)
12 million bp and 6000 genes
done
clear
C)
4.7 million bp and 4000 genes
done
clear
D)
97 million bp and 13000 genes
done
clear
View Answer play_arrow
question_answer 141) Dermatogen, periblem and plerome are
A)
permanent tissues
done
clear
B)
meristematic tissues
done
clear
C)
intercalary tissues
done
clear
D)
secondary tissue .
done
clear
View Answer play_arrow
question_answer 142) Which one is viral disease in silkworm?
A)
Flacherie
done
clear
B)
Maggot disease
done
clear
C)
Muscardine
done
clear
D)
Pebrine diseases
done
clear
View Answer play_arrow
question_answer 143)
Which one is correct sequence of following events as per their discovery? I. Formulation of the chromosome theory of inheritence. II. Experiments which proved that DNA is the genetic material. III. Mendels law of inheritance.
A)
I, III and II
done
clear
B)
I, II and III
done
clear
C)
III, I and II
done
clear
D)
II, I and III
done
clear
View Answer play_arrow
question_answer 144) Which of the following has more imbibition power?
A)
Cellulose
done
clear
B)
Hemicellulose
done
clear
C)
Fat
done
clear
D)
Protein
done
clear
View Answer play_arrow
question_answer 145) The structural and functional unit of the striated muscle fibre is called
A)
sarcolemma
done
clear
B)
sarcomere
done
clear
C)
sarcoplasm
done
clear
D)
myofibril
done
clear
View Answer play_arrow
question_answer 146) The fossils record of placoderms is found in the era
A)
Mesozoic
done
clear
B)
Coenozoic
done
clear
C)
Archaeozoic
done
clear
D)
Palaeozoic
done
clear
View Answer play_arrow
question_answer 147) An examples of gene silencing is
A)
Bt cotton
done
clear
B)
trangenic rice
done
clear
C)
Flavr savr tomato
done
clear
D)
transgenic maize
done
clear
View Answer play_arrow
question_answer 148) How many number of reducing powers are required to synthesise one molecule of glyceraldehyde phosphate?
A)
9 ATP and 6 NADPH
done
clear
B)
3ATP and 3 NADPH
done
clear
C)
6 ATP and 6 NADPH
done
clear
D)
9 ATP and 36 NADPH
done
clear
View Answer play_arrow
question_answer 149) Who purposed the central dogma?
A)
Francis Crick
done
clear
B)
William Klug
done
clear
C)
Beadle and Tatum
done
clear
D)
Watson and Crick
done
clear
View Answer play_arrow
question_answer 150) Which of the following disease is non-cancerous?
A)
BPH
done
clear
B)
Hepatitis-B virus
done
clear
C)
Carcinoma of testis
done
clear
D)
Melanoma
done
clear
View Answer play_arrow
question_answer 151) Fragile X syndrome is characterised by
A)
excessive bleeding
done
clear
B)
colour blindness
done
clear
C)
paralysis of a limb
done
clear
D)
mental retardation and abnormal facial appearance with large ears and long face
done
clear
View Answer play_arrow
question_answer 152) Where can we study mitosis?
A)
Brain
done
clear
B)
Legs
done
clear
C)
Nail base
done
clear
D)
Kidney
done
clear
View Answer play_arrow
question_answer 153) The type of joints between the human skull bones is called
A)
cartilaginous joint
done
clear
B)
hinge joint
done
clear
C)
fibrous joint
done
clear
D)
synovial joint
done
clear
View Answer play_arrow
question_answer 154) Somatostatin is a hormone secreted by hypothalamus. It inhibits the excretion of
A)
thyroxine
done
clear
B)
GH (Growth Hormone)
done
clear
C)
vasopressin
done
clear
D)
ACTH
done
clear
View Answer play_arrow
question_answer 155) Natural selection favours the leaf butterfly because it shows
A)
counter shading
done
clear
B)
batesian mimicry
done
clear
C)
mullerian mimicry
done
clear
D)
protective resemblance
done
clear
View Answer play_arrow
question_answer 156) The portion of the endometrium that covers the embryo and is located between the embryo and uterine cavity is the
A)
deidua basalis
done
clear
B)
decidua umbilicus
done
clear
C)
decidua capsularis
done
clear
D)
decidua functionalis
done
clear
View Answer play_arrow
question_answer 157) Duration of complete cardiac diastable is
A)
0.1 sec
done
clear
B)
0.4 sec
done
clear
C)
0.3 sec
done
clear
D)
0.5 sec
done
clear
View Answer play_arrow
question_answer 158) Which of the following is not the main function of lymph glands.
A)
Basophil
done
clear
B)
Neutrophil
done
clear
C)
Lymphocyte
done
clear
D)
Eosinophil
done
clear
View Answer play_arrow
question_answer 159) Which is not connected with ficory of agening?
A)
Wear and tear
done
clear
B)
Neurohormonal
done
clear
C)
Epimorphosis
done
clear
D)
Metabolic rate
done
clear
View Answer play_arrow
question_answer 160) Auxetic growth is seen in
A)
rotifers
done
clear
B)
nematodes
done
clear
C)
funicates
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 161) Biolistic technique is used in
A)
tissue culture process
done
clear
B)
gene transfer process
done
clear
C)
hybridisation process
done
clear
D)
germplasm conservation process
done
clear
View Answer play_arrow
question_answer 162) Which of the following is correctly matched?
A)
Iron age -Present age.
done
clear
B)
Bronze age - Age of agriculture, use of cloths and utensile
done
clear
C)
Mesolithic age - Age of tools stones and bones
done
clear
D)
Palaeolithic age-Age of animal husbandry language reading and write
done
clear
View Answer play_arrow
question_answer 163) Which of the following virus spreaded through blood?
A)
Hepatitis-A virus
done
clear
B)
Hepatitis-B virus
done
clear
C)
Hepatitis-E virus
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 164) A higher dose of alcohol may leads to
A)
\[C{{H}_{3}}CHO\]accumulation in liver
done
clear
B)
lowering of glucose level in blood
done
clear
C)
causes gastritis
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 165) Which one of the following is suitable for experiment on linkage?
A)
\[AA\,aa\times Aa\,BB\]
done
clear
B)
\[Aa\,Bb\times Aa\,Bb\]
done
clear
C)
\[AA\,BB\times aa\,bb\]
done
clear
D)
\[aaBB\times aaBB\]
done
clear
View Answer play_arrow
question_answer 166) Surgical procedure for child birth is called
A)
dilation and curattage (D and C)
done
clear
B)
artifical rupture of membrane
done
clear
C)
caesarean
done
clear
D)
shirodkar
done
clear
View Answer play_arrow
question_answer 167) \[\overset{\text{o}}{\mathop{\text{A}}}\,\](Angstrom) means
A)
\[{{10}^{-9}}m\]
done
clear
B)
\[{{10}^{-8}}m\]
done
clear
C)
\[{{10}^{-10}}m\]
done
clear
D)
\[{{10}^{-11}}m\]
done
clear
View Answer play_arrow
question_answer 168) The type of arrangement of flower and mode of distribution of flower on a shoot of palm tree is
A)
spikelet
done
clear
B)
spadix
done
clear
C)
spike of spikelets
done
clear
D)
compound spadix
done
clear
View Answer play_arrow
question_answer 169) The protein first hypothesis (in origin of life) is given by
A)
Sidney Fox
done
clear
B)
Thomas Cech
done
clear
C)
Cairns Smith
done
clear
D)
Sidney Altman
done
clear
View Answer play_arrow
question_answer 170) Ozone layer is present in
A)
stratosphere
done
clear
B)
Troposphere
done
clear
C)
mesosphere
done
clear
D)
ionosphere
done
clear
View Answer play_arrow
question_answer 171) Enzyme renin acts upon
A)
angiotensinogen
done
clear
B)
vasodilation
done
clear
C)
blood pressure
done
clear
D)
digestion process
done
clear
View Answer play_arrow
question_answer 172) Types of quills (flight) feathers is/are
A)
filoplume
done
clear
B)
remiges
done
clear
C)
corert
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
question_answer 173) Ischihara chart is used to detect
A)
eye sight
done
clear
B)
colour blindness
done
clear
C)
diabetes
done
clear
D)
tuberculosis
done
clear
View Answer play_arrow
question_answer 174) Coelom is present between
A)
ectoderm and endoderm
done
clear
B)
mesoderm and ectoderm
done
clear
C)
body wall and ectoderm
done
clear
D)
mesoderm and endoderm
done
clear
View Answer play_arrow
question_answer 175) The major excretory product of arthropods
A)
ammonia
done
clear
B)
urea
done
clear
C)
uric acid
done
clear
D)
trimethyl oxide
done
clear
View Answer play_arrow
question_answer 176) Structurally the amphicribal vascular bundles resemble closely to
A)
dictyosteles
done
clear
B)
solanosteles
done
clear
C)
siphonosteles
done
clear
D)
protostele
done
clear
View Answer play_arrow
question_answer 177) In a detritus food chain detritus is broken down into form of inorganic elements by detritivore. The process of breakdown of detritus is known as
A)
mulching
done
clear
B)
leaching
done
clear
C)
mineralisation
done
clear
D)
fragmentation
done
clear
View Answer play_arrow
question_answer 178) Savannah can be defined as
A)
temperate forest
done
clear
B)
tropical forest
done
clear
C)
mansoon forest
done
clear
D)
grassland with scattered trees
done
clear
View Answer play_arrow
question_answer 179) Which of the following statements is true for lysosome?
A)
Suicidal bag
done
clear
B)
Stain for acid phosphatase
done
clear
C)
Some cystolic protin contains signals directing them to lysosome
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 180)
Match the following columns. Column I Column II A. Oral contraceptives 1. Passage of sperm is blockel B. IUDs 2. Released hormone to block ovulation C. Implants/ Norplant 3. Inhibit secretion of LH and FSH D. Vasectomy 4. Prevent fertillisation implantation of embryo
Codes
A)
A-4 A-3 A-1 A-2
done
clear
B)
B-3 B-4 B-2 B-1
done
clear
C)
C-3 C-4 C-1 C-2
done
clear
D)
D-3 D-2 D-5 D-1
done
clear
View Answer play_arrow
question_answer 181) The holidays for the banks are declared as per
A)
Reserve Bank Act
done
clear
B)
Banking Regulation Act
done
clear
C)
Negotiable Instruments Act
done
clear
D)
Companies Act
done
clear
View Answer play_arrow
question_answer 182) The primary sector includes all of the following, except
A)
forestry
done
clear
B)
manufacturing
done
clear
C)
mining
done
clear
D)
agriculture
done
clear
View Answer play_arrow
question_answer 183) Planning commission of India is
A)
a constitutional body
done
clear
B)
an independent and autonomous body
done
clear
C)
a statutory body
done
clear
D)
a non-statutory body
done
clear
View Answer play_arrow
question_answer 184) Which of the following is not covered under the monetary and credit policy of RBI?
A)
Bank Rate
done
clear
B)
Repo Rate
done
clear
C)
Cash Reserve Ratio
done
clear
D)
Exchange rate of Foreign currencies
done
clear
View Answer play_arrow
question_answer 185) Which of the following is associated with white Revolution in India?
A)
Dadabhai Naoroji
done
clear
B)
Verghese kurien
done
clear
C)
Spencer Hatch
done
clear
D)
MS Swaminathan
done
clear
View Answer play_arrow
question_answer 186) During the time of which Mughal Emperor did the English East India Company established its first factory in India?
A)
Akbar
done
clear
B)
Jahangir
done
clear
C)
Shahjahan
done
clear
D)
Aurangzeb
done
clear
View Answer play_arrow
question_answer 187) What is the local name of Mohenjodaro?
A)
Mound of the Dead
done
clear
B)
Mound of the Great
done
clear
C)
Mound of the Living
done
clear
D)
Mount of the Sun
done
clear
View Answer play_arrow
question_answer 188) Which one of the following planets has largest number of natural satellites or Moons?
A)
Jupitor
done
clear
B)
Mars
done
clear
C)
Saturn
done
clear
D)
Venus
done
clear
View Answer play_arrow
question_answer 189) By which name does the Brahmaputra river enter into India?
A)
Manas
done
clear
B)
Dhansiri
done
clear
C)
Dihang
done
clear
D)
Tsangpo
done
clear
View Answer play_arrow
question_answer 190) Mullaperiyar dom whose safety has been a matter of concern is a dispute between
A)
Tamil Nadu and Andhra Pradesh
done
clear
B)
Karnataka and Kerala
done
clear
C)
Tamil Nadu and Kerala
done
clear
D)
Tamil Nadu and Karnataka
done
clear
View Answer play_arrow
question_answer 191) Maps on large-scale, representing both natural and man-made features are called
A)
Topographic maps
done
clear
B)
Thematic maps
done
clear
C)
Atlas maps
done
clear
D)
Wall maps
done
clear
View Answer play_arrow
question_answer 192) Blood group was discovered by
A)
Pavlov
done
clear
B)
Alexander Fleming
done
clear
C)
William Harvey
done
clear
D)
Landsteiner
done
clear
View Answer play_arrow
question_answer 193) The enzyme that converts glucose to ethyl alcohol is
A)
diastase
done
clear
B)
zymase
done
clear
C)
maltase
done
clear
D)
invertase
done
clear
View Answer play_arrow
question_answer 194) Chromosomes consist of
A)
DNA and lipids
done
clear
B)
RNA and amino acids
done
clear
C)
RNA and sugar
done
clear
D)
DNA and proteins
done
clear
View Answer play_arrow
question_answer 195) Which of the metals causes Itai-Itai disease?
A)
Cobalt
done
clear
B)
Copper
done
clear
C)
Chromium
done
clear
D)
Cadmium
done
clear
View Answer play_arrow
question_answer 196) Which of the following is known as graveyard of RBCs?
A)
Spleen
done
clear
B)
Liver
done
clear
C)
Bone marrow
done
clear
D)
Appendix
done
clear
View Answer play_arrow
question_answer 197) The anti-malarial drug quinine is made from a plant. The plant is
A)
neem
done
clear
B)
eucalyptus
done
clear
C)
cinvamon
done
clear
D)
cinchona
done
clear
View Answer play_arrow
question_answer 198) Conversion of chemical energy info electrical energy occur in
A)
atomic bombs
done
clear
B)
electric heaters
done
clear
C)
battery
done
clear
D)
dynamo
done
clear
View Answer play_arrow
question_answer 199) A water tank appears shallower when it is viewed from top due to
A)
rectilinear propagation of light
done
clear
B)
reflection
done
clear
C)
total internal reflection
done
clear
D)
refraction
done
clear
View Answer play_arrow
question_answer 200) Non-stick cooking utensils are coated with
A)
polystyrene
done
clear
B)
polyvinyl chloride
done
clear
C)
black point
done
clear
D)
Teflon
done
clear
View Answer play_arrow
question_answer 201) The scientist who first discovered that the Earth revolves round the Sun was
A)
Newton
done
clear
B)
Dalton
done
clear
C)
Copernicus
done
clear
D)
Eistein
done
clear
View Answer play_arrow
question_answer 202) What is correcting errors in a programme called?
A)
Compiling
done
clear
B)
Debugging
done
clear
C)
Grinding
done
clear
D)
Interpreting
done
clear
View Answer play_arrow
question_answer 203) The set of instructions which tells a computer what to do is called
A)
matter
done
clear
B)
instructor
done
clear
C)
compiler
done
clear
D)
Programme
done
clear
View Answer play_arrow
question_answer 204) Which was mainly responsible for the Bhopal Mishap in 1984?
A)
Methyl Isocyanate
done
clear
B)
Carbon di oxide
done
clear
C)
Ammonia
done
clear
D)
Carbon monoxide
done
clear
View Answer play_arrow
question_answer 205) Glycol is added to aviation gasoline because it
A)
prevents freezing of petrol
done
clear
B)
reduces consumption of petrol
done
clear
C)
reduces evaporation of petrol
done
clear
D)
increase efficiency of petrol
done
clear
View Answer play_arrow
question_answer 206) Which one of the following is used as a moderator in a nucler reactor?
A)
Radium
done
clear
B)
Ordinary water
done
clear
C)
Thorium
done
clear
D)
Heavy water
done
clear
View Answer play_arrow
question_answer 207) Global warming is a consequences of
A)
radioactive fall out
done
clear
B)
ozone depletion
done
clear
C)
green house effect 1
done
clear
D)
acid rain
done
clear
View Answer play_arrow
question_answer 208) Acid rain is due to air pollution by
A)
carbon d ioxide
done
clear
B)
carbon monoxide
done
clear
C)
methane
done
clear
D)
nitrous oxide and sulphur dioxide
done
clear
View Answer play_arrow
question_answer 209) The concept of carbon credit originated from which one of the following?
A)
Kyoto protocol
done
clear
B)
Earth summit
done
clear
C)
Martreal protocol
done
clear
D)
G-8 summit
done
clear
View Answer play_arrow
question_answer 210) A person well known as father Terasa is
A)
Aryaratne
done
clear
B)
Gerhard Fischer
done
clear
C)
Abdul Sattar Edhi
done
clear
D)
Baba Amte
done
clear
View Answer play_arrow
question_answer 211) The larest biogas plant of the world was inaugurated in
A)
USA
done
clear
B)
Ireland
done
clear
C)
Finland
done
clear
D)
Germany
done
clear
View Answer play_arrow
question_answer 212) The correct sequence order of the most polulous countries of the world is
A)
China, India, USA, Brazil, Indonesia
done
clear
B)
China, India, Brazil, Indonesia, USA
done
clear
C)
China, India, Indonesia, USA, Brazil
done
clear
D)
China, India, USA, Indonesia, Brazil
done
clear
View Answer play_arrow
question_answer 213) Who is the author of the book My life?
A)
Hillary Clinton
done
clear
B)
Bill Clinton
done
clear
C)
The Dalai Lama
done
clear
D)
Imran Khan
done
clear
View Answer play_arrow
question_answer 214) The Headquarter of European union is lituated in
A)
England
done
clear
B)
Germany
done
clear
C)
France
done
clear
D)
Belgium
done
clear
View Answer play_arrow
question_answer 215) The Cannes award is given for excellence in which of the following fields?
A)
Films
done
clear
B)
Journalism
done
clear
C)
Economics
done
clear
D)
Literature
done
clear
View Answer play_arrow
question_answer 216) The inaugural world Test Cricket Championship will take place in 2017 in
A)
England and Wales
done
clear
B)
West Indies
done
clear
C)
Australia
done
clear
D)
India, Sri Lanka and Pakistan
done
clear
View Answer play_arrow
question_answer 217) Intensity of an earthquake is measured with
A)
Barometer
done
clear
B)
Hydrometer
done
clear
C)
Polygraph
done
clear
D)
Seismograph
done
clear
View Answer play_arrow
question_answer 218) Sushil kumar won the medal at London Olympic event is a famous
A)
Table tennis player
done
clear
B)
Wrestling champion
done
clear
C)
Golf player
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 219) UN International day for natural disaster reduction is observed on
A)
6th July
done
clear
B)
15th September
done
clear
C)
13th October
done
clear
D)
18th November
done
clear
View Answer play_arrow
question_answer 220) As per census 2011, the sex ratio of India is
A)
940
done
clear
B)
933
done
clear
C)
927
done
clear
D)
937
done
clear
View Answer play_arrow
question_answer 221) The President of Indian National Congress at the time of partition of India was
A)
C. Rajagopalachari
done
clear
B)
JB Kriplani
done
clear
C)
Jawaharlal Nehru
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 222) Article 368 of the Indian Constitution provides
A)
Financial emergency
done
clear
B)
UPSC
done
clear
C)
Amendment to the constitution
done
clear
D)
Local government
done
clear
View Answer play_arrow
question_answer 223) The Right to Information Act of Parliament received the assent of the President of India on
A)
15th may, 2005
done
clear
B)
5th June, 2005
done
clear
C)
15th June, 2005
done
clear
D)
12th October, 2005
done
clear
View Answer play_arrow
question_answer 224) The number of Anglo Indians who can be hominated by the President to the Lok Sabha is
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 225) Devaluation of currency helps in promoting
A)
imports
done
clear
B)
exports
done
clear
C)
tourism
done
clear
D)
national income
done
clear
View Answer play_arrow
question_answer 226) 2018 FIFA World Cup would be held in
A)
France
done
clear
B)
Russia
done
clear
C)
Qatar
done
clear
D)
Netherlands
done
clear
View Answer play_arrow
question_answer 227) Insufficient blood supply in the human body is referred to as
A)
Hemostasis
done
clear
B)
Haemorrhage
done
clear
C)
Ischaemia
done
clear
D)
Hypermesia
done
clear
View Answer play_arrow
question_answer 228) What percentage does the Indian population comprile in the world?
A)
17.00 %
done
clear
B)
16.31 %
done
clear
C)
17.31 %
done
clear
D)
16.25 %
done
clear
View Answer play_arrow
question_answer 229) Which one among the following gases readily combines with the haemoglobin of the blood?
A)
Methane
done
clear
B)
Nitrogen dioxide
done
clear
C)
Carbon monoxide
done
clear
D)
Sulphur dioxide
done
clear
View Answer play_arrow
question_answer 230) Human blood is a viscous fluid. This visocity is due to
A)
protains in blood
done
clear
B)
platelets in plasma
done
clear
C)
sodium in serum
done
clear
D)
RB and WBC in blood
done
clear
View Answer play_arrow
question_answer 231) Who translated Ramayan into Persian in accordance with the wishes of Akbar?
A)
Abul Fazl
done
clear
B)
Faizi
done
clear
C)
Abdul Qadir Badami
done
clear
D)
Abdur-Rahim Khan-I-Khanna
done
clear
View Answer play_arrow
question_answer 232) With which of the following movements is Arun Asaf Ali associated?
A)
Non-cooperation movement
done
clear
B)
Civil Disobedient movement
done
clear
C)
Individual Satyagraha
done
clear
D)
Quit India Movement
done
clear
View Answer play_arrow
question_answer 233) Who is competent to dissolve the Rajya Sabha?
A)
Chairman of Rajya Sabha
done
clear
B)
The President
done
clear
C)
The joint-Session of Parliament
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 234) The 9th schedule to the Indian Constitution was added by
A)
First Amendment
done
clear
B)
Eighth Amendment
done
clear
C)
North Amendment
done
clear
D)
Forty Second Amendment
done
clear
View Answer play_arrow
question_answer 235) The largest planet in our Solar System is
A)
Mars
done
clear
B)
Jupiter
done
clear
C)
Saturh
done
clear
D)
Butterfly
done
clear
View Answer play_arrow
question_answer 236) The highest grade and best quality coal is
A)
Lignite
done
clear
B)
Peat
done
clear
C)
Bituminous
done
clear
D)
Anthracite
done
clear
View Answer play_arrow
question_answer 237) Which one of the following longitudes determines the Indian standard time?
A)
\[85.5{}^\circ E\]
done
clear
B)
\[86.5{}^\circ E\]
done
clear
C)
\[84.5{}^\circ E\]
done
clear
D)
\[82.5{}^\circ E\]
done
clear
View Answer play_arrow
question_answer 238) Which of the following is a land locked state?
A)
Gujarat
done
clear
B)
Andhra Pradesh
done
clear
C)
Madhya Pradesh
done
clear
D)
Tamil Nadu
done
clear
View Answer play_arrow
question_answer 239) Ctrl shift and alt are called .... keys.
A)
modifier
done
clear
B)
function
done
clear
C)
alphanumeric
done
clear
D)
adjustment
done
clear
View Answer play_arrow
question_answer 240) Storage which stores or retains data after power off is called
A)
volatile storage
done
clear
B)
none-volatile storage
done
clear
C)
sequential storage
done
clear
D)
direct storage
done
clear
View Answer play_arrow
question_answer 241) Study of disease like cancer is called
A)
osteology
done
clear
B)
oncology
done
clear
C)
karyology
done
clear
D)
nephrology
done
clear
View Answer play_arrow
question_answer 242) Bile is stored in
A)
mouth
done
clear
B)
liver
done
clear
C)
gallbladder
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 243) Alpha-Keratin is a protein, present in
A)
blood
done
clear
B)
sakin
done
clear
C)
lymph
done
clear
D)
egg
done
clear
View Answer play_arrow
question_answer 244) Atomic power plant works on the principle of
A)
fission
done
clear
B)
fusion
done
clear
C)
thermal combustion
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 245) Cylindrical lens is used by a person suffering from
A)
astigmatism
done
clear
B)
myopia
done
clear
C)
hypermetropia
done
clear
D)
presbyopia
done
clear
View Answer play_arrow
question_answer 246) The chemical used as a fixer in photography is
A)
sodium sulphate
done
clear
B)
sodium thiosulphate
done
clear
C)
ammonium persulphate
done
clear
D)
borax
done
clear
View Answer play_arrow
question_answer 247) Higher concentration of nitrogen di oxide in atmosphere air causes
A)
cancer
done
clear
B)
bronchitis
done
clear
C)
asphyxiation
done
clear
D)
corrosion
done
clear
View Answer play_arrow
question_answer 248) The first woman to become a Chief Minister of any state in India was.....
A)
Nandini Satpathy
done
clear
B)
Dr J. Jayalalitha
done
clear
C)
Sucheta kriplani
done
clear
D)
Mayawati
done
clear
View Answer play_arrow
question_answer 249) CBDT stands for
A)
continued test Ban treaty
done
clear
B)
continued test based treatments
done
clear
C)
comprehensive test ban treaty
done
clear
D)
commerical test based tariff
done
clear
View Answer play_arrow
question_answer 250) Where is the Central Food Technology Research Institute situated?
A)
Delhi
done
clear
B)
Agara
done
clear
C)
Ahmadabad
done
clear
D)
Mysore
done
clear
View Answer play_arrow
question_answer 251) When did WHO (World Health Organisation) come into force on
A)
6th April, 1948
done
clear
B)
7th April, 1948
done
clear
C)
10th April, 1948
done
clear
D)
26th April, 1948
done
clear
View Answer play_arrow
question_answer 252) Which one of the following is the biggest flower in the world?
A)
Sun flower
done
clear
B)
Lotus
done
clear
C)
Gleory Lily
done
clear
D)
Rafflesia
done
clear
View Answer play_arrow
question_answer 253) Palk strait connects India to
A)
Sri Lanka
done
clear
B)
Pakistan
done
clear
C)
Myanmar
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 254) Who is the 158th member of WTO?
A)
Russia
done
clear
B)
Vanuatu
done
clear
C)
Laos
done
clear
D)
Australia
done
clear
View Answer play_arrow
question_answer 255) Lok Sabha passed the National Food Security Bill 2013 on
A)
22rd August, 2013
done
clear
B)
23rd August, 2013
done
clear
C)
26th August, 2013
done
clear
D)
27th August, 2013
done
clear
View Answer play_arrow
question_answer 256) Direction: Select the related word from the given alternative. Botany : Plants :: Entomology?
A)
Insects
done
clear
B)
Snakes
done
clear
C)
Birds
done
clear
D)
Germs
done
clear
View Answer play_arrow
question_answer 257)
Arrange the following words according to English dictionary. (i) Billian (ii) Bifurcate (iii) Bilateral (iv) Bilirubin
A)
i, ii, iii, iv
done
clear
B)
iv, iii, ii, i
done
clear
C)
ii, iii, i, iv
done
clear
D)
ii, iii, iv, i
done
clear
View Answer play_arrow
question_answer 258) Which one set of letters when sequentially placed at the gaps in the given letters series shall complete it? cccbb_aa_ccbbbaa_c
A)
acbc
done
clear
B)
baca
done
clear
C)
baba
done
clear
D)
acba
done
clear
View Answer play_arrow
question_answer 259) Find the wrong number in the given series. 5, 27, 61, 122, 213, 340, 509
A)
27
done
clear
B)
61
done
clear
C)
122
done
clear
D)
509
done
clear
View Answer play_arrow
question_answer 260) Pointing to a man in a photograph, a woman said, His brothers father is the only son n my grandfather. How is the woman relate to the man in the photograph?
A)
Mother
done
clear
B)
Aunt
done
clear
C)
Sister
done
clear
D)
Daughter
done
clear
View Answer play_arrow
question_answer 261) In an examination, Raju got more marks than Mukesh but not as many as Priya. Priya got more marks than Gaurav and Kavita. Gaurav got less marks than Mukesh but his marks are not the lowest in the group. Who is second in the descending order of marks?
A)
Priya
done
clear
B)
Kavita
done
clear
C)
Raju
done
clear
D)
Gaurav
done
clear
View Answer play_arrow
question_answer 262) From the given alternatives, select the word which cannot be formed using the letters of the given word. TRANSLATION
A)
NATION
done
clear
B)
RATION
done
clear
C)
TRANSIT
done
clear
D)
TRANSMIT
done
clear
View Answer play_arrow
question_answer 263) Directions: Select the missing number from the given responses.
A)
1
done
clear
B)
26
done
clear
C)
39
done
clear
D)
45
done
clear
View Answer play_arrow
question_answer 264)
Directions: Select the missing number from the given responses.
A)
6
done
clear
B)
8
done
clear
C)
3
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 265) A man walks northwards and then turns left, then turns right and then left. In which direction is he moving now?
A)
West
done
clear
B)
North
done
clear
C)
East
done
clear
D)
South
done
clear
View Answer play_arrow
question_answer 266) Ashish walked 50 m towards East and took a right turn and walked 40 m. He again took a right turn and walked 50 m. How for is he from the starting point?
A)
10 m
done
clear
B)
25 m
done
clear
C)
30 m
done
clear
D)
40 m
done
clear
View Answer play_arrow
question_answer 267)
Two statements are given followed by two conclusions I and II. You have to consider the statements to be true even if they seem to be at variance from commonly known facts. You are to decide which of the given conclusions, if any follow from the given statements. Indicate your answer. Statements All the organised persons find time for rest. Sunita, inspite of her very busy schedule, finds time for rest. Conclusions I. Sunita is an organised person II. Sunita is an industrious person.
A)
Only conclusion I follows
done
clear
B)
Only conclusion II follows
done
clear
C)
Neither conclusion I nor II follows
done
clear
D)
Both conclusions I and II follow
done
clear
View Answer play_arrow
question_answer 268)
Which answer figure will complete the pattern in the question figure? Question Figures
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 269)
A piece of paper is folded and cut as shown below in the question figures. From the given figures, indicate how it will appear when opened? Question Figures
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 270)
Which of the answer figure is exactly the mirror image of the given figure, when the mirror is held on the line AB? Question Figures
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 271) Directions: Some parts of the sentence have errors and some are correct. Find out which part of a sentence has an error corresponding to the appropriate option (a), (b) and (c). If a sentence is free from errors, mark the answer (d)
A)
We requested the watchman
done
clear
B)
/ to clean up the basement
done
clear
C)
/ so that the children had enough space to play.
done
clear
D)
/No error
done
clear
View Answer play_arrow
question_answer 272) Directions: Some parts of the sentence have errors and some are correct. Find out which part of a sentence has an error corresponding to the appropriate option (a), (b) and (c). If a sentence is free from errors, mark the answer (d)
A)
Last night I dream
done
clear
B)
/I was a sheikh on the 169th floor
done
clear
C)
/ of burj khalifa
done
clear
D)
/No error
done
clear
View Answer play_arrow
question_answer 273) Directions: Out of the four alternatives, choose the one which best expresses the meaning of the given word and mark it in the answer sheet. Benevolent
A)
Beneficial
done
clear
B)
Kind
done
clear
C)
Helpful
done
clear
D)
Supportive
done
clear
View Answer play_arrow
question_answer 274) Directions: Out of the four alternatives, choose the one which best expresses the meaning of the given word and mark it in the answer sheet. Ancestors
A)
Extinct tribes
done
clear
B)
Relatives
done
clear
C)
Forefathers
done
clear
D)
Old people
done
clear
View Answer play_arrow
question_answer 275) Directions: Out of the four alternatives, choose the one which best expresses the meaning of the given word and mark it in the answer sheet. Meek
A)
Light hearted
done
clear
B)
Serious
done
clear
C)
Submissive
done
clear
D)
Benign
done
clear
View Answer play_arrow
question_answer 276) Directions: Choose the word opposite in meaning to the given word and mark it in the answer sheet. Accomplish
A)
Fail
done
clear
B)
Improper
done
clear
C)
Disagreeable
done
clear
D)
Scatter
done
clear
View Answer play_arrow
question_answer 277) Directions: Choose the word opposite in meaning to the given word and mark it in the answer sheet. Famous
A)
Obscure
done
clear
B)
Eminent
done
clear
C)
Lenient
done
clear
D)
Fabulous
done
clear
View Answer play_arrow
question_answer 278) Directions: Four alternatives are given for the idiom/phrase. Choose the alternative which best expresses the meaning of the idiom/phrase and mark it in answer sheet. To take to heart
A)
to be encouraged
done
clear
B)
to grieve over
done
clear
C)
to like
done
clear
D)
to hate
done
clear
View Answer play_arrow
question_answer 279) Directions: Four alternatives are given for the idiom/phrase. Choose the alternative which best expresses the meaning of the idiom/phrase and mark it in answer sheet. Yeomans service
A)
medical help
done
clear
B)
excellent work
done
clear
C)
social work
done
clear
D)
hard work
done
clear
View Answer play_arrow
question_answer 280) Directions: A part of the sentence is underlined which may need improvement. Alternatives are given as (a), (b) and ()c below, which may be better option. In case no improvement is need your answer is (d). John is wearing his jacket as it is very cold.
A)
taking on
done
clear
B)
getting on
done
clear
C)
putting on
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 281) Directions: A part of the sentence is underlined which may need improvement. Alternatives are given as (a), (b) and (c) below, which may be better option. In case no improvement is need your answer is (d). The host offered me tea but I denied it
A)
refused
done
clear
B)
said no
done
clear
C)
rejected
done
clear
D)
No improvement
done
clear
View Answer play_arrow
question_answer 282) Directions: Out of the four alternatives, choose the one which can be substituted for the given words/sentence. Instrument to measure atmospheric pressure
A)
metronometer
done
clear
B)
compass
done
clear
C)
pedometer
done
clear
D)
barometer
done
clear
View Answer play_arrow
question_answer 283) Directions: Out of the four alternatives, choose the one which can be substituted for the given words/sentence. Belonging to all parts of the world
A)
common
done
clear
B)
universal
done
clear
C)
worldly
done
clear
D)
international
done
clear
View Answer play_arrow
question_answer 284) Directions: In the following questions, group of four words an given. In each group, one word is correctly spelt. Find the correctly spelt word and mark it in the answer sheet. In the following questions, group of four words an given. In each group, one word is correctly spelt. Find the correctly spelt word and mark it in the answer sheet.
A)
Onvelope
done
clear
B)
Envelope
done
clear
C)
Envalope
done
clear
D)
Envelop
done
clear
View Answer play_arrow
question_answer 285) Directions: In the following questions, group of four words an given. In each group, one word is correctly spelt. Find the correctly spelt word and mark it in the answer sheet. In the following questions, group of four words an given. In each group, one word is correctly spelt. Find the correctly spelt word and mark it in the answer sheet.
A)
Comitee
done
clear
B)
Committee
done
clear
C)
Committie
done
clear
D)
Committee
done
clear
View Answer play_arrow
question_answer 286) The average of seven consecutive numbers is 20. The largest of these numbers is
A)
21
done
clear
B)
22
done
clear
C)
23
done
clear
D)
24
done
clear
View Answer play_arrow
question_answer 287) The greatest among the numbers\[\sqrt[3]{4},\sqrt{2},\sqrt[6]{3},\sqrt[4]{5}\]
A)
\[\sqrt[3]{4}\]
done
clear
B)
\[\sqrt{2}\]
done
clear
C)
\[\sqrt[6]{3}\]
done
clear
D)
\[\sqrt[4]{5}\]
done
clear
View Answer play_arrow
question_answer 288) One-fifth a number is equal to\[\frac{5}{8}\]of another number. If 35 is added to the first number, it becomes four times of the second number, The second number is
A)
25
done
clear
B)
40
done
clear
C)
70
done
clear
D)
125
done
clear
View Answer play_arrow
question_answer 289) In dividing a certain number by 342, we get 47 as remainder of the same number is divided by 18, what will be the remainder?
A)
11
done
clear
B)
13
done
clear
C)
17
done
clear
D)
19
done
clear
View Answer play_arrow
question_answer 290) The least number which when divided by 5, 6, 7 and 8 leaves a remainder 3 but when divided by 9 leaves no remainder, is
A)
1677
done
clear
B)
1683
done
clear
C)
2523
done
clear
D)
3363
done
clear
View Answer play_arrow
question_answer 291) Sakshi can do a piece of work in 20 days. Tanya is 25% more efficient than Sakshi. The number of days taken by Tanya to do the same piece of work is
A)
15
done
clear
B)
16
done
clear
C)
18
done
clear
D)
25
done
clear
View Answer play_arrow
question_answer 292) The price of a VCR is marked at Rs. 12000. If successive discounts of 15%, 10% and 5% be allowed, then at what price does a customer buy it?
A)
Rs. 8400
done
clear
B)
Rs. 8721
done
clear
C)
Rs. 8856
done
clear
D)
Rs. 8850
done
clear
View Answer play_arrow
question_answer 293) Ram bought a refrigerator with 5% discount on the labelled price. Had he bought it with 20% discount, he would have saved Rs. 1500. At what price did he buy the refrigerator?
A)
Rs. 10000
done
clear
B)
Rs. 9000
done
clear
C)
Rs. 8000
done
clear
D)
Rs. 9500
done
clear
View Answer play_arrow
question_answer 294) If \[\frac{x+y}{x-y}=\frac{5}{2},\]then value of\[\frac{x}{y}\]is
A)
\[\frac{3}{8}\]
done
clear
B)
\[\frac{8}{3}\]
done
clear
C)
\[\frac{5}{3}\]
done
clear
D)
\[\frac{3}{5}\]
done
clear
View Answer play_arrow
question_answer 295) A vendor bought toffees at 6 for a rupee. How many for a rupee must he sell to gain 20%?
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 296) If the selling price of 50 articles is equal to the cost price of 40 articles, then the loss or gain per cent is
A)
20% loss
done
clear
B)
20% gain
done
clear
C)
25% loss
done
clear
D)
25% gain
done
clear
View Answer play_arrow
question_answer 297) The ratio of the incomes of A and B is 5 : 4 and the ratio of their expenditures is 3 :2. If at the end of the year, each saves Rs. 1600, then the income of A is
A)
Rs. 3400
done
clear
B)
Rs. 3600
done
clear
C)
Rs. 4000
done
clear
D)
Rs. 4400
done
clear
View Answer play_arrow
question_answer 298) Zinc and copper are melted together in the ratio 9 : 11. What is the weight of melted mixture, if 28.8kg of zinc has been consumed in it?
A)
58 kg
done
clear
B)
60 kg
done
clear
C)
64 kg
done
clear
D)
70 kg
done
clear
View Answer play_arrow
question_answer 299) In a stream running at 2 km/h, a motorboat goes 6 km upstream and back again to the starting point in 33 min. Find the speed of the motorboat in still water.
A)
11 km/h
done
clear
B)
22 km/h
done
clear
C)
12 km/h
done
clear
D)
20 km/h
done
clear
View Answer play_arrow
question_answer 300) The difference between the compound interest and simple interest on a certain sum of money for 3yr at \[6\frac{2}{3}%\]per annum is 3 Rs. 184, then what is the sum?
A)
Rs. 13500
done
clear
B)
Rs. 12500
done
clear
C)
Rs. 11500
done
clear
D)
Rs. 10500
done
clear
View Answer play_arrow
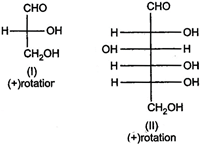
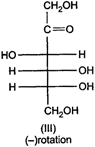
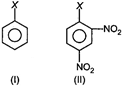 \[\underset{(\text{III})}{\mathop{{{(C{{H}_{3}})}_{3}}C-X}}\,\] \[\underset{(\text{IV})}{\mathop{{{(C{{H}_{3}})}_{3}}CH-X}}\,\]
\[\underset{(\text{III})}{\mathop{{{(C{{H}_{3}})}_{3}}C-X}}\,\] \[\underset{(\text{IV})}{\mathop{{{(C{{H}_{3}})}_{3}}CH-X}}\,\]
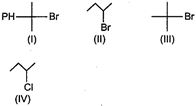
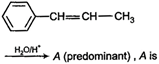
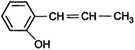
 1
1  Answer Figures
Answer Figures
 Answer Figures
Answer Figures


 Answer Figures
Answer Figures