A) six \[P-P\] single bonds
B) four \[P-P\] single bonds
C) four lone pair of electrons
D) PPP angle is \[{{60}^{o}}\]
Correct Answer: B
Solution :
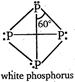 Hence, the white phosphorus has six P-P single bonds, four lone pairs of electrons and the PPP angle is \[{{60}^{o}}\].
Hence, the white phosphorus has six P-P single bonds, four lone pairs of electrons and the PPP angle is \[{{60}^{o}}\].
You need to login to perform this action.
You will be redirected in
3 sec
