question_answer 1) A body floats with one third of its volume outside water and three fourth of its volume. outside another liquid. The density of another liquid is:
A)
\[\frac{9}{4}gm/c{{m}^{3}}\]
done
clear
B)
\[4.9\text{ }gm/c{{m}^{3}}\]
done
clear
C)
\[\frac{8}{3}gm/c{{m}^{3}}\]
done
clear
D)
\[\frac{3}{8}~gm/c{{m}^{3}}\]
done
clear
View Answer play_arrow
question_answer 2) A tangential force F acts along the rim of a ring of radius R and displaces the ring through an angle \[\theta \]. The work done by the force is:
A)
\[F{{R}^{2}}\theta \]
done
clear
B)
\[\frac{FR}{\theta }\]
done
clear
C)
\[\frac{FR}{2}\theta \]
done
clear
D)
\[FR\,\theta \]
done
clear
View Answer play_arrow
question_answer 3)
For a certain mass of gas the isothermal curves between P and V at \[{{T}_{1}}\] and \[{{T}_{2}}\] temperature are 1 and 2 as shown in fig. Then:
A)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}<{{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
D)
nothing can be predicted
done
clear
View Answer play_arrow
question_answer 4) In simple harmonic motion, when the displacement is one half of the amplitude, what fraction of the total energy is kinetic :
A)
zero
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
\[\frac{3}{4}\]
done
clear
View Answer play_arrow
question_answer 5)
Two bodies M and N of equal masses are suspended from two separate massless springs of spring constants \[{{k}_{1}}\] and \[{{k}_{2}}\] respectively. If the two bodies oscillate vertically such that their maximum velocities are equal, the ratio of the amplitude of vibrations of M to that of N is :
A)
\[\frac{{{k}_{1}}}{{{k}_{2}}}\]
done
clear
B)
\[\sqrt{\frac{{{k}_{1}}}{{{k}_{2}}}}\]
done
clear
C)
\[\frac{{{k}_{2}}}{{{k}_{1}}}\]
done
clear
D)
\[\sqrt{\frac{{{k}_{2}}}{{{k}_{1}}}}\]
done
clear
View Answer play_arrow
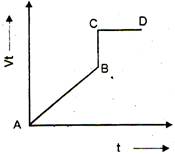
question_answer 6) The displacement time graph of a moving particle is shown below. The instantaneous velocities of the particle is negative at a point:
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
question_answer 7)
A.10 kg mass moves 3.0 m against a retarding force shown in fig. If the force is
A)
+6J
done
clear
B)
-6J
done
clear
C)
12J
done
clear
D)
-12J
done
clear
View Answer play_arrow
question_answer 8) A reference frame attached to the earth :
A)
is an inertial frame by definition
done
clear
B)
Cannot be an inertial frame because the earth is revolving around the sun
done
clear
C)
is an inertial frame because Newtons laws are applicable in this frame
done
clear
D)
Cannot be an inertial frame because the earth is rotating about its own axis
done
clear
View Answer play_arrow
question_answer 9) An iron piece fall from a height of 1 km on the ground. If all the energy is converted into heat the rise of temperature of the iron piece will be (specific heat of iron = 0.1 k cal/kg° C)
A)
\[0.233{}^\circ C\]
done
clear
B)
\[2.33{}^\circ C\]
done
clear
C)
\[23.8{}^\circ C\]
done
clear
D)
\[233{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 10) The electrical cable of copper has just one wire of radius 9 mm its resistance is 10\[\Omega \]. This single copper wire is replaced by 3 different well insulated copper wires each of radius 3 mm. The total resistance of the cable will now be equal to :
A)
90 \[\Omega \]
done
clear
B)
45 \[\Omega \]
done
clear
C)
30 \[\Omega \]
done
clear
D)
15 \[\Omega \]
done
clear
View Answer play_arrow
question_answer 11) The distance between two consecutive crests in a wave train produced in string is 5 cm. If two complete waves pass through any point per second, the velocity of wave is:
A)
2.5 cm/s
done
clear
B)
5 cm/sec
done
clear
C)
10 cm/sec
done
clear
D)
15 cm/sec
done
clear
View Answer play_arrow
question_answer 12) A charged particle moves along the axis of a current carrying selenoid. It experience :
A)
an accelerating force
done
clear
B)
a retarding force
done
clear
C)
a radial force
done
clear
D)
no force
done
clear
View Answer play_arrow
question_answer 13) A straight section \[PQ\] of a circuit lies along x- axis at \[x=-\frac{a}{2}to\,x-+\frac{a}{2}\] and carries a steady current (\[i\]), the magnetic field due to section \[PQ\] at a point \[x=+a\] will be :
A)
proportional to \[a\]
done
clear
B)
proportional to\[{{a}^{2}}\]
done
clear
C)
proportional to \[\frac{1}{a}\]
done
clear
D)
equal to zero
done
clear
View Answer play_arrow
question_answer 14) A coil has induced en-if 0.5 V produced when current is changing at the rate of 5 \[\text{A/s}\text{.}\] The self-inductance of the coil is :
A)
0.1 H
done
clear
B)
0.1 mH
done
clear
C)
10 mH
done
clear
D)
10H
done
clear
View Answer play_arrow
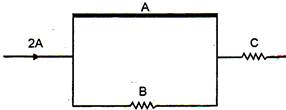
question_answer 15)
A part of circuit is shown in the given fig. The wire A is a thick copper wire and wire B and C are identical wires of manganin. The currents in wires A, B, C will be respectively :
A)
1 A, 1A, 2A
done
clear
B)
0, 2A, 2A
done
clear
C)
2A, 0, 2A
done
clear
D)
2A, 2A, 2A
done
clear
View Answer play_arrow
question_answer 16) The magnifying power of compound microscope is 32. If the magnifying power of the objective is 8, then the magnifying power of the eye peice is :
A)
4
done
clear
B)
16
done
clear
C)
24
done
clear
D)
80
done
clear
View Answer play_arrow
question_answer 17) The large aperture of objective of reflecting telescope is used to get:
A)
least spherical aberration
done
clear
B)
high resolving power
done
clear
C)
low dispersive power
done
clear
D)
high magnification
done
clear
View Answer play_arrow
question_answer 18) The focal length of the objective of a compound microscope is 2 cm and that of eye peice of microscope is 5 cm. These two lenses are separated by a distance of 25 cm. when the microscope is focused for the minimum distance of distinct vision, the magnification will be :
A)
5
done
clear
B)
30
done
clear
C)
56.5
done
clear
D)
125
done
clear
View Answer play_arrow
question_answer 19) The magnifying power of an astronomical telescope for normal vision with usual notations is :
A)
\[\frac{fo}{fe}\]
done
clear
B)
\[-fo\times fe\]
done
clear
C)
\[\frac{-fe}{fo}\]
done
clear
D)
\[-fo+fe\]
done
clear
View Answer play_arrow
question_answer 20) On making grid voltage more negative the plate resistance \[{{R}_{p}}:\]
A)
increases
done
clear
B)
decreases
done
clear
C)
does not change
done
clear
D)
changes erratically
done
clear
View Answer play_arrow
question_answer 21) If the pressure amplitude in sound wave is tripled, then the intensity of sound is increased by a factor of :
A)
3
done
clear
B)
6
done
clear
C)
9
done
clear
D)
\[\sqrt{3}\]
done
clear
View Answer play_arrow
question_answer 22) A long cylindrical copper rod carries a direct current, the magnetic field associated with the rod will be :
A)
inside the rod only
done
clear
B)
outside the rod only
done
clear
C)
both inside and outside
done
clear
D)
neither inside nor outside the rod
done
clear
View Answer play_arrow
question_answer 23) The moment of inertia of two equal masses each of mass m at seperation \[L\] connected by a rod of mass \[M\], about an axis passing through centre and perpendicular to length of rod is :
A)
\[\frac{(m+3m){{L}^{2}}}{12}\]
done
clear
B)
\[\frac{(M+6m){{L}^{2}}}{12}\]
done
clear
C)
\[\frac{M{{L}^{2}}}{4}\]
done
clear
D)
\[\frac{M{{L}^{2}}}{12}\]
done
clear
View Answer play_arrow
question_answer 24) The escape velocity from a spherical planet is \[{{u}_{es.}}\]What is the escape velocity corresponding to another planet of twice the radius and half the mean density ?
A)
\[\sqrt{2}{{u}_{es.}}\]
done
clear
B)
\[\frac{{{u}_{es}}}{\sqrt{2}}\]
done
clear
C)
\[2{{u}_{es}}\]
done
clear
D)
\[{{4}_{ue}}\]
done
clear
View Answer play_arrow
question_answer 25) The angle between the two following vectors is : \[\text{\vec{A}=3\vec{j}+4\vec{j}+5\vec{k},}\,\text{\vec{B}=3\vec{j}+4\vec{j}+5\vec{k}:}\]
A)
\[90{}^\circ \]
done
clear
B)
\[0{}^\circ \]
done
clear
C)
\[180{}^\circ \]
done
clear
D)
\[45{}^\circ \]
done
clear
View Answer play_arrow
question_answer 26) Four identical pieces of copper are painted with different types of paints. Which one would you expect to lose heat most rapidly, if these are all heated to same temperature and allowed to cool in vacuum?
A)
painted rough black
done
clear
B)
painted rough white
done
clear
C)
painted shiny white
done
clear
D)
painted shiny black
done
clear
View Answer play_arrow
question_answer 27) A wire of length \[L\] carries a current \[I.\] it is bent in the form of a circle. The magnetic moment of current loop (in amp-m ) is :
A)
\[\frac{I{{L}^{2}}}{4{{\pi }^{2}}}\]
done
clear
B)
\[I\pi {{L}^{2}}\]
done
clear
C)
\[\frac{I{{L}^{2}}}{4\pi }\]
done
clear
D)
\[4\pi I{{L}^{2}}\]
done
clear
View Answer play_arrow
question_answer 28) In an atom bomb, a temperature of about 10 million degrees is developed at the moment of explosion. In what region of the spectrum do the wavelength corresponding to maximum energy density lie if the light source is in the atom bomb?
A)
ultravoilet region
done
clear
B)
visible region
done
clear
C)
infrared region
done
clear
D)
X-ray region
done
clear
View Answer play_arrow
question_answer 29) The more rapidly fissionable isotope of curanium has an atomic mass of :
A)
234
done
clear
B)
235
done
clear
C)
236
done
clear
D)
238
done
clear
View Answer play_arrow
question_answer 30) Wave of frequency 100 Hz travels along a string towards its fixed end. When this wave travels back, after reflection a node is formed at a distance of 10 cm from the fixed end. The speed of the wave (incident and reflected) is :
A)
5 m/s
done
clear
B)
10 m/s
done
clear
C)
20 m/s
done
clear
D)
40 m/s
done
clear
View Answer play_arrow
question_answer 31) In Youngs double slit experiment, the slits are 0.5 mm apart and interference pattern is observed on a screen placed at a distance of 1m from the place containing the slits. If wavelength of the incident light is \[6000\overset{\text{o}}{\mathop{\text{A}}}\,\] then the separation between the third bright fringe and central maxima is :
A)
4mm
done
clear
B)
3.6 mm
done
clear
C)
3 mm
done
clear
D)
2.5 mm
done
clear
View Answer play_arrow
question_answer 32) The part of a transistor, which is heavily doped to produce a large number of majority carriers is :
A)
base
done
clear
B)
emitter
done
clear
C)
collector
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 33) The least count of an instrument is 0.01 cm. Taking all precautions, the most probable error in the measurement can be :
A)
0.005 cm
done
clear
B)
0.01 cm
done
clear
C)
0.05 cm
done
clear
D)
0.02 cm
done
clear
View Answer play_arrow
question_answer 34) The time period of a mass spring system having mass 2.5 kg and spring of. Force constant 10 N/m, is :
A)
\[\pi \]
done
clear
B)
2\[\pi \]
done
clear
C)
4\[\pi \]
done
clear
D)
8\[\pi \]
done
clear
View Answer play_arrow
question_answer 35) A particle of mass 50 g executes SHM with a time period of 0.18 sec. The amplitude of vibration is 10 cm, the maximum force on the particle is around :
A)
10 N
done
clear
B)
6 N
done
clear
C)
30 N
done
clear
D)
35 N
done
clear
View Answer play_arrow
question_answer 36) A Youngs double slit setup for interference is shifted from air to within water, then the fringe width :
A)
becomes infinite
done
clear
B)
decreases
done
clear
C)
increases
done
clear
D)
remains unchanged
done
clear
View Answer play_arrow
question_answer 37) The absolute temperature of a gas is increased 3 times. The root mean square velocity of the molecules will become :
A)
3 times
done
clear
B)
9 times
done
clear
C)
\[\frac{1}{3}\] times
done
clear
D)
\[\sqrt{3}\] times
done
clear
View Answer play_arrow
question_answer 38) The position \[x\] of a particle varies with time \[t\,as\,x=a{{t}^{2}}-b{{t}^{3}}.\]The acceleration of the particle will be zero at time ( equal to :
A)
\[\frac{2a}{3b}\]
done
clear
B)
\[\frac{1}{b}\]
done
clear
C)
\[\frac{a}{3b}\]
done
clear
D)
\[0\]
done
clear
View Answer play_arrow
question_answer 39) A bucket has oil of relative density 1.2 up to a height of 3 cm. On top of the oil there is a water column of height of 10 cm. If the relative density of mercury is 13.6, the pressure at the bottom of the beaker is :
A)
1 cm of Hg
done
clear
B)
5 cm of Hg
done
clear
C)
13 cm of Hg
done
clear
D)
15 cm of Hg
done
clear
View Answer play_arrow
question_answer 40) The moselay law is :
A)
\[{{u}^{2}}=z(a-b)\]
done
clear
B)
\[{{u}^{{}}}=\sqrt{a}z\]
done
clear
C)
\[u=a{{(z-a)}^{2}}\]
done
clear
D)
\[\sqrt{u}=a(z-b)\]
done
clear
View Answer play_arrow
question_answer 41) The mean distance of jupiter from the sun is nearly 5.2 times the corresponding earth-sun distance. According to Kepler? law, the period of revolution of Jupiter in its orbit will be :
A)
5 year
done
clear
B)
7.5 year
done
clear
C)
12 year
done
clear
D)
25 year
done
clear
View Answer play_arrow
question_answer 42) The equation of a transverse wave is given by \[y\] = 20 sin\[\pi \] (\[0.02x-2t\]) where \[x\] and \[y\] are in cms and \[t\] is in seconds. The wavelength in cm will be :
A)
90
done
clear
B)
100
done
clear
C)
200
done
clear
D)
5
done
clear
View Answer play_arrow
question_answer 43) A proton and an \[\alpha \] particle enter a uniform magnetic field with the same velocities. The time period of rotation of \[\alpha \] -particle will be :
A)
two times as that of proton
done
clear
B)
half as that of proton
done
clear
C)
four times as that of proton
done
clear
D)
same as that of proton
done
clear
View Answer play_arrow
question_answer 44) If 2 % of the main current is to be passed through the galvanometer of resistance G, the resistance of the shunt required is :
A)
\[\frac{G}{49}\]
done
clear
B)
\[\frac{G}{50}\]
done
clear
C)
\[49\,G\]
done
clear
D)
\[50\,G\]
done
clear
View Answer play_arrow
question_answer 45) The wire of a potentiometer has a resistance 20 and length 10 m. A battery of emf 3V is connected across the wire. The potential gradient is :
A)
0.3 V/m
done
clear
B)
0.03 V/m
done
clear
C)
3V/m
done
clear
D)
6 V/m
done
clear
View Answer play_arrow
question_answer 46) Core of transformer is made laminated to :
A)
increase permeability
done
clear
B)
increase power
done
clear
C)
increase sensitivity
done
clear
D)
avoid eddy current loss
done
clear
View Answer play_arrow
question_answer 47)
The velocity-time graph of a moving particle is shown in fig. The acceleration is maximum for part :
A)
AB
done
clear
B)
BC
done
clear
C)
CD
done
clear
D)
equal for all parts
done
clear
View Answer play_arrow
question_answer 48) The diameter of the objective of the telescope is 0.1 m and the wavelength of the light is \[6000\overset{\text{o}}{\mathop{\text{A}}}\,\]. Resolving limit of the telescope will be :
A)
\[6\times {{10}^{-5}}rad\]
done
clear
B)
\[6\times {{10}^{-4}}rad\]
done
clear
C)
\[6\times {{10}^{-3}}rad\]
done
clear
D)
\[6\times {{10}^{-6}}rad\]
done
clear
View Answer play_arrow
question_answer 49) The half-life of radium is 1620 year and its atomic weight is 226 gm/mole. The number of atoms that will decay from its 1g sample per second is :
A)
\[3.61\times {{10}^{10}}\]
done
clear
B)
\[3.6\times {{10}^{12}}\]
done
clear
C)
\[3.1\times {{10}^{15}}\]
done
clear
D)
\[31.1\times {{10}^{15}}\]
done
clear
View Answer play_arrow
question_answer 50) If the coefficient of linear expansion of a solid is 0.00009. Its coefficient of volume expansion is :
A)
0.009
done
clear
B)
0.02
done
clear
C)
0.03
done
clear
D)
0.00027
done
clear
View Answer play_arrow
question_answer 51) Which of the following is most stable among \[C{{u}^{+}},F{{e}^{+}},F{{e}^{2+}}\]and \[F{{e}^{3+}}\]?
A)
\[C{{u}^{+}}\]
done
clear
B)
\[F{{e}^{+}}\]
done
clear
C)
\[F{{e}^{2+}}\]
done
clear
D)
\[F{{e}^{3+}}\]
done
clear
View Answer play_arrow
question_answer 52) In the ground state of phosphorus total number of unpaired electrons are:
A)
\[3\]
done
clear
B)
\[5\]
done
clear
C)
\[8\]
done
clear
D)
\[9\]
done
clear
View Answer play_arrow
question_answer 53) \[H-O-H\]bond angle in \[{{H}_{2}}O\] is \[{{104.5}^{o}}\]and not \[{{109}^{o}}28\] because of:
A)
lone pair-lone pair repulsion
done
clear
B)
lone pair-bond pair repulsion
done
clear
C)
bond pair-bond pair repulsion
done
clear
D)
high electronegativity of oxygen
done
clear
View Answer play_arrow
question_answer 54) Oxidation state of \[I\] in \[{{H}_{4}}IO_{6}^{-}\] is:
A)
\[-1\]
done
clear
B)
\[+5\]
done
clear
C)
\[+6\]
done
clear
D)
\[+7\]
done
clear
View Answer play_arrow
question_answer 55) Hard water is not fit for washing clothes because:
A)
it contains impurities
done
clear
B)
it is acidic in nature
done
clear
C)
it gives precipitate with soap
done
clear
D)
it contains \[N{{a}_{2}}S{{O}_{4}}\] and \[KCl\]
done
clear
View Answer play_arrow
question_answer 56) The half life of a radioactive isotope is 2.5 our. If the initial mass of the isotope was 16 g, the mass of it remain undecayed after 10 hours will be:
A)
\[32g\]
done
clear
B)
\[4g\]
done
clear
C)
\[16g\]
done
clear
D)
\[1g\]
done
clear
View Answer play_arrow
question_answer 57) Total number of unpaired electrons in \[{{O}_{2}}\] molecule is:
A)
\[0\]
done
clear
B)
\[1\]
done
clear
C)
\[2\]
done
clear
D)
\[3\]
done
clear
View Answer play_arrow
question_answer 58) Conjugate acid of \[{{S}_{2}}{{O}_{8}}^{2-}\] is:
A)
\[{{H}_{2}}{{S}_{2}}{{O}_{8}}\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[{{H}_{2}}{{S}_{2}}O_{8}^{-}\]
done
clear
D)
\[HSO_{_{4}}^{-}\]
done
clear
View Answer play_arrow
question_answer 59) Which of the following element exhibit maximum valency?
A)
\[Cu\]
done
clear
B)
\[Os\]
done
clear
C)
\[Pt\]
done
clear
D)
\[Cs\]
done
clear
View Answer play_arrow
question_answer 60) Which of the following contains maximum ionic radius?
A)
\[{{C}^{4-}}\]
done
clear
B)
\[{{C}^{3-}}\]
done
clear
C)
\[{{O}^{2-}}\]
done
clear
D)
\[M{{g}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 61) Which of the following represents isoelectronic sequence?
A)
\[N,\,O,\,F,\,Ne\]
done
clear
B)
\[N{{a}^{+}},M{{n}^{2+}},A{{l}^{3+}},S{{i}^{4+}}\]
done
clear
C)
\[C{{l}^{-}},Ar,C{{a}^{2+}},T{{i}^{4+}}\]
done
clear
D)
\[Be,M{{g}^{2+}},Ca,S{{i}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 62) The indicator used in the titration of oxalic acid and sodium hydroxide is :
A)
phenolphthalein
done
clear
B)
methyl orange
done
clear
C)
methyl red
done
clear
D)
ammonium hydroxide
done
clear
View Answer play_arrow
question_answer 63) Nitrogen molecule is chemically less active because of the presence of....... between two nitrogen atoms:
A)
single bond
done
clear
B)
double bond
done
clear
C)
triple bond
done
clear
D)
coordinate bond
done
clear
View Answer play_arrow
question_answer 64) To dissolve argentite ore which of the following is used:
A)
\[Na[Ag{{(CN)}_{2}}]\]
done
clear
B)
\[NaCN\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[NaCl\]
done
clear
View Answer play_arrow
question_answer 65) When \[{{H}_{2}}S\] gas is passed through an acidified solution of ...... yellow precipitate is formed?
A)
Curious
done
clear
B)
\[C{{d}^{2+}}\]ions
done
clear
C)
\[N{{i}^{2+}}\]ions
done
clear
D)
Copious
done
clear
View Answer play_arrow
question_answer 66) Which of the following has same number of electrons as in \[C{{a}^{2+}}\]ion?
A)
\[C{{u}^{2+}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[Ne\]
done
clear
D)
\[M{{g}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 67) Which of the following have highest melting point?
A)
\[NaCl\]
done
clear
B)
\[NaI\]
done
clear
C)
\[NaBr\]
done
clear
D)
\[NaF\]
done
clear
View Answer play_arrow
question_answer 68) Which of the following is not an acid anhydride?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[CO\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 69) \[{{H}_{2}}S\] is more acidic than \[{{H}_{2}}O\] because:
A)
atomic number of sulphur is greater than that of oxygen
done
clear
B)
oxygen is more electronegative than sulphur
done
clear
C)
\[H-S\] bond is weaker than \[H-O\] bond
done
clear
D)
\[H-O\]bond is weaker than \[H-S\] bond
done
clear
View Answer play_arrow
question_answer 70) Which of the following halide of calcium is insoluble in water?
A)
\[CaC{{l}_{2}}\]
done
clear
B)
\[Ca{{l}_{2}}\]
done
clear
C)
\[Ca{{F}_{2}}\]
done
clear
D)
\[CaB{{r}_{2}}\]
done
clear
View Answer play_arrow
question_answer 71) Which of the following sulphur containing species can not act as oxidising agent?
A)
\[{{H}_{2}}S\]
done
clear
B)
\[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}S{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 72) Compared to the mass of lightest nucleus mass an electron is only:
A)
\[1/80\]
done
clear
B)
\[1/1800\]
done
clear
C)
\[1/800\]
done
clear
D)
\[1/1000\]
done
clear
View Answer play_arrow
question_answer 73) Decreasing order of s-character is \[sp,s{{p}^{2}},s{{p}^{3}}\]-hybrid orbitals:
A)
\[s{{p}^{3}}>s{{p}^{2}}>sp\]
done
clear
B)
\[sp>s{{p}^{2}}>s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}>sp>s{{p}^{2}}\]
done
clear
D)
\[s{{p}^{2}}>sp>s{{p}^{3}}\]
done
clear
View Answer play_arrow
question_answer 74) Silver mirror test is given by:
A)
propanone
done
clear
B)
propane
done
clear
C)
propanal
done
clear
D)
propanol
done
clear
View Answer play_arrow
question_answer 75) Total number of isomers for \[{{C}_{8}}{{H}_{8}}{{O}_{2}}\] will be:
A)
\[2\]
done
clear
B)
\[3\]
done
clear
C)
\[4\]
done
clear
D)
\[5\]
done
clear
View Answer play_arrow
question_answer 76) Aniline on heating with solid \[KOH\] and \[C{{S}_{2}}\] produced:
A)
thiourea
done
clear
B)
phenol
done
clear
C)
S-diphenyl thiourea
done
clear
D)
Schiffs base
done
clear
View Answer play_arrow
question_answer 77) Common name of phosgene is:
A)
carbon tetrachloride
done
clear
B)
phosphoryl chloride
done
clear
C)
carbonyl chloride
done
clear
D)
phosphorus tri chloride
done
clear
View Answer play_arrow
question_answer 78) Which of the following is the strongest acid:
A)
formic acid
done
clear
B)
acetic acid
done
clear
C)
trichloroacetic acid
done
clear
D)
trifluoroacefic acid
done
clear
View Answer play_arrow
question_answer 79) Identify (Y) in the series: \[CH\equiv CH\xrightarrow{{{O}_{3}}/NaOH}(X)\xrightarrow{Zn/C{{H}_{3}}COOH}(Y)\]
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
D)
\[C{{H}_{2}}OH-C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 80) Which of the following can be sulphonated easily?
A)
Benzene
done
clear
B)
Toluene
done
clear
C)
Nitrobenzene
done
clear
D)
Chlorobenzene
done
clear
View Answer play_arrow
question_answer 81) Benzene diazonium chloride on treatment with hypo phosphorus acid and water in presence of \[C{{u}^{+}}\] catalyst produces :
A)
benzene
done
clear
B)
toluene
done
clear
C)
aniline
done
clear
D)
chlorobenzene
done
clear
View Answer play_arrow
question_answer 82) Butene-1 is isomeric with 2-methyl propene. They can be distinguished by:
A)
\[B{{r}_{2}}\] solution
done
clear
B)
Tollens reagent
done
clear
C)
Baeyers reagent
done
clear
D)
\[{{O}_{3}},{{H}_{2}}O/Zn\]
done
clear
View Answer play_arrow
question_answer 83) Ethyl alcohol exhibits acidic nature on reaction with:
A)
acidic \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]
done
clear
B)
sodium metal
done
clear
C)
\[HCl\]
done
clear
D)
\[C{{H}_{3}}COOH\]
done
clear
View Answer play_arrow
question_answer 84) Which of the following cannot be nitrated directly?
A)
aniline
done
clear
B)
phenol
done
clear
C)
toluene
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 85) Adipic acid on heating form:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 86) The ratio of carbon, hydrogen and oxygen in 2-methyl benzoic acid is:
A)
\[4:2:2\]
done
clear
B)
\[2:4:1\]
done
clear
C)
\[4:4:1\]
done
clear
D)
\[4:4:2\]
done
clear
View Answer play_arrow
question_answer 87) Beckmann thermometer is used for measuring:
A)
boiling point
done
clear
B)
freezing point
done
clear
C)
both a and b
done
clear
D)
elevation in BP or depression in FP
done
clear
View Answer play_arrow
question_answer 88) An equilibrium mixture of the reaction \[2N{{O}_{(g)}}+{{O}_{2(g)}}\] \[2N{{O}_{2}}_{(g)}\] contains \[0.12moles\]of\[N{{O}_{2}}\], \[0.08moles\]of \[NO\]and \[0.64\text{ }moles\]of \[{{O}_{2}}\] in a 4 litre flask at constant temperature. The value of \[{{K}_{c}}\] this temperature is:
A)
\[7\]
done
clear
B)
\[14\]
done
clear
C)
\[28\]
done
clear
D)
\[24\]
done
clear
View Answer play_arrow
question_answer 89) A solution of \[NaCl\] in contact with atmosphere has pH of about:
A)
\[3.2\]
done
clear
B)
\[5.4\]
done
clear
C)
\[7\]
done
clear
D)
\[14\]
done
clear
View Answer play_arrow
question_answer 90) A \[0.01M\]ammonia solution is 5% ionised. , Its pH will be:
A)
\[11.80\]
done
clear
B)
\[10.69\]
done
clear
C)
\[7.22\]
done
clear
D)
\[12.24\]
done
clear
View Answer play_arrow
question_answer 91) Equal volumes of \[0.02M\,CaC{{l}_{2}}\]and \[0.0004M\] \[N{{a}_{2}}S{{O}_{4}}\]are mixed. If \[{{K}_{sp}}\] for\[CaS{{O}_{4}}=2.4\times {{10}^{-5}}\], whether there would be any precipitation?
A)
Yes
done
clear
B)
No
done
clear
C)
Unpredictable
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 92) If the vapour pressure of pure solvent is \[{{P}_{0}}\] and the vapour pressure of dilute solution of non-volatile solute is P, then the lowering in vapour pressure is:
A)
\[+ve\]
done
clear
B)
\[-ve\]
done
clear
C)
\[P/{{P}_{0}}\]
done
clear
D)
\[{{P}_{0}}/P\]
done
clear
View Answer play_arrow
question_answer 93) If \[\Delta H\] is the change in enthalpy and \[\Delta E\] is the change in internal energy, then:
A)
\[\Delta H<\Delta E\], only if the number of moles of gaseous products is greater than the number of moles of gaseous reactants
done
clear
B)
\[\Delta H<\Delta E\], only if the number of moles of gaseous products is less than the number of moles of gaseous reactants
done
clear
C)
\[\Delta H\] is always greater than \[\Delta E\]
done
clear
D)
\[\Delta H\] is always less than \[\Delta E\]
done
clear
View Answer play_arrow
question_answer 94) If the concentration of \[M{{g}^{2+}}\] is \[0.01M\], the electrode potential of \[M{{g}^{2+}}/Mg\]electrode will be (Given that \[{{E}^{o}}M{{g}^{2+}},\,Mg=-2.36V\]):
A)
\[+2.80\]
done
clear
B)
\[-2.80\]
done
clear
C)
\[4-2.41\]
done
clear
D)
\[-2.41\]
done
clear
View Answer play_arrow
question_answer 95) The rate of a chemical reaction doubles for every \[{{10}^{o}}C\]increase in temperature, if the temperature of reaction is-increased from \[{{30}^{o}}C\]to \[{{80}^{o}}C\]. The rate of reaction increases:
A)
\[16\]
done
clear
B)
\[32\]
done
clear
C)
\[64\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 96) A chemical system is in equilibrium. The addition of a catalyst would result is:
A)
increase the amount of heat evolved in the reaction
done
clear
B)
a new reaction pathway to reaction
done
clear
C)
increase in the rate of reverse reaction
done
clear
D)
increase in the rate of forward reaction
done
clear
View Answer play_arrow
question_answer 97) Number of moles occupied by \[22.4\text{ }litres\]of \[He\] gas at \[{{30}^{o}}C\]and one atmospheric pressure are:
A)
\[0.20\]
done
clear
B)
\[1.46\]
done
clear
C)
\[0.90\]
done
clear
D)
\[2.62\]
done
clear
View Answer play_arrow
question_answer 98) What maximum amount of \[BaS{{O}_{4}}\] get precipitated if \[0.5mole\]of \[BaC{{l}_{2}}\] is mixed with \[1\text{ }mole\]of \[{{H}_{2}}S{{O}_{4}}\]?
A)
\[0.5\text{ }mol\]
done
clear
B)
\[1.0\text{ }mol\]
done
clear
C)
\[1.5\text{ }mol\]
done
clear
D)
\[2.0\text{ }mol\]
done
clear
View Answer play_arrow
question_answer 99) Sodium metal crystallises at room temperature in a body centered cubic lattice with a cell edge \[a=4.29\overset{\text{o}}{\mathop{\text{A}}}\,\]. The radius of sodium atom is:
A)
\[1.40\]
done
clear
B)
\[2.65\]
done
clear
C)
\[1.85\]
done
clear
D)
\[2.15\]
done
clear
View Answer play_arrow
question_answer 100) The half life of a radioactive element is \[3.8\] days. The time at the end of which 1/20th of the element will remain undecayed will be:
A)
\[36\text{ }day\]
done
clear
B)
\[12.86\text{ }day\]
done
clear
C)
\[24.20\text{ }day\]
done
clear
D)
\[16.40\]
done
clear
View Answer play_arrow
question_answer 101) Which of the following is a retrovirus?
A)
TMV
done
clear
B)
HIV
done
clear
C)
\[{{T}_{2}}-\]phage
done
clear
D)
\[{{T}_{4}}-\]phage
done
clear
View Answer play_arrow
question_answer 102) PCR can be used for :
A)
DNA amplification
done
clear
B)
DNA cloning
done
clear
C)
Point mutation-in DNA
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 103) On the basis of Human Genome Project report; human genome contains :
A)
30,000 genes
done
clear
B)
1,00,000 genes
done
clear
C)
60,000 genes
done
clear
D)
5,000 genes
done
clear
View Answer play_arrow
question_answer 104) YACs means :
A)
Yeast allanine complexes
done
clear
B)
Yeast artificial chromosomes
done
clear
C)
Yeast articulated computer
done
clear
D)
Yeast amplified syndrome
done
clear
View Answer play_arrow
question_answer 105) The DNA of Ti-plasmid which is obtained from Agrobacterium tumefaciens is used in genetic engineering this DNA is also called :
A)
A-DNA
done
clear
B)
B-DNA
done
clear
C)
Z-DNA
done
clear
D)
T-DNA
done
clear
View Answer play_arrow
question_answer 106) PCR is:
A)
polymerase chain reaction
done
clear
B)
polycarboxylic reaction
done
clear
C)
Polynucleotide carbon reagent
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 107) DNA sequencing can be achieved by which of the following method ?
A)
Dideoxynucleotide chain termination method of Sanger
done
clear
B)
Chemical degradation method of Maxum and Gilbert
done
clear
C)
Western blotting method
done
clear
D)
Both (a) and (b) both
done
clear
View Answer play_arrow
question_answer 108) Ribosomal RNA of eukaryotic cell is transcribed in the nucleolus with the help of enzyme :
A)
RNA polymerase-I
done
clear
B)
RNA polymerase-II
done
clear
C)
RNA polymerase-III
done
clear
D)
DNA polymerase
done
clear
View Answer play_arrow
question_answer 109) Bt-cotton is a transgenic variety which is resistant to herbicide and pesticides due to presence of protoxin protein the genes of this toxic protein has taken from :
A)
Bacillus thuringinesis
done
clear
B)
Bacillus thennophillus
done
clear
C)
Bacillus thermophillus
done
clear
D)
Therinophillus aquaticus
done
clear
View Answer play_arrow
question_answer 110) During transcription RNA polymerase enzyme binds at :
A)
reulator genes
done
clear
B)
promoter genes
done
clear
C)
operator genes
done
clear
D)
structural genes
done
clear
View Answer play_arrow
question_answer 111) Jntrpns and exons are present in the genome of:
A)
bacteria
done
clear
B)
mycoplasma
done
clear
C)
yeast
done
clear
D)
blue-green algae
done
clear
View Answer play_arrow
question_answer 112) The helix of z-DNA is :
A)
right handed
done
clear
B)
left handed
done
clear
C)
parallel
done
clear
D)
single stranded
done
clear
View Answer play_arrow
question_answer 113) Chipko Movement is worlds most known eco development programme started by Sunder Lal Bahuguna (1973). It is related with:
A)
conservation of forests
done
clear
B)
afforestation
done
clear
C)
planting
done
clear
D)
population
done
clear
View Answer play_arrow
question_answer 114) Select the psychedelic drug :
A)
LSD
done
clear
B)
marijuana
done
clear
C)
bhang
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 115) Kranz anatomy is found in :
A)
leaves of \[{{C}_{3}}\] plants
done
clear
B)
leaves of \[{{C}_{4}}\]plants
done
clear
C)
stems of \[{{C}_{3}}\]plants
done
clear
D)
stems of \[{{C}_{3}}\]plants
done
clear
View Answer play_arrow
question_answer 116) \[{{C}_{40}}{{H}_{56}}\] is the empirical formula of :
A)
carotene
done
clear
B)
chlorophyll-b
done
clear
C)
xanthophyll
done
clear
D)
anthocyanin
done
clear
View Answer play_arrow
question_answer 117) The carbondioxide acceptor in \[{{C}_{3}}\]plants is :
A)
ribulose 1, 5-diphosphate (RuBP)
done
clear
B)
phosphoenol pyruvate (PEP)
done
clear
C)
phosphoglyceric acid(PGA)
done
clear
D)
ribulose monophosphate (RuMP)
done
clear
View Answer play_arrow
question_answer 118) Blood pressure is measured by :
A)
electro-cardiogram
done
clear
B)
stethoscope
done
clear
C)
phono-cardiogram
done
clear
D)
sphygmomanometer
done
clear
View Answer play_arrow
question_answer 119) Anaemia is due to deficiency of :
A)
Mg
done
clear
B)
Ca
done
clear
C)
Fe
done
clear
D)
P
done
clear
View Answer play_arrow
question_answer 120) Which one is the most infectious disease ?
A)
AIDS
done
clear
B)
Hepatitis-B
done
clear
C)
Malaria
done
clear
D)
Cough and cold
done
clear
View Answer play_arrow
question_answer 121) Which of the following is a bacterial disease ?
A)
Leprosy
done
clear
B)
Diabetes
done
clear
C)
Scurvy
done
clear
D)
Measles
done
clear
View Answer play_arrow
question_answer 122) Population whose members reproduce sexually are termed as :
A)
panimictic
done
clear
B)
Ecotype
done
clear
C)
apomitic
done
clear
D)
amphimictic
done
clear
View Answer play_arrow
question_answer 123) Genetically adapted population to a particular habitat is :
A)
Ecotone
done
clear
B)
Ecotype
done
clear
C)
Ecosphere
done
clear
D)
Ecology
done
clear
View Answer play_arrow
question_answer 124) Niche of a species in an ecosystem refers to :
A)
centre of origin
done
clear
B)
competitive ability
done
clear
C)
place of its occurrence
done
clear
D)
function at its place of occurrence
done
clear
View Answer play_arrow
question_answer 125) Zonula adherens is another name for :
A)
desmosome
done
clear
B)
mesosomes
done
clear
C)
gap junction
done
clear
D)
terminal bars
done
clear
View Answer play_arrow
question_answer 126) Who gave the electron microscopic structure of chloroplast ?
A)
Mayer
done
clear
B)
Menke
done
clear
C)
Michaelis
done
clear
D)
Meves
done
clear
View Answer play_arrow
question_answer 127) Respiratory quotient of mixed diet is :
A)
0.25
done
clear
B)
0.85
done
clear
C)
0.7
done
clear
D)
1.5
done
clear
View Answer play_arrow
question_answer 128) Measurement of rate of \[{{O}_{2}}\] utilisation by a unit volume of water over a period of time is to measure :
A)
fermentation
done
clear
B)
bio-gas generation
done
clear
C)
bio-synthetic pathway
done
clear
D)
biological oxygen demand
done
clear
View Answer play_arrow
question_answer 129) Increasing skin cancer and high mutation nate are due to :
A)
Co-pollution
done
clear
B)
\[C{{O}_{2}}\]pollution
done
clear
C)
Ozone depletion
done
clear
D)
Acid rain
done
clear
View Answer play_arrow
question_answer 130) Chemical fertilizers pollute :
A)
water resources
done
clear
B)
soil resources
done
clear
C)
both water and soil resources
done
clear
D)
neither water nor soil resources
done
clear
View Answer play_arrow
question_answer 131) Which one is present in the roots of legumes ?
A)
Rhizobium
done
clear
B)
Nostoc
done
clear
C)
Anabaena
done
clear
D)
Azospirillum
done
clear
View Answer play_arrow
question_answer 132) A green manure is :
A)
Sesbania
done
clear
B)
Wheat
done
clear
C)
Maize
done
clear
D)
Rice
done
clear
View Answer play_arrow
question_answer 133) An \[R{{h}^{-}}\] in dividual receives \[R{{h}^{+}}\] blood. The recipient becomes :
A)
sterile
done
clear
B)
dead
done
clear
C)
no reaction
done
clear
D)
iso-immunised
done
clear
View Answer play_arrow
question_answer 134) ADH controls water permeability of :
A)
collecting tube
done
clear
B)
proximal convoluted tubules
done
clear
C)
distal convoluted tubules
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 135) Part of body having a single pair of bones :
A)
vertebral column
done
clear
B)
pelvic girdle
done
clear
C)
femur
done
clear
D)
pectoral girdle
done
clear
View Answer play_arrow
question_answer 136) A dihybrid ratio of 1 : 4 : 6 : 4 :1 is obtained instead of 9 :3 :3 :1. This is an example of:
A)
supplementary genes
done
clear
B)
complementary genes
done
clear
C)
polygenic inheritance
done
clear
D)
pleiotropic genes
done
clear
View Answer play_arrow
question_answer 137) A pleiotropic gene is one which :
A)
affects more than one character
done
clear
B)
affects one character
done
clear
C)
supplements the effect of another gene
done
clear
D)
requires another gene for expression
done
clear
View Answer play_arrow
question_answer 138) In which of the following is 2000 times sweeter than sucrose ?
A)
Fructose
done
clear
B)
Glucose
done
clear
C)
Monellin
done
clear
D)
Auspartane
done
clear
View Answer play_arrow
question_answer 139) The basic unit of chitin is :
A)
N-acetyl glucosamine
done
clear
B)
glucose
done
clear
C)
galactose
done
clear
D)
fructose
done
clear
View Answer play_arrow
question_answer 140) Enzymes are absent in :
A)
algae
done
clear
B)
plants
done
clear
C)
virus
done
clear
D)
bacteria
done
clear
View Answer play_arrow
question_answer 141) Vinegar is prepared from alcohol with the help of :
A)
Azotobacter
done
clear
B)
Lactobacillus
done
clear
C)
Rhizobhium
done
clear
D)
Acetobacter
done
clear
View Answer play_arrow
question_answer 142) Fermentation of sugar to yield alcohol is carried out by :
A)
zymase
done
clear
B)
micro-organisms
done
clear
C)
decomposition of sugar
done
clear
D)
raised temperature
done
clear
View Answer play_arrow
question_answer 143) Interferon is a glycoprotein which is an :
A)
anti-bacterial
done
clear
B)
anti-viral
done
clear
C)
bacterium
done
clear
D)
anti-animal cell
done
clear
View Answer play_arrow
question_answer 144) Bio-gas contains :
A)
\[C{{H}_{4}},\text{ }S{{O}_{2}},\text{ }{{H}_{2}}\]
done
clear
B)
\[C{{O}_{2}},\text{ }{{C}_{2}}{{H}_{5}}OH,\text{ }{{H}_{2}}\]
done
clear
C)
\[C{{H}_{4}},\text{ }C{{O}_{2}},\text{ }{{H}_{2}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}CO,\text{ }{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 145) Petroleum can be replaced by :
A)
propanol
done
clear
B)
ethanol
done
clear
C)
lactic acid
done
clear
D)
acetic acid
done
clear
View Answer play_arrow
question_answer 146) Fuel wood required per person per day is :
A)
30 Kg
done
clear
B)
3 Kg
done
clear
C)
4 Kg
done
clear
D)
5 Kg
done
clear
View Answer play_arrow
question_answer 147) Gasohal contains :
A)
10-15% alcohol
done
clear
B)
20-25% alcohol
done
clear
C)
30-35% alcohol
done
clear
D)
70-80% alcohol
done
clear
View Answer play_arrow
question_answer 148) Haemophilic man marries a carrier women percentage of daughters becoming haemophilic shall be :
A)
0%
done
clear
B)
50%
done
clear
C)
75%
done
clear
D)
100%
done
clear
View Answer play_arrow
question_answer 149) Which genotype will indicate colour blindness in male ?
A)
\[{{X}^{c}}Y\]
done
clear
B)
\[{{X}^{c}}\]
done
clear
C)
\[{{X}^{c}}{{Y}^{c}}\]
done
clear
D)
\[X{{Y}^{c}}\]
done
clear
View Answer play_arrow
question_answer 150) The prothallus of Pteris is mostly :
A)
dioecious
done
clear
B)
monoecious
done
clear
C)
Synoecious
done
clear
D)
protogynous
done
clear
View Answer play_arrow
question_answer 151) A tree fern is :
A)
Adiantum
done
clear
B)
Dryopteris
done
clear
C)
Cyathaea
done
clear
D)
Asplenium
done
clear
View Answer play_arrow
question_answer 152) Maiden hair fern is :
A)
Pteris
done
clear
B)
Dryopteris
done
clear
C)
Adiantum
done
clear
D)
Masilea
done
clear
View Answer play_arrow
question_answer 153) Common between bryophytes and pteridophytes is:
A)
absence of sporophyte
done
clear
B)
gametophyte dependent on sporophyte
done
clear
C)
sporophyte dependent on gametophyte
done
clear
D)
multicellular sex organs with sterile jacket
done
clear
View Answer play_arrow
question_answer 154) In mammals the lower jaw is made tip of :
A)
maxilla
done
clear
B)
dentary
done
clear
C)
mandible
done
clear
D)
ethmoid
done
clear
View Answer play_arrow
question_answer 155) The potential, difference across the membrane of nerve fibre when it does not show any physiological activity is called resting potential. It is about :
A)
- 60 mv
done
clear
B)
- 80 mv
done
clear
C)
+ 60 mv
done
clear
D)
+ 90 mv
done
clear
View Answer play_arrow
question_answer 156) Turners syndrome is represented by :
A)
XYY
done
clear
B)
XO
done
clear
C)
XXX
done
clear
D)
XXY
done
clear
View Answer play_arrow
question_answer 157) Number of barr bodies in XXXX female would be:
A)
4
done
clear
B)
3
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 158) Find out the mismatch :
A)
Klinefelters syndrome-XO
done
clear
B)
Haemophilia-sex-linked
done
clear
C)
Downs syndrome autosomal aneuploidy
done
clear
D)
Turners syndrome-females with retarded sexual development
done
clear
View Answer play_arrow
question_answer 159) Traits controlled by genes present on X-chromosomes are called :
A)
sex-influenced
done
clear
B)
sex-linked
done
clear
C)
sex-limited
done
clear
D)
autosomal
done
clear
View Answer play_arrow
question_answer 160) Human female reaches menopause at the age of about :
A)
25 years
done
clear
B)
35 years
done
clear
C)
50 years
done
clear
D)
70 years
done
clear
View Answer play_arrow
question_answer 161) Correct sequence of hormone secretion from beginning of menstruation is :
A)
FSH, progesterone, estrogen
done
clear
B)
estrogen, FSH, progesterone
done
clear
C)
FSH, estrogen, progesterone
done
clear
D)
estrogen, progesterone, FSH
done
clear
View Answer play_arrow
question_answer 162) Homologous organs are :
A)
wings of Butter fly, flying fish and bird
done
clear
B)
paddle of Whale, front legs of Horse and arms of humans
done
clear
C)
legs of Duck, hind limbs of Pig, hind limbs of Kangaroo
done
clear
D)
tails of Bird, Monkey and Scorpion
done
clear
View Answer play_arrow
question_answer 163) Mammals are believed to have evolved from reptiles in :
A)
Triassic
done
clear
B)
Jurassic
done
clear
C)
Permian
done
clear
D)
Cretaceous
done
clear
View Answer play_arrow
question_answer 164) Origin of first mammals occured :
A)
500 million years
done
clear
B)
220 million years
done
clear
C)
1600 million years
done
clear
D)
over 1600 million years ago
done
clear
View Answer play_arrow
question_answer 165) Female gametophyte of angiosperms is represented by :
A)
embryo sac
done
clear
B)
ovule
done
clear
C)
nucellus
done
clear
D)
megaspore mother cell
done
clear
View Answer play_arrow
question_answer 166) The tissue which attaches the ovules inside the ovary is :
A)
placenta
done
clear
B)
chalaza
done
clear
C)
funicle
done
clear
D)
hilum
done
clear
View Answer play_arrow
question_answer 167) The point of attachment of funicle with the body of ovule is :
A)
nucellus
done
clear
B)
chalaza
done
clear
C)
micropyle
done
clear
D)
hilum
done
clear
View Answer play_arrow
question_answer 168) The female gametophyte of a typical dicot (Polygonum) at the time of fertilization is :
A)
8 nucleated and 8 called
done
clear
B)
4 nucleated and 4 celled
done
clear
C)
7 nucleated 8 celled
done
clear
D)
8 nucleated and 7 celled
done
clear
View Answer play_arrow
question_answer 169) End product of anaerobic respiration is :
A)
pyruvic acid
done
clear
B)
lactic acid
done
clear
C)
phosphoglyceric acid
done
clear
D)
glyceraldehyde phosphate
done
clear
View Answer play_arrow
question_answer 170) Number of wate molecules produced per molecule of glucose during aerobic respiration is:
A)
12
done
clear
B)
8
done
clear
C)
6
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 171) Alcoholic fermentation is carried out by :
A)
Saccharomyces
done
clear
B)
Lactobacillus
done
clear
C)
Clostrdium
done
clear
D)
Asprgillus
done
clear
View Answer play_arrow
question_answer 172) Anaerobic respiration of yeast produces :
A)
\[{{N}_{2}}\]
done
clear
B)
\[{{C}_{2}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 173) Dee-geo-woo-gen reported in Taiwan is a dwarf gene of :
A)
Sugarcane
done
clear
B)
Sugar beet
done
clear
C)
Wheat
done
clear
D)
Rice
done
clear
View Answer play_arrow
question_answer 174) The father of green revolution in India is :
A)
M.S. Swaminathan
done
clear
B)
B.P. Pal
done
clear
C)
P. Maheswari
done
clear
D)
Y.P.S. Bajaj
done
clear
View Answer play_arrow
question_answer 175) Prehistoric man was :
A)
food producer
done
clear
B)
food gatherer
done
clear
C)
food exporter
done
clear
D)
food importer
done
clear
View Answer play_arrow
question_answer 176) Thalidomide intake by a pregnant lady leads to one of the following abnormalities in the foetus:
A)
vaginal carcinoma
done
clear
B)
microcephaly
done
clear
C)
phocomedia
done
clear
D)
virilism
done
clear
View Answer play_arrow
question_answer 177) Which of the following is least effective in photosynthesis?
A)
Red light
done
clear
B)
Sun light
done
clear
C)
Blue light
done
clear
D)
Green light
done
clear
View Answer play_arrow
question_answer 178) The isotope of carbon used extensively for studies in photosynthesis is :
A)
\[^{13}C\]
done
clear
B)
\[^{14}C\]
done
clear
C)
\[^{15}C\]
done
clear
D)
\[^{16}C\]
done
clear
View Answer play_arrow
question_answer 179) The first step in photosynthesis is the :
A)
ionization of water
done
clear
B)
formation of ATP
done
clear
C)
excitation of chlorophyll
done
clear
D)
attachment of \[C{{O}_{2}}\] to carbon sugar
done
clear
View Answer play_arrow
question_answer 180) Ovule is straight with funiculus, embryo sac chalaza and micropryle lying on one straight line. It is :
A)
anatropous
done
clear
B)
orthotropous
done
clear
C)
amphitropous
done
clear
D)
campylotropous
done
clear
View Answer play_arrow
question_answer 181) The archesporium of ovule is :
A)
single celled lateral
done
clear
B)
single celled hypodermal
done
clear
C)
single celled central
done
clear
D)
single celled terminal
done
clear
View Answer play_arrow
question_answer 182) Anthesis means :
A)
dehiscence of anthers
done
clear
B)
opening of floral buds
done
clear
C)
emergence of anthers
done
clear
D)
growth of pollen tube inside the carpel.
done
clear
View Answer play_arrow
question_answer 183) For energy earth is :
A)
closed system
done
clear
B)
open as well as closed system
done
clear
C)
open system
done
clear
D)
neither open nor closed system
done
clear
View Answer play_arrow
question_answer 184) Pearls of valuable jewellaly are of from :
A)
Pinctada
done
clear
B)
Nacetihis
done
clear
C)
Octopus
done
clear
D)
Water mussel
done
clear
View Answer play_arrow
question_answer 185) The chemical which causes damage to WBC, bone marrow, spleen, lymph nodes and lungs is:
A)
iodine-131
done
clear
B)
strontium-90
done
clear
C)
iodine-127
done
clear
D)
calcium
done
clear
View Answer play_arrow
question_answer 186) Pithecanthropus was discovered in :
A)
Java
done
clear
B)
China
done
clear
C)
Africa
done
clear
D)
Germany
done
clear
View Answer play_arrow
question_answer 187) Fir was used for cooking and protection first by :
A)
Cro-magnon man
done
clear
B)
Peking man
done
clear
C)
Neanderthal man
done
clear
D)
Modern man
done
clear
View Answer play_arrow
question_answer 188) Bipedal locomotion is advantageous becuause it:
A)
reduces body weight
done
clear
B)
increases weight
done
clear
C)
releases for limbs for other purposes
done
clear
D)
increases speed
done
clear
View Answer play_arrow
question_answer 189) Heart pumps blood more force fully in order persons than younger ones due to :
A)
increase in elasticity of arteries
done
clear
B)
deccrease in \[{{O}_{2}}\] content of blood
done
clear
C)
decrease in eleasticity of arteries
done
clear
D)
fall in nutrient content of blood
done
clear
View Answer play_arrow
question_answer 190) Blood circulation in humans is :
A)
open
done
clear
B)
closed
done
clear
C)
semi-closed
done
clear
D)
both (a) and (b)
done
clear
View Answer play_arrow
question_answer 191) Study of protozoans is called :
A)
Parazoology
done
clear
B)
Protozoology
done
clear
C)
Zoology
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 192) Select an anadromous fish :
A)
Hippocampus
done
clear
B)
Petromyzon
done
clear
C)
Exocoetus
done
clear
D)
Anabas
done
clear
View Answer play_arrow
question_answer 193) Which sound producing organ is found in birds ?
A)
Pharynx
done
clear
B)
Larynx
done
clear
C)
Syrinx
done
clear
D)
Trachea
done
clear
View Answer play_arrow
question_answer 194) Treatment of cancer by X-rays or gamma rays is:
A)
radio-therapy
done
clear
B)
chemo-therapy
done
clear
C)
hormone-therapy
done
clear
D)
immuno-therapy
done
clear
View Answer play_arrow
question_answer 195) Leprosy is also called :
A)
Hansens disease
done
clear
B)
Infantile paralysis
done
clear
C)
Kochs disease
done
clear
D)
Pertussis
done
clear
View Answer play_arrow
question_answer 196) Which pollution causes jaundice ?
A)
Air
done
clear
B)
Water
done
clear
C)
Land
done
clear
D)
Thermal
done
clear
View Answer play_arrow
question_answer 197) Bio-degradable pollutant is :
A)
Sewage
done
clear
B)
Mercury
done
clear
C)
Plastic
done
clear
D)
Asbestos
done
clear
View Answer play_arrow
question_answer 198) Disease caused by eating fish found in water contaminated with industrial waste having mercury is :
A)
Brights disease
done
clear
B)
Minimata disease
done
clear
C)
Osteosclerosis
done
clear
D)
Hashimotos disease
done
clear
View Answer play_arrow
question_answer 199) The longest phase in meiotic division is :
A)
prophase-I
done
clear
B)
metaphase-I
done
clear
C)
anaphase-I
done
clear
D)
telophase-I
done
clear
View Answer play_arrow
question_answer 200) Two kingdom system of classification was given by :
A)
Linnaeus
done
clear
B)
John Ray
done
clear
C)
Copeland
done
clear
D)
Whittaker
done
clear
View Answer play_arrow
question_answer 201) Binomial nomenclature was mentioned for the first time in book :
A)
Systema Nature
done
clear
B)
Philosophic Zoologique
done
clear
C)
Historia Plantarum
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 202) Oxidative phosphorylation occurs during :
A)
respiration
done
clear
B)
protein synthesis
done
clear
C)
\[{{C}^{2}}\] photosynthesis
done
clear
D)
lipid synthesis
done
clear
View Answer play_arrow
question_answer 203) Iron porphyrin occurs in :
A)
phytochrome
done
clear
B)
FAD
done
clear
C)
cytochrome
done
clear
D)
anthocyanin
done
clear
View Answer play_arrow
question_answer 204) Cytochrome is :
A)
metauoflavo protein
done
clear
B)
lipid
done
clear
C)
Fe-containg porphyrin pigment
done
clear
D)
glycoprotein
done
clear
View Answer play_arrow
question_answer 205) For their functioning, cytochromes possess:
A)
Mn
done
clear
B)
Mg
done
clear
C)
Fe
done
clear
D)
Na
done
clear
View Answer play_arrow
question_answer 206) At high altitude, RBCs of human blood will :
A)
increase in number
done
clear
B)
decrease in number
done
clear
C)
decrease in size
done
clear
D)
increase in size
done
clear
View Answer play_arrow
question_answer 207) Compound double in water which does not impede oxygen transporation is :
A)
\[S{{O}_{2}}\]
done
clear
B)
\[S{{O}_{3}}\]
done
clear
C)
CO
done
clear
D)
NO
done
clear
View Answer play_arrow
question_answer 208) Which of the following has least urea ?
A)
Pulmonary vein
done
clear
B)
Hepatic vein
done
clear
C)
Hepatic portal vein
done
clear
D)
Renal vein
done
clear
View Answer play_arrow
question_answer 209) Bio-fertilizers include :
A)
nitrogen fixing cyanobacteria
done
clear
B)
nitrogen fixing bacteria
done
clear
C)
cyanobacteria, bacteria and mycorrhizal fungi
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 210) VAM is:
A)
ecto-mycorrhiza
done
clear
B)
endo-mycorrhiza
done
clear
C)
bio-herbicide
done
clear
D)
bio-insecticide
done
clear
View Answer play_arrow
question_answer 211) Mycorrhiza is a symbiotic association between :
A)
fungi and roots of higher plants
done
clear
B)
algae and fungi
done
clear
C)
fungi and bacteria
done
clear
D)
blue-green algae and roots of higher plants
done
clear
View Answer play_arrow
question_answer 212) Genetics deals with :
A)
heredity
done
clear
B)
mutations
done
clear
C)
variation
done
clear
D)
both (a) and (c)
done
clear
View Answer play_arrow
question_answer 213) A genetic clone is :
A)
hybrid produced by sexual reproduction
done
clear
B)
plants produced by asexual reproduction-
done
clear
C)
heterozygous plant produced by sexual reproduction
done
clear
D)
homozygous plant produced by sexual reproduction .
done
clear
View Answer play_arrow
question_answer 214) Milk is changed into curd by :
A)
Lactobacillus
done
clear
B)
Acrtobacter aceti
done
clear
C)
Bacillus megatherium
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 215) Restriction enzyme was discovered by :
A)
Waksman
done
clear
B)
Flemming
done
clear
C)
Berger
done
clear
D)
Smith and Nathans
done
clear
View Answer play_arrow
question_answer 216) Enzyme diastase was identified by :
A)
Hansen
done
clear
B)
Reamur
done
clear
C)
Payen and Persoz
done
clear
D)
Dubrun Faut
done
clear
View Answer play_arrow
question_answer 217) Heat killed pathogenic pneumonia like cells and live non-pathogenic cells are mixed and injected into mice. The result would be :
A)
mice develop disease and die
done
clear
B)
mice die without developing disease
done
clear
C)
mice remains healthy
done
clear
D)
50% mice develop disease and die
done
clear
View Answer play_arrow
question_answer 218) Non genetic RNA is of :
A)
two types
done
clear
B)
three types
done
clear
C)
four types
done
clear
D)
one types
done
clear
View Answer play_arrow
question_answer 219) Which is the source of latex containing liquid hydrocarbons ?
A)
Asclepiadaceae
done
clear
B)
Euphorbiaceae
done
clear
C)
Apocynaceae and Compositae
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 220) Which wood is most suitable as fuel wood ?
A)
Monocot
done
clear
B)
Dicot
done
clear
C)
Gymnosperm
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 221) Leucaena leucocephala, a new plant for social forestry is recommended for :
A)
fodder
done
clear
B)
fire wood
done
clear
C)
bio-fertilizer
done
clear
D)
hedge plant
done
clear
View Answer play_arrow
question_answer 222) A good fire wood is that which is not :
A)
resionous
done
clear
B)
smoky
done
clear
C)
highly combustible
done
clear
D)
easy to dry
done
clear
View Answer play_arrow
question_answer 223) Segregation of Mendelian factors (no linkage, no crossing over) occurs during :
A)
anaphase-I
done
clear
B)
anaphase-II
done
clear
C)
diplotene
done
clear
D)
metaphase-I
done
clear
View Answer play_arrow
question_answer 224) Gametes are never hybrid It is a statement of law of :
A)
dominance
done
clear
B)
segregation
done
clear
C)
independent assortment
done
clear
D)
random fertilization
done
clear
View Answer play_arrow
question_answer 225) Pollen grain wings of Finns are out growths of :
A)
exine
done
clear
B)
intine
done
clear
C)
exo-intine
done
clear
D)
sporangial wall
done
clear
View Answer play_arrow
question_answer 226) Sago of Cycas is given to patients with stomach disorders because it is :
A)
with high nutritive value
done
clear
B)
tastier
done
clear
C)
easily digestible with less starch
done
clear
D)
cheap
done
clear
View Answer play_arrow
question_answer 227) Gymnosperms have a mode of pollination celled:
A)
anemophily
done
clear
B)
zoophily
done
clear
C)
entomophily
done
clear
D)
hydrophily
done
clear
View Answer play_arrow
question_answer 228) Arbor vitae is composed of :
A)
Grey matter
done
clear
B)
White matter
done
clear
C)
Neuroglia cells
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 229) Jacobsons organ is found in :
A)
nose
done
clear
B)
eye
done
clear
C)
kidney
done
clear
D)
ear
done
clear
View Answer play_arrow
question_answer 230) Oxytocin hormone is produced by :
A)
pituitary
done
clear
B)
adrenals
done
clear
C)
hypothalamus
done
clear
D)
thyroid
done
clear
View Answer play_arrow
question_answer 231) Queen Victoria of England was :
A)
Haemophilic carrier
done
clear
B)
Colour blind
done
clear
C)
AIDS patient
done
clear
D)
deaf
done
clear
View Answer play_arrow
question_answer 232) Distance between two linked genes is measured in map units that depict :
A)
ratio of crossing over between them
done
clear
B)
crossing over value
done
clear
C)
number of genes between them
done
clear
D)
both (b) and (c)
done
clear
View Answer play_arrow
question_answer 233) Extra 18th autosomal chromosome results in:
A)
Edwards syndrome
done
clear
B)
Pataus syndrome
done
clear
C)
Downs syndrome
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 234) Male sex is determined in human zygote by :
A)
nutrition of mother
done
clear
B)
strength of father
done
clear
C)
chromosome composition of egg
done
clear
D)
chromosome composition of sperm
done
clear
View Answer play_arrow
question_answer 235) When the discovery is made by accident and unexpectedly the phenomenon is called :
A)
mutation
done
clear
B)
serendipity
done
clear
C)
recombination
done
clear
D)
linkage
done
clear
View Answer play_arrow
question_answer 236) Thermostate of human body is :
A)
hypothalamus
done
clear
B)
pituitary
done
clear
C)
thyroid
done
clear
D)
muscle
done
clear
View Answer play_arrow
question_answer 237) Recently discovered only proteinous pathogenic particle is called :
A)
virion
done
clear
B)
protovirus
done
clear
C)
prion
done
clear
D)
protoprotein
done
clear
View Answer play_arrow
question_answer 238) Study of the fossils comes under :
A)
Paleontology
done
clear
B)
Palaeogeography
done
clear
C)
Herpetology
done
clear
D)
Organic evolution
done
clear
View Answer play_arrow
question_answer 239) Fossils are dated by :
A)
radioactive carbon content
done
clear
B)
amount of calcium
done
clear
C)
association with mammals
done
clear
D)
stratigraphic age
done
clear
View Answer play_arrow
question_answer 240) Evolution is :
A)
development of DNA from nucleotides
done
clear
B)
development of organism through time
done
clear
C)
development of a cell from chemicals
done
clear
D)
cloning
done
clear
View Answer play_arrow
question_answer 241) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Penicillin was discovered by Sir Alexander Flemming R : Discovery of penicillin in an example of serendipity
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 242) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Cytochrome c and haemoglobin help in transporting oxygen R : Both have high affinity for oxygen.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 243) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : In a DNA molecule, the total quantity of purines equal to the total quantity of pyrimidines. R : Adenine pairs with thymine and cytosine pairs with guanine.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 244) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Electron microscope has very high resolving power. R : It has powerful magnetic lenses.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 245) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Pepsin is a conjugated enzyme. R : It requires Mg^ as prosthetic group for its activity.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 246) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Fruits soften as these ripen R : The lignin of the cell wall is hydrolysed
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 247) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Workers prior to Mendel could not work out the principles of inheritance R : They considered the individual as a whole complex of characters.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 248) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Histones ate basic proteins. R : These interact with DNA and neutralize negatively charged phosphate groups.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 249) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Lac operon model is applicable to E coli. R : E. coli is a prokaryote.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
question_answer 250) Direction: Each of the question below consists of two statements an Assertion (A) and a Reason (R) mark the appropriate response as under: A : Eiiglena shows mixotrophic life style. R : Eiiglena is autotrophic in sunlight and heterotrophic in the absence of sunlight.
A)
If both A and R are true and R is the correct explanation of A
done
clear
B)
If both A and R are true but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
Both A and R are false
done
clear
View Answer play_arrow
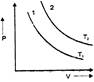
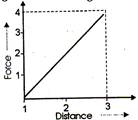 zero at the begining. How much kinetic energy is changed ?
zero at the begining. How much kinetic energy is changed ?