question_answer 1) Which of the following is true regarding beats?
A)
Frequency different, amplitude same
done
clear
B)
Frequency same, amplitude same
done
clear
C)
Frequency same, amplitude different
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 2) Moment of inertia of ring about its diameter is I. Then, moment of inertia about an axis passing through centre perpendicular to its plane is
A)
\[2I\]
done
clear
B)
\[\frac{I}{2}\]
done
clear
C)
\[\frac{3}{2}I\]
done
clear
D)
\[I\]
done
clear
View Answer play_arrow
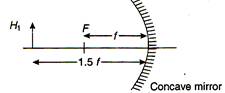
question_answer 3)
If in the following figure, height of object is \[{{H}_{1}}=+2.5cm,\] then height of image H2 formed is
A)
-5 cm
done
clear
B)
+5 cm
done
clear
C)
+7.5 cm
done
clear
D)
-7.5cm
done
clear
View Answer play_arrow
question_answer 4) In a semiconductor electron concentration is \[7\times {{10}^{13}}c{{m}^{-3}}\] and hole concentration is \[5\times {{10}^{12}}c{{m}^{-3}}.\] Then the semiconductor is
A)
n-type
done
clear
B)
p-type
done
clear
C)
intrinsic
done
clear
D)
p-n type
done
clear
View Answer play_arrow
question_answer 5) The output is low when either of the input is high, then this represents which of the following gates?
A)
OR
done
clear
B)
NOR
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
question_answer 6) Reverberation time does not depend upon
A)
temperature
done
clear
B)
volume of room
done
clear
C)
size of window
done
clear
D)
carpet and curtain
done
clear
View Answer play_arrow
question_answer 7) If\[{{B}_{H}}=\frac{1}{\sqrt{3}}{{B}_{v,}}\] find angle of dip. (where symbols have their usual meanings)
A)
\[60{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
\[45{}^\circ \]
done
clear
D)
\[90{}^\circ \]
done
clear
View Answer play_arrow
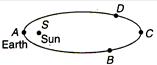
question_answer 8)
The earth moves in an elliptical orbit with the sun S at one of foci as shown in the figure. Its rotational kinetic energy is maximum at the point
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
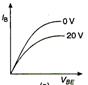
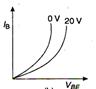
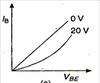
question_answer 9) Graph of input characteristic of common emitter amplifier is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 10) In a potentiometer the null point is obtained at 7th wire. If now we have to change the null point at 9th wire, what should we do?
A)
Attach resistance in series with battery
done
clear
B)
Increase resistance in main circuit
done
clear
C)
Decrease resistance in main circuit
done
clear
D)
Decrease applied emf
done
clear
View Answer play_arrow
question_answer 11) An aluminium rod and a copper rod are taken such that their lengths are same and their resistances are also same. The specific, resistance of copper is half that of aluminium, but its density is three times that of aluminium. The ratio of the mass of aluminium rod and that of copper rod will be
A)
\[\frac{1}{6}\]
done
clear
B)
\[\frac{2}{3}\]
done
clear
C)
\[\frac{1}{3}\]
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 12) If average velocity becomes 4 times then what will be the effect on rms velocity at that temperature?
A)
1.4 times
done
clear
B)
4 times
done
clear
C)
2 times
done
clear
D)
\[\frac{1}{4}\] times
done
clear
View Answer play_arrow
question_answer 13) What is the Q-value of the reaction \[p{{+}^{7}}Li{{\xrightarrow{{}}}^{4}}He{{+}^{4}}He\] The atomic masses of\[{{~}^{1}}H{{,}^{4}}He\] and \[^{7}Li\]are 1.007825\[u\], 4.002603 \[u\] and 7.016004 \[u\] respectively
A)
17.35 MeV
done
clear
B)
18.06 MeV
done
clear
C)
177.35 MeV
done
clear
D)
170.35 MeV
done
clear
View Answer play_arrow
question_answer 14) Which of the following is not a state function?
A)
Work done at constant pressure Enthalpy
done
clear
B)
Work done by conservative force
done
clear
C)
Work done by non-conservative force
done
clear
D)
A Carnots engine has an efficiency of 50% at
done
clear
View Answer play_arrow
question_answer 15) Sink temperature \[50{}^\circ C\]. Calculate the temperature of source.
A)
\[133{}^\circ C\]
done
clear
B)
\[143{}^\circ C\]
done
clear
C)
\[100{}^\circ C\]
done
clear
D)
\[373{}^\circ C\]
done
clear
View Answer play_arrow
question_answer 16) When both the listener and source are moving towards each other, then which of the following is true regarding frequency and wavelength of wave observed by the observer?
A)
More frequency, less wavelength
done
clear
B)
More frequency, more wavelength
done
clear
C)
Less frequency, less wavelength
done
clear
D)
More frequency, constant wavelength
done
clear
View Answer play_arrow
question_answer 17) Which motion does not require force to maintain it?
A)
Uniform circular motion
done
clear
B)
Elliptical motion
done
clear
C)
Uniform straight line motion
done
clear
D)
Projectile motion
done
clear
View Answer play_arrow
question_answer 18) The coefficient of friction between the tyres and the road is 0.25. The maximum speed with which car can be driven round a curve of radius 40 m without skidding is (assume \[g=10m{{s}^{-2}}\]
A)
\[40m{{s}^{-1}}\]
done
clear
B)
\[20m{{s}^{-1}}\]
done
clear
C)
\[15m{{s}^{-1}}\]
done
clear
D)
\[10m{{s}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 19) In forward bias the width of depletion layer decreases with increase in potential
A)
barrier voltage
done
clear
B)
increases with increase in potential barrier voltage
done
clear
C)
is independent of potential barrier voltage
done
clear
D)
none of the above
done
clear
View Answer play_arrow
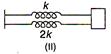
question_answer 20)
If \[{{K}_{s}},\]and \[{{K}_{p}},\] respectively are effective spring constants in series and parallel combination of springs as shown in figure, find \[\frac{{{K}_{s}}}{{{K}_{p}}}\]
A)
\[\frac{9}{2}\]
done
clear
B)
\[\frac{3}{7}\]
done
clear
C)
\[\frac{2}{9}\]
done
clear
D)
\[\frac{7}{3}\]
done
clear
View Answer play_arrow
question_answer 21)
A bob of pendulum was filled with Hg and entire Hg is drained out, then the time period of pendulum
A)
remains unchanged
done
clear
B)
decreases
done
clear
C)
increases
done
clear
D)
increases then decreases
done
clear
View Answer play_arrow
question_answer 22) The minimum force required to move a body of mass m vertically upward is
A)
mg
done
clear
B)
mg/2
done
clear
C)
more than 2mg
done
clear
D)
more than mg
done
clear
View Answer play_arrow
question_answer 23) The uncertainty in the momentum of a particle is \[{{10}^{-30}}kg-m/s.\] The minimum uncertainty in its position will be
A)
\[{{10}^{-8}}\] m
done
clear
B)
\[{{10}^{-12}}\] m
done
clear
C)
\[{{10}^{-16}}\]m
done
clear
D)
\[{{10}^{-4}}\] m
done
clear
View Answer play_arrow
question_answer 24) If distance between earth and sun becomes four times, then time period becomes
A)
4 times
done
clear
B)
8 times
done
clear
C)
1/4 times
done
clear
D)
1/8 times
done
clear
View Answer play_arrow
question_answer 25)
An air bubble is contained inside water. It behaves as a
A)
concave lens
done
clear
B)
convex lens
done
clear
C)
neither convex nor concave
done
clear
D)
cannot say
done
clear
View Answer play_arrow
question_answer 26) The power dissipated across resistance R which is connected across a battery of potential V is P. If resistance is doubled, then the power becomes
A)
\[\frac{1}{2}\]
done
clear
B)
\[2\]
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
\[4\]
done
clear
View Answer play_arrow
question_answer 27) BE/nucleon relation with mass number
A)
first decreases then increases
done
clear
B)
first increases then decreases
done
clear
C)
increases
done
clear
D)
decreases
done
clear
View Answer play_arrow
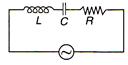
question_answer 28)
A 100 V, AC source of frequency 500 Hz is connected to an LCR circuit with L =8.1 mH, C = 12.5\[\mu \]F, R = 10 \[\Omega \] all connected in series as shown in figure. What is the quality factor of circuit?
A)
2.02
done
clear
B)
2.5434
done
clear
C)
20.54
done
clear
D)
200.54
done
clear
View Answer play_arrow
question_answer 29) The inductance of a coil is L = 10 H and resistance R = 5 \[\Omega \]. If applied voltage of battery is 10 V and it switches off in 1 millisecond, find induced emf of inductor.
A)
\[2\times {{10}^{4}}V\]
done
clear
B)
\[1.2\times {{10}^{4}}V\]
done
clear
C)
\[2\times {{10}^{-4}}V\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 30) Two bodies A and B having masses in the ratio of 3:1 possess the same kinetic energy. The ratio of linear momentum of B to A is
A)
1:3
done
clear
B)
3:1
done
clear
C)
\[1:\sqrt{3}\]
done
clear
D)
\[\sqrt{3}:1\]
done
clear
View Answer play_arrow
question_answer 31) Which of the following four fundamental forces has shortest range?
A)
Nuclear
done
clear
B)
Gravitational
done
clear
C)
Electromagnetic
done
clear
D)
Weak force
done
clear
View Answer play_arrow
question_answer 32) A proton is moving in a uniform magnetic field B in a circular path of radius a in a direction perpendicular to z-axis along which field B exists. Calculate the angular momentum, if the radius is a and charge on proton is e.
A)
\[\frac{Be}{{{a}^{2}}}\]
done
clear
B)
\[e{{B}^{2}}a\]
done
clear
C)
\[{{e}^{2}}eB\]
done
clear
D)
\[aeB\]
done
clear
View Answer play_arrow
question_answer 33) If acceleration of a panicle at any time is given by \[a=2t+5\] Calculate the velocity after 5 s, if it starts from rest.
A)
50 m/s
done
clear
B)
25 m/s
done
clear
C)
100 m/s
done
clear
D)
75 m/s
done
clear
View Answer play_arrow
question_answer 34) 8 bits form
A)
1 byte
done
clear
B)
1 diction
done
clear
C)
1 nibble
done
clear
D)
1 binary
done
clear
View Answer play_arrow
question_answer 35) A body from height h is dropped. If the coefficient of restitution is e, then calculate the height achieved after one bounce.
A)
\[{{h}_{1}}={{e}^{2}}h\]
done
clear
B)
\[{{h}_{1}}={{e}^{4}}h\]
done
clear
C)
\[{{h}_{1}}=eh\]
done
clear
D)
\[{{h}_{1}}\frac{h}{e}\]
done
clear
View Answer play_arrow
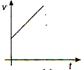
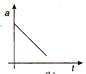
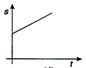
question_answer 36) A body moves with uniform acceleration, then which of the following graphs is correct?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 37) Two vectors are perpendicular, if
A)
\[\overrightarrow{\text{A}}\text{.}\overrightarrow{\text{B}}\text{.=1}\]
done
clear
B)
\[\overrightarrow{\text{A}}\text{.}\overrightarrow{\text{B}}\text{.=0}\]
done
clear
C)
\[\overrightarrow{\text{A}}\text{.}\overrightarrow{\text{B}}\text{=}\,\text{0}\]
done
clear
D)
\[\overrightarrow{\text{A}}\text{.}\overrightarrow{\text{B}}\text{=}\,AB\]
done
clear
View Answer play_arrow
question_answer 38) If coefficient of static friction is \[{{\mu }_{s}}\], and coefficient of kinetic friction is \[{{\mu }_{k}}\], which is correct?
A)
\[{{\mu }_{s}}={{\mu }_{k}}\]
done
clear
B)
\[{{\mu }_{s}}>{{\mu }_{k}}\]
done
clear
C)
\[{{\mu }_{s}}<{{\mu }_{k}}\]
done
clear
D)
Cannot predict
done
clear
View Answer play_arrow
question_answer 39) Light year is used to measure
A)
distance between stars
done
clear
B)
distance between atoms
done
clear
C)
revolution time of earth around sun
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 40) Electromagnetic waves are produced by
A)
accelerated charged particle
done
clear
B)
decelerated charged particle
done
clear
C)
charge in uniform motion
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 41) A beam of protons with velocity \[4\times {{10}^{5}}m/s\] enters a uniform magnetic field of 0.3 T at an angle of \[60{}^\circ \] to the magnetic field. Find the radius of the helical path taken by the proton beam.
A)
0.2 cm
done
clear
B)
1.2 cm
done
clear
C)
2.2cm
done
clear
D)
0.122cm
done
clear
View Answer play_arrow
question_answer 42)
Three progressive waves A, B and C are shown in the figure.
A)
B lags by \[\frac{\pi }{2}\] and C leads by \[\frac{\pi }{2}\]
done
clear
B)
B lags by \[\pi \] and C leads by \[\pi \]
done
clear
C)
B leads by \[\frac{\pi }{2}\] and C lags by \[\frac{\pi }{2}\]
done
clear
D)
B leads by \[\pi \]and C lags by \[\pi \]
done
clear
View Answer play_arrow
question_answer 43) When light passes from one medium to other then which will not change?
A)
Frequency
done
clear
B)
Wavelength
done
clear
C)
Amplitude
done
clear
D)
Velocity
done
clear
View Answer play_arrow
question_answer 44) Total internal reflection takes place
A)
when a ray moves from denser to rarer and incident angle is greater than critical angle
done
clear
B)
when a ray moves from rarer to denser and incident angle is less than critical angle
done
clear
C)
when a ray moves from rarer to denser and incident angle is equal to critical angle
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 45) A beam of light travelling along x-axis is described by the electric field \[{{\text{E}}_{\text{y}}}\text{=(600V}{{\text{m}}^{\text{-1}}}\text{)sin }\!\!\omega\!\!\text{ (t-x/c)}\] then maximum magnetic force on a charge.\[\text{q=}{{\text{2}}^{\text{e}}}\text{,}\] moving along y-axis with a speed of \[3.0\times {{10}^{7}}m{{s}^{-1}}\] is \[(e=1.6\times {{10}^{-19}}C)\]
A)
\[19.2\times {{10}^{-17}}N\]
done
clear
B)
\[1.92\times {{10}^{-17}}N\]
done
clear
C)
0.192 N
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 46) Which of the following is polar?
A)
\[I_{3}^{-}\]
done
clear
B)
\[CO_{3}^{2-}\]
done
clear
C)
\[Xe{{F}_{4}}\]
done
clear
D)
\[P{{F}_{3}}\] n the reaction,
done
clear
View Answer play_arrow
question_answer 47)
In the reaction,
A)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
B)
\[C{{U}_{2}}C{{l}_{2}}\]
done
clear
C)
\[HgS{{O}_{4}}/{{H}_{2}}S{{O}_{4}}\]
done
clear
D)
\[{{H}^{+}}/{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 48) \[Ag\] crystallises as fee. If radius of \[Ag\] is \[~144\text{ }pm\]then its density will be
A)
\[10ge{{m}^{-3}}\]
done
clear
B)
\[5\text{ }ge{{m}^{-3}}\]
done
clear
C)
\[15\text{ }ge{{m}^{-3}}\]
done
clear
D)
\[6.5\text{ }ge{{m}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 49) Which of the following process will give soap?
A)
Hydrolysis
done
clear
B)
Saponification
done
clear
C)
Neutralisation
done
clear
D)
Acidification
done
clear
View Answer play_arrow
question_answer 50) Sodium hypo halite when dissolved in water will turn
A)
blue litmus red
done
clear
B)
red litmus blue
done
clear
C)
red litmus green
done
clear
D)
no change
done
clear
View Answer play_arrow
question_answer 51) A when added to ^silica will give B. A and B are
A)
\[HF,{{H}_{2}}Si{{F}_{4}}\]
done
clear
B)
\[HF,{{H}_{2}}Si{{F}_{6}}\]
done
clear
C)
\[HCl,{{H}_{2}}SiC{{l}_{6}}\]
done
clear
D)
\[HI,{{H}_{2}}Si{{I}_{6}}\]
done
clear
View Answer play_arrow
question_answer 52)
The compound
A)
alkyne, \[{{3}^{o}}\] amine
done
clear
B)
alkene, \[{{2}^{o}}\] amine
done
clear
C)
alkene, \[{{3}^{o}}\] amine
done
clear
D)
alkyne, \[{{2}^{o}}\] amine
done
clear
View Answer play_arrow
question_answer 53) The number of geometrical isomers in \[C{{H}_{3}}CH=CHC{{H}_{2}}CH=C{{H}_{2}}\] are
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 54) IUPAC name of the compound\[C{{H}_{2}}=CH-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\underset{{{C}_{2}}{{H}_{5}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,HC{{H}_{3}}\]
A)
\[3-ethyl-4-methyl\text{ }hex-5-en-2-ol\]
done
clear
B)
\[3-methyl-4-ethyl\text{ }hex-l-en-5-ol\]
done
clear
C)
\[3-ethyl-2-hydroxy-4-methylhex-5-ene\]
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 55) In the reaction,\[C{{H}_{3}}C{{H}_{2}}COON{{H}_{2}}\xrightarrow{{{P}_{2}}{{O}_{5}}}A\xrightarrow{{{H}^{+}}/{{H}_{2}}O}B\]A and B are
A)
\[C{{H}_{3}}C{{H}_{2}}CON{{H}_{2}},\,\,C{{H}_{3}}C{{H}_{2}}CO{{O}^{-}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COON{{H}_{2}},\,\,C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CN,\text{ }C{{H}_{3}}C{{H}_{2}}CO{{O}^{-}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CN,\text{ }C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
question_answer 56) Number of a and n bonds in \[{{C}_{6}}{{H}_{5}}COOH\] are
A)
\[15\sigma ,\,\,4\pi \]
done
clear
B)
\[14\sigma ,\,\,4\pi \]
done
clear
C)
\[13\sigma ,\,\,4\pi \]
done
clear
D)
\[16\sigma ,\,\,4\pi \]
done
clear
View Answer play_arrow
question_answer 57) \[{{\Psi }^{2}}=0\]represent
A)
node
done
clear
B)
orbital
done
clear
C)
angular wave function
done
clear
D)
wave functions
done
clear
View Answer play_arrow
question_answer 58) Which of the following disaccharide has different type of linkage?
A)
Maltose
done
clear
B)
Galactose
done
clear
C)
Starch
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
question_answer 59) For a reaction,\[\Delta H=+29\,kJ\,mo{{l}^{-1}}\]\[\Delta S=-35\,kJ\,mo{{l}^{-1}}\]at what temperature the reaction will be spontaneous?
A)
\[{{828.7}^{o}}C\]
done
clear
B)
\[828.7K\]
done
clear
C)
Spontaneous at all temperature
done
clear
D)
Not possible
done
clear
View Answer play_arrow
question_answer 60) As the number of nucleon increases, binding energy per nucleon
A)
increases
done
clear
B)
decreases
done
clear
C)
first increases then decreases
done
clear
D)
first decreases then increases
done
clear
View Answer play_arrow
question_answer 61) Rate constant has the unit \[mo{{l}^{-2}}\text{ }{{L}^{2}}\text{ }{{s}^{-1}}\], then order of reaction is
A)
zero
done
clear
B)
first
done
clear
C)
second
done
clear
D)
third
done
clear
View Answer play_arrow
question_answer 62) The reaction follows the mechanism\[A+BAB\,\,\,(fast)\]\[AB+B\xrightarrow{{{k}_{2}}}A+{{B}_{2}}\,\,(slow)\]then rate law is
A)
\[r=k[A][B]\]
done
clear
B)
\[r=k[AB][B]\]
done
clear
C)
\[r=k[A]{{[B]}^{2}}\]
done
clear
D)
\[r=k{{[A]}^{2}}[B]\]
done
clear
View Answer play_arrow
question_answer 63) Ozonolysis of which of the following will give\[HCHO\]?
A)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
D)
\[C{{H}_{2}}=CHCH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 64) \[25g\] of \[MC{{l}_{4}}\] contains \[0.5mole\]chlorine then its molecular weight is
A)
\[100g\text{ }mo{{l}^{-1}}\]
done
clear
B)
\[200\text{ }g\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[150g\text{ }mo{{l}^{-1}}\]
done
clear
D)
\[400g\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 65) pH of the solution containing \[1.04\times {{10}^{-12}}M\]\[KOH\] is
A)
\[-11.9\]
done
clear
B)
\[+11.9\]
done
clear
C)
\[7\]
done
clear
D)
\[2.30\]
done
clear
View Answer play_arrow
question_answer 66) Benzene gives mainly
A)
addition reaction
done
clear
B)
elimination reaction
done
clear
C)
substitution reaction
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 67) Formula of ammonium manganate is
A)
\[N{{H}_{4}}Mn{{O}_{4~}}\]
done
clear
B)
\[{{(N{{H}_{4}})}_{2}}Mn{{O}_{4~}}\]
done
clear
C)
\[N{{H}_{4}}{{(Mn{{O}_{4~}})}_{2}}\]
done
clear
D)
\[N{{H}_{4}}M{{n}_{2}}{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 68) Heart patients can be given
A)
Mono saturated fatty acid
done
clear
B)
Trans fatty acid
done
clear
C)
Polyunsaturated fatty acid
done
clear
D)
Poly saturated fatty acid
done
clear
View Answer play_arrow
question_answer 69) For the reaction \[1g\,mole\]of \[CaC{{O}_{3}}\]is enclosed in 5 L container \[CaC{{O}_{3}}(s)\xrightarrow{{}}CaO(s)+C{{O}_{2}}(g)\]\[{{K}_{p}}=1.16\]at \[1073K\]then per cent dissociation of \[CaC{{O}_{3}}\]is
A)
\[65%\]
done
clear
B)
\[100%\]
done
clear
C)
\[6.5%\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 70) The only amino acid which is non-chiral, is
A)
lysine
done
clear
B)
proline
done
clear
C)
glycine
done
clear
D)
histidine
done
clear
View Answer play_arrow
question_answer 71) The electrolyte \[[Co(N{{H}_{3}})Cl]C{{l}_{2}}\] is which of the following type?
A)
\[1:2\]
done
clear
B)
\[1:3\]
done
clear
C)
\[2:3\]
done
clear
D)
\[4:1\]
done
clear
View Answer play_arrow
question_answer 72) In heterogeneous catalysis which of the following process takes place?
A)
Absorption
done
clear
B)
Hydration
done
clear
C)
Adsorption
done
clear
D)
Intermediate compound
done
clear
View Answer play_arrow
question_answer 73) An ideal solution is obtained by mixing\[C{{H}_{3}}CHB{{r}_{2}}\]and \[C{{H}_{2}}Br-C{{H}_{2}}Br\] in the ratio \[2:1,{{P}^{o}}_{C{{H}_{3}}CHBr}=173,\,{{P}^{o}}_{C{{H}_{2}}BrC{{H}_{2}}Br}=127\]
A)
\[158\]
done
clear
B)
\[257\]
done
clear
C)
\[137\]
done
clear
D)
\[197\]
done
clear
View Answer play_arrow
question_answer 74) For the reaction,\[{{N}_{2}}+3{{H}_{2}}\rightleftharpoons 2N{{H}_{3}}+heat\]when temperature increases cone. Of
A)
formation of \[N{{H}_{3}}\]increases
done
clear
B)
formation of \[N{{H}_{3}}\] decreases
done
clear
C)
concentration of \[{{N}_{2}}\] decreases
done
clear
D)
concentration of \[{{H}_{2}}\] decreases
done
clear
View Answer play_arrow
question_answer 75) Carbon-oxygen bond length of \[C{{H}_{2}}O(I);\]\[C{{H}_{2}}\text{O(II)}\]and \[C{{H}_{3}}OH(III)\]is in the order
A)
\[I>II>III\]
done
clear
B)
\[II>I>III\]
done
clear
C)
\[III>II>I\]
done
clear
D)
\[III>I>II\]
done
clear
View Answer play_arrow
question_answer 76)
In the table Proton Electron Charge 23 X +3 46 43 Y
X and y are
A)
\[20,-3\]
done
clear
B)
\[20,+3\]
done
clear
C)
\[26,-3\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 77) Which of the following is true for polypropylene?
A)
Propylene, condensation polymer
done
clear
B)
Propylene, addition polymer
done
clear
C)
Propylene, ammonic polymers
done
clear
D)
Propylene, cationic ammonic polymerase
done
clear
View Answer play_arrow
question_answer 78) The total number of orbital up to \[n=4\] are
A)
\[2\]
done
clear
B)
\[16\]
done
clear
C)
\[32\]
done
clear
D)
\[30\]
done
clear
View Answer play_arrow
question_answer 79) Coordination number of a body centred cubic is
A)
\[6\]
done
clear
B)
\[8\]
done
clear
C)
\[10\]
done
clear
D)
\[12\]
done
clear
View Answer play_arrow
question_answer 80) x mole of \[BaC{{O}_{3}}\]are soluble in 1 L then x is related to its solubility product by the expression
A)
\[x={{K}_{sp}}\]
done
clear
B)
\[{{x}^{2}}=\sqrt{{{K}_{sp}}}\]
done
clear
C)
\[x=\sqrt{{{K}_{sp}}}\]
done
clear
D)
\[x=\frac{{{K}_{sp}}}{2}\]
done
clear
View Answer play_arrow
question_answer 81) Which of the following do not have \[s{{p}^{2}}\]hybridised carbon?
A)
\[HCHO\]
done
clear
B)
\[C{{H}_{3}}OH\]
done
clear
C)
\[HCOOH\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 82) Isopigknic solution are those which have
A)
same vapour pressure
done
clear
B)
same concentration
done
clear
C)
same density
done
clear
D)
same osmotic pressure
done
clear
View Answer play_arrow
question_answer 83) Which of the following is a superoxide?
A)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
B)
\[K{{O}_{2}}\]
done
clear
C)
\[{{K}_{2}}O\]
done
clear
D)
\[{{C}_{3}}{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 84) The total number of isomers in \[{{C}_{6}}{{H}_{3}}C{{l}_{3}}\]
A)
two
done
clear
B)
three
done
clear
C)
four
done
clear
D)
five
done
clear
View Answer play_arrow
question_answer 85) In the reaction of \[NaH\] with water which of the following is present?
A)
\[Na{{O}^{-}}\]
done
clear
B)
\[O{{H}^{-}}\]
done
clear
C)
\[{{H}^{+}}\]
done
clear
D)
\[N{{a}^{-}}\]
done
clear
View Answer play_arrow
question_answer 86) NaH contains how many covalent bonds?
A)
Zero
done
clear
B)
Two
done
clear
C)
One
done
clear
D)
Three
done
clear
View Answer play_arrow
question_answer 87) \[{{S}_{g}}\] and \[{{S}_{x}}\] are
A)
isotopes
done
clear
B)
isotones
done
clear
C)
allotropes
done
clear
D)
isobars
done
clear
View Answer play_arrow
question_answer 88) The total number of lone pair of \[Cl{{F}_{3}}\] are
A)
\[2\]
done
clear
B)
\[9\]
done
clear
C)
\[11\]
done
clear
D)
\[15\]
done
clear
View Answer play_arrow
question_answer 89) For the reaction between \[KMn{{O}_{4}}\]and\[~{{H}_{2}}{{O}_{2}}\] the number of electrons transferred per mol of \[~{{H}_{2}}{{O}_{2}}\] are
A)
one
done
clear
B)
two
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
question_answer 90) What will be the resultant pH when \[200Ml\]of an aqueous solution of \[HCl\]\[(pH=12)\] is mixed with 300 mL of an aqueous solution of\[NaOH\] \[(pH=12)\]?
A)
\[10\]
done
clear
B)
\[11\]
done
clear
C)
\[12\]
done
clear
D)
\[14\]
done
clear
View Answer play_arrow
question_answer 91) Term Ecosystem development to ecological succession was given by
A)
Odum
done
clear
B)
Clements
done
clear
C)
R. Mishra
done
clear
D)
Blackmans
done
clear
View Answer play_arrow
question_answer 92) Root cap is absent in
A)
xerophytes
done
clear
B)
mesophytes
done
clear
C)
hydrophytes
done
clear
D)
halophytes
done
clear
View Answer play_arrow
question_answer 93) Van Mahotsav was started by
A)
K. M. Munshi
done
clear
B)
Sunder Lal Bahuguna
done
clear
C)
Binova Bhave
done
clear
D)
J. L. Nehru
done
clear
View Answer play_arrow
question_answer 94) Zoochlorellae is an example of
A)
mutualism
done
clear
B)
protocooperation
done
clear
C)
parasitism
done
clear
D)
predadon
done
clear
View Answer play_arrow
question_answer 95) Pollution caused by persistent pesticides is relatively more hazardous to which type of organisms?
A)
Herbivores
done
clear
B)
First level carnivores
done
clear
C)
Producers
done
clear
D)
Top carnivores
done
clear
View Answer play_arrow
question_answer 96) First man made cereal (i.e., Triticale) is
A)
octaploid
done
clear
B)
hexaploid
done
clear
C)
both (a) and (b)
done
clear
D)
diploid
done
clear
View Answer play_arrow
question_answer 97) First hormone produced artificially by culturing bacteria is
A)
insulin
done
clear
B)
thyroxine
done
clear
C)
testosterone
done
clear
D)
adrenaline
done
clear
View Answer play_arrow
question_answer 98) The dried fruit used for making musical instruments is
A)
snake gourd
done
clear
B)
bitter gourd
done
clear
C)
bottle gourd
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 99) Inflorescence axis is called
A)
rachis
done
clear
B)
pedicel
done
clear
C)
petiole
done
clear
D)
peduncle
done
clear
View Answer play_arrow
question_answer 100) Triploid tissue in angiosperms is
A)
nucellus
done
clear
B)
endosperm
done
clear
C)
endothelium
done
clear
D)
tapetum
done
clear
View Answer play_arrow
question_answer 101) Ramenta are characteristic of
A)
Marchantia
done
clear
B)
Funaria
done
clear
C)
Dryopteris
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 102) Single seeded winged fruit is called
A)
achene
done
clear
B)
cypsela
done
clear
C)
samara
done
clear
D)
caryopsis
done
clear
View Answer play_arrow
question_answer 103) Gynobasic style is characteristic feature of
A)
Malvaceae
done
clear
B)
Lamiaceae
done
clear
C)
Ranunculaceae
done
clear
D)
Brassicaceae
done
clear
View Answer play_arrow
question_answer 104) Source of kelp is
A)
green algae
done
clear
B)
blue-green algae
done
clear
C)
brown algae
done
clear
D)
red algae
done
clear
View Answer play_arrow
question_answer 105) Which is not present in moss sporophyte?
A)
Elaters
done
clear
B)
Foot
done
clear
C)
Seta
done
clear
D)
Columella
done
clear
View Answer play_arrow
question_answer 106) Tapetal cells are characterized by
A)
mitotic division
done
clear
B)
meiotic division
done
clear
C)
endomitosis
done
clear
D)
endomitosis as well as endopolyploidy
done
clear
View Answer play_arrow
question_answer 107) Saffron is obtained from
A)
flowers of Crocus
done
clear
B)
styles and stigma of Crocus
done
clear
C)
flowers of Carthamus
done
clear
D)
seeds of Carthamus
done
clear
View Answer play_arrow
question_answer 108) Best material for demonstrating streaming movements of protoplasm within living cells is
A)
onion peelings
done
clear
B)
staminal hair of Tradescantia
done
clear
C)
sensitive plant
done
clear
D)
pith cells
done
clear
View Answer play_arrow
question_answer 109) Nutation is shown by
A)
root
done
clear
B)
stem
done
clear
C)
tendril
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 110) Short day plant is
A)
Xanthium
done
clear
B)
Pisum
done
clear
C)
Cucumis
done
clear
D)
Avena
done
clear
View Answer play_arrow
question_answer 111) A hormone delaying senescence is
A)
auxin
done
clear
B)
cytoldnin
done
clear
C)
ethylene
done
clear
D)
gibberellins
done
clear
View Answer play_arrow
question_answer 112) Anemophily type of pollination is found in
A)
Salvia
done
clear
B)
bottle brush
done
clear
C)
Vallisneria
done
clear
D)
coconut
done
clear
View Answer play_arrow
question_answer 113) The outermost layer of maize endosperm is known as
A)
perisperm
done
clear
B)
aleurone
done
clear
C)
tapetum
done
clear
D)
endothelium
done
clear
View Answer play_arrow
question_answer 114) The net gain of ATP molecules in glycolysis is
A)
8
done
clear
B)
2
done
clear
C)
4
done
clear
D)
0
done
clear
View Answer play_arrow
question_answer 115) Maximum number ofATP is obtained from
A)
glucose
done
clear
B)
palmitic acid
done
clear
C)
malic acid
done
clear
D)
P-amino acid
done
clear
View Answer play_arrow
question_answer 116) Which of the following cell do no fYespire?
A)
Epidermal cell
done
clear
B)
Cork cell
done
clear
C)
RBC
done
clear
D)
Sieve tube cell
done
clear
View Answer play_arrow
question_answer 117) PS-I and PS-II were discovered by
A)
Robert Emerson
done
clear
B)
Blackman
done
clear
C)
Robert Mayer
done
clear
D)
Arnon
done
clear
View Answer play_arrow
question_answer 118) Maximum \[C{{O}_{2}}\] fixation is done by
A)
green plants
done
clear
B)
phytoplanktons
done
clear
C)
zooplanktons
done
clear
D)
bacteria
done
clear
View Answer play_arrow
question_answer 119) Mycorrhiza is an example of
A)
symbiosis
done
clear
B)
parasitism
done
clear
C)
saprophytism
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 120) Stomata can also open at night, present in
A)
xerophytes
done
clear
B)
gametophytes
done
clear
C)
hydrophytes
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 121) Which of the following is a macronutrient?
A)
Mo
done
clear
B)
Ca
done
clear
C)
Zn
done
clear
D)
Mn
done
clear
View Answer play_arrow
question_answer 122) Intercalary meristem results in
A)
secondary growth
done
clear
B)
primary growth
done
clear
C)
apical growth
done
clear
D)
secondary growth
done
clear
View Answer play_arrow
question_answer 123) Bulliform cells found in
A)
seeds of sunflower
done
clear
B)
leaf of wheat
done
clear
C)
pod of pea
done
clear
D)
tuber of potato
done
clear
View Answer play_arrow
question_answer 124) Complementary cells are associated with
A)
lenticels
done
clear
B)
hydathodes
done
clear
C)
rhytidome
done
clear
D)
bark
done
clear
View Answer play_arrow
question_answer 125) Jaculators are present in
A)
ruellia
done
clear
B)
ecballium
done
clear
C)
impatiens
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 126) Which is soluble RNA?
A)
HnRNA
done
clear
B)
rRNA
done
clear
C)
mRNA
done
clear
D)
tRNA
done
clear
View Answer play_arrow
question_answer 127) Export firm of the body is
A)
Golgi bodies
done
clear
B)
ER
done
clear
C)
Nucleus
done
clear
D)
Mitochondria
done
clear
View Answer play_arrow
question_answer 128) Characteristic of meiosis is
A)
two nuclear and two chromosome divisions
done
clear
B)
two nuclear and one chromosome division
done
clear
C)
one nuclear and two chromosome division
done
clear
D)
one nuclear and one chromosome division
done
clear
View Answer play_arrow
question_answer 129) Colchicine arrests which of the following stage of cell division?
A)
Prophase
done
clear
B)
Anaphase
done
clear
C)
Telophase
done
clear
D)
Metaphase
done
clear
View Answer play_arrow
question_answer 130) The endosperm in gymnosperms is
A)
haploid
done
clear
B)
diploid
done
clear
C)
triploid
done
clear
D)
polyploidy
done
clear
View Answer play_arrow
question_answer 131) Transfusion tissue is present in the leaves of
A)
Dryopteris
done
clear
B)
Cycas
done
clear
C)
Pinus
done
clear
D)
Both (b) and (c)
done
clear
View Answer play_arrow
question_answer 132) The 13-celled male gametophyte in Selaginella is
A)
12-cells of antheridium + 1-prothallial cell
done
clear
B)
10-cells of antheridium + 3-prothallial cells
done
clear
C)
9-cells of antheridium + 4-prothallial cells
done
clear
D)
7-cells of antheridium + 6-prothallial cells
done
clear
View Answer play_arrow
question_answer 133) Stomata in Funaria are found in
A)
capsule
done
clear
B)
foot
done
clear
C)
seta
done
clear
D)
no where
done
clear
View Answer play_arrow
question_answer 134) Gibberellin was first discovered from
A)
algae
done
clear
B)
fungi
done
clear
C)
bacteria
done
clear
D)
roots of higher plants
done
clear
View Answer play_arrow
question_answer 135) Heterocysts are found in
A)
Cyanophyceae
done
clear
B)
Chlorophyceae
done
clear
C)
Pheophyceae
done
clear
D)
Rhodophyceae
done
clear
View Answer play_arrow
question_answer 136) Study of reptiles is known as
A)
Serpentology
done
clear
B)
Herpetology
done
clear
C)
Icthyology
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 137) Who wrote the book Genetics and origin of species?
A)
A.I. Oparin
done
clear
B)
Th. Dobzhansky
done
clear
C)
Joseph Hooker
done
clear
D)
Charles Darwin
done
clear
View Answer play_arrow
question_answer 138) The bond present between two carbohydrates molecules is
A)
amide
done
clear
B)
hydrogen
done
clear
C)
glycosidic
done
clear
D)
phosphodiester
done
clear
View Answer play_arrow
question_answer 139) The idea that life originates from pre-existing life is referred as
A)
biogenesis theory
done
clear
B)
special creation theory
done
clear
C)
abiogenesis theory
done
clear
D)
extraterrestrial theory
done
clear
View Answer play_arrow
question_answer 140) Interferons were discovered by
A)
Zinder and Lederberg
done
clear
B)
Isaacs and Lindemann
done
clear
C)
Safferman and Morris
done
clear
D)
Temin
done
clear
View Answer play_arrow
question_answer 141) Wings of birds and butterflies are
A)
homologous organs
done
clear
B)
analogous organs
done
clear
C)
vestigial organs
done
clear
D)
grafted organs
done
clear
View Answer play_arrow
question_answer 142) Genetic drift operates in
A)
large isolated population
done
clear
B)
small isolated population
done
clear
C)
fast reproductive population
done
clear
D)
slow reproductive population
done
clear
View Answer play_arrow
question_answer 143) The modern man differs from the apes in
A)
protruding eyes
done
clear
B)
spare body hair
done
clear
C)
wearing of clothes
done
clear
D)
arms shorter than legs
done
clear
View Answer play_arrow
question_answer 144) An animal without anus is
A)
Unio
done
clear
B)
Pheretima
done
clear
C)
Fasciola
done
clear
D)
Periplaneta
done
clear
View Answer play_arrow
question_answer 145) Identify a deuterostome
A)
Leucosolenia
done
clear
B)
Dentalium
done
clear
C)
Dugesia
done
clear
D)
Ophiothrix
done
clear
View Answer play_arrow
question_answer 146) Arms are absent in
A)
sea urchin
done
clear
B)
sea cucumber
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 147) Isinglass, a type of byproduct of fish industry, is principally used for
A)
production of insulin
done
clear
B)
feeding cattle, pig and poultry
done
clear
C)
preparation of paints and varnishes
done
clear
D)
clarification vinegar, wines and beer
done
clear
View Answer play_arrow
question_answer 148) Which of the following is an egg-laying mammal?
A)
Whale
done
clear
B)
Penguin
done
clear
C)
Platypus
done
clear
D)
Kangaroo
done
clear
View Answer play_arrow
question_answer 149) Barr body of a mammal represents
A)
all heterochromatin in female cells
done
clear
B)
all heterochromatin in male and female cells
done
clear
C)
the Y-chromosome in the somatic cells of male
done
clear
D)
one of the two X-chromosomes in somatic cells of female
done
clear
View Answer play_arrow
question_answer 150) A plasmid
A)
cannot replicate
done
clear
B)
can replicate independently
done
clear
C)
shows independent assortment
done
clear
D)
lies together with chromosomes
done
clear
View Answer play_arrow
question_answer 151) Which of the following acts as middle man of the body?
A)
Plasma
done
clear
B)
Lymph
done
clear
C)
WBC
done
clear
D)
RBC
done
clear
View Answer play_arrow
question_answer 152) Example of embryonic connective tissue is
A)
Wolmans jelly
done
clear
B)
Whartons jelly
done
clear
C)
Wrights jelly
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 153) The rupture of urinary bladder is prevented by
A)
pseudo stratified epithelium
done
clear
B)
stratified columnar epithelium
done
clear
C)
stratified cuboidal epithelium
done
clear
D)
transitional epithelium
done
clear
View Answer play_arrow
question_answer 154) Which one of the following is present in the integument of frog, but not in mammals?
A)
Dermis
done
clear
B)
Mucous gland
done
clear
C)
Sweat glands
done
clear
D)
Stratum germinativum
done
clear
View Answer play_arrow
question_answer 155) A sesamoid bone is
A)
palatins
done
clear
B)
pterygoid
done
clear
C)
patella
done
clear
D)
presphenoid
done
clear
View Answer play_arrow
question_answer 156) An acromian process is characteristically found in
A)
skull of frog
done
clear
B)
sperm of mammals
done
clear
C)
pelvic girdle of mammals
done
clear
D)
pectoral girdle of mammals
done
clear
View Answer play_arrow
question_answer 157) Diastema is associated with
A)
organ of Corti
done
clear
B)
retinal cells
done
clear
C)
presence of certain teeth
done
clear
D)
absence of certain teeth
done
clear
View Answer play_arrow
question_answer 158) Which amino acid is denoted by symbol F?
A)
Phenylalanine
done
clear
B)
Proline
done
clear
C)
Tryptophan
done
clear
D)
Methionine
done
clear
View Answer play_arrow
question_answer 159) Medium in which pepsin is active?
A)
Neutral
done
clear
B)
Alkaline
done
clear
C)
Acidic
done
clear
D)
Isotonic
done
clear
View Answer play_arrow
question_answer 160) Vitamin-D is produced in human body by
A)
skin
done
clear
B)
nerves
done
clear
C)
muscles
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 161) Booklungs are respiratory organs in
A)
scorpion
done
clear
B)
prawn
done
clear
C)
snail
done
clear
D)
cockroach
done
clear
View Answer play_arrow
question_answer 162) Lungs have a large number of narrow tubes called
A)
alveoli
done
clear
B)
bronchi
done
clear
C)
bronchioles
done
clear
D)
tracheae
done
clear
View Answer play_arrow
question_answer 163) Pylangium in frog is found in
A)
conus arteriosus
done
clear
B)
sinus venosus
done
clear
C)
atrium
done
clear
D)
ventricle
done
clear
View Answer play_arrow
question_answer 164) Papillary muscles are located in
A)
heart ventricles of rabbit
done
clear
B)
dermis of mammalian skin
done
clear
C)
orbits of vertebrate eyes
done
clear
D)
pylorus of vertebrate stomach
done
clear
View Answer play_arrow
question_answer 165) A heart murmur indicates a defective
A)
bundle of His
done
clear
B)
heart valves
done
clear
C)
sinoatrial node
done
clear
D)
atrio-ventricular node
done
clear
View Answer play_arrow
question_answer 166) A nephron does not have loop of HenIe in
A)
frog
done
clear
B)
man
done
clear
C)
rabbit
done
clear
D)
dog
done
clear
View Answer play_arrow
question_answer 167) Haemodialysis is associated with
A)
liver
done
clear
B)
spleen
done
clear
C)
kidney
done
clear
D)
stomach
done
clear
View Answer play_arrow
question_answer 168) Average pH of human urine is
A)
6.0
done
clear
B)
9.0
done
clear
C)
3.0
done
clear
D)
7.0
done
clear
View Answer play_arrow
question_answer 169) Node of Ranvier is found in
A)
muscle bundles
done
clear
B)
dendrite
done
clear
C)
Right auricle
done
clear
D)
axon
done
clear
View Answer play_arrow
question_answer 170) Which of the following cranial nerve is present in rabbit but absent in frog?
A)
Glossopharyngeal
done
clear
B)
Hypoglossal
done
clear
C)
Olfactory
done
clear
D)
Optic
done
clear
View Answer play_arrow
question_answer 171) Pecten, a comb-like structure is found in the eye of
A)
fishes
done
clear
B)
frogs
done
clear
C)
birds
done
clear
D)
mammals
done
clear
View Answer play_arrow
question_answer 172) Bony labyrinth contains a fluid called
A)
perilymph
done
clear
B)
endolymph
done
clear
C)
haemolymph
done
clear
D)
lymph
done
clear
View Answer play_arrow
question_answer 173) Columella auris is found in
A)
rabbit
done
clear
B)
frog
done
clear
C)
man
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 174) Static equilibrium is maintained by
A)
utriculus
done
clear
B)
sacculus
done
clear
C)
both (a) and (b)
done
clear
D)
semicircular canals
done
clear
View Answer play_arrow
question_answer 175) Which hormone produces calorigenic effect?
A)
Thyroxine
done
clear
B)
FSH
done
clear
C)
Insulin
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 176) Treatment with alloxan destroys
A)
STH cells
done
clear
B)
alpha cells of islets of Langerhans
done
clear
C)
beta cells of islets of Langerhans
done
clear
D)
cells of Leydig
done
clear
View Answer play_arrow
question_answer 177) Hassalls corpuscles are found in
A)
thymus gland
done
clear
B)
adrenal gland
done
clear
C)
spleen
done
clear
D)
parathyroid gland
done
clear
View Answer play_arrow
question_answer 178) Bartholins glands of female correspond to which glands in male?
A)
Cowpers glands
done
clear
B)
Inguinal glands
done
clear
C)
Rectal glands
done
clear
D)
Prostate glands
done
clear
View Answer play_arrow
question_answer 179) Corpus spongiosum is found in
A)
ovary
done
clear
B)
penis
done
clear
C)
testis
done
clear
D)
uterine wall
done
clear
View Answer play_arrow
question_answer 180) Cytoplasm of ovum does not contain
A)
Golgi complex
done
clear
B)
centrosomes
done
clear
C)
mitochondria
done
clear
D)
ribosomes
done
clear
View Answer play_arrow



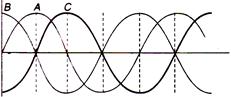 With respect to A, the progressive wave
With respect to A, the progressive wave
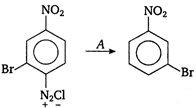 A is
A is
